Abstract
Background:
We studied the management of patients with acute upper gastrointestinal (GI) bleeding (AUGIB) at the Royal Free Hospital. The aim was to compare our performance with the national standard and determine ways of improving the delivery of care in accordance with the recently published ‘Scope for improvement’ report.
Methods:
We randomly selected patients who presented with haematemesis, melaena, or both, and had an oesophageogastroduodenoscopy (OGD) between April and October 2009. We developed local guidelines and presented our findings in various forums. We collaborated with the British Medical Journal’s Evidence Centre and Cerner Millennium electronic patient record system to create an electronic ‘Action Set’ for the management of patients presenting with AUGIB. We re-audited using the same standard and target.
Results:
With the action set, documentation of pre-OGD Rockall scores increased significantly (p ≤ 0.0001). The differences in the calculation and documentation of post-OGD full Rockall scores were also significant between the two audit loops (p = 0.007). Patients who inappropriately received proton-pump inhibitors (PPIs) before endoscopy were reduced from 73.8% to 33% (p = 0.02). Patients receiving PPIs after OGD were also reduced from 66% to 50% (p = 0.01). Discharges of patients whose full Rockall score was less than or equal to two increased from 40% to 100% (p = 0.43).
Conclusion:
The use of the Action Set improved calculation and documentation of risk scores and facilitated earlier hospital discharge for low-risk patients. Significant improvements were also seen in inappropriate use of PPIs. Actions sets can improve guideline adherence and can potentially promote cost-cutting and improve health economics.
Introduction
Acute upper gastrointestinal (GI) bleeding (AUGIB) is the most common emergency managed by gastroenterologists with an incidence of 50–150 per 100,000/year and over 4000 deaths annually in the UK [Hearnshaw et al. 2010]. Anatomically, upper GI bleeding is defined as bleeding above the ligament of Trietz. Clinically, the definition describes haematemesis, or the passage of melaena and/or firm clinical laboratory evidence of acute blood loss from the upper GI tract within a period of 10 preceding days.
There is a 7% mortality in those who present with AUGIB and 26% mortality in those who develop AUGIB during their hospital admission [British Society of Gastroenterology, 2007]. There are several factors affecting mortality such as age [Blatchford et al. 1997; Rockall et al. 1995; Zimmerman et al. 1995], comorbidities [Blatchford et al. 1997; Cameron et al. 2002; Rockall et al. 1995; Zimmerman et al. 1995] such as cardiac, liver and renal disease, as well as the presence of shock [Blatchford et al. 1997; Cameron et al. 2002; Rockall et al. 1995]. The most common causes of AUGIB are peptic ulcer disease (35–50%), gastroduodenal erosions (8–15%), oesophagitis (5–15%) and varices (10–15%) [Barkun et al. 2004; Holman et al. 1990; Marmo et al. 2008; Rockall et al. 1995].
The latest national audit [Hearnshaw et al. 2010] carried out by the National Blood Service and the British Society of Gastroenterology (BSG) highlighted a large gap between published guidelines and management pathways followed by different bleeding units. It called for service providers to bridge this gap by redesigning their existing infrastructure in order to achieve a more standardized care when managing patients presenting with AUGIB. As a response to this, the ‘Scope for improvement’ report was published by the Association of Upper GI Surgeons, the BSG, Royal College of Nursing, Royal College of Physicians and Royal College of Radiologists, with funding support from the National Patient Safety Agency [Palmer, 2011]. The report encourages Trusts to create toolkits that will enable them to manage AUGIB effectively and improve clinical outcomes. It emphasizes the need for minimum standards and local protocols, and calls for confirmed or suspected patients with AUGIB to be appropriately assessed and risk scored at presentation. It also recommends appropriate resuscitation for all patients before endoscopic intervention and systematic data collection in order to measure service provision against auditable outcomes.
We audited the management of patients with AUGIB at the Royal Free hospital (RFH) in London. The aim was to compare our performance to the standard practice at the time of this study which was set by the Scottish Intercollegiate Guidelines Network (SIGN) [Scottish Intercollegiate Guidelines Network, 2008] and was adopted by the BSG. More recently, NICE (the National Institute of Clinical Excellence) has published its guidance on the management of nonvariceal and variceal AUGIB, and once again, the need for service development and appropriate risk assessment for these patients is strongly highlighted.
Methods
Our audit was carried out in two cycles, between April and October 2009 and between September 2010 and February 2011. For the first part of the audit, we searched our endoscopy database (Unisoft, UK) for all patients who underwent an oesophogastroduodenoscopy (OGD) with the indication of melaena, haematemesis, or both in the period April–October 2009. We identified 195 patients who had an OGD, and we were able to retrieve the notes for 30 of them through medical records. The notes were randomly selected [RANDOM.ORG, Randomness and Integrity Services Ltd.].
Three of these patients did not in fact have any history of AUGIB and they were excluded from the study. The results of the remaining 27 patients were analysed in the first loop of this audit.
We examined the clinical notes of these patients and gathered further information from our Trust’s patient electronic care record (CERNER, Kansas, USA). For each patient, we looked at the initial assessment at the time of the AUGIB and their management thereon. We used the SIGN guidelines as the audit standard and full adherence to these guidelines was our audit target.
Several parameters were examined. We looked at the use of appropriate risk stratification such as the Rockall scoring system [Rockall et al. 1996a], before as well as after the OGD [Rockall et al. 1996b; Sanders et al. 2002]. We also looked at the length of hospital stay for each patient and for justification of the hospital admission based on the initial risk assessment. The use of PPIs as well as the different prescription regimens were also explored.
As part of our implementation strategy we developed local guidelines and protocols. Our results and findings were presented at the hospital’s Grand Round, clinical governance meetings and departmental induction meetings.
Although we developed a paper pro forma for the clerking of patients who present with AUGIB, we also elected to develop a novel method of implementing guidance by designing a guideline that was embedded within the clinical workflow of the electronic patient’s record. We collaborated with the British Medical Journal’s (BMJ) ‘Evidence Centre’ and ‘Cerner Millennium Electronic Patient Record System’ to create an electronic ‘action set’ for the assessment and emergency management of adult patients presenting with AUGIB. The tool was embedded in the clinical workflow and went live in September 2010.
The action set is a tool that is activated every time a patient with suspected or confirmed AUGIB is seen by a nurse or a clinician. It can be readily accessed via the hospital’s electronic care record system and it captures all activity from the moment the patient is seen and the order set is activated. Activating the action set is as simple as requesting any other investigation for the patient, such as a full blood count (Figure 1). Multiple orders can be placed simultaneously.

Activation of the action set is as simple as requesting to do a full blood count for the patient (red circle and arrow).
Every step of the patient’s journey is time-stamped and the data is collected electronically. Our local guidelines are incorporated in the interface. The evidence on which the guidelines are based is retrieved by clicking on the ‘BMJ Evidence’ links (Figures 2 and 3). In order to increase the clinical utility of the action set we have also included useful bleep numbers and phone extensions including the duty gastroenterology registrar and the endoscopy department. With the action set, one is able to request blood tests for up to 5 days, as well as thoracic/abdominal radiographs or other imaging studies such abdominal computed tomography (CT). The most important and necessary investigations as per guideline recommendations are already selected for the user (Figure 4). E-prescribing and the nurse ordering functionalities are also available.

By clicking on the evidence icon next to the instruction (red arrow) you will be automatically be directed to local antibiotic guidance or the BMJ’s Evidence Centre where the evidence summary behind the suggested instruction/recommendation will be displayed (Figure 3). Local protocols are also displayed (blue arrow).
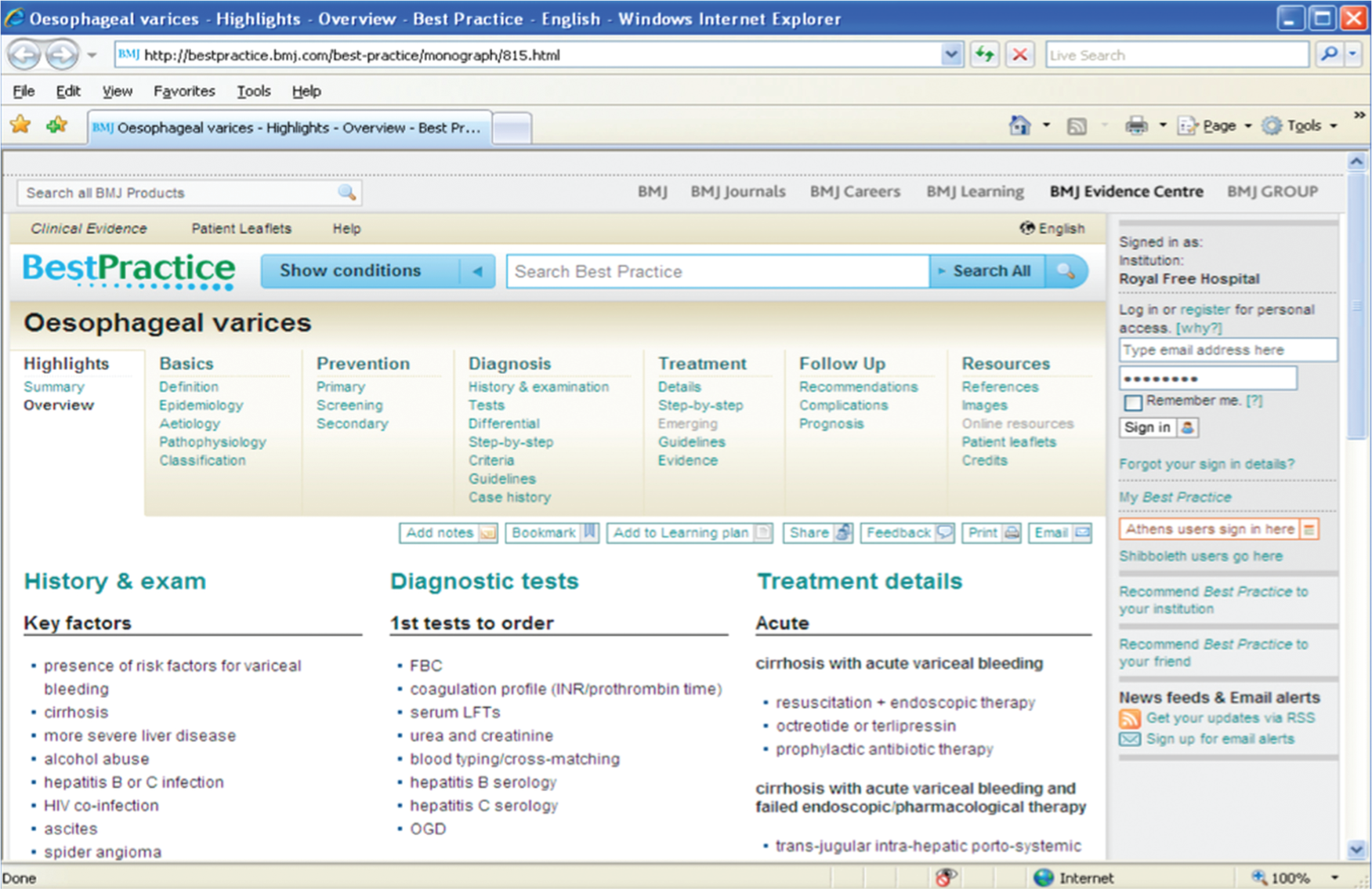
The evidence-based summary and additional information extracted from BMJ Evidence Centre.

The most important investigations needed to manage AUGIB according to the guidelines are already selected and can be requested automatically (red circle). Additional investigations can be added by ticking the box adjacently (blue arrows). The collective order set can be submitted by entering a contact/bleep number and then electronically signing off the request.
After implementation of the action set we re-audited between September 2010 and February 2011 using data that was automatically streamed and collected on a weekly basis via the action set interface. During this period, 157 patients presented to our hospital with AUGIB and were endoscoped having an indication of melaena, haematemesis, or both. The action set was activated 13 times during this period for 13 different patients. One of these activations was invalid, as the patient did not have any history of AUGIB. The results of the remaining 12 patients were analysed in the second loop of this audit. These patient’s notes were retrieved and further manual analysis was performed.
Statistical analysis was performed using IBM SPSS v20.0 software (SPSS, Inc., Chicago, IL, USA). Tests of independence to assess whether paired observations of two or more variables are independent of each other included Pearson’s chi-squared test and Fisher’s exact test.
Results
The first loop of the audit included 27 patients, 5 of which presented with variceal bleeding. The second loop included 12 patients, 1 of which had haematemesis due to bleeding varices. The mean age of presentation was 66 years and our random selection of patients included 23 (59%) male and 16 (41%) female patients (Table 1). The patients were either admitted through emergency services or bled while they were inpatients in our hospital.
Showing basic patient demographics, calculated risk scores (Rockall scores in this study) and duration of hospital stay.

(d), discharged when score was zero; F, female; M, males; N/A, not available or not applicable; OGD, oesophogastroduodenoscopy; PPI, proton-pump inhibitor.
Patient also treated for acute coronary syndrome.
Patient was an inpatient at the time of data processing.
Admitted on PPI.
Before the introduction of the action set we had observed extremely poor rates of risk assessment and stratification of patients at the point of first presentation. None of the physicians in acute medicine or A&E used a risk stratifying system (in this case the pre-Rockall score) on first encounter during the initial assessment and none of the gastroenterology/hepatology physicians calculated pre-Rockall score on first encounter when asked to review patients. Alternative systems such as the Addenbrooke or Blatchford scoring systems were not used either. Following endoscopic procedures, full Rockall scores were not recorded by any of the reviewing physicians (including the endoscopists) for any of these patients. Following implementation of the action set, documentation of pre-OGD Rockall scores increased from 3.7% (1 patient) to 50% (6 patients) (p ≤ 0.0001). The differences in the calculation and documentation of post-OGD full Rockall scores were also significant between the two audit loops (p = 0.007).
We have calculated the Rockall scores for all patients and then looked at the duration of their hospital admissions. The results for patients with pre-endoscopy Rockall score of zero (0) were analysed. Only one patient (20%) was considered for nonadmission or early discharge with outpatient follow up as per guideline recommendations. The median duration of hospital stay for the nondischarged patients was 3.5 days (range 1–5). Following implementation of the action set, discharges of patients whose Rockall score was zero significantly increased from 20% to 100% (five patients, p = 0.048).
The results for patients with post-endoscopy (full) Rockall scores 0 to 2 were also analysed and 40% (two patients) had been considered for early discharge and outpatient follow up. The median duration of hospital stay for the nondischarged patients was 5 days (range 5–9). Following implementation of the action set, discharges of patients whose full Rockall score of ≤2 increased from 40% to 100% (two patients) (p = 0.43). These findings are summarized in Table 1.
Before the introduction of the action set, PPIs were administered to our patients in various formulations and doses as part of the management of AUGIB. Prior to endoscopy, 19 patients (79%) received a PPI. Although this was better when compared with the results of the national audit [Hearnshaw et al. 2010], it was much higher than the standard set for this audit. As a result, PPIs were appropriately withheld in only five patients (21%), hence, adhering to the audit standard. No standard prescription regimen was followed by the attending physicians. Pre-endoscopy PPI data were not available for one patient (3.7%). Following the introduction of the action set, prescription of PPIs became more standardized with less formulaic variations. We found that only four patients (33%) inappropriately received pre-endoscopy PPIs and this decrease was statistically significant (p = 0.02) between the two groups.
Before the introduction of the action set, 24 patients (89%) received PPIs after their endoscopy. Only six patients (23%) had stigmata of recent haemorrhage and should have received a PPI in the first place. The remaining patients received a PPI against the guideline used as the standard for this audit. Various prescription regimens were once again used. PPIs were withheld in two patients (7.3%), who did not have stigmata of recent haemorrhage at endoscopy. Postendoscopy PPI prescription data were not available for one patient (3.7%). After the introduction of the action set, four patients (50%) received a PPI following their endoscopy and three of the patients who had an OGD (37.5%) demonstrated stigmata of recent haemorrhage. All patients with true clinical indication received a formulation of a PPI and five patients (62.5%) inappropriately received a PPI. The differences between the two groups were statistically significant (p = 0.01). These findings are summarized in Table 2.
Summary of PPI formulations prescribed to patients presenting with AUGIB.

AUGIB, acute upper gastrointestinal bleeding; IV, intravenous; OGD, oesophogastroduodenoscopy; PPI, proton-pump inhibitors; PO, per os (oral).
PPI infusions were not administered for any patients before endoscopy neither prior to nor after the introduction of the action set. Before the action set, all of the patients who had received a PPI infusion after endoscopy did so against the guidelines and endoscopic findings. Following introduction of the action set, one patient received a PPI infusion and this was appropriately prescribed. Before the action set was implemented, two PPI infusions were prescribed, both in patients who did not have stigmata of recent haemorrhage at endoscopy. The differences were not statistically significant (p = 0.4).
Discussion
As we are moving towards an era of electronic patient records and centralized digital patient management systems, novel ways have to be designed to improve and document patient care. Research and clinical guidelines are ever evolving providing a broader understanding of acute clinical conditions and up-to-date guidance on their management. We believe that action sets linked to clinical evidence databases could be an efficient way to give physicians the tools in order to provide safer patient care and promote clinical governance.
Although this audit was performed and completed before the publication of the new NICE guidelines in June 2012, the main core themes remain the same. Through this study, we were able to understand how clinicians in our Trust manage AUGIB and identify whether their actions were based on clinical evidence. By formulating local protocols, as well as by adopting the national standards, we aimed to create standardization of care for all patients presenting with AUGIB, provide a useful evidence-based resource for our doctors managing such patients and align our Trust to the ‘Scope for improvement’ report’s recommendations.
One of the limitations of this study was the number of times the action set had been activated when a patient with an AUGIB was encountered in the second audit loop. This highlights a more fundamental issue; how incredibly difficult it is to alter clinical behaviour and practice. What we are proposing is an extremely easy-to-use tool which is as simple as requesting an additional blood test. Even though the patient sample size for this audit was small, we were still able to observe statistically significant improvements in guideline adherence, initial assessment and risk scoring at presentation, identification of potential safe discharges and correct administration of PPIs before and after endoscopy. Through electronic data streaming, this tool has provided us with auditable data in order to benchmark our performance to the national standards and helped us bridge some of the deficits and meet most of the requirements underlined in the ‘Scope for improvement’ report. It strongly encourages clinical leadership and promotes clinical governance. We continue to strive to promote the action set among doctors and nurses in our hospital and we continue to audit the data streamed automatically on a weekly basis. We believe that clinical behaviour and practice can change through repeated positive reinforcement and we believe that action sets can be useful for the correct management of AUGIB.
This audit was design to assess on a local level our competency in managing correctly patients who present with AUGIB. Through our implementation strategies we wanted to promote correct guideline adherence and higher-quality care to our patients. One of the methodological limitations is that we did not design this study to have specific amount of patients in each cycle and we did not aim to achieve any particular ratio of patients in each loop, e.g. 2:1. One of the reasons for the small number in our audit is the fact that we also had to educate a huge number of doctors and present our findings in various meetings before we could go ‘live’ and start re-auditing. This invariably takes time and junior doctor rotations provided an additional challenge. Generally, our aim was to audit as many patients as possible for whom the action set was appropriately activated. Even after going ‘live’ however, this did not yield as many patients as we would have hoped. Precisely, this fact highlighted the issue of how incredibly difficult it is to alter established clinical practice.
Creating the action set was our way to deliver the best evidence-based and updated care to our patients presenting with AUGIB. The use of action or order sets has been employed in several settings with good results. They seem to provide healthcare systems with an excellent opportunity to improve patient safety using evidence-based healthcare, and may have wide applicability as a clinical decision support tool [O’Connor et al. 2009].
The action set is easily accessible as it is embedded in the clinical workflow and through it our doctors and nurses can request an order set automatically by just entering their bleep number. It provides a ‘working’ guideline and it has direct links to BMJ’s ‘Best Practice’ and ‘BMJ Evidence’ (Figures 2 and 3). This helps not only to govern clinical decision-making but also provides prescribing support for medication used along the management pathway. It can also link to the user’s BMJ learning plan and education record. One of the best features of the tool is its ability to capture the data and, hence, facilitate the auditing process. It monitors all activity throughout the patient’s journey and time stamps each step of the patient’s management. These steps can be traced back and possible deficits in the guideline implementation can be recognized. Through this system, the process of auditing becomes automatic, simpler and quicker. A flow chart of the management protocol used once the action set is activated is shown in Figure 5.

Flow chart showing the management protocol used once the action set is activated. NSAIDs, nonsteroidal anti-inflammatory drugs; PPI, proton-pump inhibitors.
Various scoring systems have been studied in the literature and have been evaluated for parameters such as rebleeding, mortality, need for blood transfusion, need for surgical intervention and hospital stay. Validated systems appearing in the literature include the Addenbrooke’s [Cameron et al. 2002], Blatchford [Chen et al. 2007; Gralnek and Dulai, 2004; Masaoka et al. 2007] and Rockall [Camellini et al. 2004; Church et al. 2006; Enns et al. 2006; Phang et al. 2000; Rockall et al. 1995; Rotondano et al. 2011; Sanders et al. 2002] scoring systems. The standard for this audit was the SIGN guidelines; hence, we used the Rockall score assessment before and after endoscopy. The NICE guidelines recommend the use of the Blatchford score at first assessment, and the full Rockall score after endoscopy. Regardless of which scoring system is used, high-risk patients should be promptly identified and offered emergency treatment. Furthermore, identifying low-risk patients may possibly facilitate earlier discharge and create more hospital beds. Through appropriate use of PPIs, rebleeding rates [Lau et al. 2000], mortality rates [Leontiadis et al. 2004] and costs can be reduced if prescribed appropriately and when indicated as per guideline recommendations [Ballard et al. 2008; Lau et al. 2007].
Improved outcomes when using order sets have been previously reported in several cases such as the appropriate administration of antibiotics in surgical patients [Braxton et al. 2010; Lau et al. 2000], improving sepsis outcomes [Thiel et al. 2009; Winterbottom et al. 2011], reducing mortality in patients with pneumonia [Fleming et al. 2009], improving outcomes in heart failure patients [Ballard et al. 2010], optimizing end-of-life care [Walker et al. 2011] and managing patients with alcohol withdrawal [Riddle et al. 2010].
We have identified the potential to expand the use of action sets for other emergencies in our Trust. This will promote the use of updated and evidence-based guidelines in a wider spectrum of acute presentations, such as asthma and chronic obstructive pulmonary disease (COPD) exacerbations, head injuries and acute coronary syndromes. Action sets may be limited by failures in electronic systems and equipment. These tools are by in no way utilities that eliminate clinical judgement and didactic lectureship. During life-threatening emergencies and staff shortages, there may not be enough time to activate action sets appropriately and clinicians will need to act on already-established knowledge on clinical guidelines and standards.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
Dr O’Beirne has received consultancy fees from Ferring pharmaceuticals. The other authors have no conflicts of interest to declare.
