Abstract
Abdominal pain is a common symptom in patients with inflammatory bowel disease (IBD) and has a profound negative impact on patients’ lives. There are growing data suggesting that pain is variably related to the degree of active inflammation. Given the multifactorial etiologies underlying the pain, the treatment of abdominal pain in the IBD population is best accomplished by individualized plans. This review covers four clinically relevant categories of abdominal pain in patients with IBD, namely, inflammation, surgical complications, bacterial overgrowth, and neurobiological processes and how pain management can be addressed in each of these cases. The role of genetic factors, psychological factors, and psychosocial stress in pain perception and treatment will also be addressed. Lastly, psychosocial, pharmacological, and procedural pain management techniques will be discussed. An extensive review of the existing literature reveals a paucity of data regarding pain management specific to IBD. In addition, there is growing consensus suggesting a spectrum between IBD and irritable bowel syndrome (IBS) symptoms. Thus, this review for adult and pediatric clinicians also incorporates the literature for the treatment of functional abdominal pain and the clinical consensus from IBD and IBS experts on pharmacological, behavioral, and procedural methods to treat abdominal pain in this population.
Keywords
Introduction
Abdominal pain is a common and frustrating symptom in patients with inflammatory bowel disease (IBD), with multifaceted pathophysiology and associated emotional suffering, disability, and high medical costs. This paper reviews the predictors and consequences of abdominal pain in adult and pediatric patients with IBD as well as pharmacological and nonpharmacological treatment options. The data on treatment options for abdominal pain in patients with IBD are limited. Since there is overlap between clinical symptoms of IBD and irritable bowel syndrome (IBS) [Long and Drossman, 2010] some management strategies have been extrapolated from the functional gastrointestinal (GI) disorder literature, IBS specifically [Grover and Drossman, 2009].
Types of abdominal pain
Abdominal pain can be categorized as visceral or somatic. Visceral pain arises internally and is characteristically diffuse, intermittent, and difficult to localize. It is caused by stretching of the viscera and deep inflammation or obstruction. Somatic pain is musculoskeletal, and is generally well localized. Both types of pain involve nociceptive fibers, can have neurobiological, psychological, and psychosocial underpinnings, and can occur in IBD.
Etiological factors underlying abdominal pain in inflammatory bowel disease
Many overlapping factors influence the perception of pain in IBD. These include inflammation, obstruction, psychological, psychosocial, neurobiological, and genetic factors [Grover and Drossman, 2009]. Although pain in IBD has traditionally been attributed to severity of inflammation, 20–50% of patients have disabling abdominal pain while they are in remission from a clinical and/or endoscopic standpoint [Edwards et al. 2001; Farrokhyar et al. 2006; Minderhoud et al. 2004; Siegel and MacDermott, 2009]. Reports of poor correlations between reported abdominal pain intensity and IBD activity indices [Schirbel et al. 2010] reinforce the complex nature of pain in IBD. All of these findings add to the growing recognition of a spectrum of symptoms between IBD (inflammation) and IBS.
Inflammation
During disease flares, 50–70% of patients with IBD will have abdominal pain [Wagtmans et al. 1998]. Gut inflammation can lead to parietal peritoneum inflammation. Subsequent transmission via somatic nociceptive afferents leads to the sensation of pain. Furthermore, subtle inflammation (and consequent abdominal pain) may exist even when standard disease activity indices suggest that disease is in remission [Keohane et al. 2010].
Development of chronic visceral pain is thought to result from changes that begin with hypersensitivity of the primary sensory neurons innervating the GI tract, which is subsequently maintained or magnified by changes in the central nervous system (CNS). Sensitization of the primary afferents has been shown to occur via changes in the intrinsic properties of sensory neurons that regulate excitability and in gene expression for nociceptive-specific membrane proteins [Malin et al. 2011]. Changes in primary afferent sensitivity not only contribute to the development of persistent pain, but also contribute to cytokine changes via neurogenic inflammation. Primary sensory neurons, especially peptidergic sensory neurons that innervate the gut, release a number of molecules that induce vasodilation, edema, and swelling [Hutter et al. 2005; Liddle and Nathan, 2004; Vera-Portocarrero and Westlund, 2005]. This function of sensory neurons, which has only recently been appreciated, significantly contributes to visceral inflammation. Intestinal inflammation can also affect motility [Garret et al. 1967; Snape et al. 1991; Vermillion et al. 1993], which can secondarily induce visceral hypersensitivity both through peripheral and central influences.
Lastly, there are some data to suggest some of the standard medications used to treat inflammation in IBD and its consequences (anemia) – aminosalicylates [Lim and Hanauer, 2010] and enteral iron formulations [Liguori, 1993] – can cause abdominal pain.
Strictures/adhesions
Patients with IBD experiencing persistent pain in the absence of disease activity may have underlying fibrotic strictures or adhesions secondary to IBD itself or following surgical interventions. The intestinal distension caused by these complications generates the sensation of abdominal pain through stretch receptor activation.
Small intestinal bacterial overgrowth
In patients with Crohn’s disease (CD), dysmotility secondary to inflammation and postinflammatory changes [Annese et al. 1997], stasis secondary to partial obstruction, fistulae, or blind loops secondary to surgery are all possible etiologies of small intestinal bacterial overgrowth (SIBO). The resulting distension and potential intestinal irritation from bacteria can lead to pain via the previously identified mechanisms. Unfortunately, screening for SIBO in patients with CD is limited by the lack of accurate diagnostic modalities; in particular, H2 breath tests and small bowel aspirate assays have low specificity [Camilleri, 2011].
Neurobiological
The perception of pain involves the integration of neurobiological pathways within the enteric, peripheral, and central nervous systems. These neurobiological pathways are clearly affected by inflammation. Andersson and colleagues demonstrated a direct correlation between rectal hypersensitivity and inflammation [Andersson et al. 2003]. Adult and animal studies have suggested a role for inflammatory effects on enteric neurons in pain generation in IBD [Beyak and Vanner, 2005; Geboes and Collins, 1998]. This role has been substantiated through intestinal tissue analysis of patients with IBD, which demonstrated enteric nervous system abnormalities including glial cell hypertrophy and hyperplasia [Lakhan and Kirchgessner, 2010]. These changes are more marked in CD than ulcerative colitis (UC) [Steinhoff et al. 1988].
Regarding patients with persistent pain despite normal disease activity, Jacobson and colleagues have shown that microscopic gut inflammation can stimulate visceral neuronal signaling, thereby potentially leading to visceral hypersensitivity [Jacobson et al. 1995]. For example, the cytokine, interleukin-1β (IL-1β), which is released during states of inflammation (i.e. uncontrolled disease activity and flares) in IBD, can indirectly induce glial cells thereby increasing prostaglandin E2 release. Prostaglandin E2 then increases the sensitivity of myenteric neurons to bradykinin, which is involved in IBD [Kraneveld et al. 2008]. Mast cells may play a role in stimulating visceral neuronal signaling as well, even when IBD is quiescent. For example, van Hoboken and colleagues conducted a study showing sigmoid biopsy mast cell presence, density, and close proximity to nerve endings may play a role in mediating visceral hypersensitivity in adult patients with IBD [van Hoboken et al. 2011]. In their small study, roughly equivalent amounts of patients with UC in remission (Mayo score of zero) and controls (healthy subjects undergoing routine colonoscopy) were compared in terms of degree of rectal hypersensitivity, amount of mucosal mast cells (per 100 crypts), proximity of mast cells to nerve endings, and degree of mast cell degranulation. All of these aforementioned parameters were significantly increased in the patients with UC who were in remission compared with the control population. Additionally, there was a significant (albeit weak) correlation between mast cell density and pain perception in patients with UC who were in remission. Thus, post-inflammatory changes of the enteric nervous system can lead to visceral pain, though the mediators are yet unknown [Lakhan and Kirchgessner, 2010].
Autonomic dysregulation has been described in IBD and IBS [Ananthakrishnan et al. 2010; Tougas, 2000]. This dysregulation may be due to chronic inflammation through nitric oxide induced nerve damage [Kraneveld et al. 2008]. Furthermore, as research has indicated that patients with IBS may have alterations in brain regulatory centers (e.g. the anterior cingulate cortex) [Tillisch et al. 2011], there also may be CNS changes in patients with IBD and persistent pain. Structural and functional brain imaging studies are needed to understand how inflammation affects central processing of pain in the IBD population.
Abnormalities in neuroendocrine signaling involving the hypothalamic–pituitary–adrenal (HPA) axis have been described in IBD and IBS. In IBD, a stress response mediated by the HPA axis has been postulated to mediate the development of inflammation [Reber, 2012]. While in IBS, HPA axis dysfunction and exaggerated stimulation responses have been demonstrated [Myers and Greenwood-Van Meerveld, 2009].
Psychological
Although they are overlapping, as detailed in Figure 1, the directionality between neurobiological (central and peripheral) and psychological (e.g. depression and anxiety) underpinnings of abdominal pain within IBD and the functional pain literature is currently unclear [Elsenbruch, 2011]. Psychological factors, in addition to psychosocial and genetic factors, may influence differences in sensory processing between those with and without perceived abdominal pain. There are significant rates of depression in IBD, ranging from 16% to 44% [Fuller-Thomson and Sulman, 2006; Graff et al. 2009; Szigethy et al. 2007] in adult and pediatric studies combined. In one of the largest studies examining the relationship between depression and IBD, Fuller-Thomson and Sulman noted a depression rate of 16% and a significant association between depression and report of pain within their IBD cohort [Fuller-Thomson and Sulman, 2006]. In addition, mood disorders are associated with persistent symptoms, including pain, in patients with IBD who are in remission. Factors that have been shown to be associated with increased pain perception include increased worrying, limited coping ability, and negative cognitive distortions [Farrokhyar et al. 2006]. While data demonstrate that psychological conditions are common in patients with IBD and comorbid pain, the exact mechanisms leading to the perception of pain in these patients still need to be identified.
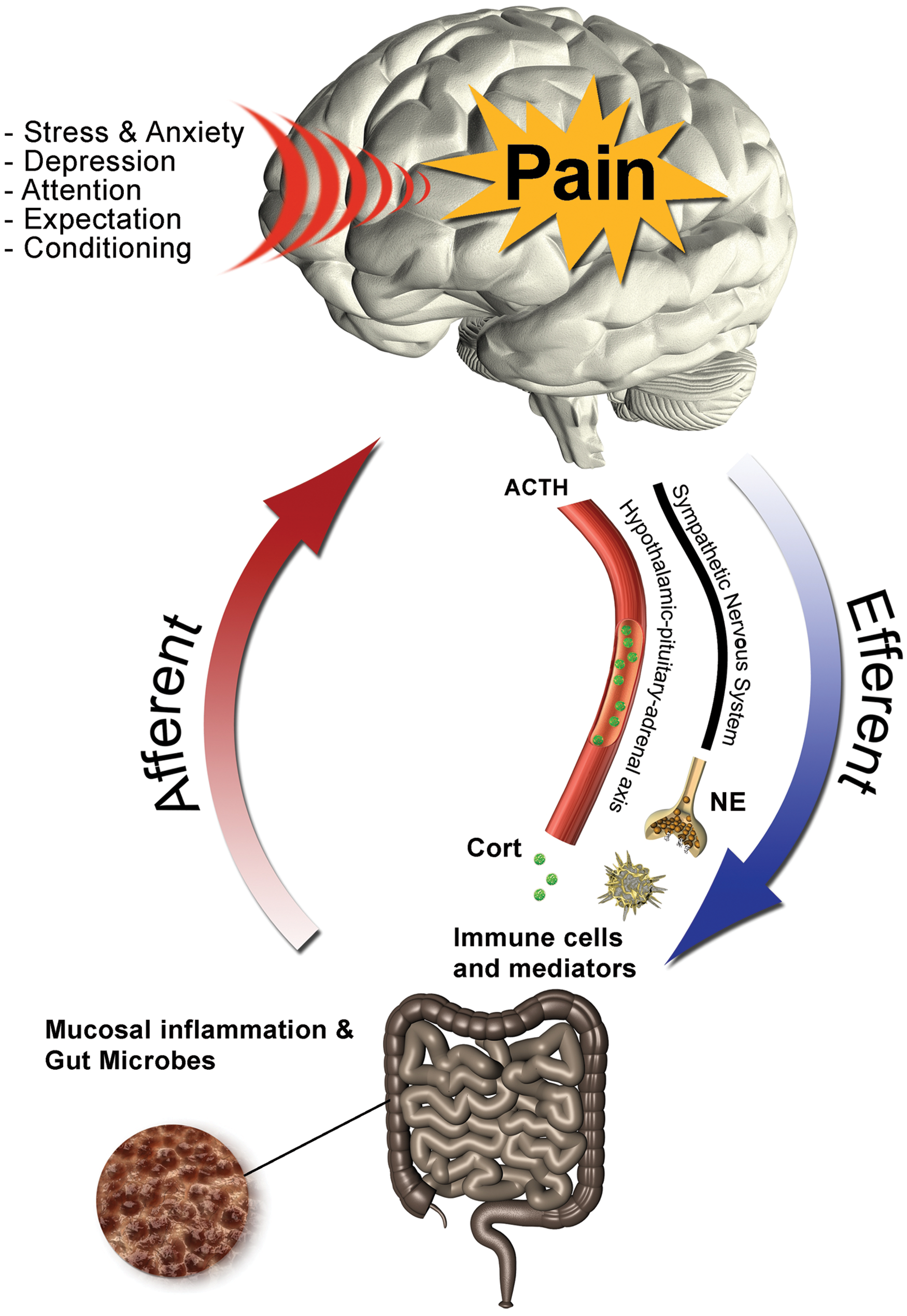
The multidimensional bidirectional relationship between luminal, neurobiological, psychological, and psychosocial factors and abdominal pain in inflammatory bowel disease (IBD). ACTH, adrenocorticotropic hormone; NE, norepinephrine. (Reproduced from Eisenbruch [2011] with permission.)
Research has shown high rates of anxiety, depression, and other comorbid psychiatric conditions in patients with functional abdominal pain [Hertig et al. 2007]. These comorbidities may play a role in visceral hypersensitivity in IBD, as demonstrated in Faure and Giguère’s small study of pediatric patients with CD and coexisting functional GI disorders (including functional abdominal pain, according to the Rome II criteria) [Faure and Giguère, 2008]. These patients had increased rectal hypersensitivity demonstrated via low rectal sensory thresholds for pain compared to healthy controls. Seven out of the eight patients with CD in their study had anxiety; which was thus postulated to be a mediator of visceral hypersensitivity.
Approximately 60% of patients with IBS seeking medical care have an anxiety disorder diagnosis [Drossman et al. 1999; Whitehead et al. 2002]. In a study comparing differences in the report and evaluation of pain, Elsenbruch and colleagues demonstrated that adult patients with IBS and comorbid anxiety and depression reported greater perceived abdominal pain yet did not experience changes in rectal sensory thresholds [Elsenbruch et al. 2010]. Thus, it is possible that patients with IBD and comorbid psychological disorders have a heightened response to visceral sensations, leading to an increase in pain perception. This postulate is reinforced by Mussell and colleagues who demonstrated that a depressive coping style in particular was more predictive than medical variables for disease-related concerns (including reports of abdominal pain) [Mussell et al. 2004]. In addition, research has shown catastrophic thinking negatively influences the perception of pain [Vervoort et al. 2006].
Psychosocial
In adults and children with IBD, psychosocial stressors on patients and their families are known to exacerbate pain. Life stressors have a negative impact on coping and may influence pain perception. Furthermore, there are data to suggest that early life trauma may be associated with the development of visceral hypersensitivity [Drossman, 2011]. Adding to the complexity of these interactions, research in adults has shown that chronic stress may lead to increased disease reactivation risk, which may in turn lead to greater pain [Levenstein et al. 2000]. In a pediatric study, Engström demonstrated that parental distress can lead to negative ways of coping with pain and poor quality of life for patients, which may lead to increased symptom report and escalation of medical care on the part of the clinician [Engström, 1991].
Genetic
Genetic components may account for approximately 50% of the variance in pain processing as measured by quantitative sensory testing [Buscher et al. 2006; Diatchenko et al. 2005; Giesecke et al. 2004]. There are a growing number of potential genes implicated in the etiopathogenesis and severity of IBD (e.g. NOD2 and CARD15) [Cuffari, 2010] and IBS [polymorphisms in the serotonin transporter gene (5-HTTLPR), certain G-protein coupled receptors, cholecystokinin-receptor and tumor necrosis factor (TNF)] [Hotoleanu et al. 2008]. In addition, transient receptor potential vanilloid type-1 (TRPV1) has been shown to play an important role in visceral hypersensitivity. Akbar and colleagues showed increased TRPV1 nerve fibers in patients with quiescent IBD and IBS-like symptoms including pain [Akbar et al. 2010]. To date, functional genetic polymorphisms associated with pain processing have not been systematically studied in IBD.
Consequences of abdominal pain in inflammatory bowel disease
The consequences of abdominal pain in IBD can be devastating. They include, but are not limited to, psychological morbidity, stress, impaired quality of life, interrupted psychosocial functioning [Lix et al. 2008], and potentially high costs for utilization of medical resources for workup and management. Effective and individualized management of abdominal pain in IBD is thus critically important.
Abdominal pain management
Given the complex interplay between multiple factors leading to abdominal pain and its significant consequences, effective management is critical. In addition, with improved medical treatments for IBD, there is growing recognition of functional abdominal pain (as in IBS) within the IBD population. Patients with persistent pain despite little to no inflammation have visceral hypersensitivity from postinflammatory changes and central dysregulation [Long and Drossman, 2010]. When IBS is defined by pain and diarrhea out of proportion to the observed disease activity [Grover and Drossman, 2009], IBS symptoms occur two to three times more often in patients with IBD than the general population. Simren and colleagues showed that roughly 60% of patients with CD and 30% of patients with UC report IBS-like symptoms. The clinician should recognize and appreciate the potential overlap of symptoms between IBD and IBS. Since patients will have variable contributions and overlap of the above factors to their pain, we stress that pain management should be customized. This section reviews pharmacological, behavioral, and procedural methods to alleviate abdominal pain in IBD.
Pharmacological treatment
Inflammation
Standard inflammatory bowel disease specific anti-inflammatory agents
Since one of the cardinal features of intestinal inflammation is abdominal pain, inducing remission using standard of care IBD treatments (e.g. aminosalicylates, antibiotics, corticosteroids, enteral therapy, immunomodulators, and biological response modifiers) can alleviate inflammation-related pain in most patients.
Antispasmodics
Antispasmodics have some utility in providing a bridge of pain relief as remission is induced and aiding with pain if standard medical therapies are unable to control inflammation. As pain from inflammation or partial obstruction usually manifests with intestinal spasms, anticholinergic antispasmodic medications are frequently employed as analgesic agents. Ford and colleagues published the most recent meta-analysis on utility of antispasmodics for IBS [Ford et al. 2008]. Overall antispasmodics were found to be more efficacious than placebo for functional abdominal pain. Of the medications studied by their group, hyoscine (hyoscyamine) was found to be the most efficacious and is the only one available in US markets. Practically speaking, the antispasmodics used most frequently are hyoscyamine and dicyclomine.
Unfortunately, though antispasmodics seem to be a reasonable choice for pain management associated with inflammation or partial obstruction, there is no definitive evidence supporting their usage in IBD [Makharia, 2011]. Furthermore, one must be wary of potential side effects, including fulminant obstruction in an already partially obstructed gut or worsening dysmotility in a gut that is already prone to motility problems secondary to inflammation and its sequelae.
Nonsteroidal anti-inflammatory drugs
Nonsteroidal anti-inflammatory drugs (NSAIDs) are commonly used to treat abdominal pain and musculoskeletal symptoms associated with IBD [Kefelakes et al. 2009]. However, multiple studies have demonstrated that NSAIDs may increase the risk for IBD relapse [Kefelakes et al. 2009; Cryer, 2005; Felder et al. 2000 ], flares [Evans et al. 1997], and overall increase in disease activity [Bonner et al. 2000].
Cyclooxygenase-2 inhibitors
After NSAIDs were found to be associated with IBD exacerbations, cyclooxygenase-2 (COX-2) inhibitors were proposed as a treatment for pain by way of decreasing inflammation in the gut. Mechanistically, COX-2 is much more responsive to inflammatory stimuli such as lipopolysaccharide and other proinflammatory cytokines (including IL-1 and TNF-α) [Eckman et al. 1997] than COX-1 [Funk, 2001]. In IBD, COX-2 mRNA is expressed in inflamed intestinal tissue by epithelial cells and lamina propria mononuclear cells and in the neural cells of the myenteric plexus [Roberts et al. 2001]. The expression of COX-2 mRNA does not occur in the normal gut or neural cells of patients without IBD. As COX-2 mRNA expression is specific to IBD, COX-2 inhibitors have been used to treat abdominal pain in this population.
Unfortunately, COX-2 inhibitors have not been shown to be beneficial in terms of safety, toxicity, or even anti-inflammatory effect. Case reports in the early 2000s noted associations between COX-2 inhibitors and IBD reactivation [Bonner, 2001; Gornet et al. 2002]. Larger studies thereafter demonstrated rates of disease exacerbation ranging from 7% to 19% following treatment with COX-2 inhibitors [Mahadevan et al. 2002; Reinisch et al. 2001; Biancone et al. 2003]. Specifically, those studies examined the use of celecoxib and rofecoxib. Additional research studies using celecoxib and etoricoxib did not demonstrate acute anti-inflammatory effects by those COX-2 inhibitors [Sandborn et al. 2004; El Miedany et al. 2006]. Regardless of the criticisms of COX-2 inhibitors, it is important to note there are no trials specifically investigating the utility of these medications for abdominal pain associated with IBD.
Opiates
Traditionally, narcotics have been used acutely after surgical resection of the intestinal tract in IBD and to treat pain due to inflammation in IBD. Approximately 5–13% of patients with IBD are on chronic narcotics in the outpatient setting [Edwards et al. 2001; Cross et al. 2005]. Risk factors for outpatient narcotic use in IBD include psychiatric comorbidities such as anxiety and depression, history of abuse, female gender, and a high degree of clinical symptoms [Edwards et al. 2001; Cross et al. 2005; Hanson et al. 2009]. Although there are limited data regarding rate of narcotic use in hospitalized patients, Lian and colleagues showed that 20% of patients with UC required narcotics prior to colectomy [Lian et al. 2010]. In another study, it was demonstrated that 70% of hospitalized patients with IBD were on narcotics, and all of those patients had overlapping symptoms of IBS and IBD. Risk factors for inpatient narcotic use in IBD include a diagnosis of CD, substance abuse, psychiatric factors, and the presence of IBS symptoms [Long et al. 2012].
Unfortunately, narcotic use, particularly chronic use, has multiple potentially detrimental consequences in IBD. Problems with narcotic use in IBD include nausea, vomiting, constipation, intestinal pseudo-obstruction, reduced GI motility, narcotic bowel syndrome [Grunkemeier et al. 2007], postoperative serious infection and mortality in CD [Lichtenstein et al. 2006], fulminant colitis [Gan and Beck, 2003], and toxic megacolon in UC [Hartong et al. 1977].
Narcotic bowel syndrome and treatment
Narcotic bowel syndrome is an under-recognized condition characterized by chronic or recurrent abdominal pain in patients treated acutely or chronically with narcotics. The abdominal pain itself is progressive, paradoxically worsens or is only partially responsive at best to narcotics at current or escalating doses, and worsens when the narcotic doses wane. Pathophysiological mechanisms include bimodal G-protein inhibitory (GI/GO) and excitatory (GS) receptors activated by narcotics, modulating descending antinociceptive pathways (involving cholecystokinin and the endogenous opioid dynorphin), and with chronic use, increased spinal cord glial activation [Grunkemeier et al. 2007]. Narcotic bowel syndrome should be suspected in a patient with worsening abdominal pain despite chronic or increasing narcotic doses. Grunkemeier and colleagues succinctly detail treatment for this condition [Grunkemeier et al. 2007]. Specifically, their treatment utilizes a biopsychosocial model of pain with narcotic taper along with treatment of narcotic side effects (e.g. constipation) and withdrawal symptoms (e.g. anxiety) while initiating a tricyclic antidepressant (TCA) or serotonin-norepinephrine reuptake inhibitor (SNRI).
Strictures/adhesions
The pathogenesis of pain arising from intestinal strictures has been discussed previously. There are no published studies of pain management for this complication of IBD. Clinical consensus suggests there is utility in dietary modification (e.g. low residue, low fiber, or liquid diet), and antispasmodics in moderation. The dietary modification physically works by being less resistant to passage through the area of narrowing, leading to less intestinal distension, and therefore, a reduction in pain. Antispasmodics lessen the magnitude of the intestinal contractions as particulate material passes through the narrowed intestine.
Small intestinal bacterial overgrowth
There are limitations of current diagnostic modalities of SIBO. If suspected, an empiric trial with a relatively nonabsorbable antibiotic (e.g. metronidazole, rifaximin, neomycin) is the ideal diagnostic and treatment modality [Quigley and Quera, 2006; Lauritano et al. 2009].
Neurobiological
Since the research demonstrating the neurobiological changes associated with IBD and abdominal pain is relatively new, data supporting specific treatments for pain due to underlying neurobiological factors are limited. To facilitate an organized discussion for this review, psychotropic treatments for abdominal pain are classified under the ‘neurobiological’ category while behavioral treatments are classified under ‘psychological’. We acknowledge this distinction is not entirely correct, as behavioral treatments may affect visceral neuronal signaling and psychotropic medications may affect psychological status.
Psychotropics
There are few trials demonstrating the utility of psychotropic medications on visceral pain. Unfortunately, many of those that have been conducted do not examine abdominal pain as a primary outcome but rather focus on the effects of psychotropic medication (e.g. antidepressants) on relapse rates and inflammation [Mikocka-Walus et al. 2006, 2009]. Since visceral hypersensitivity exists in IBD and IBS, many clinicians rely on the current IBS literature for management of visceral pain in IBD [Grover and Drossman, 2011]. The neurochemicals targeted by these drugs such as serotonin and norepinephrine are involved in visceral motility and sensation. However, it is unclear whether they directly impact nociception or their beneficial effects are mediated by decreasing anxiety and depression often comorbid with chronic abdominal pain. TCAs, selective serotonin-reuptake inhibitors (SSRIs), and SNRIs have been studied for their effect on visceral pain within the functional GI disorders. Comparison studies between these two classes of medications are thus far inconclusive [Rahimi et al. 2009].
Tricyclic antidepressants
A meta-analysis examining the use of low-dose TCAs in adult patients with IBS suggested they have some utility in alleviating abdominal pain [Rahimi et al. 2009]. However, the authors noted they did not include a well designed trial by Drossman and colleagues because it examined all types of functional GI disorders (not limited to IBS) [Drossman et al. 2003]. In their randomized placebo-controlled trial, Drossman and colleagues evaluated the utility of desipramine for treating functional abdominal pain, moderate–severe IBS, and functional constipation. They found significant benefit using desipramine despite a roughly 20% non-adherence rate. This finding may suggest that inclusion of nonadherent subjects in previous studies may have resulted in an underestimation of the ability of TCAs to reduce functional abdominal pain symptoms.
The use of TCAs for abdominal pain in the pediatric literature is promising but results remain inconclusive due to a limited number of studies in this area. In their review, Kaminski and colleagues [Kaminski et al. 2011] examined two pediatric studies that evaluated the effects of amitriptyline on pediatric patients with functional GI disorders [Bahar et al. 2008; Saps et al. 2009]. Neither study found a difference between amitriptyline and placebo; however, Saps and colleagues found amitriptyline was more helpful in the mild to moderate pain category. Thus, Kaminski and colleagues concluded that amitriptyline has not been shown to be beneficial for functional abdominal pain but noted that more studies need to be done [Kaminski et al. 2011].
To date, there have not been any trials comparing various types of TCAs for functional abdominal pain. In spite of the existing lack of research data, an interview study noted that gastroenterologists anecdotally reported that TCAs (e.g. amitriptyline, dothiepin, prothiaden, doxepin, imipramine, nortriptyline) reduced pain [Mikocka-Walus et al. 2007]. Clinical consensus is that psychotropic medications may affect visceral pain by decreasing inter-neuron signaling from pain fibers. Based on the current literature and clinical consensus, we recommend a trial of low-dose TCA for functional abdominal pain in IBD.
Selective serotonin-reuptake inhibitors
Several studies have been conducted examining the utility of SSRI treatment for abdominal pain in patients with IBD; however, many of the studies were limited by small sample sizes and the assessment tools used for pain rating. One such SSRI trial for patients with IBD demonstrated the possible therapeutic effect of paroxetine on abdominal pain. Unfortunately, extrapolation of the study findings are limited by the study’s small sample size (n = 8), open-label design, failure to differentiate between CD and UC, and because the pain rating was not an independent assessment, rather, it was embedded in a quality of life measure [Walker et al. 1996].
SSRIs have been used in trials examining people with IBS with and without depression [Friedrich et al. 2010]. Because of the variation in psychiatric comorbidity and the difficulty in isolating improvement in abdominal pain, the efficacy of SSRIs is unclear. Kuiken and colleagues and Tack and colleagues have shown some efficacy of citalopram in treating functional abdominal pain, though the latter study has been criticized for being underpowered [Kuiken et al. 2003; Tack et al. 2006]. More recently, Ladabaum and colleagues found no utility for low-dose citalopram (an SSRI) for improving abdominal pain in adults with IBS who were not depressed [Ladabaum et al. 2010]. In pediatrics, Campo and colleagues demonstrated utility of citalopram for abdominal pain [Campo et al. 2004]. It is notable that dosing in the study by Campo and colleagues began at 10 mg/day but was escalated to 40 mg/day by week 4 if there was no symptomatic improvement. Based on the current literature, we conclude that SSRIs are a promising yet understudied class of medications for visceral pain within IBD and the functional GI disorders.
Serotonin-norepinephrine reuptake inhibitors
Clinicians have noted low-dose SNRIs (e.g. duloxetine and venlafaxine) to be more efficacious than SSRIs, though the literature continues to emerge in this area [Drossman, 2008]. In a small open-label trial, Brennan and colleagues noted 12 weeks of duloxetine (60 mg per day) significantly ameliorated abdominal pain in adults with IBS who were not depressed [Brennan et al. 2009].
Gabapentin and pregabalin
Gabapentin and pregabalin are centrally acting agents. Their mechanism is thus far unclear, but they are postulated to be centrally acting voltage-gated calcium channel modulators. Although they are structurally similar to γ-amino butyric acid (GABA), which is a major inhibitory neurotransmitter in the CNS, gabapentin and pregabalin are functionally inactive at GABA receptors, are not metabolically converted into GABA agonists, and have no effect on GABA synthesis, uptake, or degradation [Gale and Houghton, 2011]. They reduce neuropathic pain by attenuating the release of many different neurotransmitters [Taylor et al. 2007].
There are no trials investigating the utility of gabapentin or pregabalin for abdominal pain relief in either IBD or IBS. A review of the literature revealed several trials outside of IBD and IBS, the results of which suggest future clinical trials with these medications for abdominal pain (especially visceral hypersensitivity) will have encouraging outcomes. For example, Houghton and colleagues found 3 weeks of pregabalin compared with placebo improved rectal hypersensitivity in patients with IBS [Houghton et al. 2007]. By the end of the small study rectal distension thresholds in the patients with IBS taking pregabalin were at the levels of control patients. Side effects (i.e. somnolence and dizziness) were mild and transient. The authors extrapolated the findings and suggested utility of pregabalin for other types of visceral hypersensitivity within IBS. Chua and colleagues’ abstract noted that pregabalin prevented the development of secondary hyperalgesia in the proximal esophagus after distal esophageal acidification but had no effect on the site of exposure [Chua et al. 2009]. This study suggested a centrally acting mechanism for pregabalin for ameliorating or preventing visceral hypersensitivity. In molecular studies, gabapentin and pregabalin have been shown to inhibit the substance P mediated activation of transcription factor nuclear factor κB (NFκB) [Park et al. 2008]. Gale and Houghton postulated that these findings may suggest utility for gabapentin and pregabalin for patients with pain at sites of prior inflammation or sensitization (where there may be an upregulation of the NFκB signaling pathway) [Gale and Houghton, 2011].
Abdominal pain in chronic pancreatitis also has a complex etiology, in particular, related to neuropathic pain [Ceyhan et al. 2009; Dimcevski et al. 2007; Drewes et al. 2008; Olesen et al. 2010]. Olesen and colleagues investigated and found efficacy for adjuvant pregabalin for abdominal pain relief in adults with chronic pancreatitis [Olesen et al. 2011]. Further clinical studies are required to evaluate the efficacy and tolerability of these medications in the treatment of abdominal pain from visceral hypersensitivity in IBD and IBS. For now, they are reasonable for adjunctive management of central and inflammatory-mediated visceral hypersensitivity.
Psychological approach
As discussed earlier, there is growing evidence suggesting symptomatic overlap between IBD and IBS [Bielefeldt et al. 2009]. Since both groups suffer similar impairment, it is often difficult to differentiate inflammatory, structural, or psychological mechanisms for pain. Thus, much of the behavioral psychotherapeutic treatments for IBD and associated visceral pain have come from the IBS literature.
Cognitive behavioral therapy
Cognitive behavioral therapy (CBT) is an intervention that teaches the patient to recognize the relationship between thoughts, feelings, and behaviors. Often the goal of CBT is to train an individual to examine his or her negative cognitions, understand how they may impact his or her behavior and emotions, and ultimately work to change this pattern. Typical CBT interventions are brief and last from 4 to 12 sessions. There are no trials to date investigating the utility of CBT on functional pain in IBD. Thus, much of clinical practice has been extrapolated from the IBS literature, where the data are quite promising. CBT for functional abdominal pain has been shown to be efficacious in adults compared with supportive therapy [Drossman et al. 2003] and pediatrics compared with standard medical care alone [Robins et al. 2005; Duarte et al. 2006].
Given the utility of CBT for functional abdominal pain within the functional GI disorders, this psychotherapeutic intervention may be efficacious for functional abdominal pain in patients with IBD.
Hypnotherapy
The data on hypnotherapy as a treatment modality for patients with IBD have thus far consisted of case reports and case series. Although there are some suggestions that hypnotherapy may affect symptom report, namely abdominal pain, psychological state, and quality of life [Emami et al. 2009; Keefer and Keshavarzian, 2007], the effect on pain perception still needs to be examined. In the non-IBD literature, Wilson and colleagues and Vlieger and colleagues have shown efficacy of hypnotherapy for functional abdominal pain in adult and pediatric patients with IBS, respectively [Wilson et al. 2006; Vlieger et al. 2007]. Several years later, Vlieger and colleagues demonstrated the short-term gut-directed hypnotherapy performed on pediatric patients with IBS in their previous study [Vlieger et al. 2007] had long-lasting effects after a mean of 4.8 years of follow up – with the patients in the hypnotherapy group having statistically significant lower pain intensity and pain frequency than those who received standard medical treatment plus supportive therapy [Vlieger et al. 2012]. Preliminary data suggest that it is a reasonable, but not first-line, option for psychotherapeutic treatment in patients with IBD.
Psychosocial
Coping skill modification
As discussed above, coping ability, skill type, and manner influence reported perception of abdominal pain. McCormick and colleagues examined the utility of a coping skills intervention on the perception of abdominal pain. Adolescent female participants with IBD were randomized to either an intervention consisting of a 1-day intensive coping skills treatment followed by 6 weeks of web-based modules for the patient and parent or a wait list control group [McCormick et al. 2010]. Although they did not find differences in abdominal pain between or within groups pre- or post-intervention they did note a reduction in somatization (measured by the Children’s Somatization Inventory; [Walker and Garber, 2003]). The study was again limited by a small sample size, high attrition rate, and failure to measure IBD activity. Given the limitations of this study, the paucity of data in the area of coping skill modification, and the unclear distinction between somatization and abdominal pain perception, for now modification of coping skills should be an adjunctive behavioral treatment for abdominal pain in IBD.
Stress management
García-Vega and Fernandez-Rodriguez sought to examine the relationships between stress and abdominal pain in patients with IBD by comparing the impact of manualized and self-directed stress management programs [Garcia-Vega and Fernandez-Rodriguez, 2004]. Those who received both stress management interventions notably reported significant changes in abdominal pain after the intervention. Unfortunately, psychological factors and IBD activity were not assessed in the study. The most recent study on stress management in IBD (INSPIRE study) [Boye et al. 2011] did not include abdominal pain as a specific outcome.
The relationship between stress and abdominal pain in IBD is likely bidirectional. Though it is plausible that stress management may help to decrease abdominal pain in IBD, until further research is done, stress management should remain an adjunctive treatment for abdominal pain in IBD.
Procedural methods
The IBD literature examining procedural interventions for the treatment of abdominal pain is extremely limited. Procedural treatments include the use of acupuncture, nerve blocks, and transcutaneous electrical nerve stimulation (TENS) for pain. Though these are not considered first-line treatments, they may have benefit in certain patients with IBD. Studies examining these procedures are described below.
Acupuncture
Acupuncture has been shown to reduce cortisol levels and affect serotonergic, cholingergic, and glutamatergic pathways in IBS [Yoon et al. 2011]. The existing functional literature on acupuncture demonstrates no difference between acupuncture and standard medical care for abdominal pain [Lim et al. 2006; Reynolds et al. 2008], and a potential placebo response for global IBS symptom and quality of life improvement [Lembo et al. 2009; Schneider et al. 2006].
There are several problems with studying acupuncture therapy. As the nature of acupuncture therapy is an individualized treatment it is difficult to standardize treatment protocols. Furthermore, there is some concern that the current sham procedures are inadequate and that better blinding procedures need to be designed [Wu, 2010]. Based on the current controversies in acupuncture efficacy, we do not recommend this procedure for relieving abdominal pain in IBD.
Nerve blocks
Celiac plexus nerve blocks are an effective treatment for pain associated with chronic pancreatitis and pancreatic cancer [Penman and Rösch, 2009] and have been used for many years. The use of nerve blocks for the management of intractable abdominal pain in the absence of inflammation is a possibility. The literature examining their utility for the treatment of functional GI disorders is limited [Kageshima et al. 1992], and there are no data available investigating their use in IBD. Given the complex nature of pain within both IBD and IBS, it is unlikely that blocking one pathway leading to pain perception will have efficacy.
Transcutaneous electrical nerve stimulation
Sylvester and colleagues were among the first to show efficacy of using TENS for functional abdominal pain in an open trial of 29 adults [Sylvester et al. 1986]. In a study of patients with functional dyspepsia, Zhou and colleagues noted a significant improvement in abdominal pain after TENS compared with placebo [Zhou et al. 2009]. Animal models have corroborated this decrease in abdominal pain and correlated it to decreased spinal N-methyl-D-aspartate receptor phosphorylation [Tian et al. 2008]. For nonfunctional abdominal pain, the literature does not point to TENS utility. For example, El-Rakshy and colleagues showed no differences in postoperative pain or narcotic requirement with TENS versus placebo after intra-abdominal surgery [El-Rakshy et al. 2009]. Given the limited nature of these data, we recommend considering adjunctive TENS for abdominal pain that is not due to inflammation.
Future directions
Although there is a growing body of data pertaining to visceral pain management in IBD, further research is warranted. Future trials should focus on uncovering the underlying mechanisms driving abdominal pain in patients with IBD, as these remain incomplete. These trials will lead to the development of future therapeutic agents that should be tested in large-scale randomized controlled trials.
When developing trials for pediatric populations, investigators must account for the impact of developmental plasticity on the central, peripheral, and enteric nervous systems. Clinically, we have found these changes to be more marked during puberty. Studies on the developmental pathways to abdominal pain (and associated visceral hypersensitivity), new classes of anti-inflammatory IBD medications, further trials on psychotropic medications, TENS stimulation, acupuncture, and even deep brain stimulation are welcomed. Exploring the impact of physical activity and exercise in this population should also be explored.
Recommendations for providers
Patients with unrelenting abdominal pain are some of the most challenging patients for practitioners to manage. When abdominal pain occurs in the absence of inflammation it can lead to multiple provider visits and potential patient frustration that their pain is not being taken seriously. However, there are extensive data to suggest that pain frequently occurs in the absence of objective data. Unfortunately, there is no ‘quick fix’ for many of these patients, and they need to be informed that successful management of abdominal pain may be an ongoing process. A strong patient–clinician alliance is crucial for managing abdominal pain in patients with IBD. Figure 2 outlines the spectrum of potential treatments given relative contributions of factors associated with abdominal pain perception in IBD.

Overview of treatment options for abdominal pain in inflammatory bowel disease (IBD). 5-ASA, 5-aminosalicylic acid; IBS, irritable bowel syndrome; TNF, tumor necrosis factor. (Courtesy of Dr. Douglas Drossman.)
We recommend the following algorithm for patients with IBD and abdominal pain (see Figure 3). The clinician should always consider overlap of visceral hypersensitivity and compounding psychological and psychosocial factors when screening for inflammatory and obstructive disease leading to pain. This consideration will facilitate more efficacious treatment plans tailored to the varying and individually unique contributions of these overlapping factors. In addition to standard IBD-specific treatments, medical therapy for pain arising from inflammation or strictures/adhesions could include antispasmodics [i.e. hyoscyamine (hyoscine), dicyclomine (dicycloverine)] as needed. Empiric treatment for SIBO should be considered in the presence of known small intestinal disease and history of surgery, particularly with side-to-side anastomoses. The patient should be weaned off narcotics if they have been taking them chronically [Grunkemeier et al. 2007]. If a stricture refractory to anti-inflammatory therapy or adhesion is suspected, surgical consultation should be done. If the abdominal pain is felt to be secondary to visceral hypersensitivity, one should consider prescribing a low-dose TCA or SNRI with concomitant CBT.

Clinical algorithm for abdominal pain management in inflammatory bowel disease (IBD). CBT, cognitive behavioral therapy.
Regardless of whether or not the pain is primarily due to inflammatory or partially obstructive etiologies, behavioral treatment should be considered based on clinical degree of visceral hypersensitivity and impact of abdominal pain on the patient’s life. Coping skills and stress management teaching should be considered only adjunctive to other therapies at this point. There are insufficient data from the IBD, IBS, or general GI literature to recommend acupuncture, spinal nerve blockage, or TENS.
Conclusion
Abdominal pain in IBD has multiple etiologies, is complex, and the literature delineating possible treatment modalities is limited. The significant overlap between IBD and IBS lends to the application of the IBS literature to pain management algorithms in IBD; however, further research is needed to develop better treatments of abdominal pain in IBD. Many treatments discussed in this review show promise in reducing abdominal pain in IBD, yet to fully understand this phenomenon, the field must continue to move forward.
Footnotes
Acknowledgements
The present work benefited from the input of Dr Douglas A. Drossman, Professor of Medicine and Co-Director of the University of North Carolina Center for Functional GI and Motility Disorders, and Dr Miguel D. Regueiro, Associate Professor of Medicine and Co-Director of the University of Pittsburgh Inflammatory Bowel Disease Center who provided valuable comments, ideas, and assistance to this manuscript.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declare no conflicts of interest in preparing this article.
