Abstract
Keywords
Introduction
Constipation is one of the most prevalent gastrointestinal complaints, estimates of UK prevalence varying from 8.2% to 52% [Wald et al. 2010; National Horizon Scanning Centre, 2008; Klaschik et al. 2003]. Part of the reason for such disparate estimates is the difficulty in defining constipation, which varies between patients and healthcare professionals and between studies. The NHS Clinical Knowledge Summary on constipation defines the condition as defaecation that is unsatisfactory, because of infrequent stools, difficult stool passage or seemingly incomplete defaecation [NHS Clinical Knowledge Summaries, 2010]. A study of 1055 factory workers in the UK in 1965 [Connell et al. 1965] found that 99% of the working population maintained a bowel frequency of between three bowel movements per day and three per week. On the basis of this evidence, constipation is sometimes defined as a frequency of fewer than three bowel movements per week [Higgins and Johanson, 2004].
Another factor contributing to difficulty in estimating the prevalence of the condition is that it may occur chronically in patients, but it may also be a transient short-term condition. There are many causes of constipation: it may be due to a prolonged colon passage or defaecation disorder, another disease, medication such as opioid therapy, or factors such as diet, fluid intake, immobility and lack of exercise [Klaschik et al. 2003]. Pregnancy is also known to increase the risk of constipation. Bradley and colleagues estimated that, during pregnancy, one in four women experience constipation [Bradley et al. 2007].
Laxatives as a treatment for constipation are among the most widely used of all medications [Xing and Soffer, 2001]. These medicines are available over the counter from pharmacies as well as by prescription from a general practitioner (GP). There are four main types of laxative: bulk-forming agents; osmotic laxatives; stimulant laxatives; and lubricants. Bulk-forming agents, such as ispaghula husk, are organic polymers with various water-holding capacities: they increase the intraluminal volume by retaining water, which stimulates motility and speeds the transit of luminal contents through the colon. Stimulant laxatives, such as senna, stimulate intestinal motility and affect epithelial transport of water and electrolytes [Klaschik et al. 2003; Xing and Soffer, 2001].
Macrogol and lactulose are osmotic laxatives, although the latter also promotes bacterial fermentation. They are not absorbed during their transit through the bowel and the water that is bound to them remains within the stool, thus aiding the relief of constipation. Macrogol is the international non-proprietary name for polyethylene glycol and is available in two different types, 3350 and 4000. These numbers represent the average molecular weight of the polyethylene glycol. It can be formulated either with or without electrolytes, which are added in an attempt to rectify the electrolyte depletion that can occur in some patients. The addition of electrolytes has an adverse effect on the taste of the product, however, and this can affect patient compliance.
The laxatives most commonly prescribed by GPs are lactulose, macrogol, senna, ispaghula husk, docusate sodium, bisacodyl and glycerol suppositories. In 2009, prescriptions for these products made up over 95% of all laxative prescriptions in England [NHS Information Centre, 2010]. However, there are no definitive treatment guidelines for constipation in adults and treatment of this condition may present many challenges, especially in pregnancy, in which there is an additional need to ensure the safety of treatments [Tytgat et al. 2003]. The NICE clinical guidelines on routine antenatal care give advice on managing common problems in pregnancy; the recommended treatment for constipation is a change in diet. No recommendations are given for treating constipation that is not improved by a change in diet [NICE Clinical Guidance, 2010].
The British National Formulary (BNF) recommends that, if diet and lifestyle changes do not control constipation in pregnancy, then moderate doses of poorly absorbed laxatives may be used; bulk-forming laxatives are recommended to be tried first, followed by an osmotic laxative such as lactulose, or a stimulant laxative such as senna if necessary [British National Formulary, 2010]. However, a consensus document on the use of laxatives in pregnancy has concluded that macrogols meets the criteria for the ideal laxative for use in pregnancy [Tytgat et al. 2003].
Despite the high prevalence of constipation in the UK and the related cost implications, the condition has not been widely studied in large patient groups. This study is the first investigation of constipation in the general population and in pregnancy, and also of the prescribing trends of laxatives within these patient groups, using the UK General Practice Research Database (GPRD).
Materials and methods
Study design
This was a cohort study designed to characterize the population of patients with GP-diagnosed constipation in each year between 2005 and 2009. A cohort of patients was extracted from the GPRD for each year. Patients were included in the study if they were aged 18 or older and had a diagnosis of constipation or faecal impaction within the study year, identified using a list of relevant READ codes (Appendix). The READ codes used were those for constipation, including constipation symptoms, chronic, acute and functional constipation, and faecal impaction.
Patients were included in the study only if their medical record met the acceptable standard of quality defined by the GPRD as being suitable for inclusion in research. For example, patients were required to have a valid registration date and a viable birth year. This ensured that patients with poor-quality or noncontiguous medical records were not included in the cohort. Only patients from practices that were up to standard were selected; such practices were those that had met the data quality criteria required by GPRD at the beginning of the study period. Patients were excluded if they were registered in a practice for which the latest data collection date was before the end of the study period, or if the patient transferred out of the practice before the end of the study year. This was to ensure that patient follow up was complete.
Therapies of interest were the seven most commonly prescribed laxatives: lactulose, macrogol, senna, ispaghula husk, docusate sodium, bisacodyl and glycerol. All available brands were included for each substance, and the therapies were identified using a list of codes (Appendix). Macrogol 3350 and macrogol 4000 were both included within the macrogol prescriptions.
Pregnancies can be identified within the longitudinal record of GPRD patients [Devine et al. 2010]. An algorithm was used to identify pregnancies, which combined pregnancy codes (Appendix) within the clinical details in GPRD with codes entered into the maternity module of the database. These codes were used to define a current ongoing pregnancy, and the patient records were examined for constipation diagnoses and laxative prescribing concurrent with the pregnancy.
The size of the study population was determined by the number of patients within the GPRD database with a diagnosis of constipation. A feasibility study showed that approximately 45,000 patients would be included in each year cohort. This number is large enough to give extremely precise estimates of the proportion of patients prescribed laxatives.
Data source
The GPRD is a database of longitudinal patient primary care records, containing anonymized data on demographics, diagnoses, referrals, prescribing and health outcomes for patients from almost 500 GP practices in the UK (over 3 million currently registered patients) [Jick et al. 1991]. The database contains approximately 6% of UK patients, and the geographical distribution is representative of the UK population [Garcia Rodriguez and Gutthann, 1998]. Validation studies have confirmed the high data quality and completeness of clinical records within the GPRD [Khan et al. 2010; Jick et al. 2003; Garcia Rodriguez and Gutthann, 1998]. A recent systematic literature review of studies using the GPRD reported that the median proportion of diagnoses correctly coded was 89% [Herrett et al. 2010]. The LUCK (Laxative Usage in patients with GP-diagnosed Constipation in the UK) study received approval from the Independent Scientific Advisory Committee at the Medicines and Healthcare products Regulatory Agency (Protocol number 10_078).
Data analysis
Data were extracted using GPRD OnLine Data (GOLD) and analysed using SAS® (SAS Institute Inc, North Carolina, USA) software version 9.2.
The prevalence of GP-diagnosed constipation was calculated for each year from 2005 to 2009. The patient population was summarized for each year by comedications, comorbidities, age and sex. Prescribing trends for each product were summarized by product and by patient age and sex. Regional differences in prescribing trends were examined using the GPRD-defined regions of the UK. The number of prescriptions issued per patient was summarized by product and by patient age and sex. The prescribing trends of laxatives during pregnancy were examined similarly.
Results
Diagnoses of constipation in primary care
Within a population of 3.8 million patients in the GPRD, the prevalence of GP-diagnosed constipation ranged from 12 per 1000 persons in 2005 to 12.8 per 1000 persons in 2009. Overall, the prevalence in women was almost twice that in men; in 2009 the prevalence was 9.1 per 1000 people in men and 16.5 per 1000 people in women.
The prevalence of GP-diagnosed constipation increased with age, from 4.0 per 1000 in patients aged 18–29 years to 52.1 per 1000 in patients aged over 75 in 2009. This pattern did not change during the 5-year study period. The prevalence of constipation in 2009 was higher in women than in men in the younger age groups, but higher in men than in women in patients over the age of 75 (Figure 1). There was a peak in prevalence amongst females aged between 30 and 44, possibly reflecting an increase in the risk of constipation during pregnancy or an increase in the likelihood of consulting a GP regarding constipation during pregnancy.
Prevalence of GP-diagnosed constipation in 2009 by age and sex.
Between 2005 and 2009, the number of patients with GP-diagnosed constipation in the GPRD increased with increasing age in both male and female patients (Figure 2). In each age group there were more female patients than male. Although there were more female patients than male among patients aged over 75, the prevalence of GP-diagnosed constipation was higher in males than in females in this age group, reflecting the higher number of female patients within this age group.
Number of patients with GP-diagnosed constipation by age and sex (2005–2009).
Patients with a diagnosis of constipation between 2005 and 2009.

There are a number of diagnostic codes for constipation; there are several general constipation codes and a number of more specific codes for the type of constipation. The code used to record constipation was examined for the cohorts of patients; however, although there are READ codes available for a GP to record the type of constipation with which a patient presents, it appears that these specific codes are not often used, and a general constipation code is used more commonly.
Opiate usage and cancer diagnoses were investigated in patients with a diagnosis of constipation, as opiate usage is known to cause constipation [Klaschik et al. 2003]. The cohort was examined for any prescriptions for opioids within the year of interest. A total of 44% of patients in the 2009 cohort had a prescription for an opioid within 2009. This proportion was similar across the 5 study years.
Diagnoses of constipation during pregnancy
Pregnant patients with a diagnosis of constipation in 2005 and 2009.

The average age of patients with GP-diagnosed constipation with a recorded pregnancy was 29.2 (SD 6.0) in 2005 and 31.1 (SD 7.1) in 2009. In 2005, 25% of these patients had a recorded history of GP-diagnosed constipation prior to their pregnancy, and 29% of patients had prior GP-diagnosed constipation in 2009.
Laxative prescribing in primary care
Amongst the six medications studied, lactulose was the most commonly prescribed overall. The pattern of prescribing changed over the 5-year study period, however; the percentage of patients with GP-diagnosed constipation who were prescribed lactulose and senna decreased and the proportion prescribed macrogol increased (Figure 3). Between 2005 and 2009 macrogol moved from being the least prescribed of the three most common laxatives to the most commonly prescribed; 29% of patients received a prescription for macrogol in 2005 and 47% received one in 2009.
Percentage of constipation cohort prescribed each type of laxative. *Ispaghula, Docusate Sodium, Bisacodyl or Glycerol.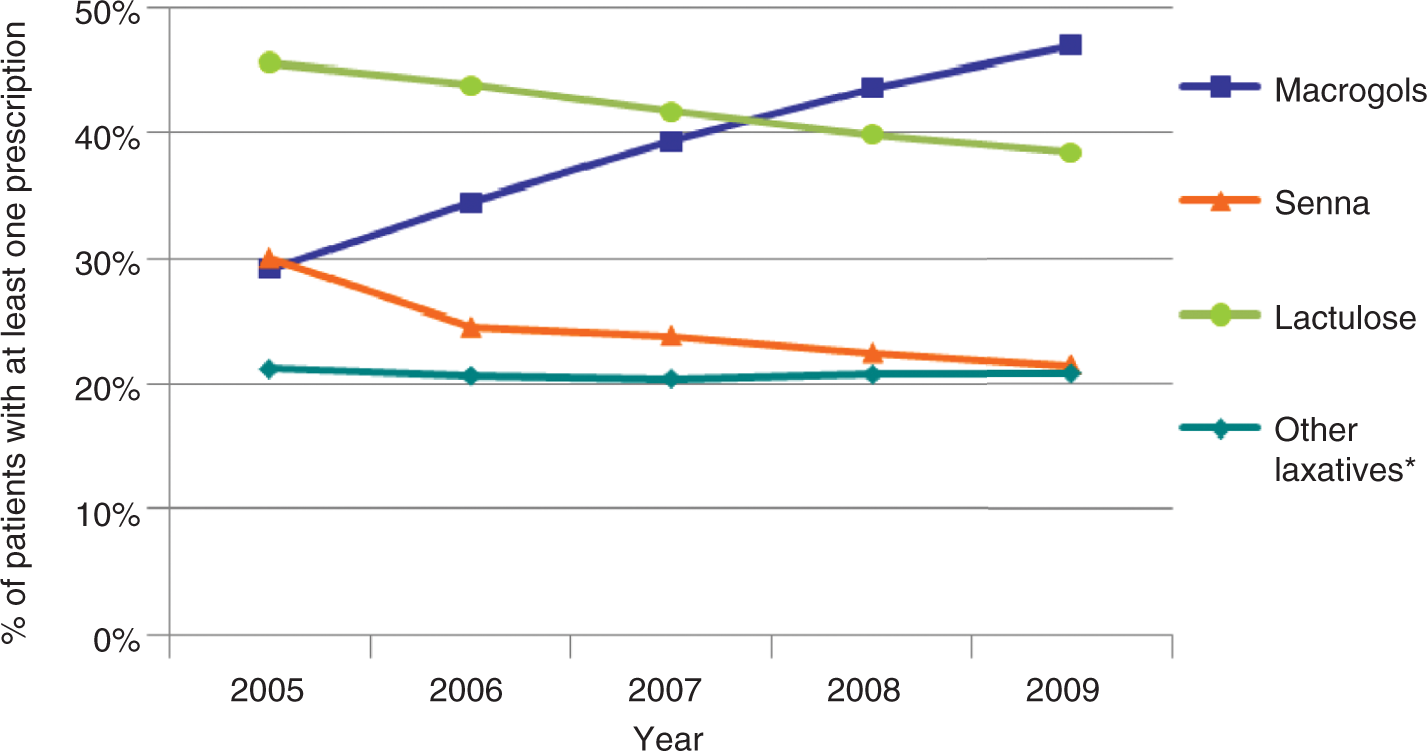
Characteristics of patients by prescribed laxative.

Ispaghula, docusate sodium, bisacodyl or glycerol suppositories.
Patients with a diagnosis of constipation but no prescription for a laxative were, on average, younger than patients prescribed laxatives: 56.6 years (SD 20.8) in 2009 compared with an overall average age of 61.5 (21.1). This is probably due to the fact that patients under the age of 60 pay a prescription charge in England, and laxatives bought over the counter in a pharmacy are likely to be cheaper than this charge. Patients prescribed senna tended to be older [67.2 (19.6) in 2009], and this group of patients had a slightly higher proportion of males: 38% compared with 35% overall. Patients prescribed lactulose or other laxatives were, on average, younger than patients prescribed macrogol or senna: 62.2 (22.0) and 62.7 (20.4) compared with 64.5 (20.0) for macrogol and 67.2 (19.6) for senna.
Over 76% of patients within the constipation cohort were prescribed at least one of the three most common laxatives (macrogol, senna and lactulose) within the year of diagnosis. In 2009, 32% of patients were prescribed at least two of these three types of laxative during 2009 (Figure 4). The number of prescriptions each patient received in a year increased with age.
Number of patients with each combination of treatments within 2009.
For the seven most commonly prescribed laxatives, the number of prescriptions each patient was prescribed within a 1-year period for the 2009 cohort of patients with GP-diagnosed constipation is summarized in Figure 5. Over 50% of patients who were prescribed each drug within 2009 received only one prescription. A total of 20% of patients prescribed senna were prescribed more than six prescriptions during 2009, compared with 10% of patients prescribed macrogol and 14% of those prescribed lactulose.
Number of patients by number of prescriptions in 2009.
In 2009 within the GPRD, 48,752 patients werediagnosed with constipation (prevalence of 0.13 per patient year). In this group of patients the total number of GP consultations in which constipation was diagnosed was 67,493. The average number of consultations per year that included a diagnosis of constipation was 1.3 (SD 0.86) and 19% of patients consulted a GP about constipation at least twice during this 1-year period.
The prescribing trends for the three most common laxatives within the cohort of patients with GP-diagnosed constipation were examined by UK region. There was a shift in prescribing from senna and lactulose to macrogol across all regions of the UK, with the biggest shift in the south-east, south central and south-east coast of England. In these areas macrogol was the most commonly prescribed laxative by 2009.
Laxative prescribing in pregnancy
Table 2 shows laxative prescribing in pregnant patients in 2005 and 2009. In 2005, 33% of pregnant patients with GP-diagnosed constipation were prescribed laxatives, rising to 44% in 2009. Lactulose was the laxative most commonly prescribed in pregnancy, but the percentage of laxative-treated patients prescribed lactulose dropped from 81% in 2005 to 64% in 2009. The percentage of pregnant patients treated with laxatives who were prescribed macrogols rose from 13% in 2005 to 32% in 2009. Patients treated with laxatives during pregnancy were more likely to have consulted a GP regarding constipation prior to their pregnancy than patients who were not treated with laxatives during their pregnancy (in 2009, 34% of those treated compared with 27% of those not treated).
Discussion
The prevalence of GP-diagnosed constipation has remained constant over the 5-year study period. The prevalence figures produced in this study are likely to be a considerable underestimate of the actual prevalence of constipation in the UK. This is because constipation can be a relatively minor complaint for which patients can self-medicate with a change in diet or can buy over-the-counter medications from a pharmacy.
The prevalence of GP-diagnosed constipation was found to be higher in female patients in all age groups under 75, but in patients aged over 75 the prevalence was higher in males. This may reflect a lower frequency of GP visits in general amongst younger males or an actual lower prevalence of constipation in men under the age of 75.
A high proportion of patients in this cohort had a record of opioid use, suggesting the possibility of medication-induced constipation in these patients. GPs prescribing opiates are aware that these agents can cause constipation, so may coprescribe laxatives or be more likely to inquire during their consultations whether the patient is constipated. Constipation in these patients could also be more severe or chronic than in patients not being prescribed opiates, which would mean that these patients are more likely to mention it to the GP.
There are other medications that are known to cause constipation, such as anticholinergic agents, tricyclic antidepressants, calcium channel blockers, diuretic drugs and NSAIDs, which were not investigated in this study. Further studies are needed to fully explain the proportion of medication-induced constipation within the GP-diagnosed constipation cohort.
Age is another factor that affects whether a patient consults a GP or a pharmacist. In both male and female patients the prevalence of GP-diagnosed constipation in patients aged 75 and older was much higher than the prevalence in younger patients. The results of this study suggest that older patients are either more likely to suffer from constipation or more likely to consult their GP if they do suffer from constipation, as the number of diagnoses and prescriptions was higher in the older age groups. Older patients would be likely to suffer from more comorbidities than younger patients and may therefore consult their GP more, and may therefore be more likely to mention constipation symptoms within a consultation. It is also possible that younger patients may prefer to visit a pharmacy for minor illness.
It is likely that patients with more severe and more chronic cases of constipation would be those more likely to be consulting a GP, although it was not possible to conclude this from this study as the diagnostic codes for constipation do not specify severity.
The trend in prescribing of laxatives by GPs to patients diagnosed with constipation has changed during the 5-year study period. The use of senna and lactulose products has decreased, while prescriptions for macrogol have increased steadily. Macrogol is now the most commonly prescribed laxative to patients with GP-diagnosed constipation in the UK. This pattern is also reflected in pregnant women; the proportion of patients prescribed macrogols for constipation in pregnancy has increased and the proportion prescribed lactulose and other laxatives has decreased.
The results of this study indicate that the most common laxatives prescribed in pregnancy are lactulose and macrogols, with lactulose being replaced by macrogols in the 5-year study period, reflecting the trend in the general population. The database does not allow us to examine the reasons GPs may have had for preferentially prescribing macrogols, so more research is needed to establish whether this might be due to their experience suggesting that macrogol is more effective, or to a perception that macrogol it is a safer product because of its mode of action.
There are no definitive guidelines on laxative prescribing in pregnancy, but the BNF suggests that if laxatives are required in pregnancy, bulk-forming laxatives should be tried first, followed by an osmotic laxative such as lactulose, followed by a stimulant such as senna if needed. Although the BNF suggests lactulose as the osmotic laxative to be prescribed, the results of this study suggest that GPs are increasingly confident in prescribing macrogols in pregnancy, and that therefore this advice is perhaps outdated. This is in line with the Summary of Product Characteristics for Macrogol 4000, which states that the product is suitable for use in pregnancy.
In 2009 there were approximately 67,000 GP consultations in which constipation was discussed and diagnosed. Although these consultations may also have been used to discuss otherconditions, the condition still represents a significant use of GP resources. Projecting thesefigures up to the whole UK population, itis estimated that there are over one millionGP consultations regarding constipation every year.
Footnotes
Acknowledgements
The authors would like to thank Dr Jas Kalsi for medical input into the writing up of the study, and Lindsay Vye and Eryl Lloyd for continued support of the study.
Appendix
Constipation codes. Pregnancy Codes.
Medical Code
Number of clinical events
READ code
Term
1028
635856
19C..00
Constipation
2004
609583
19C..11
Constipation symptom
5803
183861
J520z00
Constipation NOS
1709
36286
J520.00
Constipation - functional
10687
6428
J503100
Faecal impaction
20450
5381
19CZ.00
Constipation NOS
6364
2352
J520100
Chronic constipation with overflow
23641
819
J520000
Acute constipation
25797
815
J520200
Chronic constipation without overflow
26022
580
J520300
Drug induced constipation
15939
213
E264500
Psychogenic constipation
24180
150
J520y00
Other specified constipation

Current pregnancy codes
medcode
readcode
readterm
127
62…00
Patient pregnant
6184
ZV22.00
[V]Normal pregnancy
5709
62…13
Pregnancy care
3030
4654
Urine pregnancy test positive
5044
13H7.00
Unwanted pregnancy
4536
621..11
Pregnancy confirmed
13165
621..00
Patient currently pregnant
1771
L182.00
Anaemia during pregnancy, childbirth and the puerperium
6715
6219
Patient ? pregnant
10306
Z22C314
Weeks pregnant
5778
67A..00
Pregnancy advice
9408
L10y.11
Bleeding in early pregnancy
49519
Z229.00
Observation of position of pregnancy
1668
L182500
Iron deficiency anaemia of pregnancy
14899
621Z.00
Patient pregnant NOS
15567
6218
Pregnant -unplanned-not wanted
1130
L210.00
Twin pregnancy
29631
6222
Antenatal care: 2nd pregnancy
15033
ZV61900
[V]Other unwanted pregnancy
7517
621C.00
Unplanned pregnancy
1357
L210100
Twin pregnancy - delivered
2638
L1…00
Pregnancy complications
3766
L10..00
Haemorrhage in early pregnancy
3191
L16y500
Abdominal pain in pregnancy
20240
6216
Pregnant - planned
10185
L13..00
Excessive pregnancy vomiting
9754
Z22A400
Early stage of pregnancy
29593
6223
Antenatal care: 3rd pregnancy
14925
L10z.00
Early pregnancy haemorrhage NOS
10775
8B74.00
Iron supplement in pregnancy
3421
L12z300
Unspecified hypertension in preg/childb/puerp - not deliv
16215
6211
Pregnant - urine test confirms
11760
L13z.00
Unspecified pregnancy vomiting
16611
ZV22300
[V]Pregnant state, incidental
12890
Z227.00
Confirmation of pregnancy
12837
7F2B100
Ultrasound monitoring of early pregnancy
15318
6214
Pregnant - on history
18500
Z22D100
Viable pregnancy
14842
6217
Pregnant - unplanned - wanted
14994
6174
Pregnant, sheath failure
13672
8HHf.00
Refer to early pregnancy unit
16775
L16E.00
Pregnancy pruritus
36903
67AZ.00
Pregnancy advice NOS
13968
584D.00
Antenatal ultrasound confirms intra-uterine pregnancy
6649
L166800
Urinary tract infection complicating pregnancy
22193
Z229100
Intrauterine pregnancy
35158
Z225.00
Normal pregnancy
67975
L166.00
Genitourinary tract infections in pregnancy
1850
7F06012
Shirodkar suture in pregnancy
22183
957..11
Prescription exempt form-preg
21119
L182300
Anaemia during pregnancy - baby not yet delivered
10184
67A3.00
Pregnancy smoking advice
13759
445..00
Serum pregnancy test (B-HCG)
5693
L16A.00
Glycosuria during pregnancy
23495
L265.00
Small-for-dates fetus in pregnancy
61835
L161z00
Oedema or excessive weight gain in pregnancy NOS
10205
ZG9..00
Advice relating to pregnancy and fertility
15338
621A.00
Pregnancy unplanned ? wanted
37701
Z22A.00
Observation of pattern of pregnancy
53685
L410500
Varicose veins of legs in pregnancy
43140
67A2.00
Diet in pregnancy advice
21849
L031.00
Tubal pregnancy
30365
Z22AA00
Wanted pregnancy
15061
L13..12
Hyperemesis of pregnancy
17947
62a..00
Pregnancy review
26286
L18A000
Cholestasis of pregnancy
20439
L123.00
Transient hypertension of pregnancy
9986
Z212.11
Pregnancy care
23421
615C.00
IUD failure - pregnant
14644
L166z11
UTI - urinary tract infection in pregnancy
35912
ZV22200
[V]Pregnancy confirmed
25131
Z22AD11
Reported conception - pregnancy
28103
Z22A300
Concealed pregnancy
15433
L21..00
Multiple pregnancy
30618
Z22AB00
Unplanned pregnancy
26201
Z22AC00
Pregnancy with uncertain dates
35859
67A5.00
Pregnancy alcohol advice
20197
L211.00
Triplet pregnancy
14877
621B.00
Pregnant - ? planned
3029
L166500
Infections of kidney in pregnancy
41122
L10zz00
Early pregnancy haemorrhage NOS
15418
L166300
Genitourinary tract infection in pregnancy - not delivered
46270
ZV22z00
[V]Unspecified pregnant state
51298
6215
Pregnant - on abdom. palpation
36006
L16z.00
Pregnancy complication NOS
10278
L180800
Diabetes mellitus arising in pregnancy
2602
L166.11
Cystitis of pregnancy
10261
L2…00
Risk factors in pregnancy
37693
13Hd.00
Teenage pregnancy
2937
L175.11
Rubella contact in pregnancy
97034
67AE.00
Folic acid advice in first trimester of pregnancy
24603
L10y.00
Other haemorrhage in early pregnancy
15634
L166z00
Genitourinary tract infection in pregnancy NOS
29692
615C.11
Pregnant, IUD failure
38882
L123600
Transient hypertension of pregnancy
20118
62O..12
Static weight gain pregnancy
25254
Z21..00
Care relating to reproduction and pregnancy
28107
L161.00
Oedema or excessive weight gain in pregnancy no hypertension
50421
Z22A900
Unwanted pregnancy
39117
L126500
Eclampsia in pregnancy
14651
13H8.00
Illegitimate pregnancy
29205
4453
Serum pregnancy test positive
48552
L010.11
Anembryonic pregnancy
42614
Z22..00
Pregnancy observations
32975
6166
Pregnant, diaphragm failure
29746
8B75.00
Vitamin supplement - pregnancy
35592
6213
Pregnant - V.E. confirms
35509
624..00
A/N care: precious pregnancy
65834
ZV23200
[V]Pregnancy with history of abortion
23438
L2z..00
Risk factors in pregnancy NOS
27451
L166000
Genitourinary tract infection in pregnancy unspecified
45965
Z22C311
Pregnancy duration
50058
13SZ.00
Pregnancy benefit NOS
38346
ZV22000
[V]First normal pregnancy supervision
33708
L182100
Anaemia during pregnancy - baby delivered
34173
L12B.00
Proteinuric hypertension of pregnancy
41587
L210z00
Twin pregnancy NOS
30351
67A6.00
Drugs in pregnancy advice
12521
L161.11
Excessive weight gain in pregnancy
22557
Z22B100
Single pregnancy
31162
L165.00
Asymptomatic bacteriuria in pregnancy
15065
8B7..11
Pregnancy vitamin/iron prophyl
22215
Z22A200
High risk pregnancy
44729
L031000
Fallopian tube pregnancy
32493
Z22AD00
Presentation of pregnancy
26866
L2y..00
Other specified risk factors in pregnancy
27740
L16..00
Other pregnancy complication NEC
35646
L123z00
Transient hypertension of pregnancy NOS
45729
Z22D.00
Observation of viability of pregnancy
52583
L168.00
Fatigue during pregnancy
23334
L162.11
Albuminuria in pregnancy without hypertension
29786
L416600
Haemorrhoids in pregnancy
42015
L16y.00
Other pregnancy complications
30817
6212
Pregnant - blood test confirms
44057
ZV23111
[V]Pregnancy with history of hydatidiform mole
23751
L266.00
Large-for-dates fetus in pregnancy
40978
Z22A700
Surrogate pregnancy
49884
6761
Diabetic pre-pregnancy counselling
56451
L13zz00
Unspecified pregnancy vomiting NOS
6580
ZV23800
[V]Supervision of high-risk pregnancy due to social problems
38771
Lyu2200
[X]Other venous complications in pregnancy
18258
L167.00
Liver disorder in pregnancy
10173
Z23D200
Pregnant abdomen observation
33441
L511.00
Maternal care for viable fetus in abdominal pregnancy
46577
66AX.00
Diabetes: shared care in pregnancy - diabetol and obstet
24063
62O7.00
Pregnancy prolonged - 41 weeks
23373
L150.00
Post-term pregnancy
54942
L123100
Transient hypertension of pregnancy - delivered
21196
ZV23.00
[V]High-risk pregnancy supervision
27980
L166700
Infections of the genital tract in pregnancy
36235
67A4.00
Pregnancy exercise advice
49855
Q015100
Fetus or neonate affected by twin pregnancy
34639
L180100
Diabetes mellitus during pregnancy - baby delivered
37048
6282
A/N care:10yrs+since last preg
36394
L16C.00
Pregnancy induced oedema + proteinuria without hypertension
52048
Z22AB11
Accidental pregnancy
38022
Z22CF00
Date symptom of pregnancy first noted
50512
L031z00
Tubal pregnancy NOS
25501
Z22A100
Low risk pregnancy
16017
L166100
Genitourinary tract infection in pregnancy - delivered
30302
7F…12
Pregnancy operations
12230
L263900
Maternal care for fetal tachycardia during pregnancy
65555
L132.00
Late vomiting of pregnancy
34938
L212.00
Quadruplet pregnancy
38556
67A7.00
Pregnancy dental advice
45413
L263A11
Maternal care for reduced fetal heart rate during pregnancy
35514
624Z.00
A/N care: precious preg. NOS
40633
Z22A600
Teenage pregnancy
41033
Z22C511
EDC - Estimated date of conception
23831
ZV22y00
[V]Other specified pregnant state
40964
L192.00
Continuing preg after intrauterine death one fetus or more
54866
L030.00
Abdominal pregnancy
55338
L123000
Transient hypertension of pregnancy unspecified
32950
L03y100
Cornual pregnancy
25872
L16yz00
Other pregnancy complication NOS
46082
K5Cz.11
Habitual aborter-not pregnant
49559
L180300
Diabetes mellitus during pregnancy - baby not yet delivered
70993
L16Az00
Glycosuria during pregnancy NOS
24170
L15..00
Prolonged or post-term pregnancy
26055
L263A00
Maternal care for fetal bradycardia during pregnancy
39359
L385.00
Failed or difficult intubation during pregnancy
40357
L2D..00
Retained intrauterine contraceptive device in pregnancy
47608
L177.00
Infections of bladder in pregnancy
53097
Z22C313
Duration of pregnancy
61301
L10y000
Other haemorrhage in early pregnancy unspecified
20623
ZV4J000
[V]Problems related to unwanted pregnancy
50440
Z22A500
Biochemical pregnancy
27833
L13y.00
Other pregnancy vomiting
49407
L2B..00
Low weight gain in pregnancy
53167
L188300
Abnormal GTT during pregnancy - baby not yet delivered
54107
L10z000
Early pregnancy haemorrhage NOS unspecified
37038
Z23D100
Girth of pregnant abdomen
53849
L16y000
Other pregnancy complication unspecified
37348
Lyu2500
[X]Other specified pregnancy-related conditions
43000
L16D.00
Excessive weight gain in pregnancy
59276
L032.00
Ovarian pregnancy
60345
M240500
Alopecia of pregnancy
61284
L210200
Twin pregnancy with antenatal problem
23995
L15..11
Post-term pregnancy
46009
Q0…00
Fetus/neonate affected by maternal problem unrelated to preg
41625
L13yz00
Other pregnancy vomiting NOS
54577
L210000
Twin pregnancy unspecified
47080
L162.00
Unspecified renal disease in pregnancy
48534
ZV23100
[V]Pregnancy with history of trophoblastic disease
52221
L03y000
Cervical pregnancy
53921
L161000
Oedema or excessive weight gain in pregnancy, unspecified
56252
ZV22100
[V]Other normal pregnancy supervision
52685
ZV23000
[V]Pregnancy with history of infertility
63344
Z22C500
Estimated date of conception
68004
Q01..00
Fetus/neonate affected by maternal complication of pregnancy
24944
L163300
Pregnancy care of habitual aborter
36421
L167z00
Liver disorder in pregnancy NOS
38312
L162.12
Nephropathy NOS in pregnancy without hypertension
55730
ZV22.11
[V]Supervision of normal pregnancy
59634
L411512
Vaginal varices in pregnancy
64366
L412500
Superficial thrombophlebitis in pregnancy
26852
ZV23400
[V]Pregnancy with other poor obstetric history
65783
L030000
Delivery of viable fetus in abdominal pregnancy
69327
L10yz00
Other haemorrhage in early pregnancy NOS
41504
L13y000
Other pregnancy vomiting unspecified
48304
L10z200
Early pregnancy haemorrhage NOS - not delivered
61408
L120300
Benign essential hypertension in preg/childb/puerp-not deliv
64247
Z235.00
Observation of shape of pregnant abdomen
25230
9Ea2.00
Less 24 wk involv risk injury physic/mentl health preg woman
58142
L16A000
Glycosuria during pregnancy unspecified
59588
L411500
Genital varices in pregnancy
61563
L161300
Oedema or excessive weight gain in pregnancy - not delivered
72835
Q01z.00
Fetus/neonate affected by maternal complic pregnancy NOS
96757
L411513
Vulval varices in pregnancy
56953
Z235400
Pendulous pregnant abdomen
64292
L16y300
Other pregnancy complication - not delivered
53661
L13z200
Unspecified pregnancy vomiting - not delivered
64125
L09yz00
Other specified complication NOS follow abortive pregnancy
64523
L091z00
Delayed/excess haemorrhage NOS following abortive pregnancy
65256
L164.00
Peripheral neuritis in pregnancy
67098
L183300
Drug dependence during pregnancy - baby not yet delivered
67863
Z22B.00
Observation of quantity of pregnancy
69722
ZV23z00
[V]Unspecified high-risk pregnancy
73559
L13z000
Unspecified pregnancy vomiting unspecified
43344
ZV61800
[V]Illegitimate pregnancy
44734
62O8.00
Pregnancy prolonged - 42 weeks
49502
L228.00
Multiple pregnancy with malpresentation
53490
L178.00
Infections of urethra in pregnancy
54677
L167200
Liver disorder in pregnancy - not delivered
55618
ZV23600
[V]Supervisn/pregnancy wth history insufficnt antenatal care
73917
L10y200
Other haemorrhage in early pregnancy - not delivered
44770
L191.00
Continuing pregnancy after abortion of one fetus or more
59313
Z22BA00
Contin pregnancy after intrauterine death of sibling fetus
63751
L16A300
Glycosuria during pregnancy - not delivered
64099
L168000
Fatigue during pregnancy unspecified
66649
L41z500
Venous complication of pregnancy, unspecified
69011
L132000
Late pregnancy vomiting unspecified
71730
L123300
Transient hypertension of pregnancy - not delivered
73455
Q015200
Fetus or neonate affected by triplet pregnancy
94804
67AB.00
Preg. prescription exempt adv.
37573
Z22B900
Continuing pregnancy after abortion of sibling fetus
41959
L171300
Maternal gonorrhoea in pregnancy - baby not yet delivered
60309
Q0y..00
Maternal problems unrelated preg affecting fetus/neonate OS
61935
L185.11
Congenital heart disease in pregnancy
67164
L18z300
Medical condition NOS in pregnancy - baby not yet delivered
68694
Z22C.00
Observation of measures of pregnancy
69815
62H3.00
Rh screen - 1st preg. sample
72883
Lyu2100
[X]Other vomiting complicating pregnancy
73914
L412511
Thrombophlebitis of legs in pregnancy
91254
Z235200
Rounded pregnant abdomen
21467
L03y200
Membranous pregnancy
29623
62H4.00
Rh screen - 2nd preg. sample
35855
L15z.00
Prolonged pregnancy NOS
40825
Z22B800
Undiagnosed multiple pregnancy
54701
ZV23y00
[V]Other specified high-risk pregnancy
55889
L175300
Maternal rubella during pregnancy - baby not yet delivered
57059
L187300
Orthopaedic disorder in pregnancy - baby not yet delivered
61466
Z22A211
HRP - High risk pregnancy
62358
L167000
Liver disorder in pregnancy unspecified
66390
Q0z..00
Maternal problem unrelated preg affecting fetus/neonate NOS
69599
L21y.00
Other multiple pregnancy
69686
L417000
Cerebral venous thrombosis in pregnancy
91888
L181300
Thyroid dysfunction in pregnancy - baby not yet delivered
93895
Z22B500
Quintuplet pregnancy
94473
L211z00
Triplet pregnancy NOS
99247
L13y200
Other pregnancy vomiting - not delivered
49193
67A7.11
Care of teeth advice -in preg.
51956
L212200
Quadruplet pregnancy with antenatal problem
54293
Q015.00
Fetus or neonate affected by multiple pregnancy
54938
L162000
Unspecified renal disease in pregnancy unspecified
59650
Z235300
Transversely enlarged pregnant abdomen
60877
L263800
Maternal care for fetal decelerations during pregnancy
61576
L211000
Triplet pregnancy unspecified
64500
L183.11
Pregnancy and drug dependence
66594
L186.11
Heart disease during pregnancy
67698
L150000
Post-term pregnancy unspecified
67893
L2C..00
Malnutrition in pregnancy
72014
L212z00
Quadruplet pregnancy NOS
72019
L132200
Late pregnancy vomiting - not delivered
73727
L168z00
Fatigue during pregnancy NOS
92579
L162.13
Uraemia in pregnancy without hypertension
93303
ZV22400
[V]Supervision of other normal pregnancy
96743
L122300
Other pre-exist hypertension in preg/childb/puerp-not deliv
97349
L121300
Renal hypertension in preg/childbirth/puerp - not delivered
99237
L411511
Perineal varices in pregnancy
99980
L168300
Fatigue during pregnancy - not delivered
37163
L150z00
Post-term pregnancy NOS
67728
L150200
Post-term pregnancy - not delivered




Therapy Codes
Macrogols. Senna. Lactulose. Ispaghula. Docusate Sodium. Bisacodyl. Glycerol.
Product code
Number of events
Product
Substance
5201
1132924
MOVICOL sachets [NORGINE]
potassium chloride/macrogol 3350/sodium bicarbonate/sodium chloride
6581
108523
macrogol compound npf oral powder 13.8 g
potassium chloride/macrogol 3350/sodium bicarbonate/sodium chloride
6599
76211
MOVICOL PAEDIATRIC PLAIN oral powder [NORGINE]
potassium chloride/macrogol 3350/sodium bicarbonate/sodium chloride
6119
11695
MOVICOL HALF oral powder [NORGINE]
potassium chloride/macrogol 3350/sodium bicarbonate/sodium chloride
10069
11380
IDROLAX powder 10 g [SCHWARZ]
macrogol 4000
38390
6906
LAXIDO ORANGE oral powder [GALEN]
potassium chloride/macrogol 3350/sodium bicarbonate/sodium chloride
10237
5952
macrogol compound npf half strength sugar free oral powder
potassium chloride/macrogol 3350/sodium bicarbonate/sodium chloride
10261
3805
macrogol npf oral powder 10 g
macrogol 4000
10125
1767
macrogol 4000 powder 10 g
macrogol 4000
35443
1608
MOVICOL PLAIN sachets [NORGINE]
potassium chloride/macrogol 3350/sodium bicarbonate/sodium chloride
39532
658
MOVICOL CHOCOLATE sachets [NORGINE]
potassium chloride/macrogol 3350/sodium bicarbonate/sodium chloride
39660
283
macrogol compound npf oral powder 13.7 g
potassium chloride/macrogol 3350/sodium bicarbonate/sodium chloride
40791
264
macrogol compound npf sugar free oral powder
potassium chloride/macrogol 3350/sodium bicarbonate/sodium chloride
12915
246
macrogol with sodium sulphate + electrolytes powder
potassium chloride/macrogol 3350/sodium bicarbonate/sodium chloride/sodium sulphate
7030
209
polyethylene glycol with electrolytes oral powder
potassium chloride/macrogol 3350/sodium bicarbonate/sodium chloride
39734
175
LAXIDO NATURAL oral powder [GALEN]
potassium chloride/macrogol 3350/sodium bicarbonate/sodium chloride
39702
152
macrogol compound npf oral powder
potassium chloride/macrogol 3350/sodium bicarbonate/sodium chloride
41776
3
MOLAXOLE powder for oral solution [MEDA]
potassium chloride/macrogol 3350/sodium bicarbonate/sodium chloride
30905
2
GOLYTELY powder
potassium chloride/macrogol 3350/sodium bicarbonate/sodium chloride/sodium sulphate


Product code
Number of events
Product
Substance
Formulation
BNF
52
2455687
senna tablets 7.5 mg
sennoside
tablets
Stimulant laxatives
2494
295499
MANEVAC granules [HFA]
senna fruit/ispaghula husk
granules
Bulk-forming laxatives/ Stimulant laxatives
1858
219088
senna syrup 7.5 mg/5 ml
sennoside
syrup
Stimulant laxatives
7105
81606
senna oral solution 7.5 mg/5 ml
sennoside
oral solution
Stimulant laxatives
3672
36598
SENOKOT syrup 7.5 mg/5 ml [RECKITT B]
sennoside
syrup
Stimulant laxatives
6034
20106
ispaghula husk with senna fruits granules 54.2% + 12.4%
senna fruit/ispaghula husk
granules
Bulk-forming laxatives/ Stimulant laxatives
5897
14995
senna tablets 15 mg
sennoside
tablets
Stimulant laxatives
5210
9743
senna granules
sennoside
granules
Stimulant laxatives
6324
8689
SENOKOT granules [RECKITT B]
sennoside
granules
Stimulant laxatives
14215
4312
SENOKOT syrup [RECKITT B]
sennoside
syrup
Stimulant laxatives
171
1708
SENOKOT tablets [RECKITT B]
sennoside
tablets
Stimulant laxatives
9890
1479
senna tablets 12 mg
sennoside
tablets
Stimulant laxatives
17587
666
sennosides-total elixir
senna leaf
elixir
Stimulant laxatives
16030
612
senna chewable tablet 15 mg
sennoside
chewable tablet
Stimulant laxatives
14292
507
SENOKOT HI-FIBRE ORANGE granules [RECKITT B]
ispaghula husk
granules
Bulk-forming laxatives
5987
378
CALIFIG SYRUP OF FIGS elixir [MERCK CONS]
senna leaf
elixir
Stimulant laxatives
28003
369
SENNA tablets 7.5 mg [ACTAVIS]
sennoside
tablets
Stimulant laxatives
35847
329
SENOKOT DUAL RELIEF tablets [RECKITT B]
aloes (aloe)/taraxacum (dandelion root)/cascara/senna leaf/fennel seed
tablets
Stimulant laxatives/Herbal remedy
28122
283
SENNA tablets 7.5 mg [TEVA]
sennoside
tablets
Stimulant laxatives
20452
116
SENOKOT HI-FIBRE LEMON granules [RECKITT B]
ispaghula husk
granules
Bulk-forming laxatives
28831
107
SENOKOT MAX STRENGTH tablets 15 mg [RECKITT B]
sennoside
tablets
Stimulant laxatives
16150
63
SENOKOT PHARMACY syrup [RECKITT B]
sennoside
syrup
Stimulant laxatives
14143
50
EX-LAX SENNA pill [NOVARTIS]
sennoside
pill
Stimulant laxatives
20747
44
SENOKOT
Unknown
21570
43
EX-LAX SENNA tablets 15 mg [NOVARTIS]
sennoside
tablets
Stimulant laxatives
23084
42
senna with cascara tablets 32 mg + 130 mg
cascara/senna leaf
tablets
Stimulant laxatives
41565
22
SENNA tablets [FAMILY H]
sennoside
tablets
Stimulant laxatives
15181
12
NYLAX WITH SENNA tablets [RECKITT B]
sennoside
tablets
Stimulant laxatives
33700
10
SENNA tablets 7.5 mg [HILLCROSS]
sennoside
tablets
Stimulant laxatives
22303
7
SENNA FRUIT 12.4%/ISPAGHULA SEED 54.2%
Unknown
10287
6
X-PREP liquid [NAPP]
senna leaf
liquid
Stimulant laxatives
16484
5
SENOKOT DIRECT RELIEF suppository 4 g [RECKITT B]
glycerol
suppository
Stimulant laxatives
30150
2
SURE-LAX SENNA chewable tablet 15 mg [POTTER'S]
sennoside
chewable tablet
Stimulant laxatives
28836
1
AGIOLAX granules [RADIOL]
senna fruit/ispaghula husk
granules
Bulk-forming laxatives/Stimulant laxatives
31988
1
NYLAX tablets [CROOKES]
bisacodyl/phenolphthalein/senna
tablets
Stimulant laxatives
32858
1
SENNA tablets 7.5 mg [ASPAR]
sennoside
tablets
Stimulant laxatives
36256
1
SENNA
Unknown
39230
1
DUAL LAX EXTRA STRONG tablets [LANE]
aloin/cascara extract/senna leaf
tablets
Stimulant laxatives


Product code
Number of events
Product
Substance
Formulation
BNF
12
4051008
lactulose solution 3.35 g/5 ml
lactulose
solution
Osmotic laxatives
4613
1614999
lactulose solution 3.1–3.7 g/5 ml
lactulose
solution
Osmotic laxatives
4695
43057
lactulose solution (flavoured) 3.35 g/5 ml
lactulose
solution (flavoured)
Osmotic laxatives
5010
12193
lactulose sachets 10 g
lactulose
sachets
Osmotic laxatives
9489
8814
LACTUGAL solution [INTRAPHARM]
lactulose
solution
Osmotic laxatives
19524
8187
LACTULOSE
Unknown
16088
6669
LACTULOSE solution 3.1–3.7 g/5 ml [IVAX]
lactulose
solution
Osmotic laxatives
28877
1856
LACTULOSE solution 3.1–3.7 g/5 ml [TEVA]
lactulose
solution
Osmotic laxatives
9650
1222
DUPHALAC DRY powder 10 g [SOLVAY]
lactulose
powder
Osmotic laxatives
34015
1221
LACTULOSE solution 3.1–3.7 g/5 ml [BERK]
lactulose
solution
Osmotic laxatives
18423
488
REGULOSE solution [NOVARTIS]
lactulose
solution
Osmotic laxatives
8911
313
DUPHALAC solution [SOLVAY]
lactulose
solution
Osmotic laxatives
34055
141
LACTULOSE solution 3.1–3.7 g/5 ml [HILLCROSS]
lactulose
solution
Osmotic laxatives
27708
35
LACTULOSE solution 3.1–3.7 g/5 ml [GEN (UK)]
lactulose
solution
Osmotic laxatives
4559
12
LEMLAX solution 3.28 g/5 ml [CO-PHARMA]
lactulose
solution
Osmotic laxatives
33678
6
LACTULOSE solution 3.1–3.7 g/5 ml [KENT]
lactulose
solution
Osmotic laxatives
41638
6
LACTULOSE solution 3.1–3.7 g/5 ml [SOLVAY]
lactulose
solution
Osmotic laxatives
26590
5
LAXOSE solution [BERK]
lactulose
solution
Osmotic laxatives
32598
5
LACTULOSE solution 3.1–3.7 g/5 ml [SANDOZ]
lactulose
solution
Osmotic laxatives
34360
3
LACTULOSE solution 3.1–3.7 g/5 ml [NOVARTIS]
lactulose
solution
Osmotic laxatives

Product code
Number of events
Product
Substance
Formulation
BNF
1227
719146
ispaghula husk gluten-free sugar-free effervescent granules
ispaghula husk
sugar-free effervescent granules
Bulk-forming laxatives
2337
74909
ispaghula husk gluten-free sugar free powder 3.4 g
ispaghula husk
sugar free powder
Bulk-forming laxatives
6430
60013
ispaghula husk gluten-free sugar free powder 3.5 g
ispaghula husk
sugar free powder
Bulk-forming laxatives
5598
47783
mebeverine hydrochloride with ispaghula husk sachets 135 mg + 3.5 g
ispaghula husk/mebeverine hydrochloride
sachets
Bulk-forming laxatives/Other antispasmodics
6034
21204
ispaghula husk with senna fruits granules 54.2% + 12.4%
senna fruit/ispaghula husk
granules
Bulk-forming laxatives/Stimulant laxatives
2582
19004
ispaghula husk gluten-free sugar free effervescent powder 3.6 g
ispaghula husk
sugar free effervescent powder
Bulk-forming laxatives
6851
15952
ispaghula husk gluten-free sugar free granules
ispaghula husk
sugar free granules
Bulk-forming laxatives
8559
8400
ISPAGHULA HUSK 90 % GRA
Unknown
14618
3254
ispaghula husk gluten-free sugar-free effervescent granules
ispaghula husk
sugar-free effervescent granules
Bulk-forming laxatives
1655
2649
ISPAGHULA HUSK 66 % GRA
Unknown
13171
1949
ispaghula husk gluten-free granules
ispaghula husk
granules
Bulk-forming laxatives
11124
851
ispaghula husk gluten-free powder 3.4 g
ispaghula husk
powder
Bulk-forming laxatives
11243
308
ispaghula husk gluten-free sugar free effervescent powder 6 g
ispaghula husk
sugar free effervescent powder
Bulk-forming laxatives
25032
34
ISPAGHULA HUSK EFFERVESCENT SACHET
Unknown
34800
32
ISPAGHULA HUSK GLUTEN-FREE sugar free effervescent powder 3.5 g [HILLCROSS]
ispaghula husk
sugar free effervescent powder
Bulk-forming laxatives
22303
7
SENNA FRUIT 12.4%/ISPAGHULA SEED 54.2%
Unknown
37647
7
ispaghula husk with lactobacillus and bifidobacteria oral powder
ispaghula husk/lactobacillus acidophilus/
bifidobacterium bifidum
oral powder
Bulk-forming laxatives/Unlicensed product
24523
6
ISPAGHULA HUSK ORANGE SACHET
Unknown
20683
4
ISPAGHULA HUSK SACHET
Unknown
25637
4
ISPAGHULA HUSK
Unknown
29829
1
ISPAGHULA HUSK MICRONISED + DEXTROSE
Unknown


Product code
Number of events
Product
Substance
Formulation
BNF
2468
417366
docusate sodium capsules 100 mg
docusate sodium
capsules
Stimulant laxatives/Faecal softeners
5215
16627
docusate sodium sugar free oral solution 50 mg/5 ml
docusate sodium
sugar free oral solution
Stimulant laxatives/Faecal softeners
3558
14562
docusate sodium sugar free paediatric oral solution 12.5 mg/5 ml
docusate sodium
sugar free paediatric oral solution
Stimulant laxatives/Faecal softeners
2699
7880
docusate sodium tablets 100 mg
docusate sodium
tablets
Stimulant laxatives
9510
3942
docusate sodium ear drops 0.5%
docusate sodium
ear drops
Removal of ear wax
17989
408
docusate sodium enema 120 mg
docusate sodium
enema
Stimulant laxatives
13999
214
docusate sodium with glycerol enema 90 mg + 3.78 g/5 ml
docusate sodium
enema
Stimulant laxatives
24073
69
docusate sodium ear drops 5%
docusate sodium
ear drops
Removal of ear wax
15002
37
docusate sodium with glycerol ear drops
docusate sodium/glycerol
ear drops
Removal of ear wax
24109
36
docusate sodium with sorbitol enema
docusate sodium/sorbitol
enema
Faecal softeners
23668
34
DOCUSATE SODIUM
Unknown
21613
8
DOCUSATE SODIUM
Unknown
22260
4
docusate sodium and bisacodyl tablets
bisacodyl/docusate sodium
tablets
Stimulant laxatives
28791
3
DOCUSATE SODIUM S/F ORAL
Unknown
Product code
Number of events
Product
Substance
Formulation
BNF
2451
400750
bisacodyl enteric coated tablets 5 mg
bisacodyl
Oral
Stimulant laxatives
2771
99721
bisacodyl suppository 10 mg
bisacodyl
Rectal
Stimulant laxatives
2770
22219
bisacodyl paediatric suppository 5 mg
bisacodyl
Rectal
Stimulant laxatives
11565
1300
bisacodyl rectal solution 2.74 mg/ml
bisacodyl
Rectal
Stimulant laxatives/Unlicensed medicinal product (specials)
12012
954
BISACODYL 10 MG TAB
Unknown
34016
219
BISACODYL tablets 5 mg [CELLTECH]
bisacodyl
Oral
Stimulant laxatives
33799
155
BISACODYL tablets 5 mg [HILLCROSS]
bisacodyl
Oral
Stimulant laxatives
12079
66
bisacodyl with dioctyl sodium sulphosuccinate tablets
bisacodyl/docusate sodium
Oral
Stimulant laxatives
36071
27
BISACODYL suppository 10 mg [DANIEL]
bisacodyl
Rectal
Stimulant laxatives
34772
16
BISACODYL tablets 5 mg [APS]
bisacodyl
Oral
Stimulant laxatives
32322
6
BISACODYL suppository 10 mg [HILLCROSS]
bisacodyl
Rectal
Stimulant laxatives
34027
6
BISACODYL tablets 5 mg [IVAX]
bisacodyl
Oral
Stimulant laxatives
33798
5
BISACODYL tablets 5 mg [ACTAVIS]
bisacodyl
Oral
Stimulant laxatives
22260
4
docusate sodium and bisacodyl tablets
bisacodyl/docusate sodium
Oral
Stimulant laxatives
38425
3
BISACODYL suppository 10 mg [CELLTECH]
bisacodyl
Rectal
Stimulant laxatives
27532
2
BISACODYL
Unknown
34352
1
BISACODYL enteric coated tablets 5 mg [SOVEREIGN]
bisacodyl
Oral
Stimulant laxatives
41685
1
BISACODYL suppository 10 mg [MARTINDALE]
bisacodyl
Rectal
Stimulant laxatives

Product code
Number of events
Product
Substance
Formulation
BNF
4364
131673
glycerol suppository 4 g
glycerol
Rectal
Stimulant laxatives
2989
43410
glycerol suppository 1 g
glycerol
Rectal
Stimulant laxatives
3160
17494
glycerol suppository 2 g
glycerol
Rectal
Stimulant laxatives
13999
214
docusate sodium with glycerol enema 90 mg + 3.78 g/5 ml
docusate sodium
Rectal
Stimulant laxatives
41730
15
GLYCEROL suppository 4 g [MARTINDALE]
glycerol
Rectal
Stimulant laxatives

