Abstract
Background:
Two studies demonstrated the efficacy and safety of naldemedine in adult patients with chronic non-cancer pain and opioid-induced constipation (OIC). However, no studies have compared the efficacy of peripherally acting µ-opioid receptor antagonists in patients with adequate and inadequate responses to prior OIC therapy with laxatives. This
Methods:
Patients with OIC were randomized to once-daily treatment with naldemedine 0.2 mg or placebo. The primary efficacy endpoint was the proportion of responders [⩾3 spontaneous bowel movements (SBMs)/week and an increase from baseline of ⩾1 SBM/week for ⩾9 weeks of the 12-week treatment period and ⩾3 weeks of the final 4 weeks of the 12-week treatment period]. Additional endpoints included change in SBM frequency, change in frequency of SBMs without straining, proportion of complete SBM (CSBM) responders, change in CSBM frequency, and time to first SBM. Treatment-emergent adverse events (TEAEs) were assessed.
Results:
The analysis included 538 (317 PLRs, 221 non-PLRs) and 537 (311 PLRs, 226 non-PLRs) patients in the naldemedine and placebo arms, respectively. There were significantly more responders in the naldemedine PLR (46.4%;
Conclusion:
This integrated analysis further supports the efficacy and tolerability of naldemedine in the treatment of OIC and demonstrates a consistent effect in both PLR and non-PLR subgroups.
[ClinicalTrials.gov identifier: NCT01965158 and NCT01993940]
Introduction
Opioid-induced constipation (OIC) is the most frequent adverse effect associated with opioid analgesic treatment. 1 This condition, defined by Rome IV criteria as new or worsening symptoms of constipation that occur when initiating, changing, or increasing the dose of opioid therapy, 2 results from the activation of µ-opioid receptors, which have extensive distribution throughout the enteric nervous system in the gastrointestinal (GI) tract.1,2 The reported prevalence of OIC is typically around 50% among patients taking opioids for chronic cancer pain and non-cancer pain.3–7 In one prospective study in cancer patients, about half developed OIC within 2 weeks of beginning opioid therapy. 7
Unlike other adverse effects associated with opioid therapy, OIC persists unabated over time, even with use of laxatives. 8 Opioids interfere with motility, secretion, and sphincter function.9–13 Traditional laxatives (stool softeners, osmotics, stimulants, salines, and rectal options) are often used for treatment of OIC but do not target µ-opioid receptors, to which opioids bind in the myenteric and submucosal nerve plexi of the GI tract; therefore, they do not directly block these adverse effects on GI function.11,14 Studies have reported that 40%–94% of patients taking laxatives for management of OIC have an inadequate response.3,14 As a result, these patients may experience a range of GI symptoms that have a negative impact on quality of life, activities of daily living, and work productivity.3,14 In an effort to reduce GI symptoms, patients may skip or reduce the opioid dose, leading to inadequate pain relief.3,14,15
Naldemedine is an oral, once-daily, peripherally acting µ-opioid receptor antagonist (PAMORA) approved in Japan, the United States (US), and Europe for the treatment of OIC in adults.16–19 The efficacy and safety of once-daily naldemedine 0.2 mg
The objective of this
Methods
Study design
COMPOSE-1 and COMPOSE-2 [ClinicalTrials.gov identifiers: NCT01965158 and NCT01993940, respectively] were identically designed randomized, multinational, double-blind, placebo-controlled, parallel-group phase III studies conducted in 68 and 69 centers, respectively, between August 2013 and June 2015. 20 Study protocols were approved by independent ethics committees at each site and all institutional review boards (Supplemental Table S1). The studies were performed in accordance with the principles of the Declaration of Helsinki and all applicable local Good Clinical Practice guidelines and regulations. All patients provided written informed consent. The data from COMPOSE-1 and COMPOSE-2 were integrated for this analysis.
Integrated study population baseline demographics and medical characteristics (ITT population).

Non-PLR, naldemedine 0.2 mg
BM, bowel movement; BMI, body mass index; BSS, Bristol Stool Scale; ITT, intent to treat; MED, morphine equivalent dose; non-PLR, non-poor laxative responder; OIC, opioid-induced constipation; PLR, poor laxative responder; SBM, spontaneous bowel movement; SD, standard deviation.
Participants
The methodology of the COMPOSE-1 and COMPOSE-2 studies has been described previously. 20 Briefly, patients were aged 18–80 years with chronic non-cancer pain for ⩾3 months who were on a stable opioid regimen (⩾30 mg morphine equivalents/day) for at least 1 month prior to screening. Patients were not currently using, or were willing to discontinue, laxatives. After discontinuation of laxatives and a 2- to 4-week screening/qualification period to verify OIC symptomatology and eligibility, enrolled patients were randomized in a 1:1 ratio to 12 weeks of once-daily treatment with naldemedine 0.2 mg or matching placebo (Figure 1). 20 Criteria for OIC during screening included: ⩽4 spontaneous bowel movements (SBMs) in qualifying period; ⩽3 SBMs in any 7-day period during screening, and ⩾1 incidence of straining, hard, or lumpy stools, sensation of incomplete evacuation, or sensation of anorectal obstruction/blockage with ⩾25% of bowel movements. Key exclusion criteria were evidence/history of bowel structural abnormalities, strictures, obstructions, or history of bowel surgery; medical conditions affecting GI transit; history of chronic constipation unrelated to opioid use; and no history of laxative use for OIC.
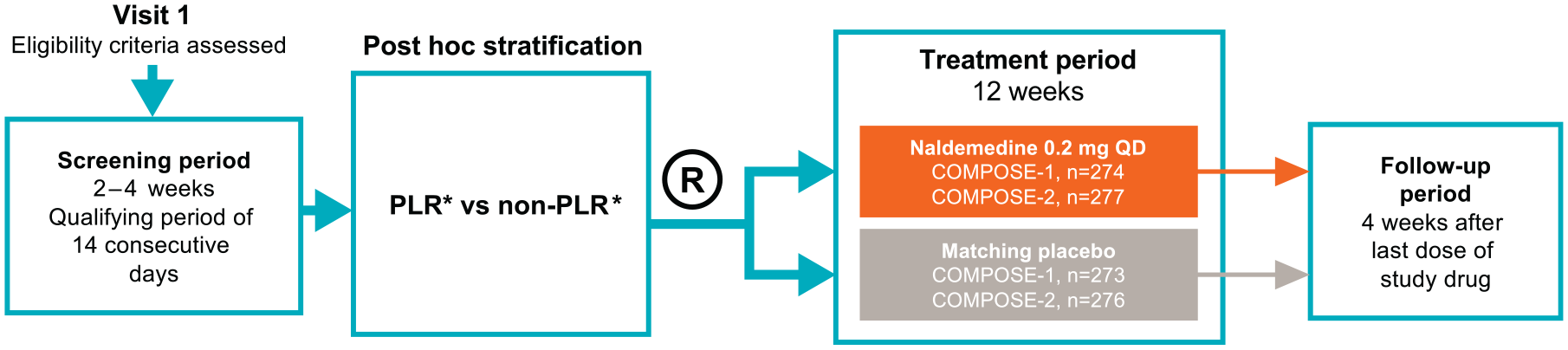
Study design. PLRs were defined as patients who were on laxative therapy prior to entering the study and who stopped its use within 30 days prior to screening. Non-PLRs were defined as patients who stopped laxatives >30 days prior to screening or who only received rescue laxative at or after screening.
Assessments
The primary efficacy endpoint was the proportion of responders, defined as patients with ⩾3 SBMs per week and an increase from baseline of ⩾1 SBM per week for ⩾9 weeks of the 12-week treatment period and ⩾3 weeks of the final 4 weeks of the 12-week treatment period. 20 Additional efficacy endpoints included the least-squares mean (LSM) change from baseline in the frequency of SBMs per week to each week of the treatment period; the change from baseline in frequency of SBMs without straining (defined as a straining score of 0) per week to the last 2 weeks of the treatment period; the proportion of complete SBM (CSBM) responders, with CSBM responders defined as having ⩾3 CSBMs per week and an increase from baseline of ⩾1 CSBM per week for ⩾9 weeks of the 12-week treatment period and ⩾3 weeks of the final 4 weeks of the 12-week treatment period; the change from baseline in the frequency of CSBMs per week to each week of the treatment period; and the time to first SBM after the initial dose of study drug.
Safety was assessed using incidences of treatment-emergent adverse events (TEAEs), TEAEs in the GI disorders system organ class (SOC), and major cardiovascular events. 20 Adverse events were coded to SOC and preferred term using the Medical Dictionary for Regulatory Activities, version 16.0. TEAEs were assessed for the population comprising all randomized patients who received at least one dose of study medication. 20
Statistical analysis
For the purpose of integration, efficacy data included in this analysis are from first dose up to 12 weeks of treatment in the COMPOSE-1 and COMPOSE-2 studies and do not include data collected during the follow-up periods. Efficacy analyses were based on the intent-to-treat population comprising all randomized patients. Safety analyses were based on the safety population comprising all randomized patients who received at least one dose of study medication.
20
The data presented here represent a
Comparisons were made between the naldemedine PLR and placebo PLR subgroups and the naldemedine non-PLR and placebo non-PLR subgroups, as well as the PLR and non-PLR subgroups for patients treated with naldemedine. Analysis of the primary efficacy outcome (proportion of SBM responders) and the proportion of CSBM responders used the Cochran–Mantel–Haenszel test adjusted for opioid dose strata. A mixed-effect repeat-measures model was used for change from baseline in SBM and CSBM frequency at each week. 20 Analysis of covariance using the opioid dose strata as a covariate was applied to the change from baseline in frequency of SBMs without straining. 20 Time to first SBM was evaluated using the generalized Wilcoxon test adjusted by study. The Clopper–Pearson method was used to calculate confidence intervals (CIs) for differences between the PLR and non-PLR subgroups for efficacy endpoints. 20 Incidences of TEAEs were summarized descriptively and presented along with treatment differences and 95% confidence intervals (CIs) for the PLR and non-PLR subgroups. All statistical analyses were conducted using SAS version 9.2.
Results
Study population
The integrated efficacy analysis of COMPOSE-1 and COMPOSE-2 included 549 and 546 patients in the naldemedine and placebo arms, respectively. Of these, 11 patients in the naldemedine group and 9 patients in the placebo group could not be categorized as PLR or non-PLR. Overall, 317 PLR patients were randomized to naldemedine and 311 to placebo. A total of 221 and 226 non-PLR patients were randomized to naldemedine and placebo, respectively. A summary of the integrated patient demographics, baseline characteristics, and opioid use is presented in Table 1.
Patient groups were generally well matched between naldemedine and placebo and between PLR and non-PLR subgroups with no statistical differences between groups, although a lower proportion of patients in the non-PLR group receiving naldemedine was White, and a higher proportion was Black, relative to the other study arms. Additionally, a higher proportion of PLR patients
Efficacy
Primary efficacy endpoint
The proportion of SBM responders in the integrated efficacy population are illustrated in Figure 2. Similar to the overall group, there were significantly more responders in the naldemedine PLR (46.4%;

Spontaneous bowel movement responders in PLR and non-PLR subgroups by laxative response at baseline (primary endpoint).
Additional efficacy endpoints
A significant increase in change from baseline in SBM frequency with naldemedine was established at week 1 (the first on-treatment assessment)
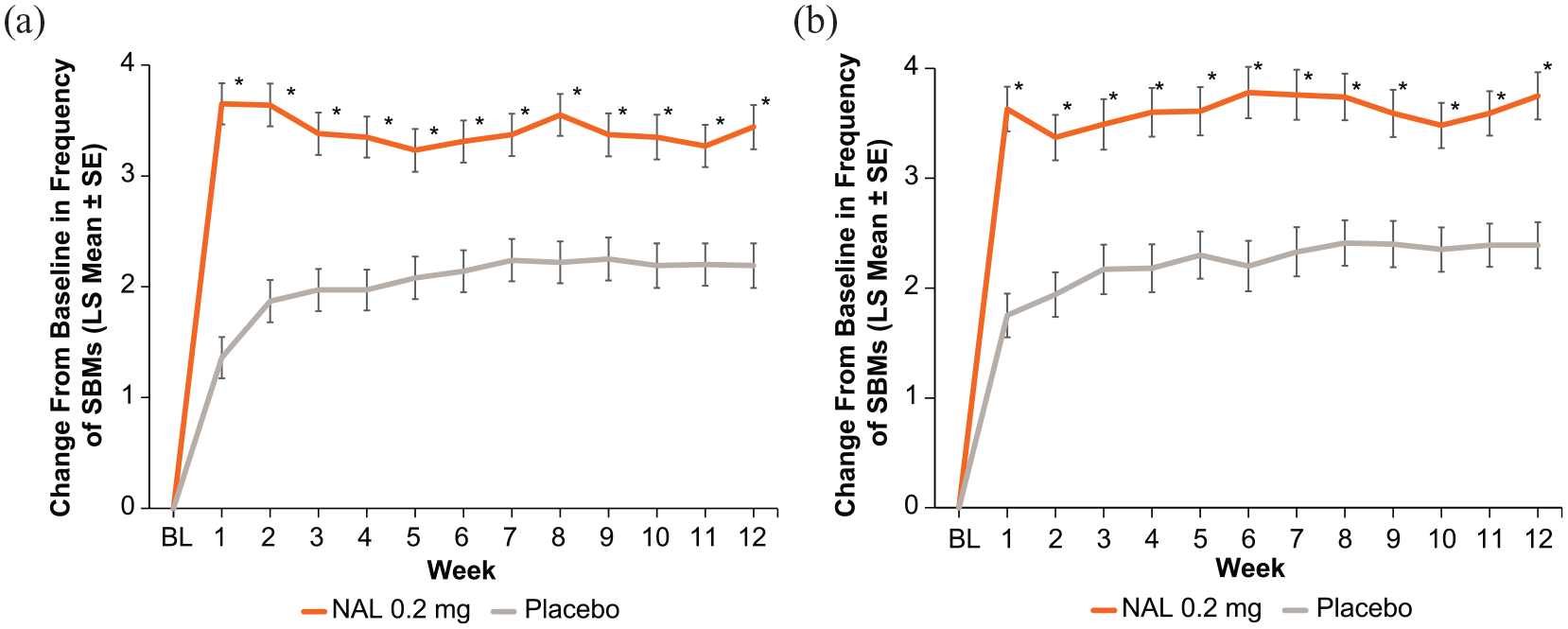
Change from baseline to each week in frequency of spontaneous bowel movements in (a) PLR and (b) non-PLR subgroups.
Similar to the overall group, an increase from baseline to the last 2 weeks of treatment in the frequency of SBMs without straining were observed in the PLR (LSM: 1.6;

Change from baseline in frequency of spontaneous bowel movements without straining per week to the last 2 weeks of the treatment period in PLR and non-PLR subgroups by laxative response at baseline.
The proportion of patients with improvement in CSBMs was similar to the improvement found in SBMs. There were significantly more responders in the PLR (25.9%;
The time to first SBM was significantly shorter for the naldemedine groups

Kaplan–Meier estimate of time to first spontaneous bowel movement in (a) PLR and (b) non-PLR subgroups and (c) in PLR/non-PLR subgroups treated with naldemedine. Vertical lines represent censored time.
Safety
There was no notable difference in the overall incidence of TEAEs in the PLR subgroups treated with naldemedine or placebo (Table 2). Treatment-related TEAEs occurred in 22.2% and 17.0% of PLR patients in the naldemedine and placebo groups, respectively, compared with 19.1% and 9.7% in the non-PLR naldemedine and placebo groups, respectively. TEAEs leading to discontinuations occurred in 4.8% and 3.5% of PLR patients in the naldemedine and placebo groups, respectively, compared with 5.5% and 0.9% in the non-PLR naldemedine and placebo groups, respectively. For both treatment-related TEAEs and TEAEs leading to discontinuation, significantly higher rates were observed with naldemedine
Summary of TEAEs by laxative response at baseline (safety population).

Both cardiac events were myocardial infarctions.
CI, confidence interval; MACE, major adverse cardiovascular event; NA, not available; non-PLR, non-poor laxative responder; PLR, poor laxative responder; TEAE, treatment-emergent adverse event.
The incidence of serious TEAEs was comparable between naldemedine and placebo in the PLR and non-PLR subgroups (Table 2). There were few serious treatment-related TEAEs, with three in the PLR subgroup treated with naldemedine (1%), one each in the PLR placebo group (0.3%) and non-PLR naldemedine subgroup (0.5%), and none in the non-PLR placebo group. One patient each (0.3%) in the PLR naldemedine and placebo arms had major adverse cardiovascular events of myocardial infarction. There was one death in the PLR naldemedine subgroup (0.3%) due to an opioid overdose.
Discussion
This
Comparison between PLR and non-PLR subgroups
The proportion of responders to naldemedine was slightly but significantly higher in the non-PLR subgroup than in the PLR subgroup, although this finding should be interpreted with caution because the analysis did not include a placebo comparator. No other significant differences in efficacy outcomes were observed between the PLR and non-PLR subgroups in patients treated with naldemedine. We hypothesized that PLRs represent a subgroup of patients that are less compliant or that have pre-existing comorbidities, such as defecation disorders, that may make them less responsive to medications for constipation in general. Although the results of this analysis do not support our hypothesis, the lack of a pronounced difference between the PLR and non-PLR subgroups is not unexpected given that PAMORAs such as naldemedine act directly in the GI tract by reversing the effect of opioids on µ-opioid receptors. 19 Laxatives, by contrast, do not address the underlying pathophysiology of OIC. 19
The presence and severity of OIC symptoms at baseline were generally similar between PLR and non-PLR subgroups. It is also notable that PLR and non-PLR subgroups received the same ranges of opioid doses, underscoring the fact that even low-dose/weak opioids may result in OIC.20,25 On the primary efficacy endpoint, the placebo response rate appeared somewhat lower in the PLR subgroup (30.2%) than in the non-PLR subgroup (38.9%). This observation of a lower placebo response may be indicative of a slightly less responsive population, although the difference may have been due to chance.
Adverse events
Naldemedine 0.2 mg treatment once daily was generally well tolerated for up to 12 weeks in both the PLR and non-PLR subgroups, with the most common TEAEs in both subgroups being GI related. This latter observation is consistent with the mechanism of action of naldemedine.17,19 The incidences of treatment-related TEAEs and adverse events leading to discontinuation were significantly higher among naldemedine-treated patients
Definition of PLR
The definition of PLR varies across studies of patients with non-cancer pain and OIC, making comparisons difficult. A prospective study of patients treated with naloxegol prespecified the definition of PLR as those who took medication from one or more laxative classes for a minimum of 4 days within 2 weeks before screening and whose symptoms were rated as moderate, severe, or very severe in at least one of four stool symptom domains on the baseline laxative-response questionnaire.26,27 In contrast, a retrospective study examining characteristics of patients with non-cancer pain and OIC defined PLR as those who had <3 bowel movements and ⩾1 symptom from the Patient Assessment of Constipation Symptoms questionnaire scored moderate, severe, or very severe, despite sufficient laxative use (at least one laxative agent on ⩾4 occasions during a 2-week period). 14 Development of a standard definition for PLR is further complicated by the fact that many patients express satisfaction and willingness to continue laxative therapy despite experiencing no or minimal relief of symptoms. 28 Nonetheless, response rates in the above mentioned naloxegol study were consistent with the results reported here, with comparable response rates in patients with PLR and in the overall population.
Limitations
A limitation of this current analysis is that it was a
Conclusion
Data from this integrated analysis further support the efficacy and tolerability of naldemedine in the treatment of OIC and demonstrate that naldemedine has a consistent effect regardless of whether or not a patient had a poor response to laxatives.
Supplemental Material
sj-docx-1-tag-10.1177_17562848211032320 – Supplemental material for Naldemedine is effective in the treatment of opioid-induced constipation in patients with chronic non-cancer pain who had a poor response to laxatives
Supplemental material, sj-docx-1-tag-10.1177_17562848211032320 for Naldemedine is effective in the treatment of opioid-induced constipation in patients with chronic non-cancer pain who had a poor response to laxatives by Martin E. Hale, James E. Wild, Tadaaki Yamada, Takaaki Yokota, Jan Tack, Viola Andresen and Asbjørn Mohr Drewes in Therapeutic Advances in Gastroenterology
Footnotes
Acknowledgements
Editorial and writing support was provided by Adrienne Drinkwater of Peloton Advantage, LLC, an OPEN Health company, and funded by Shionogi Inc.
Author contributions
Study design: AMD
Data analysis: AMD
Data interpretation: All authors
Manuscript preparation: AMD
Manuscript review and revisions: All authors
Final approval of manuscript: All authors
Conflict of interest statement
MH was a clinical trial investigator, consultant to Shionogi Inc. and received a stipend for review of the clinical study report.
JW received a stipend from Shionogi Inc. for review of the clinical study report.
T Yamada is an employee of Shionogi Inc. who may or may not own stock options.
T Yokota is an employee of Shionogi & Co., Ltd. who may or may not own stock options.
JT has provided scientific advice to Allergan, Kyowa Kirin, Shionogi, and Shire, has been a speaker for Allergan and Kyowa Kirin, and has received a research grant from Kyowa Kirin related to opioid-induced constipation.
VA has received speaker and/or consultant fees from Allergan, Bayer, Falk, Ferring, Hexal, Kyowa Kirin, 4M-Medical, Sanofi, and Shionogi.
AMD has no conflicts to declare.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: These studies were sponsored by Shionogi Inc., Florham Park, NJ. BDSI owns full rights to commercialize Symproic® (naldemedine) tablets 0.2 mg in the US for opioid-induced constipation.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
