Abstract
Pulmonary artery aneurysms (PAAs) are a rare pathology with potentially devastating consequences. In this case series, we describe two cases of clinically isolated pulmonary arteritis and concisely review the literature surrounding this new clinical entity. Two Caucasian women, aged 65 and 73, were referred to our cardiovascular surgery center with incidental PAAs, both growing to over 65 mm in main pulmonary artery diameter. Both participants underwent serial thoracic CT-angiograms to monitor the pulmonary artery aneurysms. Both pulmonary artery aneurysms showed an interval increase in diameter, so they were repaired surgically. The main pulmonary artery was excised in both cases and replaced with a synthetic graft. No pre- or postoperative corticosteroids were administered. Neither of our patients had systemic symptoms of giant cell arteritis or elevated inflammatory markers. In both cases, only the main pulmonary artery was replaced to minimize surgical complexity and risk; however, the left pulmonary artery of one patient began to further dilate 5 years postoperatively. Currently, no reintervention is planned. These cases represent mounting evidence for the existence of clinically isolated pulmonary arteritis leading to a pulmonary artery aneurysm. This entity is analogous to clinically isolated aortitis and may represent a subclinical smoldering vasculitis. These rare aneurysms can progress over time and require surgical intervention. In the absence of guidelines for pulmonary artery aneurysm intervention, the present cases will serve to guide further management of this rare disease.
Plain Language Summary
Isolated inflammation of the pulmonary artery (large blood vessel that supplies the lung) has not been well described. We describe two cases which were initially followed with repeat imaging and eventually grew to be larger and subsequently underwent surgery. There are no guideline recommendations for management of this rare condition. These cases with similar features may be used to guide management of additional cases.
Background
Defining pulmonary artery aneurysms
Pulmonary artery aneurysms (PAAs) are a rare phenomenon with potentially devastating consequences. PAAs represent a focal dilation of all three layers of the pulmonary artery (PA) wall—the tunica intima, media, and adventitia. 1 Its most-feared complication is rupture, with a mortality rate of 50%–100%. 1 In addition, PAAs can compress nearby structures, leading to acute coronary syndrome (via coronary artery compression), hoarseness (recurrent laryngeal nerve compression), and respiratory distress (bronchus compression). However, PAAs are often asymptomatic and found incidentally on imaging; even those whose PAAs have ballooned to 70 mm in diameter may have few clinical complaints (mean normal PA diameter of 32 mm).2,3
PAAs are assessed with computed tomography (CT) angiograms, but there are no standardized criteria for diagnosis or intervention. Several definitions have been proposed: first, using a general size threshold to define a PAA (ranging from 27 to 32 mm); using a tailored size indexed to patient body surface area; or based on a focal dilation 1.5-fold the normal PA diameter.3,4 A 2022 study conducted an in-depth analysis of PA dimensions, accounting for body surface area, age, and sex. They concluded that PA diameters <45 mm are not aneurysmal. 3
There are no consensus guidelines for when to surgically repair PAAs, especially if asymptomatic. Suggestions have included: if the PAA diameter exceeds a threshold (e.g., 55 mm); a growth rate >5 mm within 6 months; PA pressure >50 mmHg; and the presence of associated complications.2,4
Clinically isolated pulmonary arteritis
Our cases involve clinically isolated pulmonary arteritis (CIPA). This name mirrors consensus-based terminology for clinically isolated aortitis. 5 Just as clinically isolated aortitis is thought to represent a smoldering vasculitis with subclinical presentation, CIPA may represent the same phenomenon in the PA. Both CIPA and clinically isolated aortitis are subcategories of the broader entity “clinically isolated vasculitis.” Aneurysms form secondary to inflammatory disruption of the vessel’s elastic lamina.
The CIPA cases we present demonstrated granulomatous/giant cell patterns of inflammation. This histology is similar to that found in giant cell arteritis (GCA) with marked infiltrates of CD4+ T cells and tissue macrophages, forming multinucleate giant cells. 6 However, GCA is a clinical diagnosis, and neither of our patients met the criteria for this diagnosis 7 : absent were any clinical symptoms, physical findings of GCA, or elevated systemic markers of inflammation.
There have been case reports of pulmonary arteritis that would fit with a diagnosis of CIPA since 2014. Specifically, we have identified seven similar cases to our own, all reported in the past decade (Table 1).8–14 In these cases, the main PA diameter ranged from 51 to 90 mm. One report featured an aneurysm of the interlobar right PA measuring 41 mm (mean = 31 mm). In all cases, the PAAs were discovered incidentally or presented symptomatically with angina, acute coronary syndrome, or progressive dyspnea. When there were symptoms, they were attributable to localized effects of the aneurysm, for example, pulmonary valve regurgitation or compression of the coronary arteries. There were no systemic symptoms of GCA such as headache, polymyalgia rheumatica, or fever. Generally, there were no lab findings of systemic inflammation or a personal history of vasculitis. There were a few exceptions: first, one case featured an elevated C reactive protein (CRP) of 5.3 mg/L (<5.0 mg/L) in the context of an non-ST elevation myocardial infaction presentation secondary to PAA compression of the coronary arteries 12 ; second, one case featured a patient with a background of granulomatosis with polyangiitis (GPA). 10 In this patient, anti-neutrophil cytoplasmic antibody titers were negative, and the PAA showed non-necrotizing pathology, a picture at odds with typical GPA pathology.10,15 For our review, we opted to restrict our literature search to cases with histopathology. Otherwise, it would not have been possible to compare the pathophysiology of the aneurysms to our cases. In none of the papers we reviewed was a biopsy taken of the PAA without complete excision of the PAA and/or grafting. Therefore, all the cases in Table 1 were managed with the PAA surgically.
Prior case reports of clinically isolated pulmonary arteritis.

F, assigned female sex at birth; M, assigned male sex at birth; PAA, pulmonary artery aneurysm; rPA, right pulmonary artery.
The first report using the terminology “clinically isolated PAA due to granulomatous vasculitis” was a Canadian report from 2018. 11 In this case, a PAA was discovered incidentally in a 74-year-old female and grew from 57 to 64 mm in a 6-month interval, prompting surgical repair.
In summary, there have been seven cases of CIPA with granulomatous patterns of vasculitis, all reported in the last decade.8–14 In these cases, there were no definitive clinical signs or lab markers to diagnose GCA or other autoimmune disease. Steroids were not used to treat the PAAs in any of these cases, owing to the absence of systemic inflammation. All PAAs were treated surgically. There were no long-term adverse outcomes reported post-surgically, and no accounts of aneurysm recurrence. However, long-term follow-up was not available for every case.
Case presentation
We present two prospective cases of clinically isolated PAA (Table 2; Figures 1 and 2), which presented to our academic institution for surgical consideration. This research was performed in accordance with the Declaration of Helsinki, and a waiver of institutional review board was granted (REB waiver number 1030804). The reporting of this study conforms to CARE guidelines (Supplemental Material). 16 Written informed consent for treatment and participation in this case report was obtained from both patients. Both cases were in women over the age of 65, whose aneurysms grew greater than 65 mm in diameter (Figure 3). Both aneurysms were discovered incidentally during unrelated diagnostic imaging. Both patients received a thorough rheumatologic assessment and workup, which showed no systemic signs, symptoms, or markers of inflammatory or autoimmune disease, ruling out Behçet’s or other systemic vasculitis, sarcoidosis, or infectious etiology. Neither patient underwent a Positron Emission Tomography (PET) or Magnetic Resonance Imaging (MRI) scan, as it was felt systemic therapy was not indicated in the absence of clinical symptoms, regardless of potential results. Both patients were followed with serial CTs. Although initially the aneurysms seemed stable, both patients’ aneurysms eventually showed interval dilation, prompting surgical repair. Specifically, aneurysm diameter increased by over 5 mm from baseline imaging in both patients (Figure 3). Histology showed non-necrotizing granulomatous inflammation in both cases (Figures 4 and 5).
Summary of patient information.
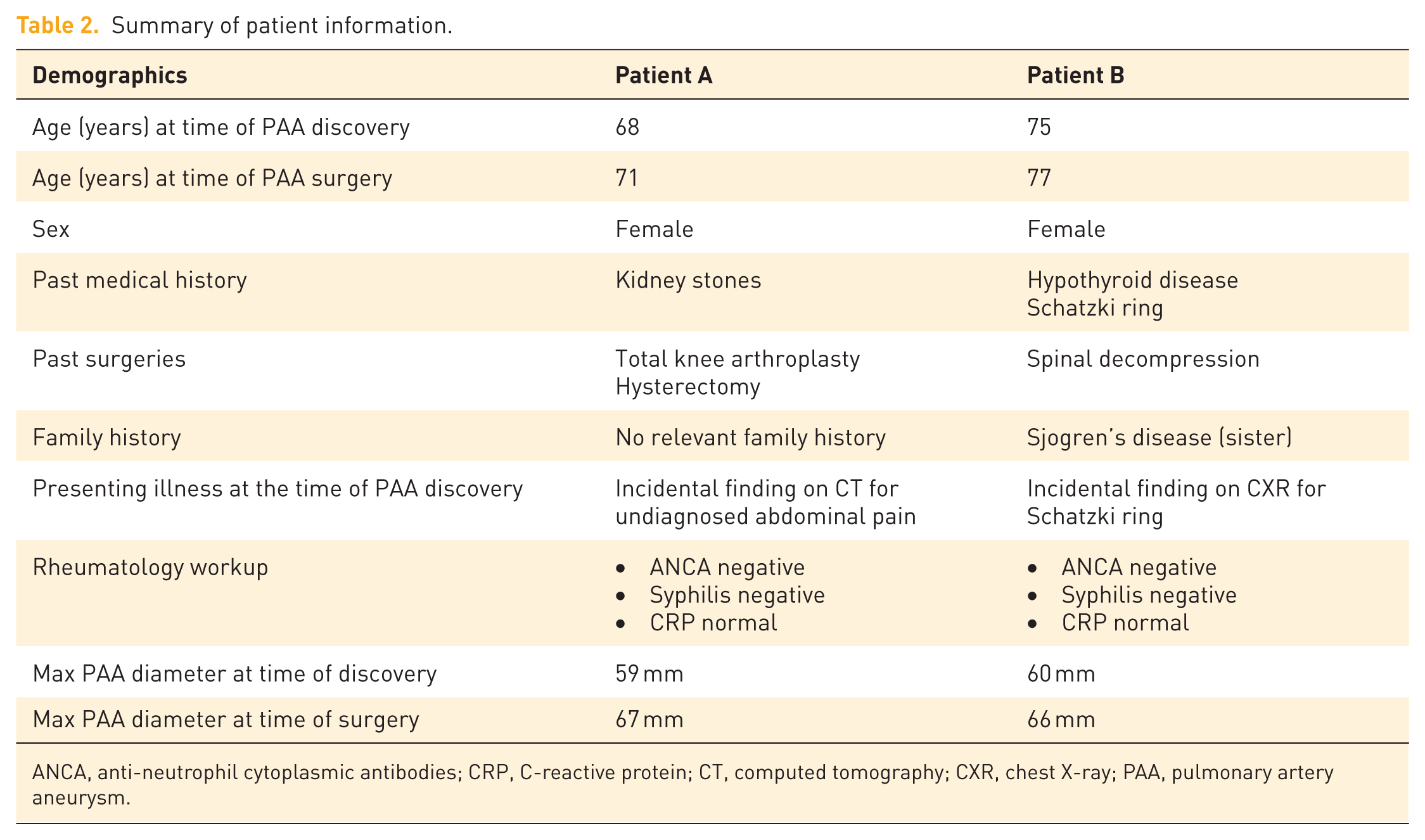
ANCA, anti-neutrophil cytoplasmic antibodies; CRP, C-reactive protein; CT, computed tomography; CXR, chest X-ray; PAA, pulmonary artery aneurysm.

Patient A imaging before and after PAA repair. (a, b) Coronal slices of Patient A’s thorax at the level of maximum PAA diameter, before (a) and after PAA repair (b). (c, d) Axial slices at the level of maximum PAA diameter, before (c) and after PAA repair (d). Red double-headed arrow is 67 mm; green double-headed arrow is 26 mm. Images were obtained using a contrast-enhanced CT thorax protocol (ECG-gated).

Patient B imaging before PAA repair. (a) Coronal slice of Patient B’s thorax at the level of maximum PAA diameter. (b) Axial slice at the level of maximum PAA diameter. Red double-headed arrow is 66 mm. Images were obtained from the same contrast-enhanced CT, thorax protocol (ECG-gated).

Growth of pulmonary artery aneurysms. Scatter plot showing the growth in diameter of PAAs over time. The Y-axis represents the maximum PAA diameter in millimeters, as measured on contrast CT scans. The X-axis shows the number of months preceding surgical repair of the PAA. The green line represents the 90th percentile of female pulmonary artery size, 27 mm, as per the Framingham Heart Study. Values for the PAA diameter are shown next to their respective data points for clarity.

Pathology of Patient A’s pulmonary artery aneurysm. (a) Elastic stain of PA section showing loss of structure of elastic fibers within the tunica media. TM, TA, and TI are identified. Original magnification ×20. (b) Gross specimen of a PA fragment, showing irregular intimal thickening. (c) H&E stain of PA section showing transmural non-necrotizing granulomatous inflammation. Original magnification ×20. (d) Enlarged image of the rectangular area at left, showing giant cells (e.g., GC) and many epithelial histiocytes. Original magnification ×100.
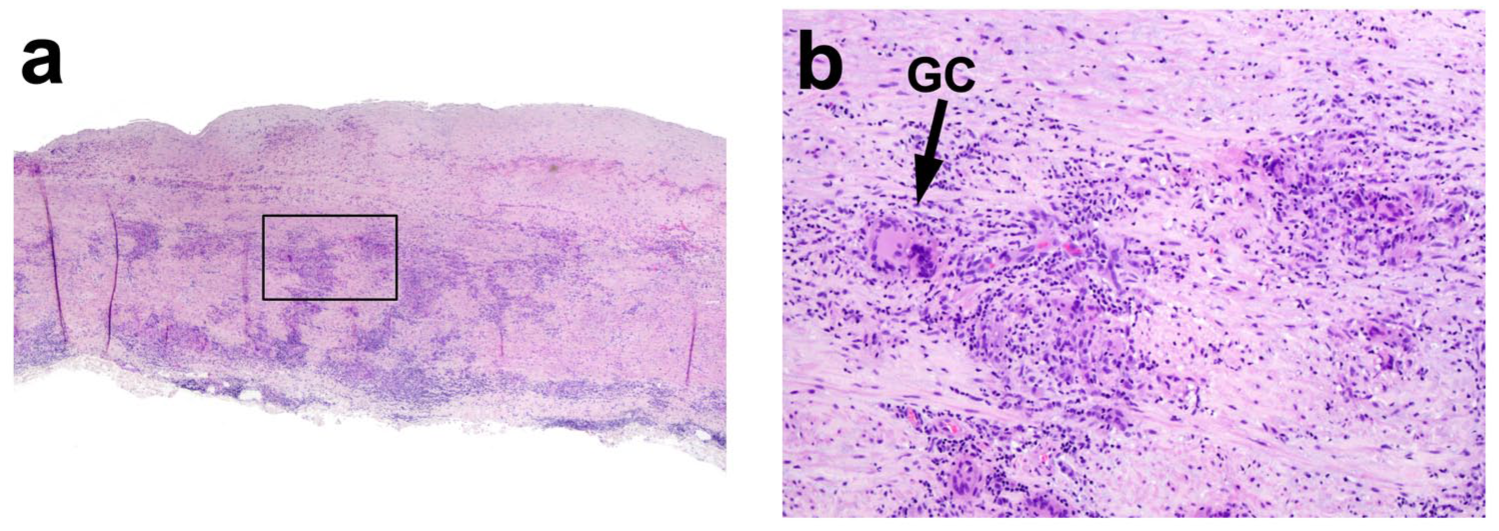
Pathology of patient B’s pulmonary artery aneurysm. (a) H&E stain of PA section showing transmural non-necrotizing granulomatous inflammation. Original magnification ×20. (b) Enlarged image of the rectangular area at left, showing giant cells (e.g., GC) and many epithelial histiocytes. Original magnification ×100.
Patient A
Patient A was a 68-year-old female at the time of PAA discovery and 71 at the time of PAA repair. A CT chest-abdomen-pelvis for unrelated abdominal pain incidentally revealed a PAA 59 mm in diameter. There was no history of autoimmune disease. Eventually, the aneurysm’s diameter increased to 67 mm. Furthermore, the right PA was 34 mm in diameter (average = 25 mm). 3 Given the increase in size of the main PAA, the decision was made for surgical repair.
Patient A’s examination was unremarkable. There were no symptoms of GCA. At the time of aneurysm discovery, her systolic blood pressure averaged below 140/90 mmHg and her BMI was 23 kg/m2. Investigations were also unremarkable, with an echocardiogram showing only mild pulmonary valve regurgitation. Infectious panels, including syphilis, were negative. Erythrocyte Sedimentation Rate (ESR) and C-Reactive Protein (CRP) were within normal limits. Postoperatively, there continued to be no evidence of systemic vasculitis, so no immunosuppressive therapy was initiated.
Patient A was admitted to the Queen Elizabeth II Health Sciences Centre in Halifax, Nova Scotia, Canada, in June 2021 for elective PAA repair. Her PAA was replaced with a 26 mm Dacron graft via a median sternotomy on cardiopulmonary bypass. While the right PA appeared enlarged (34 mm) and ectatic, it was not replaced owing to the increased surgical complexity. Her postoperative course was uncomplicated.
Two years following her operation, Patient A was doing well clinically with no shortness of breath, chest pain, headache, or symptoms of GCA. However, imaging follow-up showed that her right PA remained enlarged. Her left PA had increased in size from 18 to 25 mm (2 years postoperatively), suggesting possible disease progression. There is currently no plan for surgical reintervention.
Patient B
Patient B was a 75-year-old female at the time of PAA discovery and 77 at the time of PAA repair. During workup to assess her Schatzki ring, a CT scan revealed a main PAA (diameter 60 mm). There was no personal history of autoimmune disease, infection, or rheumatologic condition.
Patient B’s examination was unremarkable. Her BMI was 22 kg/m2, and her BP was 140/72 mmHg. There were no findings of GCA or labs suspicious for inflammatory vasculitis. Infectious panels, including syphilis and CRP, were negative. An echocardiogram revealed only mild mitral regurgitation. While initial interval imaging showed the PAA was growing very slowly (60–62 mm), several months later, follow-up imaging revealed the diameter of the aneurysm to have grown from 62 to 66 mm (6 mm greater than the baseline of 60 mm), prompting surgical intervention. Patient B was admitted to the Queen Elizabeth II Health Sciences Centre in Halifax, Nova Scotia, Canada, in October 2023 for elective PAA repair. The PAA was replaced with a 26 mm Dacron graft similarly to Patient A. The postoperative course was uncomplicated. Two years following her operation, the appearance of the graft, pulmonary trunk, and branch pulmonary arteries has remained stable.
Pathology
The pathology of the PAA tissue was similar for both patients (Figures 4 and 5). Gross specimens of the PA wall demonstrated a classic “tree-bark” texture, reflecting irregular intimal thickening. 17 Light microscopic examination of the PA wall showed transmural non-necrotizing granulomatous inflammation, with destruction of the medial elastic fibers providing a mechanism for aneurysm formation. There were no identifiable microorganisms with tissue special stains (Ziehl–Neelsen and Grocott–Gömöri’s methenamine silver). The pathology we observed was distinct from other large vessel vasculitides; Behçet disease typically lacks giant cells 18 and Takayasu arteritis is typified by very extensive fibrosis and inflammation at the tunica adventitia in particular. 19
Discussion
The most striking clinical feature of these two aneurysms is that they were silent: Patient B had a first-degree relative with Sjogren disease, but otherwise there were no identified risk factors for arteritis in either case. Moreover, there were no relevant symptoms or lab abnormalities. We posit that these presentations are consistent with the new diagnostic entity of clinically isolated pulmonary arteritis (CIPA). A major challenge with this diagnosis is that it is difficult to counsel patients with regard to the utility and timing of surgical intervention.
To improve patient counseling in cases of CIPA, it would be beneficial to have standardized metrics to guide surgical intervention in this population. For instance, it would be very useful to quantify the size of a patient’s aneurysm relative to a population average. Unfortunately, there is no standardized measurement of PA diameter. Much of the literature cites the Framingham study values, which only utilized a single CT slice (per patient) for quantifying average PA diameter. More recent work strongly suggests the Framingham study underestimated average PA size by approximately 7 mm. We suggest adopting these recommendations from Berger et al., who posit that any main PA diameter below 45 mm would represent an ectasia, rather than an aneurysm. 3
With regard to counseling patients on whether watchful waiting or surgical resection of their asymptomatic aneurysm is more appropriate, there is no consensus guideline. That said, the majority of literature is in alignment with the recommendations of Kreibich et al. 2 In asymptomatic patients, such as our two cases, we agree it is reasonable to repeat the CT every 6 months to establish growth rate, then increase the imaging interval if the aneurysm is stable in size. The larger estimates for mean PA diameter (as discussed above) are aligned with an initial approach of watchful waiting, especially when aneurysms are less than 55 mm and stable over time. 2 By contrast, if asymptomatic PAAs are larger than 55 mm in diameter or grow more than 5 mm in 6 months, intervention should be considered.
Strengths and limitations
The strength of this series is that it confirms the existence of CIPA, which has been reported in the literature, however, with non-standardized terminology. Our main limitation includes a lack of a comparator of patients who were managed without surgery with alternative pharmacotherapy; as such, we cannot make strong recommendations on the management of patients with CIPA. (The availability of a tissue diagnosis was predicated on surgical intervention in the cases we reviewed.) We hope this report will help standardize case descriptions of CIPA, and our experience with favorable outcomes will guide further management of this condition.
Conclusion
We present a rare pair of cases featuring clinically occult PAAs. For several years, there have been an increasing number of similar reports, though they have yet to be united under one diagnostic framework. Our case series brings the total to nine documented cases of this emerging clinical entity: clinically isolated pulmonary arteritis with granulomatous patterns of inflammation. The management of our cases consisted of vigilance and surgical replacement of the main PA upon interval dilation of the aneurysm. Both patients tolerated the intervention well. We did not use corticosteroids postoperatively as there was no lab evidence of systemic inflammation. We hope this report will help guide the management of similar cases in the future.
Supplemental Material
sj-docx-1-tak-10.1177_17539447251410305 – Supplemental material for A rare pair: two cases of clinically isolated pulmonary artery aneurysm
Supplemental material, sj-docx-1-tak-10.1177_17539447251410305 for A rare pair: two cases of clinically isolated pulmonary artery aneurysm by Donovan Cassidy-Nolan, Claudia Cote, Dominique de Waard, Mathieu Castonguay, Volodko Bakowsky and Christine Herman in Therapeutic Advances in Cardiovascular Disease
Footnotes
Acknowledgements
First, we acknowledge and thank the patients and their families for their generous participation in this research process; it is our privilege to work with them. We would also like to acknowledge the support of the administrative and education team at Dalhousie University Division of Cardiac Surgery for all their support with this project.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
