Abstract
Liver injury in Takayasu arteritis (TA) is a rare phenomenon. Most symptoms are nonspecific, and the exact pathogenesis remains to be elucidated. Early diagnosis and new treatment methods are important for an improved prognosis. A summary of the clinical information and mechanistic analyses may contribute to making an early diagnosis and development of new treatment methods. A PubMed search was conducted using the specific key words “Takayasu arteritis” and “liver” or “hepatitis” or “hepatic”. Symptoms and treatment of TA with an accompanying liver injury were reviewed retrospectively. Many factors are presumed to be involved in the mechanism of TA with liver injury, including the immune response, genes, infections, and gut microbiota. There are several lines of evidence indicating that immune dysfunction is the main pathogenic factor that triggers granuloma formation in TA patients. However, the role of genetics and infections has not been fully confirmed. Recently, the gut microbiota has emerged as an essential component in the process. We reviewed in detail the current concepts that support the complex pathogenesis of TA accompanied by liver injury, and we presented recent theories from the literature. Finally, we discussed future research directions of liver injury in TA.
Introduction
Takayasu arteritis (TA) is a chronic nonspecific rare granulomatous vasculitis that predominantly occurs in the aorta and its main branches, such as carotid and vertebral arteries. Chronic inflammation may induce intimal hyperplasia and medial and intimal fibrosis, resulting in luminal stenosis. 1 Although the disease has a worldwide distribution, it is generally thought to be more common among Asian populations; the prevalence of TA is estimated to be 40 per million in Japan and 0.9 per million in US, and these variations may be derived from geographical and genetic differences between the populations. 2 TA is associated with a profound inflammatory process, which is possibly associated with an autoimmune disorder, but its precise etiology remains largely unknown. 3 The clinical manifestations of TA are nonspecific and mainly depend on the type of lesion and the degree of inflammation. 4 The 1990 American College of Rheumatology diagnosis criteria indicate that the lack of specific signs makes diagnosing this condition extremely challenging and that the disease activity is difficult to evaluate. Thus, early diagnosis and treatment of TA are a challenge, and the disease usually remains active throughout its course, leading to continuous damage accumulation and short- and long-term morbidity and mortality.1,5
Current treatment strategies for TA mainly involve the use of corticosteroids. However, only a small number of patients are completely cured, and a sizable subset of patients relapse after treatment is gradually tapered. 6 Thus, these relapsed patients require additional treatment options. 7 Methotrexate (MTX) is an immunosuppressant that is most commonly used in these patients, and it has been reported to be a promising treatment option to achieve and maintain remission in refractory TA patients. 8 Other drugs, such as leflunomide, have also been reported to cause quick induction and sustained remission of TA, especially in refractory cases, and therefore, these drugs should be considered to be an alternative treatment for TA. 9 Additional biotherapeutic agents, such as tumor necrosis factor (TNF) inhibitors, have recently been approved for TA treatment. However, neither provide much more benefit than corticosteroids.10,11 Therefore, early diagnosis and new treatment methods are important for improving patient prognosis. However, some TA patients initially present with liver injury, making an early diagnosis difficult. The purpose of this review was to investigate the clinical features and pathogenesis of liver injury in patients with TA.
Methods
A literature review was performed by searching PubMed using the key terms “Takayasu arteritis” and “liver” or “hepatitis” or “hepatic”. We identified five cases of abnormal liver function in TA (Table 1).12–15
TA patients with accompanying liver injury.

TA, Takayasu arteritis; AST, aspartate transferase; ALT, alanine transaminase; ALP, alkaline phosphatase; GGT, γ-glutamyltransferase; NA, details not available.
Clinical course
The five cases in these four reports12–15 showed that in TA patients with liver injury, the most common complaints were nonspecific symptoms, such as fatigue, intermittent fever, headache, weight loss, anorexia, nausea, and abdominal pain. The diagnosis was established through a combination of symptoms, physical examination, imaging results that indicated vasculitis, and liver biopsy.
Pathogenesis of liver injury in TA
The mechanism of liver injury in TA has never been discussed. The basic pathological feature of TA is granulomatous inflammation (Figure 1). It has been reported that the etiology of hepatic granulomas, including vasculitis and hepatic granuloma disease, could lead to biochemical abnormalities in the liver.16,17 Thus, we conclude that TA causes liver injury via the formation of granulomas (Figure 2).

Liver pathology in a patient with TA. Most of the mononuclear cells have infiltrated, and granulomas of different sizes were localized in the lobules. Multiple epithelioid cells aggregated in the granuloma, and a few mononuclear cells were observed in the granuloma. There was no fibrous envelope around the granuloma, and acid-fast staining was negative.

Possible mechanisms of liver injury in TA.
Immune component
The exact pathogenic sequence of granuloma formation in TA is not completely understood, and functional dysregulation resulting from the immunological alterations may contribute to the formation of granulomas in TA. The immune response to pathogen exposure has been considered to be a potential etiological factor (Figure 3).

Schematic representation of the possible mechanism of granuloma formation in Takayasu’s arteritis. TA lesions are mostly generated in the adventitia through the vasa vasorum. Different antigens such as infection stimuli trigger the expression of 65 kDa HSP, which initiates an inflammatory response. The expression of 65 kDa HSP in aortic tissue may contribute to the activation of dendritic cells. Dendritic cells expressing specific HLA molecules (Glu63 and Ser67) in the adventitia in response to a stimulus have not been identified. Dendritic cells activate T cells through Toll-like receptors. After interacting with dendritic cells, T cells increase activate macrophages and release IFN-γ and TNF-α, promoting prolonged multinucleated giant cell activation. Multinucleated giant cells cause granuloma formation, ultimately leading to tissue damage. Activated macrophage release IL-6, IL-23, VEGF (leading to neovascularization), and PDGF (which results in intimal proliferation). Th17 cells induced by IL-23 attract and activate neutrophils in the vessel wall, causing vascular lesions. IL-6 and IL-9, which likely contribute to vessel wall damage. γδ T cells and NK cells expressing the immune receptor NKG2D recognize the stress ligand MICA that is expressed on vascular smooth muscle, leading to damage in the intima layer. CD8+ T cells may also cause necrosis through the release of perforin. By activating endothelial cells and inducing complement-mediated cytotoxicity, AAECA may also contribute to the pathogenesis of TA.
Different studies have suggested that mechanisms that are mediated by humoral immunity and cell immunity are strongly implicated in inflammation and tissue damage in TA. 18 T cells were reported to be present in greater amounts in the aorta of TA patients, and the T cells were associated with histological disease activity and with prednisone use in TA. 19 The decrease in the number of T regulatory cells (Treg) cells, which is a T cell subset that is needed to maintain immune homeostasis, further indicates that the T cell-mediated immune response is involved. 20 In addition, other immune cell types, such as macrophages and natural killer (NK) cells, also infiltrate the tissue in TA. 19 Seko et al. 21 revealed that major histocompatibility class I chain-related A (MICA), which is induced by external stress and binds to NKG2D receptors on infiltrating cells, was increased in aortic tissue of patients with TA. Moreover, it has been proposed that the differential release of proinflammatory cytokines such as interleukin (IL)-23 may also have a substantial influence on TA pathogenesis. 22 Serum concentrations of TNF-α, IL-6, IL-9, IL-18, and IL-17 in patients with TA are frequently observed to be higher than those in healthy people.23,24 The IL-6 concentration is increased in the peripheral blood and aortic tissues of TA patients. 25 IL-6 has been proposed to induce fibrogenesis of aortic adventitial fibroblasts via the JAK2/STAT3 and JAK2/Akt pathways. 26 The effective treatment of refractory TA with tocilizumab, an anti-IL-6 receptor monoclonal antibody, further confirms the involvement of IL-6 in TA. 27
Moreover, although B lymphocytes are absent in TA lesions, the number and frequency of peripheral blood CD19(+)/CD20(−)/CD27(high) antibody-secreting cells (B cells) have been reported to be significantly higher in patients with active TA than in healthy donors; three patients with active refractory TA and expansion of plasmablasts were successfully treated with B cell depletion therapy. 28 Zanwar et al. 29 reported serum levels of B cell survival factors activation factor (BAFF) and A proliferation inducing ligand (APRIL) correlated with disease activity (Indian Takayasu Clinical Activity Score-ESR) or the TA damage score, suggesting a probable role for B cells in TA pathogenesis. Additionally, Mutoh et al. 30 reported that a 34-year-old man with TA who was refractory to four biologic agents had been successfully treated with an anti-CD20 monoclonal antibody (rituximab), and the outcome supports the hypothesis that B cell depletion therapy can be a useful option for refractory TA.
Genetic component
In addition to immune function studies, genome-wide association studies have revealed the heritability of TA in the human leukocyte antigen (HLA) region, and common susceptibility factors have also been reported in response-regulated genes. 31
The HLA region, a 3.6-Mb segment of the human genome at 6p21, has been associated with more than 100 diseases and primarily with autoimmune diseases. 32 Kitamura et al. 33 showed that the HLA-B52 molecule and the B39 molecule could contribute to different clinical manifestations by binding different antigenic peptides to cause inflammation. Thus, it is suggested that the HLA-B molecule may play an important role in pathogenesis or determining clinical manifestations of TA. 33 Another meta-analysis also demonstrated that the HLA-B*52 allele may significantly contribute to TA susceptibility. 34 Glu and Ser at positions 63 and 67, which have been identified as essential amino acid residues in the HLA-B molecule, are also thought to determine the patient’s susceptibility to TA. 35 Moreover, patients with HLA-B52 in Turkey seem to have more severe left ventricular involvement than those without it, further indicating a possible relationship between TA and HLA-B52. 36 Umemura and coworkers 37 revealed that KIR/HLA genotypes confer susceptibility and progression in patients with autoimmune hepatitis. In another study, the HLA-B*27 subtype was reported to specifically determine targeting and viral evolution of a hepatitis C virus-specific CD8+ T cell epitope. 38
Outside the HLA region, several genes that are involved in regulating the immune response have been associated with TA. Renauer et al. 39 identified novel genetic susceptibility loci for TA with a genome-wide level of significance in IL-6 (rs2069837). The role of IL-6 gene polymorphisms was also considered to be associated with susceptibility to hepatitis in a previous study. 40 IL-1 family member 10, which was recently renamed IL-38, exerts anti-inflammatory effects by effectively inhibiting inflammatory mediator production (IL-17A and IL-22).41,42 Afzal et al. 43 also revealed that different IL-10 gene polymorphisms may lead to an imbalance between the proinflammatory and anti-inflammatory cytokine responses, which may influence the development of hepatitis. Nakajima et al. 44 reported the rs6871626 single nucleotide polymorphism in IL-12B may cause increased IL-12p40 and IL-12p70 expression. These enhanced cytokines might play roles in TA pathophysiology. 44 In Chinese Han hemodialysis patients, IL-12B polymorphisms were reported to potentially contribute to hepatitis C susceptibility. 45 Therefore, the involvement of these genes and single nucleotide polymorphisms might be important in the pathogenesis of liver injury in TA.
Infection
Compelling evidence strongly suggests that infections are significantly involved in the development of TA. Some authors have suggested that infection by several types of microorganisms, such as Mycobacterium tuberculosis and Chlamydia trachomatis, could be involved in TA pathogenesis, but supporting evidence remains inconclusive. In clinical studies, Zhang et al. 46 demonstrated that the percentage of tuberculosis (TB) history in patients with giant cell arteritis was higher than that in the Chinese general population. IS6110 (which is used to identify the M. tuberculosis complex) and HupB (which is used to establish the differences between M. tuberculosis and Mycobacterium bovis) genes have been reported have a higher frequency in aortic tissues of TA patients. 47 Patients with multidrug-resistant TB are generally considered to be at risk of hepatitis C infection. 48 Additionally, C. trachomatis has been investigated in association with Cogan’s syndrome (CS) and TA, but in most cases, this link remains hypothetical.49,50 Sarcoidosis, which is a systemic granulomatous disease of unknown etiology that can affect any organ, was also recently reported to be concomitantly developed with TA, but it is not clear whether these findings are typical or a result of chance alone.51,52 Hepatic granulomas are a common finding in systemic sarcoidosis, but most patients remain asymptomatic. 53 Fetzer et al. 54 revealed that sarcoidosis may present as liver dysfunction, cirrhosis, or portal hypertension. Sarcoid-related liver disease may progress and can manifest without, alongside, or significantly after a diagnosis of pulmonary disease. 54 Thus, although many infectious diseases may lead to liver injury in TA, the evidence remains insufficient and further research is required in the future.
Gut microbiota
Gut microbiota is involved in immune system development. The gut microbiota has been associated with a variety of human diseases, such as autoimmune encephalomyelitis and cancer.55,56
An accumulating amount of evidence has shown that gut microbiota plays an important role in the development of autoimmune diseases such as autoimmune lupus.57,58 The composition and function of gut microbiota have been reported to play an important role in the process of inflammatory bowel disease, 59 and Kawasaki disease, which is vasculitis of the small- and medium-sized vessels, has been reported to be associated with the HLA-B genotype. 60 Esposito et al. 61 reported that a higher number of Gram-positive cocci (genera Streptococcus and Staphylococcus), Eubacterium, Peptostreptococcus, and HSP60-producing Gram-negative microbes have been found in stool samples from children with Kawasaki disease. Ye et al. 62 revealed a distinct gut microbiome signature in Vogt–Koyanagi–Harada patients. Vogt–Koyanagi–Harada disease is a multisystemic autoimmune disorder that is characterized by granulomatous panuveitis, and it is associated with genotype HLA-DRA. Ye et al. 62 showed an exacerbating effect of this gut microbiome on experimental autoimmune uveitis. Various microorganisms (both bacteria and viruses) have been detected in giant cell arteritis patients even in normal aortic tissues, suggesting that blood vessels may not be sterile but are inhabited by their own unique microbial ecosystem. 63 Additionally, a wide variety of bacteria, including oral bacteria, was found to colonize aortic aneurysms. 64 Therefore, vascular dysbiosis may also be involved in the development of TA.
Numerous studies suggest that alterations in human microbiota may be involved in the pathogenesis of vasculitides as a result of the aberrant activation of the innate and adaptive immune responses. 65 Akiyama et al. 66 thought that gut microbiota may be associated with HLA-B52-positive TA. In another study, gut microbiota was considered to be a new insight that was related to TA pathogenesis and that may contribute to the design of novel therapeutic approaches. 67
Thus, several studies have shown that the changes in gut microbiota may be related to the pathogenesis of vasculitis such as TA. However, research in this area is rare, and further research is needed to better understand the impact of microbiota dysbiosis on TA progression, and to develop prevention or treatment strategies.
Discussion
Currently, treatment of granulomatous liver disease (which is defined by the histologic findings on liver biopsy) depends on the underlying diagnosis, and it is still associated with many problems. 68 Kumar et al. 53 revealed that corticosteroid therapy may improve symptoms and liver chemistry but not alter the long-term outcome of hepatic sarcoidosis. Treatment of hepatic granulomata that is caused by primary biliary cirrhosis currently includes licensed therapies (ursodeoxycholic acid and obeticholic acid) and off-label therapies (fibric acid derivatives, budesonide). 69 These treatment methods can significantly improve liver function, after which the liver may produce bile acids that could regulate gut microbiota through activate farnesoid X receptor (FXR) via the gut–liver axis (Figure 4). 70 Glucocorticoids remain the major treatment method in patients with TA, but this therapy leads to many complications and some patients relapse and require immunosuppressive agents. 7 Thus, there is an urgent need to better understand the mechanisms of liver injury in TA to develop novel and effective therapeutic strategies.
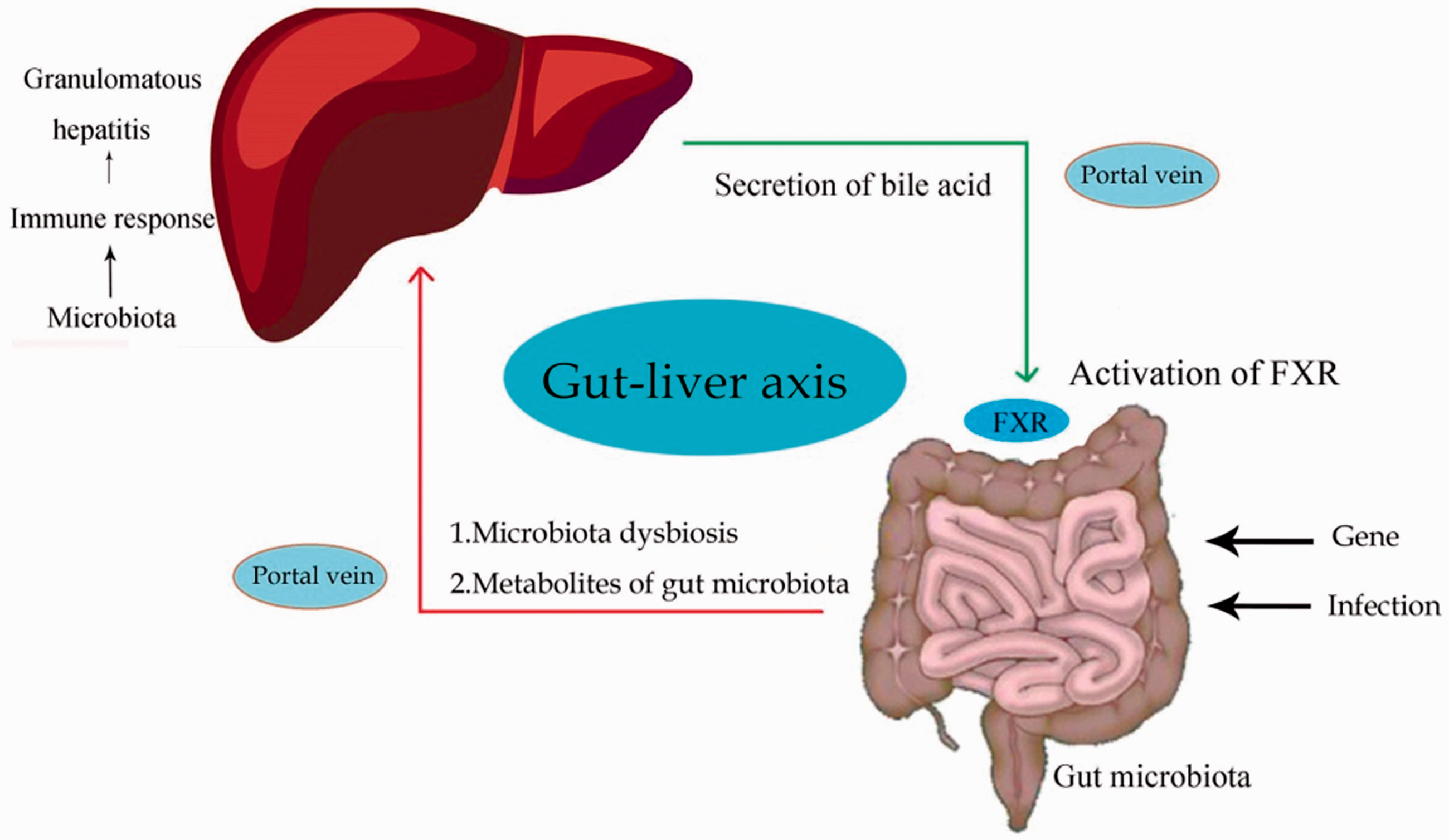
Schematic depicting the potential interaction between gut microbiota and the liver. The composition and function of gut microbiota may be influenced by different factors such as genes and infection. After the onset of gut dysbiosis, the gut microbiota and their products may reach the liver through the portal circulation. Then, the gut microbiota causes an immune response in the liver and formation of granulomatous hepatitis. When liver function recovers to normal, the liver can regulate the gut microbiota composition by secretion of bile acid through activation of the farnesoid X receptor (FXR).
In this review, we summarized the clinical information and pathological features of liver injury in TA. Recognition of the importance of the immune response as a central role of liver injury in TA indicates that anti-inflammatory agents are required for treatment. Inflammation in liver injury can be treated with anti-inflammatory drugs such as prednisone. Genetic data support the role of genes throughout the course of TA and suggest that individualized treatment strategies are needed for future treatment of TA liver injury. During sarcoidosis, microbes or their products may trigger the immune response leading to sarcoid granuloma formation. 71 The human microbiota was also reported to play an important role in pathogen colonization resistance, and it has been proposed to play a potential role in TB granuloma formation. 72 In addition, fecal microbiota transplantation, which is a novel method to manipulate gut microbiota, was reported to be an effective therapy for inflammatory bowel disease and recurrent Clostridium difficile infection.73,74 In patients with TA that was caused by gut microbiota dysbiosis, the use of fecal microflora transplantation to manipulate gut microbiota may become a new treatment method. Further study such as using high throughput sequencing on stool samples from TA patients is needed to clarify TA pathogenesis and to find novel treatment methods.
Footnotes
Authors contributions
KK wrote the manuscript. BC critically revised the manuscript. YS and YLL conceived the study. All authors approved the final manuscript.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
