Abstract
Background:
Inflammation significantly impacts disease progression and prognosis in acute pulmonary embolism (APE); however, the optimal method to quantify this inflammation for prognostic purposes remains unclear.
Objectives:
We aimed to identify the most effective blood-based inflammatory marker for predicting short-term mortality in patients with APE.
Methods:
We retrospectively analyzed 301 patients with APE. We compared the predictive performance of nine inflammatory markers for 30-day mortality, focusing on the inflammatory burden index (IBI), and contrasted it with the standard clinical score (sPESI).
Results:
After adjusting for potential confounders, a higher IBI was strongly and independently associated with an increased risk of 30-day mortality. IBI demonstrated superior predictive ability compared with all other inflammatory markers and the sPESI score. The relationship between IBI and mortality risk was linear; higher IBI values corresponded to greater mortality risk.
Conclusion:
IBI is a robust and independent predictor of short-term survival in patients with APE. Calculated from routine blood tests (C-reactive protein, neutrophil, and lymphocyte counts), it offers a simple, rapid, and cost-effective tool that may enhance early risk stratification and guide personalized treatment decisions in clinical practice. Further validation in independent cohorts is required to confirm its prognostic utility.
Keywords
Introduction
Acute pulmonary embolism (APE) is a life-threatening cardiovascular emergency associated with high mortality, particularly within the first 30 days after onset.1–3 Accurate prognostic assessment is essential for the identification of high-risk patients. Although current guideline-recommended tools that rely on imaging, laboratory tests, and various scoring systems are valuable, they are often complex and resource-intensive, underscoring the need for simpler and more cost-effective prognostic indices.4,5
Systemic inflammation plays a key role in the pathophysiology and progression of APE.6–9 This observation has prompted growing interest in hematologic inflammatory markers. 10 C-reactive protein (CRP), an acute-phase protein synthesized by the liver, reflects the magnitude of the inflammatory response and has been consistently associated with mortality across conditions ranging from heart failure to severe infections.11,12 Conversely, lymphocytes play a central role in adaptive immunity and immunoregulation. Lymphopenia, a hallmark of physiological stress and poor nutritional status, has been identified as a strong predictor of adverse outcomes in various patient populations, including those with cardiovascular disease and critical illness.13,14 Integration of these markers into composite indices, such as the C-reactive protein-to-albumin ratio (CAR) and the prognostic nutritional index, has enhanced risk stratification in diverse clinical settings,12,15 demonstrating the synergistic advantage of combining markers of inflammation and immune-nutritional status. This growing body of evidence supports further investigation of integrative inflammatory indices in APE. Recently, the inflammatory burden index (IBI) has emerged as a promising prognostic biomarker in oncology. 16 Its formula (CRP × neutrophil/lymphocyte) is designed to reflect, in a more holistic manner, the balance between pro-inflammatory (CRP, neutrophils) and anti-inflammatory (lymphocytes) components. 17 Conceptually, the IBI offers distinct advantages over existing inflammatory markers. Unlike the simplified pulmonary embolism severity index (sPESI), a clinical composite score, the IBI is a continuous and objective laboratory measure that directly quantifies systemic inflammatory burden—a central factor in APE pathogenesis.16–19 This design captures the interplay between pro-inflammatory forces (CRP, neutrophils) and anti-inflammatory capacity (lymphocytes) more comprehensively than two-parameter ratios such as the neutrophil-to-lymphocyte ratio (NLR) or CAR.15,17,20 By integrating the humoral inflammatory response (CRP) with the cellular immune component (NLR), the IBI provides a conceptually more comprehensive assessment of the inflammatory burden.12,16,20 In practice, this approach may offer greater sensitivity for identifying high-risk patients who might otherwise be misclassified by less integrative markers.
However, the prognostic utility of the IBI in APE remains unexplored. Therefore, this study aimed to systematically evaluate and compare the prognostic performance of nine composite inflammatory markers in predicting short-term mortality in APE. We hypothesized that the IBI would outperform other markers and the sPESI, providing a novel approach to risk stratification.
Study design and methodology
Study design
This study was a single-center, retrospective cohort investigation conducted at the Affiliated Yongchuan Hospital of Chongqing Medical University.
Study population
Patients diagnosed with APE and hospitalized for treatment between January 2018 and December 2024 were extracted from the database of the Information Center, the Affiliated Yongchuan Hospital of Chongqing Medical University. All data were anonymized during extraction in compliance with national and institutional regulations. The data presented consist solely of aggregated, anonymized results from which no individual participants can be identified. Diagnosis was established according to the 2019 European Society of Cardiology guidelines for the diagnosis and management of APE. Patients were categorized into two groups—the death group and the survival group—according to whether death due to APE occurred within 30 days. Inclusion criteria were as follows: (1) confirmed diagnosis of APE; (2) complete clinical examination and medical history; and (3) age greater than 18 years. Exclusion criteria included the following: (1) use of anticoagulant therapy within 4 weeks; (2) contraindications to anticoagulation or thrombolysis; (3) chronic pulmonary embolism without new-onset APE; (4) hematologic disorders, severe hepatic or renal insufficiency, or severe infection; and (5) pregnancy or lactation. These exclusion criteria were applied to minimize potential confounding from conditions that independently affect inflammatory markers or survival, and to ensure data completeness for accurate analysis. Nevertheless, this approach may have introduced selection bias by excluding critically ill patients with complex comorbidities, potentially limiting the generalizability of the findings to broader APE populations. Figure 1 illustrates the selection process for the study cohort.

Study cohort selection process.
Data acquisition
Data on age, sex, and comorbidities (chronic lung disease, heart failure, malignancy, lower extremity venous thrombosis, diabetes, prior surgery, and cerebrovascular disease) were extracted. Initial routine blood results (neutrophil, lymphocyte, platelet counts, etc.), biochemical indices, and coagulation parameters were obtained after symptom onset. Vital signs, including arterial blood gas parameters, respiratory rate, heart rate, and systolic blood pressure, were recorded at presentation. All measurements were obtained prior to the initiation of APE treatment.
sPESI scoring criteria were as follows: (1) age >80 years; (2) presence of malignancy; (3) chronic cardiac insufficiency or pulmonary disease; (4) heart rate ⩾ 110 bpm; (5) systolic blood pressure <100 mmHg; and (6) arterial oxygen saturation (SaO2) <90%. Each criterion contributes one point, resulting in a total score ranging from 0 to 6. 4
Nine composite inflammatory markers were calculated 16 :
① CAR = C-reactive protein (mg/L)/albumin (g/L)
② CALLY = [Albumin (g/L) × Lymphocyte Count (×109/L)]/[10 × CRP (mg/L)]
③ PAR = Platelet (×109/L)/albumin (g/L)
④ NAR = Neutrophil (×109/L)/albumin (g/L)
⑤ LCR = Lymphocyte (×109/L)/C-reactive protein (mg/L)
⑥ PLR = Platelet (×109/L)/lymphocyte (×109/L)
⑦ NLR = Neutrophil (×109/L)/lymphocyte (×109/L)
⑧ SII = Platelet (×109/L) × neutrophil (×109/L)/lymphocyte (×109/L)
⑨ IBI = C-reactive protein (mg/L) × neutrophil (×109/L)/lymphocyte (×109/L)
Statistical analyses
Normality of continuous variables was assessed using the Kolmogorov–Smirnov test. Variables with a normal distribution were expressed as mean ± standard deviation, whereas non-normally distributed data were reported as median (interquartile range, IQR). Independent t-tests or Mann–Whitney U tests were applied according to data distribution. Chi-squared tests were used to compare categorical variables. For Cox regression analysis, continuous variables with right-skewed distributions and large ranges (e.g., IBI, platelet-to-lymphocyte ratio (PLR)) were log10-transformed to mitigate extreme values.21,22 Confounders were selected for adjustment based on clinical relevance (e.g., age, sex) and significant univariable association with the outcome (p < 0.05 in Table 1). Multicollinearity among potential covariates was assessed, and variables with a variance inflation factor (VIF) ⩾10 were excluded from multivariable models to ensure stability. 23 Three nested Cox proportional hazards models were constructed: Model 1 (unadjusted), Model 2 (adjusted for age and sex), and Model 3 (fully adjusted for clinically relevant and statistically significant variables with low multicollinearity, as detailed in the footnote of Table 2). 24 The proportional hazards assumption for the key variable (LogIBI) and for the overall models was evaluated using Schoenfeld residuals. No significant deviation from the proportional hazards assumption was detected (global test, p > 0.05), supporting the validity of the Cox model results (Figure S1-1 and S1-2). Receiver operating characteristic (ROC) curves were plotted to determine the optimal cutoff value of IBI for predicting short-term clinical outcomes in patients with APE. DeLong’s test was used to compare the area under the ROC curves (AUCs) of IBI with those of other inflammatory markers, including sPESI. Kaplan–Meier survival curves were plotted, and log-rank tests were performed to compare cumulative survival between IBI groups. Restricted cubic spline (RCS) analyses were conducted to examine potential nonlinear associations between LogIBI and 30-day mortality in APE, adjusting for confounders such as age, sex, and comorbidities. Subgroup analyses were performed to assess the influence of potential confounders. All statistical analyses and visualizations were conducted using SPSS (IBM Corp., Armonk, NY, USA) version 26.0, MedCalc (MedCalc Software Ltd., Ostend, Belgium) version 22.0, and RStudio (Posit Software, PBC, Boston, MA, USA) version 4.4.2. p values <0.05 were considered statistically significant. Given the exploratory nature of comparing multiple inflammatory markers, numerous statistical comparisons were performed (e.g., DeLong’s tests for AUC differences). This approach increases the risk of Type I error (false positives) due to multiple testing. Therefore, p values from these comparative analyses (particularly those in Table 4) should be interpreted with caution, and the findings regarded as hypothesis-generating and requiring validation in future studies.
Baseline characteristics of APE.

p < 0.05.
COX regression survival analysis in patients with APE.

Model 1: Unadjusted for confounders. Model 2: Adjusted for age and gender confounders. Model 3: Adjusted for age, gender, erythrocyte count, platelet count, albumin, PAR, D-dimer, fibrinogen, creatinine, troponin, NT-proBNP, blood lactic acid, cerebrovascular disease, surgery, lower extremity venous thrombosis, and diabetes confounders. LogIBI#, LogPLR#, and LogNT-proBNP#, IBI, PLR, and NT-proBNP were treated with a base-10 logarithmic transformation.
β, coefficient; APE, acute pulmonary embolism; CI, confidence interval; COX, proportional hazards model; HR, hazard ratio; IBI, inflammatory burden index; NT-proBNP, the N-terminal pro-brain natriuretic peptide; SE, standard error; sPESI, a simplified version of the pulmonary embolism severity index.
p < 0.05.
Sample size calculation
Due to the retrospective nature of this study using the database of the Information Center, the incidence of missing data was low. We employed a complete-case analysis approach, meaning that only patients with complete data for all variables of interest were included in the final analysis. No data imputation techniques were applied. As this was a retrospective cohort study utilizing all available eligible data from our hospital’s database of the Information Center between January 2018 and December 2024, no a priori sample size calculation was performed. However, a post hoc power analysis was conducted using G*Power 3.1.9.7 to evaluate the statistical power for detecting the observed effect of LogIBI on 30-day mortality. Based on the final cohort of 301 patients (54 events), an odds ratio of 4.86 for LogIBI (approximating the hazard ratio from Cox model 3), an event rate of 17.9%, a significance level of 0.05, and accounting for 19 covariates in the model (R² other X = 0.3), the achieved statistical power was >99.9%. This indicates that the study sample provided sufficient power to detect the observed association (Figure S2-1–S2-3).
This manuscript was prepared in accordance with the STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) guidelines. A completed checklist has been provided as Supplemental Material.
Results
Baseline characteristics of APE
A total of 301 patients with APE were included, comprising 247 (82.1%) in the survival group and 54 (17.9%) in the death group. As summarized in Table 1, non-survivors were generally older and exhibited a higher prevalence of heart failure and chronic lung disease. They also demonstrated greater hemodynamic instability, elevated markers of end-organ damage, and significantly higher inflammatory burdens across all measured parameters compared with survivors. All differences were statistically significant (p < 0.05).
COX regression analysis of composite inflammatory indicators predicting 30-day survival
To ensure model robustness, variables with right-skewed distributions and large differences between their maximum and minimum values (PLR, SII, IBI, NT-proBNP) were log10-transformed. Variables showing multicollinearity (VIF ⩾10), including CALLY, CAR, CRP, LCR, NAR, SII, leukocyte count, hemoglobin, hematocrit, and neutrophil count, were excluded (Table S1). The final multivariable analysis focused on IBI, sPESI, NLR, and PLR. Cox proportional hazards models were constructed with incremental adjustments: Model 1 (unadjusted), Model 2 (adjusted for age and sex), and Model 3 (fully adjusted for clinical and laboratory confounders, as detailed in Table 2). Both IBI and sPESI were identified as independent predictors of 30-day survival across all models.
The AUC of the ROC curve evaluated the predictive performance of composite inflammatory markers
ROC curve analysis demonstrated that all composite inflammatory markers, except PAR, significantly predicted mortality among patients with APE. Among these, IBI exhibited the highest discriminatory power, with an AUC of 0.870 (95% CI: 0.827–0.906) (Table 3, Figure 2). In pairwise comparisons using DeLong’s test (unadjusted for multiple testing), IBI significantly outperformed all other inflammatory markers and the sPESI score (Table 4).
Region beneath the ROC curve for nine composite inflammatory markers and sPESI.
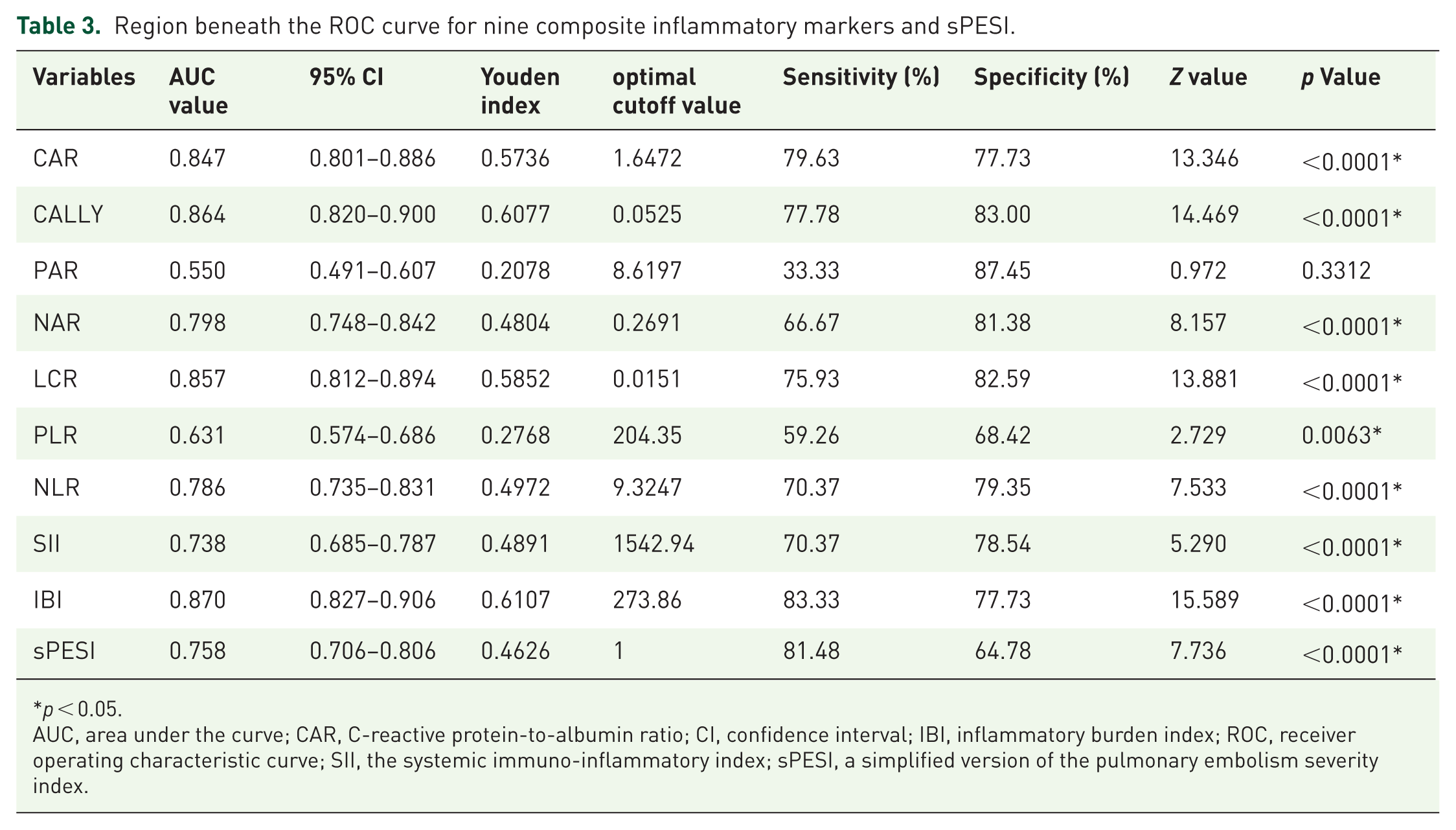
p < 0.05.
AUC, area under the curve; CAR, C-reactive protein-to-albumin ratio; CI, confidence interval; IBI, inflammatory burden index; ROC, receiver operating characteristic curve; SII, the systemic immuno-inflammatory index; sPESI, a simplified version of the pulmonary embolism severity index.

ROC curves for nine composite inflammatory markers and sPESI.
Comparative analysis of the area under the curve of IBI with other composite inflammatory markers and sPESI using DeLong’s test.

p < 0.05.
CAR, C-reactive protein-to-albumin ratio; IBI, inflammatory burden index; SII, the systemic immuno-inflammatory index; sPESI, a simplified version of the pulmonary embolism severity index.
Survival analysis of IBI in APE
Based on the optimal cutoff value of 273.86, patients were stratified into low-IBI (n = 201) and high-IBI (n = 100) groups. The high-IBI group demonstrated greater disease severity across multiple clinical parameters (Table 5). Kaplan–Meier survival analysis confirmed significantly poorer 30-day survival in the high-IBI group (45.0% vs 4.5%; p < 0.001) (Figure 3, Table 6).
Patient characteristics stratified by IBI levels in APE.
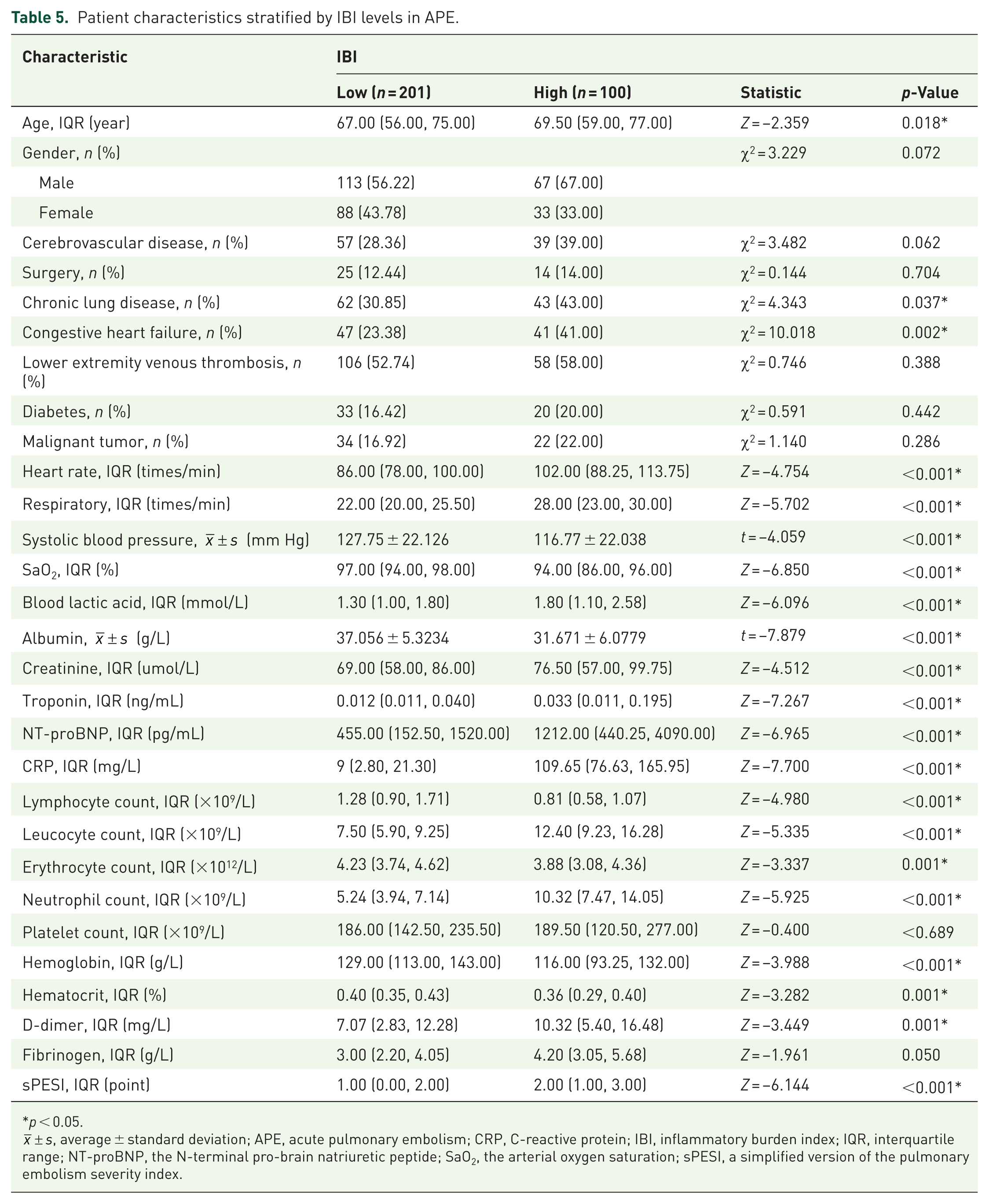
p < 0.05.

Kaplan–Meier survival analysis of APE by IBI.
Kaplan–Meier survival analysis of APE by IBI.

p < 0.05.
APE, acute pulmonary embolism; IBI, inflammatory burden index.
RCS curve analysis of IBI
After adjusting for key confounders (age, sex, and comorbidities), RCS analysis demonstrated a significant linear association between LogIBI levels and short-term mortality risk (p-overall <0.0001; p-nonlinear = 0.2948). The hazard ratio for mortality increased approximately linearly with each unit increase in LogIBI (Figure 4).

Using RCS curves to demonstrate the impact of IBI on 30-day fatality rate in APE patients.
Subgroup analysis
Each 1-unit increase in LogIBI was associated with a fourfold increase in 30-day mortality risk (HR 4.17, 95% CI 2.84–6.11, p < 0.001). No significant interaction effects were observed across any subgroups (Figure 5).

Subgroup analysis of the association between IBI and short-term prognosis in APE.
Discussion
IBI as a novel prognostic marker in APE
Inflammatory processes play a critical role in the progression and prognosis of APE.25–27 Neutrophils and lymphocytes are key mediators of this response: neutrophils exert pro-inflammatory and pro-thrombotic effects, 28 whereas lymphocytes regulate and attenuate inflammation. 29 CRP serves as a key inflammatory marker, with elevated levels correlating with right ventricular dysfunction in patients with APE.30,31 Our findings align with these mechanisms, demonstrating significantly higher CRP and neutrophil levels alongside lower lymphocyte counts in non-survivors. This consistent pattern supports that an exaggerated inflammatory response predicts adverse outcomes in APE.
Our study confirms that the IBI is an independent predictor of short-term mortality in APE, with an AUC of 0.870 (optimal cutoff = 273.86; sensitivity = 83.3%; specificity = 77.7%). The IBI exhibited a clear dose–response relationship with mortality risk, which remained consistent across all subgroups and was not significantly confounded by demographic or comorbidity variables. As an objective biomarker derived from routine laboratory tests, the IBI offers a rapid and accessible means of assessing inflammatory burden and facilitating early risk stratification in patients with APE.
Clinical implications and future perspectives
Our findings highlight the potential clinical utility of the IBI across multiple domains. In emergency settings, the IBI can be calculated rapidly at the time of admission using routine blood tests. 16 An IBI value >273.86 may serve as an objective warning indicator, prompting immediate intensive monitoring and expedited evaluation, even before completion of a full sPESI assessment. This approach may help identify occult high-risk patients who might otherwise be under-triaged.32,33 For therapeutic decision-making, the IBI may refine risk stratification among patients with intermediate sPESI scores, potentially guiding management intensity—where high-IBI values warrant closer monitoring and low-IBI values support standard care.2,34 It may also assist in decision-making regarding early discharge and outpatient management for low-risk patients. Serial measurements of IBI during hospitalization could provide a dynamic assessment of the resolution of inflammatory burden and treatment response. A declining IBI trend may indicate a favorable therapeutic response, whereas a persistent elevation or increase could suggest treatment failure, complications (e.g., new infarction), or an increased risk of early adverse events, thereby warranting re-evaluation.35,36 These potential applications require validation in prospective, multicenter studies to establish the impact of the IBI on clinical outcomes.
The field of APE prognostication is evolving toward multiparametric integration, with recent studies exploring novel biomarkers such as plasma osmolality, the Naples prognostic score, and various radiological indices.34,35,37–39 However, interpreting these diverse parameters remains challenging. Artificial intelligence (AI) and machine learning offer transformative potential by processing high-dimensional data, capturing complex nonlinear relationships, and identifying subtle predictive patterns.40,41 The successful application of deep learning models for predicting APE-related mortality demonstrates this potential. 42 Future intelligent systems may integrate clinical scores, biochemical biomarkers such as the IBI, and imaging data. In this context, the IBI could serve as a valuable input variable for AI-driven decision-support tools, thereby enabling more precise risk assessment. 43
Limitations of the study
This study has several limitations that should be acknowledged. Its retrospective, single-center design inherently carries a risk of selection bias and unmeasured confounding. Although the exclusion criteria were necessary to control for major confounders, they may have resulted in a cohort that underrepresents the most complex cases, thereby potentially limiting the generalizability of the findings. In addition, performing multiple statistical comparisons among the nine inflammatory markers without adjustment for multiplicity increases the risk of Type I error, implying that some observed significant differences (Table 4) may be attributable to chance. Furthermore, the sample size was determined by the availability of eligible patients in the institutional database during the study period rather than through a priori power calculation. Most importantly, the predictive performance of the IBI identified in this study lacks external validation in an independent, preferably multicenter, cohort. Consequently, the promising results of this study should be regarded as preliminary and hypothesis-generating. These limitations underscore the need for future prospective, multicenter studies to validate the prognostic utility of the IBI in APE and to establish more universally applicable cutoff values.
Conclusion
This study demonstrates that the IBI, a novel inflammatory biomarker, serves as an independent predictor of short-term prognosis in APE. In internal comparisons, the IBI demonstrated superior predictive performance compared with other composite inflammatory markers and the sPESI score, positioning it as a promising tool for prognostic assessment. Further prospective, multicenter studies are required to validate its generalizability and clinical utility.
Supplemental Material
sj-docx-1-tak-10.1177_17539447251407467 – Supplemental material for A novel predictor of short-term prognosis in acute pulmonary embolism: inflammatory burden index
Supplemental material, sj-docx-1-tak-10.1177_17539447251407467 for A novel predictor of short-term prognosis in acute pulmonary embolism: inflammatory burden index by Lingwei Huang, Dan Xiao, Meimei Yang, Zixing Wu and Fei He in Therapeutic Advances in Cardiovascular Disease
Supplemental Material
sj-docx-2-tak-10.1177_17539447251407467 – Supplemental material for A novel predictor of short-term prognosis in acute pulmonary embolism: inflammatory burden index
Supplemental material, sj-docx-2-tak-10.1177_17539447251407467 for A novel predictor of short-term prognosis in acute pulmonary embolism: inflammatory burden index by Lingwei Huang, Dan Xiao, Meimei Yang, Zixing Wu and Fei He in Therapeutic Advances in Cardiovascular Disease
Footnotes
Acknowledgements
The authors express their sincere gratitude to the Chongqing Municipal Health Commission Science and Health Joint Project (2021) and the Yongchuan District Science and Technology Bureau Technology Innovation and Application Development Program (2021) for their financial support of this study. We also thank the Science and Technology Department of Yongchuan Hospital, Chongqing Medical University, for their valuable administrative support and coordination throughout the study.
Declarations
Supplemental material
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
