Abstract
This review aims to describe the sex differences in heart failure (HF) patients, with a particular emphasis on the effect of cardiovascular ageing. Additionally, it takes into consideration the sex-related variation in cardiovascular health and physiology and the role ageing plays in HF and its implications in drug therapy. The pharmacokinetics and pharmacodynamics of the common HF medications, classified according to the established sub-types, are summarised with respect to sex-specific documented findings. Despite numerous studies confirming significant differences in HF outcomes according to sex, there are no current guidelines that consider patients’ sex in medical therapy of HF. Moreover, females are significantly under-represented in research trials, as well as under-treated in clinical practice, which hinders our understanding of HF in this demographic. Most of the current knowledge on sex-specific HF therapies is driven by secondary analyses of studies not primarily undertaking sex-specific analyses. Therefore, we propose a multi-faceted approach, including increased awareness among healthcare providers and more inclusive research to create a personalised care plan accounting for sex differences in HF management. Given the highlighted knowledge gaps, it is paramount for new research efforts to account for the different sex phenotypes in HF.
Introduction
Advancing age is strongly associated with increasing cardiovascular disease (CVD) burden, highlighting the importance of understanding the effects of ageing and frailty on CVD management.1,2 Approximately one-quarter of the first CVD clinical presentations are represented by heart failure (HF), with this syndrome exhibiting a similar sex distribution but increasing drastically in prevalence amongst older people. 3 Based on the left ventricular ejection fraction (LVEF), different HF sub-categories are recognised: heart failure with reduced ejection fraction (HFrEF), consisting of LVEF <40%, heart failure with mildly reduced ejection fraction (HFmrEF), with 40% < LVEF < 50%, heart failure with preserved ejection fraction (HFpEF), representing a 50% < LVEF < 65%, and the newly emerging proposed sub-category of heart failure with supra-normal ejection fraction (HFsnEF).4,5 Subsequently, HF presents different phenotypes, each with distinct underlying characteristics. HFrEF is marked by significant systolic dysfunction, often linked to conditions like ischaemic heart disease (IHD) or dilated cardiomyopathy. Patients with HFmrEF display a mix of systolic and diastolic dysfunction, often considered a transitional stage. HFpEF is characterised by diastolic dysfunction, where the heart’s ability to relax and fill is impaired. 4 HFsnEF is not a well-described phenotype, which is currently not supported in international guidelines with respect to diagnosis and management, where, despite a high ejection fraction, patients experience HF symptoms due to abnormalities such as increased myocardial stiffness. 5 Each phenotype is linked to a different pathophysiological pathway, thus prompting the introduction of tailored management approaches.
Studies of the European Society of Cardiology (ESC) Heart Failure Long-Term Registry showed that HFrEF patients were younger, predominantly male, with worse functional class, suffering more from ischaemia and dilated cardiomyopathy, and less from hypertension, or valvular pathology. Conversely, HFpEF patients exhibited opposing characteristics, while including a higher incidence of tachycardia-associated cardiomyopathies. HFmrEF patients had mixed baseline characteristics, leaning more toward HFrEF concerning sex and age distribution. Interestingly, while the prevalence and mortality of symptomatic HFpEF and HFrEF are comparable, older females were more frequently diagnosed with HFpEF.6–8 In fact, studies have shown that females have twice the risk of HFpEF compared to males. 9 Similarly to the presentation in HFpEF, HFsnEF strongly favours the female sex, while the aetiology is less likely to be linked with IHD. 5 Thus, it can be concluded that there are sex-specific pathophysiological mechanisms involved in the development of HFpEF and HFsnEF such as oestrogen synthesis dysregulation in post-menopausal females, metabolic factors and myocardial ageing and remodelling.10,11
Given that adjusting for age plays a key role in the risk prediction modelling for HFpEF, it can be hypothesised that the healthy cardiovascular ageing process is related to the pathophysiological mechanisms of this myocardial dysfunction. 12 The physiology of the ageing myocardium includes left ventricular (LV) stiffening and wall thickening (through increased collagen accumulation and fibroblast activity as well as ongoing reactive fibrosis), reduced diastolic function and reduced cardiac output reserve while maintaining normal function at rest. 13 Strikingly, the aberrant cellular pathways noted in HFpEF are also represented by hypertrophy, eventually leading to diastolic maladaptive mechanisms and dysfunction. 14 Conversely, females are significantly less likely to present with HFrEF. 15 This could be partly explained by sex-related ageing processes, occurring to a greater extent in the males, respectively: increased cardiomyocyte loss, secondary to necrosis, apoptosis and autophagy, as well as reduced stem cell regeneration. 16 Additionally, IHD significantly contributes to the development of HFrEF, with males having a notably higher incidence of IHD. Moreover, while the exact pathophysiology of HFsnEF remains unknown, there is a well-described link to the smaller LV chamber size, as well as LV hypercontractility leading to increased metabolic demand and increased passive stiffening, reflective in the higher end-systolic and diastolic elastance. Interestingly, as these worsen with age, they are disproportionately worse in females. Furthermore, it has been hypothesised that microvascular and sympathetic dysfunction may also contribute to the development of HFsnEF, as both processes manifest more prominently in ageing females. 17
Given that various aetiologies of HF lead to different phenotypes, it is important to mention the sex disparities of the causative factors of HF: IHD, hypertrophic, dilated, arrhythmogenic cardiomyopathy, being more prevalent in the male population, while hypertensive, atrial fibrillation, diabetic complications presenting more commonly in females (Figure 1). 18
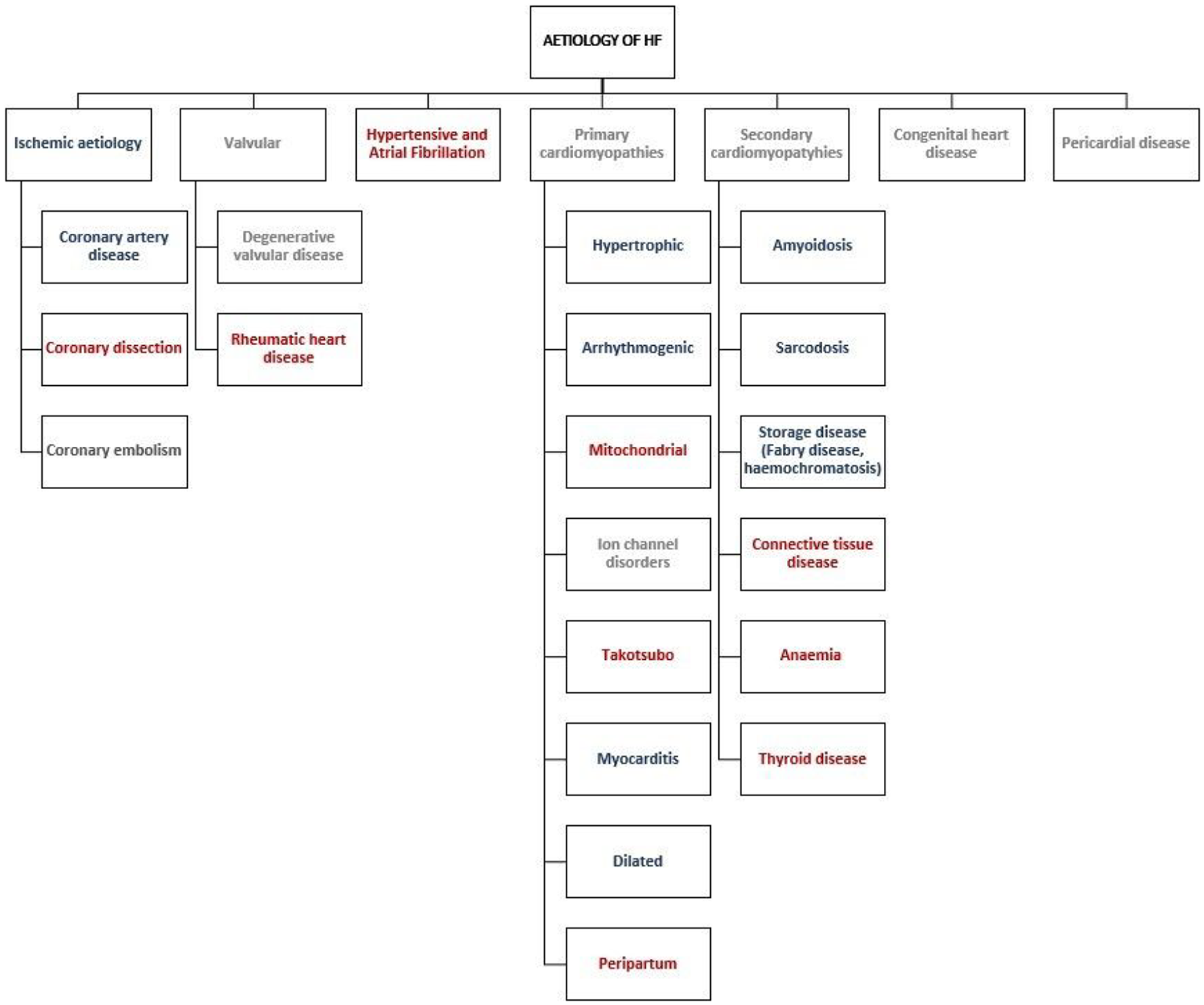
Sex distribution according to aetiologies of heart failure.
Nonetheless, the differences between the two sexes in the background of HF are not simply limited to a biological perspective, with ageing being a cornerstone contributor, but also reflect the social understanding and clinical management of these groups of patients. It has been well established that females are both under-represented in randomised control trials (RCTs) and under-treated in the clinical setting with respect to CVD. 19 Consequently, current international guidelines relating to the treatment of HF are largely based on a study population comprising middle-aged males. This is particularly problematic as males and females differ significantly in their myocardial structure and functioning, undergoing distinct ageing processes, as well as in the pharmacokinetics and pharmacodynamics of HF medication. 20
While in recent times, more attention has been cast over the well-defined sex-specific biological and pharmacological processes, there are still limited evidence-based tools that guide a sex-specific medical approach in CVD. This review aims to synthesise the existing literature, focusing on the relationship between sex and drugs available for the treatment of HF and outline the effect of sex-specific ageing on the pathophysiological mechanisms and clinical course of the condition.
Sex-based differences in cardiovascular ageing and pathophysiological mechanisms of HF
There are numerous differences between the male and female cardiovascular systems, starting from an early age, and accentuating as individuals progress through life. The distinct anatomy and physiology characterising the two sexes can be divided into cardiac differences, vascular differences, mismatch in environmental exposure and hormonal traits.21,22
Sex-specific cardiac differences
Females have smaller LV dimensions and a diminished stroke volume, leading to decreased cardiac output. While this could be matched by the lower female body size, this difference remains evident even after accounting for this parameter. 23 Similarly, females require a higher heart rate and pulsatile load to generate the same cardiac output and tissue perfusion as their male counterparts. 24
Moreover, ageing results in myocardial wall thickening independent of sex. Strikingly, females have a more accelerated process of cardiac muscle hypertrophy associated with ageing, developing concentric rather than eccentric remodelling, characteristic of male cardiovascular ageing. 25 Furthermore, despite myocardial stiffening being a hallmark feature of ageing, it occurs predominantly in females. This pathophysiological process might be related to the increased local inflammation and fibrosis of the female myocardium. 26
In the first six decades of life, males are at a much greater risk of developing hypertension; however, the sex distribution reverses in older people. 27 Additionally, it has been noted that there is a higher degree of adverse remodelling in hypertension and a lesser reversal of LV hypertrophy once treatment is initiated in females. 28 Premenopausal females also have lower cholesterol and triglyceride values compared to the age-matched male population. Despite this relative protection earlier in life, the hormonal changes in menopause result in a reversal of this biological advantage.29,30 Notably, lipid-lowering strategies, particularly statins, benefit both males and females to a similar degree. 31 Nonetheless, statins are less well tolerated by females. 32
Then the link between pregnancy and CVD is well documented. There are numerous metabolic and pro-inflammatory abnormalities during pregnancy, such as gestational diabetes and hypertensive disorders, which may lead to adverse long-term effects such as atheroma formation and endothelial dysfunction. Hypertensive pregnancy disorders are associated with a two-fold increased risk of incident HF. This increased risk is not only attributed to a higher probability of developing peri/post-partum cardiomyopathies but also to pathological processes years after the pregnancy. 33 According to NHS-II, an observational study following females from 1989 to 2015, there was also a strong correlation between gestational diabetes and CVD later in life, mediated by lifestyle factors, such as weight gain, diet and physical activity. 34
Sex-specific vascular differences
Males develop obstructive arterial disease at an earlier stage in life, the incidence of thin cap fibroatheroma being much greater in males compared to females under the age of 70. Moreover, females also present with more stable plaque. Consequently, females develop angina and myocardial infarction approximately 10 years after the male population. 35 In terms of the calcification of the coronary arteries, females have once again a physiological advantage in earlier life, with an abrupt increase in calcium score after menopause. 36
Conversely, females are more often affected by non-obstructive vascular conditions, such as endothelial dysfunction and microvascular spasm. Females also have smaller diameter, more tortuous coronary arteries, which occasionally create issues in the invasive medical management of cardiac conditions. 37 While there is contrasting evidence on the sex-based complications risk of percutaneous coronary interventions (PCI), a large observational study analysing the clinical outcomes of patients receiving PCI for coronary chronic total occlusions revealed that females develop much more commonly major adverse coronary events. Similar to age-related myocardial stiffness, vascular smooth muscle also undergoes gradual stiffening, affecting predominantly females. 38 Hence, the already discussed trend of blood pressure throughout life in the two sexes could be explained.
Sex-specific hormonal influences
Oestrogen has been hypothesised to protect cardiovascular health via mechanisms related to the antioxidant and anti-platelet effects, as well as positive metabolic regulation. Additionally, there is a definite discrepancy in the cardiovascular autonomic response, which relies on the oestrogen-mediated effect in females, reducing sympathoadrenal responsiveness. 39 The influence of oestrogen results in females exhibiting reduced sympathetic activity, generating a degree of protection against adverse outcomes associated with sympathetic response, such as arrhythmias, elevated blood pressure and increased afterload observed in HF. Females are also subject to intense physiological and hormonal changes during pregnancy. 40 Pregnancy alone can influence the anatomy and physiology of the myocardium. For instance, parity has been strongly correlated to diastolic dysfunction 10 years after birth. This could set further fundamental understanding of the increased predilection of HFpEF in females. In addition, ovarian ageing and menopause play an important role in the accelerated maladaptive cardiovascular mechanisms occurring in the sixth decade of life in females. 11 The post-menopausal sudden sharp decline in the sex hormones in females has been associated with a rise in lipid levels, atherosclerosis and other metabolic imbalances in middle and later life. 40
Sex-based differences in the management of HF
Despite recent improvements in CVD survival, the CVD burden remains high. HF incidence has been plateauing in recent years, despite a significant change in its distribution. While HFrEF incidence is decreasing, this is counteracted by a rise in HFpEF.41,42 This is particularly problematic given the relatively unexplored field of medical management of HFpEF. As noted in this review, this sub-type of HF affects with predilection older people and females, which might lead to worse outcomes in these populations given the scarce supporting evidence of medications in this clinical scenario. However, interestingly the prognosis of HF is frequently illustrated as being similar or even slightly better in females, in the absence of a clear explanation. 43
While there are numerous sex-based differences in the anatomy and physiology of the cardiovascular system, the outcomes of CVD heavily depend on medical management. In terms of HF, the treatment pathway is divided between the medical management of HFrEF and the one of HFpEF. While there is a vast amount of supporting literature concerning the treatment of HFrEF, HFpEF remains relatively understudied with little evidence regarding the benefits of drug therapy. However, sodium-glucose co-transporter 2 inhibitors (SGLT2is) have been recently established as an effective management option for HFpEF. 44 Moreover, HFmrEF and HFsnEF remain difficult to characterise due to the lack of evidence for these sub-groups.
Sex-differences in HFrEF
The management of HFrEF has been extensively documented. It relies on guideline-directed medical therapy (Table 1), as well as cardiac rhythm management, cardiac rehabilitation programmes and addressing comorbidities.
- Neutral; - favours males; - favours females.
ACEi, angiotensin-converting enzyme inhibitor; ARB, angiotensin receptor blocker; HF, heart failure; HFpEF, heart failure with preserved ejection fraction; HFrEF, heart failure with reduced ejection fraction; MRA, mineralocorticoid receptor antagonist; SGLT2i, sodium-glucose co-transporter 2 inhibitor.
Medical therapy
Angiotensin-converting enzyme inhibitors and angiotensin receptor blockers
Some of the most used classes of medications in HFrEF and hypertension are represented by angiotensin-converting enzyme inhibitors (ACEis) and ARBs. 45 When considering the pharmacokinetics parameters (area-under-the-curve (AUC), elimination half-life), multiple trials suggest that there are no significant differences between males and females when assessing the activity of ACEis, ARBs alone or in combination with neprilysin inhibitors.46–50 In terms of ageing, some studies indicate a significantly higher AUC in older people, while others describe minimal age-related variations.50,51
The sex-specific clinical outcomes of the above-mentioned medications have been extensively studied (Table 1). Noteworthy, all the RCTs depicted have been analysed post hoc in relation to sex-specific results, while the original study designs were not powered to assess the difference in outcomes between males and females.
Nonetheless, a meta-analysis including data from multiple emerging studies documenting the sex-specific effects on mortality and hospitalisations of ACEi or ARB suggests that females do not achieve the same mortality benefits compared to males. This supports previous literature, such as the SOLVD and SAVE RCTs.52,53 However, it is important to note that these trials only enrolled 12% and 17% females and were therefore not powered to determine such sex-specific treatment estimates. Strikingly, there is conflicting evidence with respect to the sex-specific effect of ACEi/ARB drug management (Table 1). For instance, CHARM is an RCT analysing the effects of candesartan on all-cause death, HF hospitalisations and cardiovascular mortality. Notably, females benefitted more from the ARBs compared to their male counterparts. Consequently, while the effects of ACEi and ARBs have been vastly researched, there remains ambiguity on the subject, with ARBs appearing advantageous when administered to the female population, as opposed to ACEi which shows inconsistencies in the results obtained. In terms of the effects of ageing on the ACEi/ARB therapy, elderly patients, in particular, the female sub-groups appear to respond less to these drugs. 51
There are also notable differences between males and females in the reporting of adverse drug reactions. For instance, a recent observational study, including over a quarter million patients, demonstrated females have a 1.31-fold increased risk of reporting adverse drug reactions from ACEi. 61
Beta-blockers
Beta-blockers are an effective first-line treatment for HFrEF. Numerous studies documented that their pharmacokinetics are strongly associated with sex and age.20,62,63 Firstly, due to the difference in body size, blood flow to the liver, and the activity of CYP2D6 enzyme (metabolising several beta-blockers, e.g. metoprolol), plasma levels of certain beta-blockers are significantly raised (up to 40% for metoprolol) in the female sex. 64 Unfortunately, there is limited evidence on more commonly prescribed beta-blockers. Moreover, the metabolism is heavily influenced by sex hormones. Females using hormonal therapy experience even greater increases in beta-blocker concentrations. 62 Despite a multitude of dose equivalency studies, supporting adjusting the doses for both sex and geriatric patients, no current guidelines or prescription information reflect these findings. Thus, toxicity and adverse reactions (bradycardia, hypotension) are more common in the female population and older people.65,66
Numerous large-scale studies confirmed similar mortality rates of female and male patients on beta-blockers (Table 1).67–69 Another study (BEST) revealed conflicting evidence, the outcomes differing according to aetiology: ischaemic (males have a survival benefit) and non-ischaemic (females have a survival benefit). 70 This finding could be explained by females being under-treated in acute coronary syndrome, as well as the fact that females tend to be frailer, older and more comorbid. Moreover, BEST studied the effects of bucindolol, which might act differently compared to commonly prescribed beta-blockers.
A recent meta-analysis supports beta-blockers’ clinical benefits irrespective of sex. It emphasises dose adjusting by age and sex for optimal results. 71
Mineralocorticoid receptor antagonists
Mineralocorticoid receptor antagonists (MRAs) seem to be beneficial in both the female and male sub-groups, despite notable differences in the baseline characteristics of the population analysed.72,73 Females were older, with worse renal function, higher incidence of hypertension and higher body mass index, whereas males had lower LVEF, higher incidence of co-morbidities and better functional class.
However, limited contrasting literature suggested that females respond better to this therapy and have improved anti-fibrotic effects. 74 Another experimental model on rodents confirmed a greater benefit of eplerenone therapy in the female specimens. 75
Nonetheless, a notable issue in patients presenting with HFrEF is the presence of co-morbidities such as diabetes or kidney disease. These conditions increase the risk of developing hyperkalaemia, which is one of the significant side effects of steroidal MRAs. This adverse reaction appears to manifest in an even distribution across both sexes. 72 Hence, alternatives such as non-steroidal MRAs have been developed, which are less likely to cause electrolyte disturbances. 76 For instance, finerenone was studied and shown to have a similar effect to more traditionally used MRAs. 77
Sodium-glucose co-transporter 2 inhibitor
SGLT2is are a relatively new class of HF medications, initially being developed for treating diabetes. Consistently, similar results (HF recorded events, hospitalisation and all-cause mortality) in females and males suffering from HF have been noted (Table 1).78,79 Interestingly, the study population differed in terms of baseline characteristics (Figure 2). Moreover, when administering this class of medications in the acute scenario, for patients suffering from myocardial infarction, once again similar results have been achieved in males and females alike. 80 Importantly, these RCTs analysed a much more diverse population and presented with a more balanced number of female and male participants.

Differences in baseline characteristics of DAPA and DELIVER between males and females.
Other: digoxin, ivabradine
Digoxin is a commonly prescribed HF and atrial fibrillation medication, despite its known difficult-to-control therapeutic target. Being renally excreted, it heavily relies on an optimal glomerular filtration rate (GFR). Females and older people have a decreased GFR, thus excreting the drug slower. 81 Moreover, males and females vary significantly in terms of fat percentage and body mass index. When adjusting for these parameters, females and males present with similar pharmacological findings. 82
However, in clinical practice, digoxin exhibits a more dangerous side-effect profile and mortality risk for the female population.83,84 This has been linked to increased toxicity in females, given that oftentimes doses are not adjusted to body weight and renal clearance. In fact, females are at a 1.4-fold increased risk of hospitalisation for digoxin toxicity compared to their male counterparts. 85 Interestingly, despite these findings, digoxin is more frequently prescribed to females, especially in the background of atrial fibrillation.86,87 Noteworthy, the concomitant use of digoxin and hormone replacement therapy is associated with a five-fold increase in the risk of coronary heart disease. 89 Furthermore, the presence of fewer sodium pumps in muscle in females could explain the worse outcomes in females, particularly with regard to the risk of life-threatening arrythmias. 20 While increasing age has been linked to a higher number of hospitalisations for suspected digoxin poisoning and increased risk of falls, the medication is equally effective across age groups.88–91
Ivabradine is another medication used in HFrEF. Unfortunately, the disproportionate number of male participants (76% and 83%) in the studies analysing ivabradine’s effect causes difficulty in interpreting the results for the female population. Based on the available literature, females and males benefit similarly from this medical management. 92 In terms of the antianginal properties of ivabradine, it appears that neither age nor sex affects the reduction of self-reported angina attacks. 92 Regardless, more evidence is required to confidently assert the effect of ivabradine in females.
Cardiac rhythm management
Alongside the standard medical therapy, cardiac rhythm management also improves mortality, morbidity and the quality of life for specific groups of HFrEF patients with notable sex disparities (Table 2). 93 Similar to the guideline-directed medical therapy research, studies analysing the impact of cardiac rhythm management on HFrEF recruited a predominantly male population, thus not being powered to detect sex-specific effects.
The notable sex-based findings of cardiac rhythm management in HFrEF.

HFrEF, heart failure with reduced ejection fraction; IHD, ischaemic heart disease; LVEF, left ventricular ejection fraction.
Especially in the early disease process, sudden cardiac death (SCD) attributed to ventricular arrhythmias is a major cause of mortality, particularly when an ischaemic aetiology is present, thus highlighting the importance of analysing this form of therapy. Nonetheless, it is important to note that the incidence of SCD is strikingly lower in women, irrespective of the aetiology of CVD. 99 Possible explanations for this discrepancy, consist of females experiencing less severe forms of the disease, and having fewer co-morbidities that could increase the risk of SCD, however repeatedly even when correcting for these parameters, a notable difference in the rates of SCD across the two sexes remains. 95
Females experience more often asystole and pulseless electrical activity, whereas males experienced predominately ventricular fibrillation and ventricular tachycardia. Thus, males experience more commonly shockable rhythms, which could benefit from ICD therapy. Hence, there is some contrasting evidence from RCTs, some suggesting that in fact males have a survival benefit with this form of therapy (Table 3).
Comparing outcomes based on sex in large-scale studies on ACEi and ARB in patients with HFpEF.

ACEi, angiotensin-converting enzyme inhibitor; ARB, angiotensin receptor blocker; HF, heart failure; HFpEF, heart failure with preserved ejection fraction; HFrEF, heart failure with reduced ejection fraction.
As described in Table 3, males are treated more often with CRT, and in fact this difference in offering this therapy between sexes appears to rise. Despite this, females exhibit a better response to this type of therapy. While this could again be linked with the aetiology of the disease, with IHD creating more scar tissue, which could negatively impact the electrical conduction and thus overall effectiveness of biventricular pacemakers, another underlying explanation to the selective advantage in females, would be the smaller myocardial size which leads to faster conduction across the heart. 100
Sex differences in HFpEF
Most of the HF drug therapies, apart from SGLT2i, have not been shown to alter the disease progression in HFpEF. Nonetheless, sex-specific findings have been noted when investigating the effects of these medications. These could form our fundamental understanding of a sex-tailored approach in HFpEF management.
ACEi and ARB
PEP-CHF is an RCT that focuses on the effects of perindopril on patients diagnosed with HF and presenting with diastolic dysfunction. While the study presented insufficient power for its primary endpoint (composite of all-cause mortality, unplanned HF-related hospitalisations), it outlines the potentially beneficial effects of ACEi on HFpEF patients. Sub-group analysis revealed no major differences in outcomes between the two sexes. 101
I-PRESERVE is another RCT investigating the effect of irbesartan on HFpEF (Table 3). Interestingly, this is one of the few studies enrolling a larger number of females (59%) compared to males (41%). Females appeared to have a lower risk of all-cause events (including deaths and hospitalisations), thus benefitting more from irbesartan therapy. 102 Another RCT analysing the impact of ARBs on clinical outcomes of HFpEF is represented by CHARM-Preserved. This study confirmed that females experienced a lower death rate on candesartan therapy. 103 While ARBs have yet to be recommended for HFpEF, it is important to note their positive effect, particularly on the female population.
Sacubitril-valsartan
Interestingly, an RCT, PARAGON-HF, investigating the outcomes of sacubitril-valsartan therapy in patients with HFpEF revealed there might be an increased advantage of the sacubitril-valsartan therapy over ACEis and ARB. Moreover, the balanced representation of participants across both sexes enhances the credibility of the results. 104 This has been confirmed by a recently published meta-analysis and large cohort studies, which highlighted the benefit of sacubitril-valsartan therapy in female patients (improved hospitalisation rates, mortality from cardiovascular causes, functional class improvement). PARAGON-HF prompted further research aimed at gaining a deeper understanding of which specific sub-groups of HFpEF patients might derive the greatest benefits from sacubitril-valsartan therapy. Refining patient selection criteria, focusing on the female population, could yield significant results regarding the reduction of HFpEF-associated morbidity and mortality. This could represent the stepping stone to the beginning of sex-specific HF management.
Beta-blockers
Beta-blockers have been investigated in the background of HFpEF, with no convincing evidence of improving outcomes. This has been mirrored in two large-scale RCTs: SENIORS and J-DHF. However, a meta-analysis suggested that while HF hospitalisations are not improved by this medication, all-cause mortality might be positively influenced by starting HFpEF patients on beta blockers. 105
Interestingly, the use of beta-blockers is elevated in females with this condition compared to their male counterparts. It might be explained by the fact that females more commonly suffer from atrial fibrillation (AF), where beta-blockade is a first-line therapy for rate control management. 106
Mineralocorticoid receptor antagonists
Likewise, to beta-blockers, numerous studies have investigated the effects of MRA on HFpEF patients. From these, the largest RCT is represented by TOPCAT. Post hoc analysis of the study highlighted that there was no notable difference between males and females. Nonetheless, overall MRAs seem to be adding no or little benefit in terms of reduction of hospitalisations and mortality, while at the same time increasing the risk of adverse events, such as hyperkalaemia. 107 In addition, the sub-analysis comparing the older (age over 75 years) and younger population (age below 75 years) revealed no significant changes in the primary endpoints. 72
While steroidal MRAs, such as aldosterone, are of little benefit to this sub-group of HF patients, non-steroidal alternatives (such as finerenone) have been recently shown to positively influence survival and hospitalisation rates. 108 Despite this, one study suggested that the effect of the non-steroidal MRAs is difficult to extrapolate for the female population given that oftentimes study designs do not take into consideration the sex-based differences in pathophysiology. 109 A post hoc analysis on two large trials, FIDELIO-DKD and FIGARO-DKD, which enrolled patients with diabetic kidney disease, and excluded patients with symptomatic HFrEF, suggested that this medication reduces CVD burden, presenting with homogeneity in effect across both sexes. 110 A minority of these patients (7.7%) presented with an HF phenotype (HFpEF, HFmrEF or asymptomatic HFrEF). 45 These findings were supported by a recent trial investigating the effects of this drug on HFpEF and HFmrEF. 108
Sodium-glucose co-transporter 2 inhibitor
SGLT2i is the only recommended medical treatment for HFpEF as reflected by the 2023 ESC guidelines. 45 Notably, multiple large RCTs (DELIVER-HF, PRESERVED-HF) have documented the sex-specific clinical outcomes of dapagliflozin in recent years. The primary outcomes analysed in these studies (reduction in HF hospitalisation rates and mortality) are similar across the two groups. 71 EMPEROR-Preserved is another RCT focusing on the effects of empagliflozin on HFpEF patients. 111 It further also outlined the uniform effect of SGLT2i across the two sexes. Similarly to HFrEF, notable distinctions emerged between males and females concerning their baseline characteristics (Figure 2). Considering the adverse drug reaction discordance, females aged over 50 years suffer increasingly more urogenital infections in the background of diabetes. 112
Sex differences in HFmrEF
HFmrEF is a poorly analysed entity in the study of CVD. Clinically, there is considerable overlap between presentations of HFmrEF and patients suffering from either HFrEF or HFpEF, resulting from differences in inclusion criteria between clinical trials. Notably, the epidemiology of HFmrEF appears to be similar to patients presenting with HFrEF, with features such as male predominance, increased coronary artery disease burden and younger age at presentation. In terms of clinical outcomes, it has been outlined that males have a 2.2-fold increased risk of death at 90 days after diagnosis, despite a similar risk of cardiovascular events. Importantly, at 1 year, the all-cause mortality appeared similar between the two sexes. 113
There is a paucity of data considering the medical management of this condition, with no RCT analysing solely the effect of medications on HFmrEF patients. As a result, there is little evidence to support the use of medications, which is reflected in the guidelines. However, several RCTs investigating various HF management options included patients with an LVEF over 40%/45%, therefore including a minority of patients with HFmrEF. The outcomes in these studies were not presented according to LVEF, making it difficult to interpret the response of HFmrEF patients to these treatments. It is important to consider that a significant amount of the patients present with co-morbidities such as hypertension and atrial fibrillation. Therefore, it is common that these patients will be receiving some form of medical therapy from the above described.
Moreover, in the 2023 focused update on the ESC Guidelines regarding HF, emerging studies on SGLT2is (mainly EMPEROR-Preserved and DELIVER) led to the recommendation of this class of drugs in this HF phenotype. This recommendation as a class I medical therapy was based on the reduction of HF hospitalisations, however notably SGLT2Is not presenting with a statistically significant impact on cardiovascular death. 45
Additionally, the role of non-steroidal MRAs (mainly finerenone) is being studied in the background of HFmrEF. A preliminary study suggests that finerenone results in a reduction of cardiovascular deaths and a slowing of the progression of HF. 108
Sex differences in HFsnEF
Moreover, in recent years, a newly proposed and still controversial sub-category of HF, started emerging, respectively HFsnEF. According to the 2021 ESC Heart Failure guidelines, there is a clear indication that if LVEF is abnormally elevated, this should prompt investigations for conditions such as amyloidosis or hypertrophic obstructive cardiomyopathy and other high output states; however, there is a current debate about this is likely only represents a very small proportion of people presenting with supra-normal LVEF. 93
This poorly recognised condition has been associated with overall increased mortality and worse outcomes,114,115 although there is conflicting evidence showing similar mortality rates between HFsnEF and other HF phenotypes. 116 Of note, a recent large national registry showed worse outcomes in females in this subpopulation of HF. 117 Furthermore, interestingly, supra-normal ejection fraction is commonly found among patients with coronary artery disease; however, it is a negative predictor only in the female subpopulations. 118
Distinctively, this HF presentation can also be delineated through the relatively low natriuretic peptide (BNP). This impacts on establishing appropriate tools for diagnosis and prognosis in this category of patients, as well as establishing appropriate drug therapies. Hence, particularly, in well-compensated patients, diagnosis could be challenging in the absence of typical echocardiographic findings and possibly normal BNP levels. Nonetheless, specific biomarkers such as c-c motif chemokine ligand-23 and interleukin-8 have been suggested to act as unique biochemical signatures of the condition. 119
Interestingly, the genotype of patients with HFsnEF has also been studied, leading to the development of a genetic risk score. This research identified 16 single nucleotide polymorphisms associated with an increased LVEF, some of which had not previously been linked to other cardiac pathologies. 120
Unfortunately, there is a lack of literature analysing potential management options in this group of patients. Furthermore, patients are less likely to be prescribed commonly encountered HF medications (e.g. β-blockers). 5 While HFsnEF might appear more similar in presentation and epidemiology to HFpEF, sub-analysis of EMPEROR-Preserved revealed that the effect of empagliflozin is reduced in people with LVEF >65%. 121 Nonetheless, the results have been heavily scrutinised given the known challenges of post hoc sub-group analysis. Among the 9718 patients included in the data set, only 865 had LVEF >65%, which were evenly distributed to the placebo and empagliflozin arm of the study. In the placebo arm, only 45 patients had a first hospitalisation for HF, which was the main object of study in this analysis. Hence, it can be concluded that there is an insufficient number of participants to draw a robust conclusion regarding the dampening effect of SGLT2is in this sub-population. 122
Conclusion
HF epidemiology, prognosis and treatment outcomes are characterised by important sex differences. For instance, females have a smaller stroke volume and cardiac output, presenting with concentric myocardial thickening related to ageing. Additionally, females present disproportionately more often with non-ischaemic pathologies than males. Moreover, hormonal influences of menopause result in accelerated maladaptive cardiovascular mechanisms, such as increased incidence of hypertension later in life in females and myocardial stiffening. All of the above impact the manifestation of HF in the two genders. Despite this, HF management has remained relatively understudied amongst females.
Early studies of HF drugs notably included very few female participants, with some trials excluding them entirely. This raises questions about whether HF treatments are equally effective and safe for both sexes. While most medications appear to benefit both, disparities have been observed. Initial studies on ACEis suggested a greater benefit for males, though newer evidence shows similar effects across the two sexes, likely due to a more balanced sub-group analysis. ARBs tend to be more effective in females, particularly in HFpEF cases, while digoxin is consistently more toxic and linked to higher mortality in this sub-population. Despite these findings, many trials highlight the need to adjust medications based on weight and renal function, a practice often overlooked in the clinical setting, leading to potentially higher risks of adverse drug reactions and poorer outcomes for females.
Nonetheless, the need for a true sex-tailored approach to HF management remains unmet, in the absence of robust evidence. However, the existing literature should prompt further evaluation of the sex-specific outcomes in CVD with respect to the commonly prescribed HF medications. Furthermore, another direction for future research should aim at finding therapies suitable for the treatment of HFpEF and HFmrEF. Finally, HFsnEF is likely underdiagnosed given its very recent description in the literature with a yet unclear diagnostic and management pathway. These considerations warrant a greater focus on this form of HF, with an emphasis on the potential sex-related differences that may underlie this disease process.

