Abstract
Ivabradine is a pure heart-rate lowering drug that is nowadays used, accordingly to the last ESC Guidelines, to reduce mortality and heart failure (HF) hospitalization in patients with HF with reduced ejection fraction and in symptomatic patiens with inappropriate sinus tachycardia. Moreover, interesting effect of ivabradine on endothelial and myocardial function and on oxidative stress and inflamation pathways are progressively emerging. The aim of this paper is to highlight newer evidences about ivabradine effect (and consequently possible future application of the drug) in pathological settings different from guidelines-based clinical practice.
Keywords
Pharmacology of ivabradine and current indications
Ivabradine is a pure heart rate (HR)-lowering drug. It acts specifically blocking the If (“funny”) current in the sinoatrial node, 1 where myocytes generate a spontaneous slow diastolic depolarization and a subsequent action potential. Ivabradine is highly selective for If channels (A), blocking them by entering their pore from the intracellular side in a concentration-dependent manner only when the channel is open.2,3 It reduces the firing rate of the pacemaker cells in the sinoatrial node without affecting the duration of the action potential and without interfering with other ionic currents.2,4 Acting only on pacemaker cells, ivabradine is not effective during atrial fibrillation. Moreover, If inhibition is dependent on the frequency of channel opening, the voltage, and the sodium concentration; because of this, the effect in enhanced at higher HR. 3
In chronic heart failure (HF), the increase in HR following sympathetic activation is related to greater morbidity and mortality, 5 although tachycardia is one of the first compensatory mechanisms of left ventricular (LV) contractile dysfunction. A novel aspect of ivabradine is its selectivity and specificity for pacemaker If channels 6 : ivabradine does not affect myocardial contractility, preserves cardiac output, and significantly enhances stroke volume and systolic stroke work, without affecting blood pressure, unlike beta-blockers and non-dihydropyridine calcium channel blockers. 7 Many clinical trials have demonstrated both a significant reduction in nonfatal myocardial infarction and a decrease in hospitalization rate for HF in patients LV systolic dysfunction and sinus rhythm with HR ⩾ 70 bpm treated with ivabradine.8,9 In those patients, the drug was shown to improve LV reverse remodelling, 10 quality of life, 11 and exercise capacity. 12 Several sub-studies obtained from the SHIFT trial highlighted the positive effects of ivabradine on LV volumes and ejection fraction (LVEF) on ventricular-arterial coupling (VAC), and, especially, on aortic elastance (Ea).9,10,13 On the other hand, in patients who had stable coronary artery disease (CAD) without clinical HF, the use of ivabradine did not show an improvement in the risk of cardiovascular events in the SIGNIFY trial, 14 suggesting reduced beneficial effect in the absence of LV dysfunction and the related neurohormonal activation.
According to the results of the SHIFT and BEAUTIFUL trials,8,9 in the latest European Society of Cardiology (ESC) guidelines for the management of HF ivabradine is recommended in class IIa to reduce CV mortality and HF hospitalization in symptomatic patients with LVEF ⩽ 35% and resting HR ⩾ 70 bpm in sinus rhythm despite the maximum tolerated dose of beta-blocker, angiotensin-converting-enzyme (ACE) inhibitor (or angiotensin receptor blocker) and mineralocorticoid receptor antagonist (or angiotensin receptor blocker) (level of evidence B), or in those not tolerating or having contraindications to beta-blocker, already receiving ACE inhibitor (or angiotensin receptor blocker) and mineralocorticoid receptor antagonist (or angiotensin receptor blocker) (level of evidence C). 15
Ivabradine is also recommended alone or in association with a beta-blocker in symptomatic patients with inappropriate sinus tachycardia (class IIa, level of evidence B). 16 Recent evidence shows that this drug can be an effective alternative to amiodarone in children with post-operative junctional ectopic tachycardia, the commonest tachyarrhythmia in the early post-operative period in children undergoing open-heart surgery.17–19
Effect of ivabradine on endothelial function
In a recent study, our workgroup demonstrated the efficacy of ivabradine (5 mg two times a day on top of optimized medical therapy for HF) in the improvement of echocardiographic stroke volume, VAC, and endothelial function evaluated with EndoPAT device in patients affected by chronic HF with LV dysfunction. 20
Many previous studies have shown the interaction between HR, Ea, and arterial stiffness, both in animal models and in humans.21–29 These data are consistent with ours, suggesting that blood vessels can adapt to increased mechanic wall stress changing their structure, benefitting from ivabradine-induced HR reduction, with a final improvement of Ea and VAC. According to previous evidence, the effect might be predominantly on the arterial system and less on LV end-systolic elastance (Ees). 30 Similar findings about endothelial function and Ea were recently confirmed in patients with CAD, in which ivabradine improved arterial stiffness, endothelial function, aortic distensibility, and aortic cyclic strain on magnetic resonance imaging (MRI) over a period of 6 months. 31 An increased HR is a marker of CAD progression, correlates with cardiovascular mortality,32–35 and is associated with coronary plaque rupture and subclinical inflammation.36–38 Ivabradine has been shown to reduce oxidative stress in the myocardial wall, circulating angiotensin II levels and expression of angiotensin II receptor 1.39–41 Moreover, it decreases plasma values of IL-6, tumor necrosis factor-α (TNF-α), MCP-1, norepinephrine, and prevents endothelial nitric oxide synthase (eNOS) uncoupling.39–46 In ApoE-deficient mice, ivabradine showed a protective effect against atherosclerosis development, enhancing anti-apoptotic, anti-inflammatory, and pro-endothelium repair genes, with a concomitant decrease of expression of genes that increase endothelium permeability and favor inflammation and apoptosis. 47
Effect of ivabradine on myocardial function
Mechanisms leading to vascular and endothelial dysfunction (i.e., oxidative stress and pro-inflammatory status) mostly overlap pathogenetic pathways that can drive to diastolic dysfunction and HF with preserved LVEF (HFpEF). 48 The effect of ivabradine was recently demonstrated positive in relation to LV diastolic function: drug-induced HR lowering led to an improvement in mitral inflow E/A ratio and deceleration time (DT) and pulmonary vein flow S/D ratio in HFpEF patients. 49 The gain in diastolic filling related to an improvement in stroke volume and LVEF. An upgrade in diastolic function was also noted in ischaemic patients undergoing exercise stress echocardiography before and after ivabradine introduction; the same was shown for chronotropic reserve and ischaemic threshold (greater duration of exercise and reduced anginal symptoms). 50 Tachycardia induced by sympathetic activation in acute HF can lead to a further reduction in stroke volume. Adding ivabradine to inotropic drug treatment in ischaemic acute HF or cardiogenic shock was shown to optimize the lusitropic effect of dobutamine and was associated with an improved diastolic function (enhanced LV early relaxation and prolonged diastolic time), leading to an increased stroke volume compared with omecamtiv mecarbil. 51
In a murine model, ivabradine was shown to be effective in preventing progression from coxsackievirus-induced myocarditis to dilated cardiomyopathy. 52 This preventive and therapeutic effect of the drug is related to inhibition of the p38 MAPK pathway, downregulation of inflammatory responses (in particular, a reduction of the proinflammatory cytokines TNF-α, IL-1β, and IL-6 was observed), and reduction of collagen expression, with a consequent reduction in the fibrogenesis. 53 Moreover, ivabradine could attenuate the expression of Caspase-3 by downregulation of Bax and upregulation of Bcl-2 to prevent the deterioration of cardiac function resulting from ventricular myocyte loss by cardiomyocyte apoptosis. 54
Conclusion
There is strong evidence for the potentially negative effect of increased HR in endothelial and vascular function and in myocardial function. Ivabradine has a pure HR-lowering effect with a clear impact in the hemodynamic setting, but newer evidence has emerged about its putative influence on many cellular mechanisms. For this reason, this drug is increasingly considered as a potential reversing pharmacological molecule, demonstrating correlation with improvement in oxidative and inflammatory status, endothelial function, arterial stiffness, and diastolic function. Table 1 summarizes the results of the newer cited clinical trials (published after our last work) conducted using ivabradine in pathological settings different from guideline-based clinical practice. It must be highlighted that those studies were conducted in small cohorts of patients, with a short time of drug administration and without long-term follow up and prognostic evaluation. Those results are really encouraging and bring hope for a wider use of ivabradine in different clinical settings, but stronger evidence from large prospective clinical trials is still needed.
Results of recent clinical trials.
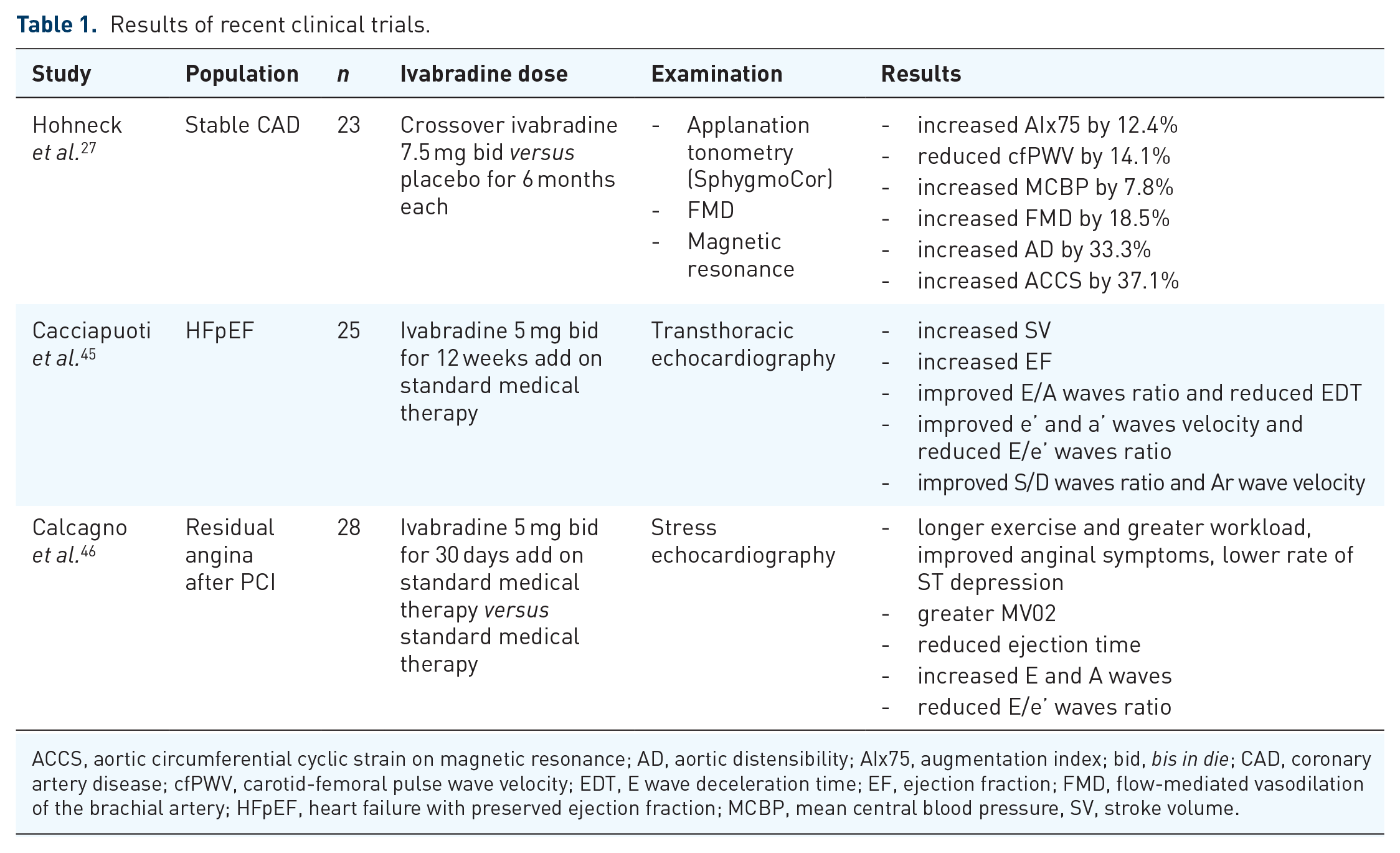
ACCS, aortic circumferential cyclic strain on magnetic resonance; AD, aortic distensibility; AIx75, augmentation index; bid, bis in die; CAD, coronary artery disease; cfPWV, carotid-femoral pulse wave velocity; EDT, E wave deceleration time; EF, ejection fraction; FMD, flow-mediated vasodilation of the brachial artery; HFpEF, heart failure with preserved ejection fraction; MCBP, mean central blood pressure, SV, stroke volume.
