Abstract
Background:
To review data from the pivotal phase III trials evaluating the efficacy and safety of direct oral anticoagulants (DOACs)
Methods:
A PubMed literature search (January 2009 to January 2017) was performed using the terms ‘dabigatran’, ‘rivaroxaban’, ‘apixaban’, ‘edoxaban’, ‘atrial fibrillation’, ‘RE-LY’, ‘ROCKET AF’, ‘ARISTOTLE’, and ‘ENGAGE AF-TIMI 48’. All primary publications and secondary analyses in special populations at increased risk of stroke or bleeding from the pivotal phase III clinical trials were evaluated.
Results:
Available secondary analyses indicate no treatment interactions with regards to stroke or systemic embolic event (SEE) prevention for any of the DOACs in the patient subgroups, including patients with advanced age, impaired renal function, diabetes, prior stroke, concomitant antiplatelet therapy, heart failure, prior stroke, history of hypertension, myocardial infarction (MI), coronary artery disease, and peripheral artery disease (PAD). Although higher bleeding incidence was reported with dabigatran and rivaroxaban in patients aged 75 years and over with apixaban in patients with diabetes, and with rivaroxaban in patients with previous MI or PAD, no changes in dosing are recommended.
Conclusions:
Overall, results of secondary analyses indicate that the recommended dosing strategy for each of the DOACs produces a consistent anticoagulant effect across a diverse patient population, including those at increased risk of stroke or bleeding.
Keywords
Introduction
For more than 50 years, warfarin was the only available oral anticoagulant in the US for the prevention of stroke and systemic embolic events (SEEs) in patients with nonvalvular atrial fibrillation (NVAF). Although warfarin therapy reduces the risk of stroke by 68%, 1 it has several limitations, including a delayed onset of action, considerable pharmacokinetic (PK) variability, interactions with food and numerous drugs, a narrow therapeutic range, and require routine monitoring.2,3 In contrast to warfarin, direct oral anticoagulants (DOACs) have a predictable PK profile and are administered at a fixed dosage without the requirement for routine monitoring.
As a class, DOACs have similar efficacy
This review describes key findings of the pivotal international phase III trials that led to the approval of DOACs in the US. This review also critically summarizes the outcomes of these trials in relation to patient subgroups that may affect the PK profile of DOACs or that are at an elevated risk for stroke or bleeding, including patients with impaired renal function, previous heart failure or stroke, diabetes, hypertension, or vascular disease, as well as patients on concomitant therapy, elderly patients, and female patients. Finally, the impact of these findings on dosing considerations for DOACs will be briefly reviewed.
Clinical trial results
The efficacy and safety of each of the DOACs for stroke prevention in atrial fibrillation (AF), in comparison with dose-adjusted warfarin (international normalized ratio 2.0–3.0), was evaluated in international phase III trials.6
–9 Details of trial designs, key baseline patient characteristics, along with the primary efficacy and safety results are provided in Table 1. The primary efficacy endpoint for all of the trials was the rate of stroke or SEE in the DOAC arm
Pivotal international phase III trials comparing DOACs
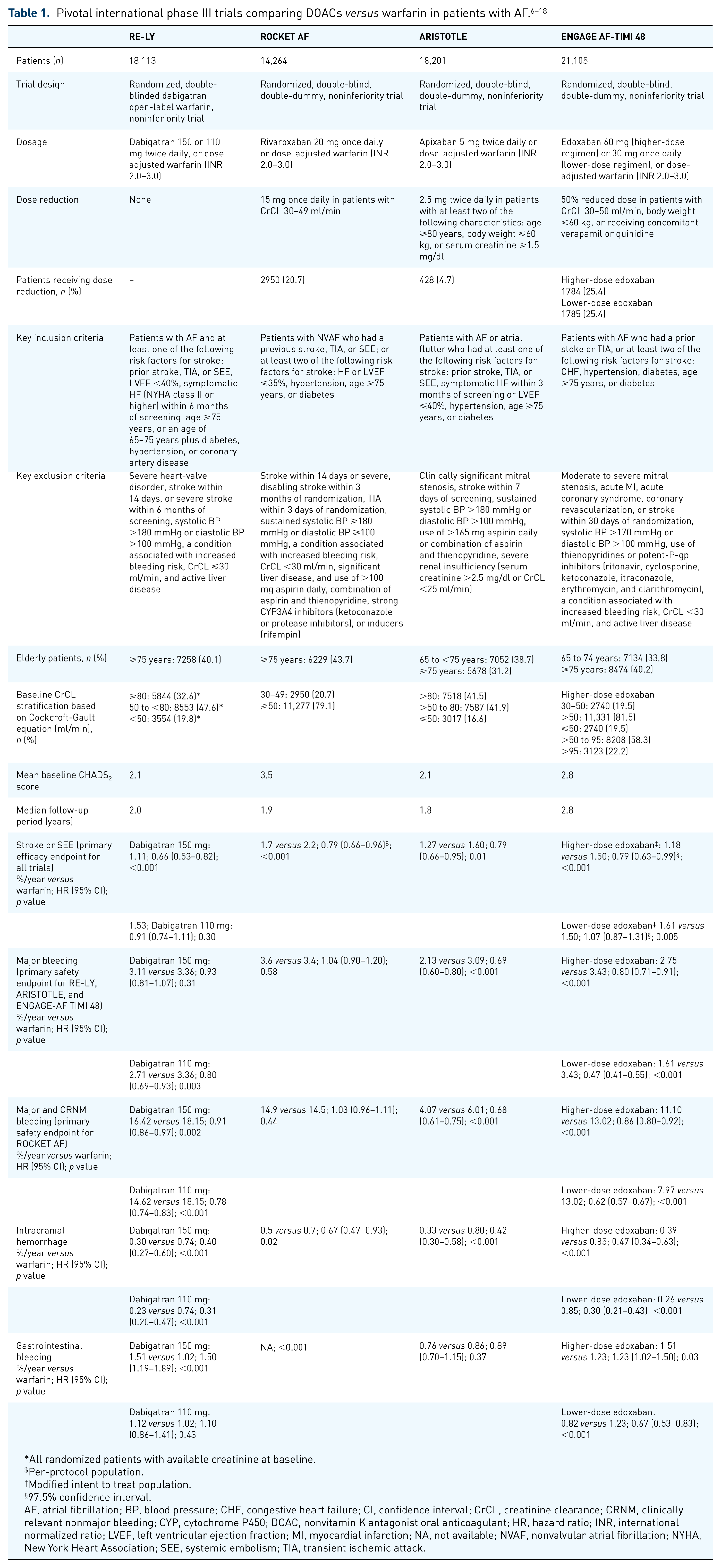
All randomized patients with available creatinine at baseline.
Per-protocol population.
Modified intent to treat population.
97.5% confidence interval.
AF, atrial fibrillation; BP, blood pressure; CHF, congestive heart failure; CI, confidence interval; CrCL, creatinine clearance; CRNM, clinically relevant nonmajor bleeding; CYP, cytochrome P450; DOAC, nonvitamin K antagonist oral anticoagulant; HR, hazard ratio; INR, international normalized ratio; LVEF, left ventricular ejection fraction; MI, myocardial infarction; NA, not available; NVAF, nonvalvular atrial fibrillation; NYHA, New York Heart Association; SEE, systemic embolism; TIA, transient ischemic attack.
DOAC efficacy and safety in patients with AF and impaired renal function
Among patients with AF, impaired renal function is associated with an increase in thromboembolic and bleeding risk.19 –21 Additionally, all DOACs are subject to renal clearance to varying degrees. Subgroup analyses of the four pivotal trials were conducted to determine whether renal function affected the efficacy and safety of DOACs, relative to warfarin in patients with AF.10 –13
A relative reduction in stroke or SEE risk with dabigatran 150 mg twice daily, rivaroxaban daily, or apixaban twice daily

Treatment outcomes in special populations. (a) Dabigatran 150 mg
With regard to the primary safety outcome, there was no interaction between treatment and renal function for dabigatran 150 mg twice daily, rivaroxaban daily, or higher-dose edoxaban daily in either the prespecified or the exploratory analysis. Across renal function subgroups, major bleeding incidence was lower with apixaban
DOAC efficacy and safety in patients with AF on concomitant amiodarone or antiplatelet therapy
Amiodarone, a P-glycoprotein (P-gp) and cytochrome P450 3A4 (CYP3A4) inhibitor, is an antiarrhythmic agent frequently used in patients with AF. In ROCKET AF, 1144 (8.0%) patients received amiodarone at baseline.
26
Of the 17907 (98.4%) patients for whom baseline amiodarone use was known in ARISTOTLE, 2051 (11.3%) received amiodarone.
27
In ENGAGE AF-TIMI 48, 2492 (11.8%) patients received concomitant amiodarone at randomization.
28
With regard to stroke or SEE incidence, or the primary safety outcome, there was no interaction between treatment and concomitant amiodarone use with rivaroxaban, apixaban, or higher-dose edoxaban
Many patients with AF receive antiplatelet therapy for coronary artery disease (CAD) or other comorbid conditions. Relative to anticoagulation alone, concurrent administration of warfarin and antiplatelet agents is associated with increased bleeding risk in patients with AF.
29
In RE-LY, 6952 (38.4%) patients received concomitant antiplatelet therapy with aspirin or clopidogrel at some point during the study.
30
ROCKET AF included 5205 (36.5%) patients who reported receiving aspirin on a daily basis at baseline.
31
A total of 4434 (24.4%) patients received concomitant aspirin on day 1 of the ARISTOTLE trial.
32
An analysis of patients in the ENGAGE AF-TIMI 48 trial by their use of single antiplatelet therapy 3 months after the start of treatment included a total of 19,909 patients, of whom 4912 (24.7%) received concomitant single antiplatelet therapy.
22
The relative decrease in stroke or SEE rate with dabigatran 150 mg twice daily, rivaroxaban daily, apixaban twice daily, or higher-dose edoxaban daily
DOAC efficacy and safety in patients with AF and previous heart failure or stroke
RE-LY included 4904 (27.1%) patients with previous heart failure (HF), defined as the occurrence of HF symptoms [⩾New York Heart Association (NYHA) functional class II] in the 6 months prior to screening in patients with a history of prior admission for congestive heart failure (CHF). 33 In ROCKET AF, 9033 (63.7%) patients with HF [a history of HF or left ventricular ejection fraction (LVEF) <40%] were included. 34 In ARISTOTLE, HF status and left ventricular function were documented for 14,671 (80.6%) patients, of whom 5072 (34.6%) had HF (a history of symptomatic CHF or the presence of CHF within 3 months of screening) and 2736 (18.6%) had left ventricular systolic dysfunction (LVSD; LVEF ⩽40% or documented moderate or severe left ventricular dysfunction). 35 Of note, although ROCKET AF classified patients with LVEF up to 35% as having HF, in the subsequent subanalysis the LVEF was changed to 40% or less. 35 ENGAGE AF-TIMI 48 enrolled 8145 (38.6%) patients with HF, defined as a history or ongoing HF stage C or D as specified by American College of Cardiology/American Heart Association criteria. 36
The relative decrease in stroke or SEE risk with dabigatran 150 mg twice daily, rivaroxaban daily, apixaban twice daily, or higher-dose edoxaban daily
A history of stroke is associated with an elevated risk for stroke among patients with AF, and with increased bleeding risk among those receiving anticoagulation. RE-LY included 3623 (20.0%) patients with AF who had a prior stroke or transient ischemic attack (TIA) with neurological symptoms lasting less than 24 h. 37 ROCKET AF enrolled 7468 (52.4%) patients with a history of stroke or TIA. 38 In ARISTOTLE, previous stroke or TIA was reported in 3436 (18.9%) patients. The ENGAGE AF-TIMI 48 trial included 5973 (28.3%) patients with previous stroke or TIA. 39 Relative to warfarin, each of the DOACs was similar for the primary efficacy and safety outcome in patients with or without prior stroke or TIA.37 –39
DOAC efficacy and safety in patients with AF and hypertension
Among patients with AF, hypertension is not only a risk factor for stroke, but is also associated with increased bleeding risk among those receiving anticoagulation.20,40,41 Overall, 14,283 patients had hypertension [systolic blood pressure (BP) 120 to ⩽180 mmHg and diastolic BP 80 to ⩽100 mmHg] during RE-LY.
42
In ROCKET AF, baseline hypertension status and screening systolic BP were known for 14,256 patients, of whom 12,902 (90.5%) had hypertension; hypertension was controlled (screening systolic BP <140 mmHg) in 7963 (61.7%) patients, and uncontrolled (screening systolic BP ⩾140 mmHg) in 4939 (38.3%).
43
ARISTOTLE included 15,916 (87.4%) patients with a history of hypertension that required treatment. The relative decrease in stroke or SEE risk with dabigatran 150 mg twice daily, rivaroxaban daily, or apixaban twice daily
DOAC efficacy and safety in elderly patients with AF
Elderly patients with AF are at an increased risk for stroke, as well as elevated bleeding risk when receiving anticoagulation.20,41 Approximately, 30–40% of patients included in the pivotal phase III trials were at least 75 years of age (Table 1).14 –17
With regard to stroke or SEE incidence, there was no interaction between treatment and age groups with dabigatran 150 mg twice daily, rivaroxaban daily, apixaban twice daily, or higher-dose edoxaban daily
DOAC efficacy and safety in patients with AF and diabetes
Diabetes is a risk factor for stroke among patients with AF.40,41 The RE-LY, ROCKET AF, and ARISTOTLE trials included 4221 (23.3%), 5695 (39.9%), and 4547 (25.0%) patients with diabetes mellitus, respectively.31,44,45 The relative reduction in stroke or SEE risk with dabigatran 150 mg twice daily, rivaroxaban daily, and apixaban twice daily
DOAC efficacy and safety in patients with AF and vascular disease
Vascular disease, comprising MI, peripheral artery disease (PAD), or aortic plaque, is a risk factor for stroke in AF. In RE-LY, 5650 (31.2%) patients with a history of CAD or prior MI were enrolled (dabigatran 110 mg,
For stroke or SEE prevention, the relative benefits of anticoagulation with dabigatran 150 mg twice daily, rivaroxaban daily, or apixaban twice daily
A total of 839 (5.9%) patients with PAD were enrolled in ROCKET AF,
50
and 841 (4.0%) in ENGAGE AF-TIMI 48.
51
Regardless of PAD status, stroke or SEE incidence was comparable for patients receiving rivaroxaban or higher-dose edoxaban
DOAC efficacy and safety in female patients with AF
The risk for stroke is greater in women than in men with AF.
41
For stroke or SEE incidence, the relative benefit of rivaroxaban, apixaban, or higher-dose edoxaban
Dosing considerations for renal function
Dosing recommendation for stroke and SEE prevention in NVAF are provided in Table 2. In general, the FDA-approved dosing regimens for dabigatran (150 mg), rivaroxaban, and apixaban55
–57 are consistent with the dosing strategies employed in the RE-LY, ROCKET AF, and ARISTOTLE trials,6,7,9 and with the findings of secondary analyses, which demonstrated that the relative benefit of these DOACs
For edoxaban, secondary analysis by renal function suggested a nonsignificant trend toward lower relative efficacy for the higher-dose edoxaban regimen
The US FDA recommends a dose reduction for patients to 75 mg twice daily for patients with CrCL 30–50 ml/min and receiving concomitant P-gp inhibitors based on pharmacokinetics despite the fact that RE-LY did not include a dose reduction.6,55 ROCKET AF did not enroll patients with CrCL 15–30 ml/min, but the US FDA suggests that 15 mg rivaroxaban once daily in these patients will result in serum concentrations of rivaroxaban similar to those in patients with normal renal function.
56
Similarly, ARISTOTLE and ROCKET AF did not enroll patients with end-stage renal disease on dialysis. However, based on the results of pharmacodynamics studies, the US FDA recommends the administration of 5 mg apixaban twice daily or 15 mg rivaroxaban once daily in patients with intermittent hemodialysis.56,57 All four DOACs are substrates of P-gp, while rivaroxaban, apixaban, and edoxaban undergo CYP3A4 metabolism. Secondary analysis of ROCKET AF and ENGAGE AF-TIMI 48 demonstrated that the concomitant P-gp and CYP3A4 inhibitor amiodarone did not affect the relative primary efficacy and safety outcomes for rivaroxaban or edoxaban
Conclusion
DOACs have been approved for the reduction of stroke and SEE risk in patients with NVAF based on the findings of the pivotal phase III randomized control trials. Available secondary analyses indicate no treatment interactions for any of the DOACs in patient subgroups at an increased risk for stroke, with regard to stroke or SEE prevention. Overall, results of secondary analyses indicate that the recommended dosing strategy for each of the DOACs produces a consistent anticoagulant effect across a diverse patient population. However, as the phase III clinical trials were not designed to specifically assess the efficacy and safety of DOACs in these high-risk population subgroups, the findings of the exploratory analyses should be considered hypothesis generating.
Footnotes
Funding
Medical writing support was provided by Sameera Kongara, PhD, of AlphaBioCom, LLC, King of Prussia, PA, and funded by Daiichi Sankyo, Inc.
Conflict of interest statement
Dr Stacy is a speaker and a consultant for Janssen Pharmaceuticals. Dr Richter reports no conflicts of interest.

