Abstract
Drug-induced QT interval prolongation may increase the risk of sudden cardiac death or ventricular arrhythmias (SCD/VA), and therefore affects the safety profile of medications. Administrative databases can be used to inform pharmacoepidemiologic drug safety studies for such rare events. In order to compare event rates between studies, validated operational definitions of these events are needed. We conducted a systematic literature review in PubMed to identify algorithms for SCD/VA. Twenty-two studies were included in the review. Fifteen (68%) studies evaluated International Classification of Diseases, 9th revision (ICD-9) based medical data, of which six utilized a common, validated operational definition. This algorithm was based on principal hospitalization discharge diagnosis or first-listed emergency department visit diagnosis, with an average positive predictive value (PPV) of 85%. Four studies evaluated ICD-9 based death data, of which three utilized a common algorithm with an average PPV of 88%. Further validation of ICD, 10th revision algorithms are needed. In conclusion, we identified a validated algorithm for SCD/VA in medical data, as well as in death data. As such, to ensure comparability between new research and the existing literature, pharmacoepidemiologic research in this area should utilize common, validated algorithms, such as the ones identified in our review, to operationally define these events.
Introduction
Sudden cardiac arrest is a severe condition in which the heart stops beating suddenly and unexpectedly and often leads to sudden cardiac death outside of the hospital or in the emergency room. 1 Unexpected sudden cardiac death is rare, with an estimated incidence ranging from 50 to 100 per 100,000 individuals per year in Europe and North America.2,3 Acute ventricular arrhythmia may account for over 80% of sudden cardiac deaths. 4
Retrospective pharmacoepidemiologic studies are used to assess risks attributed to drug exposures.5,6 Even though the relationship between QT interval prolongation and sudden cardiac death or ventricular arrhythmias (SCD/VA) remains putative, drug-induced QT interval prolongation may increase the risk of SCD/VA and therefore affects the safety profile of medications, which can result in black box warnings and drug withdrawals.7,8 Terfenadine and cisapride were withdrawn in the US in 1998 and 2000 respectively due to their association with QT prolongation and ventricular arrhythmia.9,10 More recently, in 2013, azithromycin received a black box warning for potential risk of QT prolongation and fatal cardiac arrhythmias based on the findings of a large retrospective pharmacoepidemiologic study.11,12
In this era of big data, particularly for studies of rare outcomes, such as SCD/VA, a valid operational definition of the outcome is needed. Physician review of an electrocardiogram (ECG) is the gold standard for identifying QT prolongation, however it has limited use in retrospective database studies. 13 First, ECG results are often not available in administrative databases. Further, a study identifying events by manual medical record review of ECGs and physician notes would likely be underpowered to quantify the exposure–outcome relationship. Lastly, ECG results are not available for cardiac events which occurred outside of the hospital and would result in underestimation of these events.
A number of retrospective pharmacoepidemiologic drug safety studies have sought to quantify the association between these rare outcomes and different medication exposures, however the algorithms identifying SCD/VA have varied between studies.14–18 Further the performance of these varying algorithms has not been compared. The objective of this systematic literature review was to identify the operational definitions of ventricular arrhythmias and sudden cardiac death used in retrospective database studies and compare validation results between algorithms.
Methods
A systematic literature review in the PubMed electronic databases was conducted to identify retrospective studies from peer-reviewed journals. A predetermined search strategy developed by researchers from the US Food and Drug Administration (FDA) Sentinel Initiative (Supplemental Table 1) was used to select qualifying studies published between 1 January 2000 and 31 August 2016. 19 The FDA Sentinel Initiative has sought to improve the surveillance of safety events related to marketed medications, through the creation of robust databases and improved monitoring methods. 20 This literature review was limited to studies in humans and published in English. Other inclusion criteria were retrospective studies in administrative databases; studies that identified SCD/VA; and studies that specified codes for SCD/VA. Titles and abstracts of studies identified by the search were screened against the inclusion criteria. Qualifying or uncertain studies from title and abstract review underwent further full-text review for selection. References of selected studies were also examined for inclusion. Data extracted included operational definitions and administrative codes used for identifying SCD/VA. Other aspects of study design including publication year, study setting and population, and sample size were also collected. Validation methods and results were extracted when validation was conducted. This literature review was conducted according to the standards set forth in the Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) statement. 21
Results
The search strategy identified 1237 studies. After title and abstract review, 57 studies were selected for full-text review, after which 22 (39%) studies were selected for inclusion. Figure 1 shows the literature search, review, and selection process.

Flow chart of literature search and selection process.
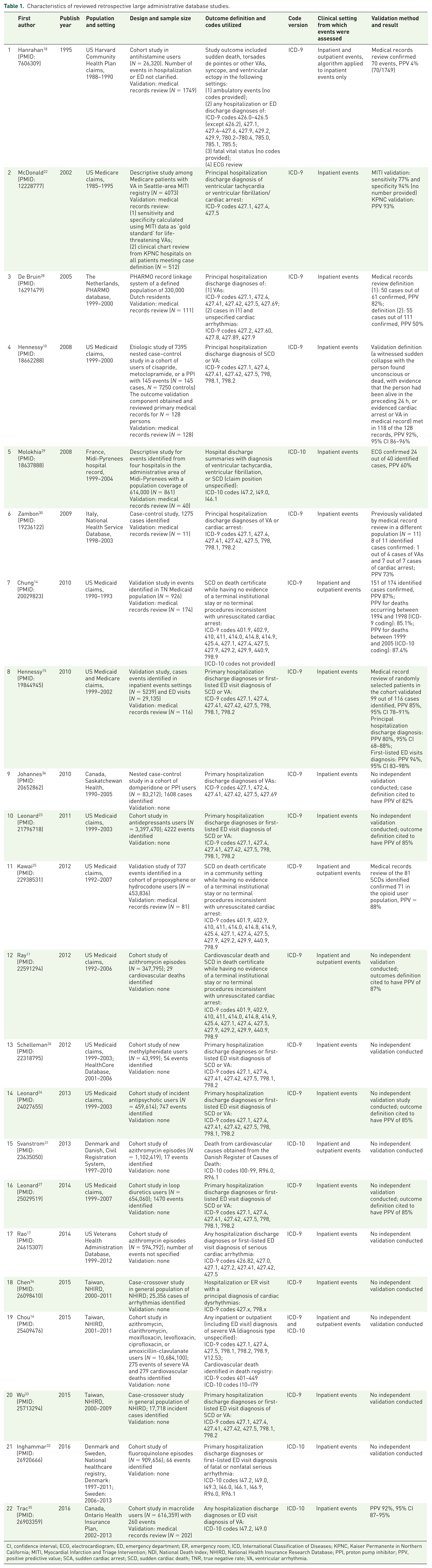
Table 1 describes the characteristics of the included studies. More than half of the included studies were published after 2012, which was the year a previous literature review of validated methods for identifying SCD/VA was published.
19
Twelve of the included studies (55%) were conducted in US populations, using Medicare or Medicaid data (
Characteristics of reviewed retrospective large administrative database studies.
CI, confidence interval; ECG, electrocardiogram; ED, emergency department; ER, emergency room; ICD, International Classification of Diseases; KPNC, Kaiser Permanente in Northern California; MITI, Myocardial Infarction and Triage Intervention; NDI, National Death Index; NHIRD, National Health Insurance Research Database; PPI, proton pump inhibitor; PPV, positive predictive value; SCA, sudden cardiac arrest; SCD, sudden cardiac death; TNR, true negative rate; VA, ventricular arrhythmia.
Algorithms applied to medical data
SCD/VA was identified from medical data in 15 studies (68.2%) using ICD-9 and 3 studies (14%) using ICD-10. Shared by six ICD-9 medical data studies, the most frequently adopted algorithm (Table 2) included diagnosis codes identified from inpatient discharges and emergency department (ED) visits.10,15,23,24,27,30 Validation was conducted in three of these studies by confirmation of SCD/VA through medical record review. When principal inpatient discharge diagnosis and first-listed ED visit diagnosis were assessed together, the PPVs ranged from 73% to 100%.10,15,30 Considering both principal and nonprincipal inpatient discharge diagnoses, the algorithm was found to have a 92% positive predictive value (PPV) for identifying SCD/VA in one study. 10 Another study identified a higher PPV with principal ED visit diagnosis (94%) than with principal discharge diagnosis (80%). 15
Variations of this algorithm have also been used in several studies. One variation of the algorithm (excluded ICD-9 code 798) was used in a Taiwanese study (National Health Insurance Research Database) and a Medicaid/HealthCore study but neither study evaluated the validation of the algorithm.26,33 Another variation (only included ICD-9 codes 427.1, 427.4, and 427.5) of this algorithm identified ventricular arrhythmias from principal inpatient discharge diagnoses using Medicare data. 22 This study validated the reduced algorithm in a registry cohort of confirmed life-threatening ventricular arrhythmia cases, resulting in a sensitivity of 77% and specificity of 94%. This algorithm also demonstrated a PPV of 93% as validated by medical record review of 30 cases. 22 A third variation (with additional ICD-9 code 427.69) identified SCD/VA from inpatient hospitalizations in the Netherlands. Medical record review demonstrated a PPV of 82%. Addition of codes for unspecified cardiac arrhythmia codes lowered the PPV to 50%.22,28
The next most common algorithm for arrhythmias was utilized in two studies, one from the Netherlands and the other from Canada.28,36 The algorithm identified ventricular arrhythmias and cardiac arrest from hospital stays with primary diagnoses of arrhythmias. Only the Dutch study validated the algorithm, where a review of medical records was used to verify the ICD-9 codes used for ventricular arrhythmia and cardiac arrest (PPV 82%) and ICD-9 codes for unspecified cardiac arrhythmias (PPV 10%). 28 The five other studies each used a different algorithm to identify SCD/VA. The Harvard community health plan study used select ICD-9 subcodes under 426, 427, 429, 780, and 785 from hospital stays and ED visits, with a low PPV of 4%. 18 The United Healthcare study included office visits and inpatient stays with diagnoses of arrhythmic events (ICD-9 codes 426.x, 427.x) but this approach also had low a PPV of 10% based on a review of medical records. 9 Three other algorithms were used in two of the Taiwanese studies and the Veterans Health Administration study, and none of these studies validated their operational definition.16,17,34
ICD-10 diagnosis codes for acute ventricular arrhythmia were used in three studies (13.6%). A French study, 29 a Denmark-Sweden binational study, 32 and one of the Canadian studies 35 each used a different algorithm for identifying QT prolongation in hospital stays or ED visits. The Canadian study used ICD-10 codes I47.2 and I49.0 and a manual chart review was conducted in 202 charts resulting in a PPV of 92%. 35 The French study used ICD-10 codes I46.1, I47.2, and I49.0 from hospital discharge summaries. In comparison with ECG records, this algorithm had a PPV of 60%. 29 The binational study used ICD-10 codes I47.2, I49.0, I49.3, I46.0, I46.1, I46.9, R96.0, and R96.1 and no validation was conducted. 32
Algorithms applied to death data
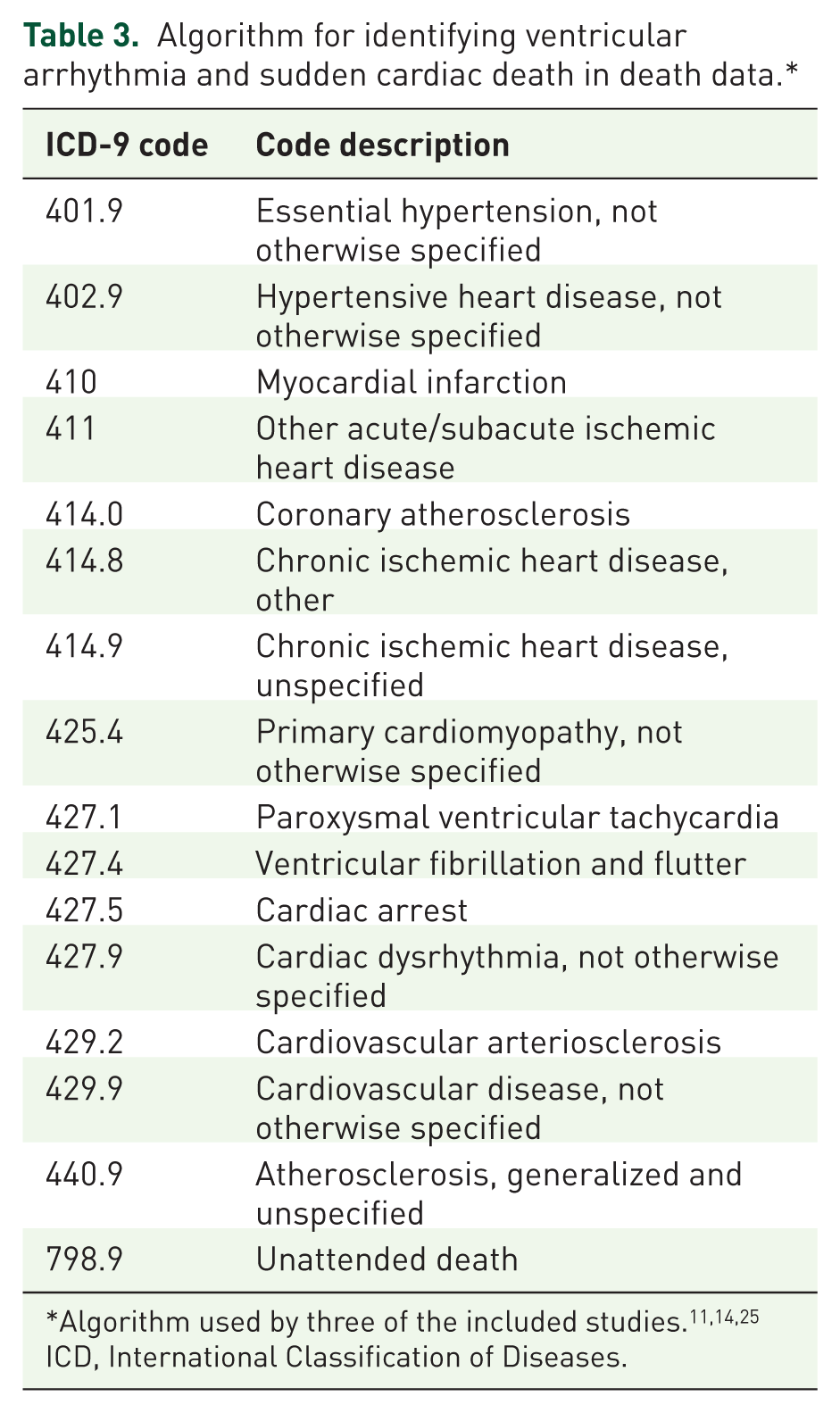
Five of the included studies (23%) utilized death certificate or death registry data to identify SCD (ICD-9,
Another algorithm conducted with Taiwanese death registry data defined the study outcome as unspecific cardiovascular death (ICD-9 codes 401–449 and ICD-10 codes I10-I79). 16 The Danish death registry study identified death from cardiovascular causes using ICD-10 codes. 31 Codes used in the Danish study were I00-I99 (diseases of the circulatory system) and R96.x (other sudden death, cause unknown). 31 Neither study performed validation of their algorithms.
Discussion
We identified validated operational definitions for identifying ventricular arrhythmias and sudden cardiac death in retrospective database studies. The most frequently used algorithms in medical data (Table 2) and death registries (Table 3) had an average PPV of 85% and 88%, respectively. These two algorithms were validated in various patient populations, including Medicaid and Medicare, administrative claims databases, and several European and Canadian databases. For ICD-9 algorithms in medical data, the PPV was highest when limited to principal inpatient or ED discharge diagnoses, and addition of less specific subcodes decreased the PPV.9,10 Though one ICD-10 algorithm in medical data was found to have a high PPV, this algorithm has not been used or validated in another database. 35 Less than a quarter of the included studies identified SCD from death certificates or death registries, however one algorithm using ICD-9 codes was found to have a high PPV for identifying sudden cardiac death among patients with a low risk of unexpected death. 14
Our study has a few limitations. First, the literature search terms might miss some other studies that also identified ventricular arrhythmias and sudden cardiac death in retrospective databases. We attempted to address this limitation by examining the references of reviewed studies for additional studies and found no additional eligible studies in the references. Second, the true incidence of ventricular arrhythmias and sudden cardiac death are unknown, since such events may originate in the outpatient setting and therefore the diagnosis may not be recorded in medical data or may be absent as a cause of death. Hence the incidence and prevalence are likely to be underestimated. As such, validation studies usually only calculate the PPV but not sensitivity or specificity. However, as a PPV is the proportion of true positives in those that tested positive, validated algorithms with high PPV may accurately identify cases captured in retrospective administrative databases.
Conclusions
Our study identified a validated algorithm for ventricular arrhythmias and sudden cardiac death in medical data, as well as a validated operational definition for sudden cardiac death in death data. Consistency between studies is necessary for establishing causal relationships between medications and rare adverse events, such as ventricular arrhythmias and sudden cardiac death. Further, transparency in the reporting of these algorithms is essential for understanding differences between studies and should include lists of codes and sufficient data source details (primary
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declare that there is no conflict of interest.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.