Abstract
Hyperuricemia leads to endothelial dysfunction and insulin resistance, and has been associated with diseases such as hypertension. Antihypertensive drugs modify serum uric acid levels, however, few data are available about their combinations on uricemia. In this study we evaluate the effect of two combinations of losartan, with amlodipine or with hydrochlorothiazide, on serum uric acid levels in hypertensive patients.
Methods:
A total of 60 hypertensive patients were randomized in two groups; group LA received losartan/amlodipine (100/5 mg) once a day, whereas LH group received losartan hydrochlorothiazide (100/12.5 mg) once a day for 3 months. In both groups serum uric acid levels were measured at the beginning and end of the study. Patients were evaluated monthly for blood pressure (BP) and adverse events. Statistical analysis was performed with a two-way analysis of variance (ANOVA) for repeated measures.
Results:
All patients experienced a significant reduction of BP to the same extent (LA 155/94 to 123/79, LH 157/92 to 124/78 mmHg, p > 0.05). In the LA group, serum uric acid decreased from 6.5 ± 1.6 to 4.6 ± 1.3 mg/ml (p = 0.0001), whereas in the LH group there was a nonsignificant increase from 5.82 ± 1.4 to 5.85 ± 1.5 mg/ml, (p = 0.936). When both groups were compared, we found a significant reduction (p < 0.00013) on serum uric acid levels in the LA group.
Conclusions:
Both combinations decrease BP values to the same extent, however, LA combination showed a reduction on serum uric acid levels, which may contribute to a reduction in the metabolic risk in hypertensive patients.
Introduction
Uric acid is the end-product of purine metabolism in humans; hyperuricemia has been associated with activation or the renin–angiotensin system. Also, a chronic increase in uricemia reduces nitric oxide levels in endothelial cells because uric acid reacts with the radical, resulting in the formation of nitrosated uric acid and depletion of nitric oxide; both pathways lead to endothelial dysfunction, inflammation, insulin resistance and an increase in blood pressure (BP) [Rubio-Guerra et al. 2016].
In fact, several epidemiological studies have shown that hyperuricemia is associated with hypertension, cardiovascular disease and metabolic syndrome [Nagahama et al. 2015]. Approximately 25% of patients with hypertension have hyperuricemia, while approximately 30% of patients with hyperuricemia or gout suffer hypertension [Nishida et al. 2013].
Antihypertensive agents may modify serum uric acid levels; it is well know that thiazides increase uricemia [Roush et al. 2014], whereas losartan decreases it [Nishida et al. 2013]. The role of calcium channel blockers are unclear. Tsouli and colleagues reported that nifedipine and verapamil have no effect, whereas amlodipine and felodipine decrease serum uric acid levels [Tsouli et al. 2006]. Recent studies suggest that hyperuricemia induced by thiazides can be associated with increased cardiovascular risk [Reungjui et al. 2008].
Despite the availability and efficacy of antihypertensive drugs, almost 70% of hypertensive patients do not reach the recommended treatment target of <140/90 mmHg with monotherapy, making combination therapy necessary to achieve those targets with minimal adverse effects [Rubio-Guerra et al. 2009]. Clinicians have many drug choices; the combinations of inhibitors of the renin–angiotensin-aldosterone system (RAAS) plus calcium antagonists, or a thiazide are efficacious and well tolerated, and both represent a real addition to the available antihypertensive treatment options [Rubio-Guerra et al. 2009]. However, few data are available on the effect of antihypertensive combination therapy on serum uric acid levels.
The aim of this study was to evaluate the effect of two combinations of losartan, with amlodipine or with hydrochlorothiazide, on serum uric acid levels in hypertensive patients.
Methods
We evaluated 60 hypertensive patients referred from primary care clinics with uncontrolled hypertension (>140/90 mmHg) despite more than 3 months of treatment with monotherapy, or with stage 2 uncontrolled hypertension without previous drug therapy (Figure 1). All patients were randomized in two groups of 30 patients each; group LA received losartan/amlodipine (100/5 mg) once a day for 3 months, whereas the LH group received losartan hydrochlorothiazide (100/12.5 mg) once a day for 3 months. A washout period was not conducted because the patients lacked an adequate response to previous antihypertensive treatment.
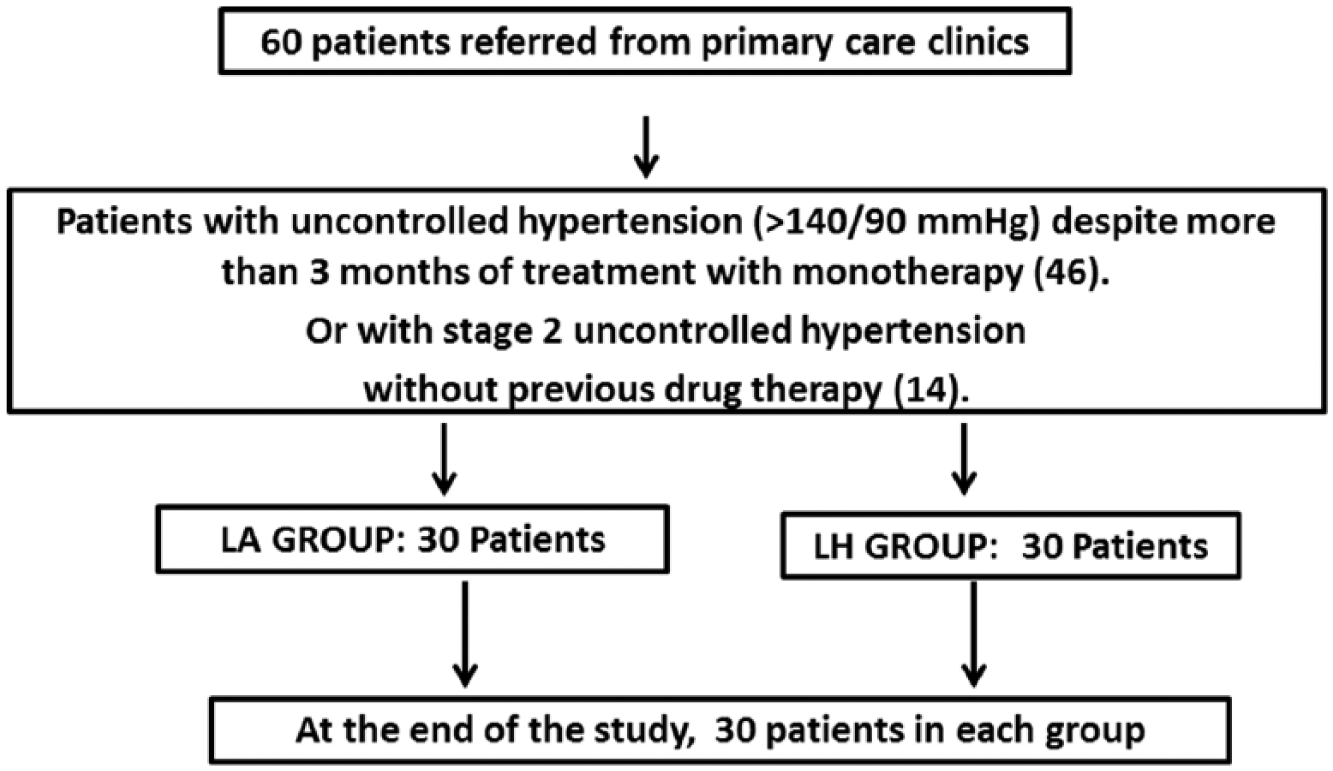
Flow chart of the selection process.
Patients were evaluated monthly for BP and adverse events. At each evaluation, BP was recorded in triplicate with a mercurial sphygmomanometer in the sitting position after a 5-min rest and at 3-min intervals; an average of the three measurements was recorded. Heart rate was also recorded at each evaluation.
Fasting serum uric acid (enzymatic method) was measured at the beginning and end of the study in both groups. Serum glucose (glucose oxidase), creatinine (JAFFE), lipid profile (CHODPAP) and triglycerides (triglyceride-pap) were also performed in all subjects. All venous samples were collected in the morning after a 12 h overnight fast. The determinations were performed by personnel blinded to the study.
Patients with any of the following diagnoses were excluded from the study: decompensate diabetes mellitus (fasting blood glucose > 250 mg/dl); heart, hepatic, or renal failure; evidence of valvular heart disease; heart block or cardiac arrhythmia; secondary hypertension; acute coronary syndrome or cerebrovascular disease 6 months before the beginning of the study; hyperkalemia, bradycardia, history of angioedema, autoimmune disease, cancer or chemotherapy, hypersensitivity and pregnancy; or a history of alcohol or psychotropic drug abuse.
The study was conducted with the approval of the Research and Medical Ethics Committee of our hospital, in accordance with the Helsinki Declaration. Participants gave informed, written consent before their inclusion in the study protocol.
Statistical analysis
Data are presented as mean ± standard deviation; the statistical analysis was performed with a two-way analysis of variance (ANOVA) for repeated measures. A p < 0.05 was considered significant.
Results
Basal characteristics of patients are shown in Table 1. No difference was found between the two groups.
Basal characteristics of patients.

LA, losartan/amlodipine; LH, losartan hydrochlorothiazide; Ns, not significant.
All patients experienced a significant reduction of BP to the same extent (LA 155/94 to 123/79, LH 157/92 to 124/78 mmHg, p > 0.05). Overall, two patients in each group required titration of dose. Previous antihypertensive treatment is depicted in Table 2.
Previous antihypertensive treatment.

ACE, angiotensin converting enzyme; ARB, angiotensin receptor blocker; LA, losartan/amlodipine; LH, losartan hydrochlorothiazide.
Serum uric acid levels decreased in the LA group from 6.5 ± 1.6 to 4.6 ± 1.3 mg/ml (p = 0.0001), whereas the LH group had a nonsignificant increase in uricemia, from 5.7 ± 1.4 to 5.75 ± 1.5 mg/ml, (p = 0.936). When both groups were compared, we found a significant reduction (p < 0.00013) in serum uric acid levels in the LA group.
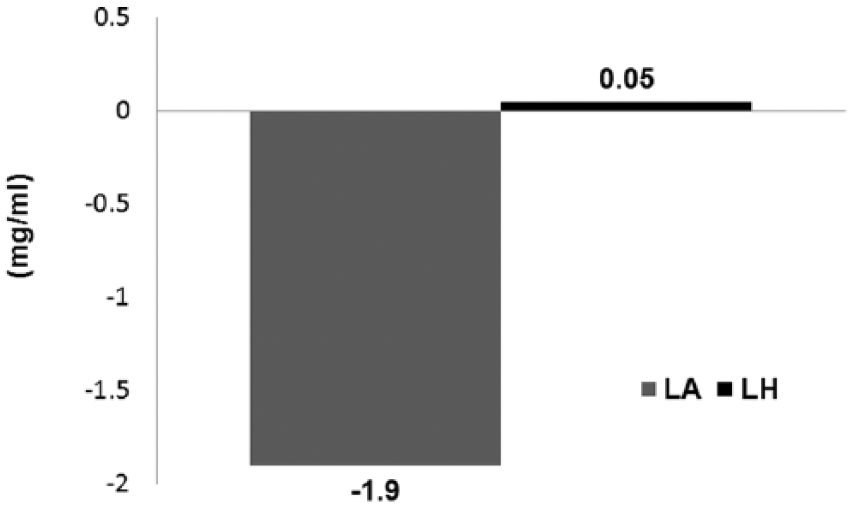
Effect of LA or LH treatment on serum uric acid levels.
Serum creatinine levels were reduced in the LA group from 0.77 ± 0.19 to 0.62 ± 0.15 mg/ml (p = 0.001), whereas in the LH group no changes on such levels were found, 0.78 ± 0.45 to 0.78 ± 0.36 (p > 0.05), when both groups were compared, a significant difference was observed in the LA group (p = 0.028).
Discussion
To achieve therapeutic targets and reduce cardiovascular risk in patients uncontrolled with monotherapy, a combination of antihypertensive agents is recommended, most hypertension guidelines recognize that most patients will require two or more antihypertensive drugs. Furthermore, guidelines suggest the initial use of combination therapy if BP is > 160/100 mmHg, or to combine drugs with different mechanism of action when the goal of BP < 140/90 mmHg is not achieved with monotherapy [Rubio-Guerra et al. 2009].
In this study we found a significant reduction of serum uric acid levels with losartan plus amlodipine, whereas the combination losartan and hydrochlorothiazide induced a modest, but insignificant, increase in such levels.
Losartan reduces serum uric acid levels because the drug blocks the uric acid transporter (URATI) in renal tubules, whereas other angiotensin receptor blockers (ARBs) such as valsartan may increase them [Nishida et al. 2013]. On the other hand, thiazide diuretics increase uricemia by increasing the net renal reabsorption of urate. Several studies have been performed evaluating the effect of losartan on diuretic-induced hyperuricemia. Matsumura and colleagues found a significant reduction of serum uric acid levels when losartan was added to thiazide therapy when compared with treatment with others ARBs plus a diuretic [Matsumura et al. 2015]; whereas other studies have found a discreet increase in uric acid levels with that combination [Hosoya et al. 2012], as in our study.
Calcium channel blockers decrease serum uric acid levels [Tsouli et al. 2006; Nishida et al. 2013]. In fact, in the A Coronary disease Trial Investigation Outcome with Nifedipine (ACTION) gastro-intestinal therapeutic system (GITS) trial, nifedipine reduced uric acid levels [Ruilope et al. 2007]. The possible pathways are: an increase in nitrogen monoxide excretion [Masaki et al. 2016], renal vasodilatory effects [Ruilope et al. 2007], and a reduction in the production of uric acid precursors such as hypoxanthine in skeletal muscle in patients with hypertension or insulin resistance [Mizuta et al. 2010; Masaki et al. 2016]. Yagi and colleagues found that irbesartan plus a high dose of amlodipine (10 mg) decreased serum uric acid concentrations in patients shifted from treatment with an ARB plus a low or regular dose of a calcium channel blocker [Yagi et al. 2015]. In the COLM study, both combinations of olmesartan, with amlodipine or with hydrochlorothiazide, increased uricemia in elderly patients, although the increase with thiazide was significantly higher than with the amlodipine combination [Saruta et al. 2015]. Indeed, both ARBs lack the effect of losartan on uric acid.
We found a nonsignificant increase in uricemia with the LH combination, while LA significantly reduced serum uric acid levels. We hypothesize that the individual effect of both drugs on uricemia are counteracted in the combination of LH, whereas the capacity of amlodipine to lower serum uric acid levels was added to the uricosuric effects of losartan. The result is the significant reduction in uricemia that we observed. Our results agree with those of Oshikawa and colleagues. They found that LA in combination reduced uricemia in patients with serum uric acid levels >5.6 mg/ml [Oshikawa et al. 2014]. They also found that the LH combination increases uric acid in patients with levels <5.6 mg/ml. Both findings are similar to our study, where LH produced a nonsignificant increase in uricemia in our patients, with uric acid levels of 5.8 mg/ml.
The combinations of an ARB plus a diuretic, or with a calcium channel blocker, have a synergistic effect on BP [Rubio-Guerra et al. 2009]. Several studies [Oshikawa et al. 2014; Martinez-Martin et al. 2011] have shown that both combinations decrease BP to the same extent in hypertensive patients, as we found in this work. However, beyond the effects on BP reduction, our results have therapeutic implications; LA in combination seems to be a good option for the treatment of hypertensive patients with gout, and also for patients with asymptomatic hyperuricemia.
We found that creatinine levels were reduced in the LA group. Nishiwaki and colleagues also found a significant reduction on creatinine with the LA combination, perhaps due to a renoprotective effect of the combination between an ARB and a calcium channel blocker in hypertensive patients by reduction of intraglomerular pressure [Nishiwaki et al. 2013]. Certainly, several authors have described a renoprotective effect of both RAAS inhibitors and calcium channel blockers by different mechanisms unrelated to their antihypertensive effects [Tabur et al. 2015; Brenner et al. 2001; Rubio-Guerra et al. 2008]. No change was noted in our LH group. Usually, when thiazide therapy is initiated, a modest increase in creatinine levels may occur due to volume contraction [Reungjui et al. 2008], and such an effect was found in the Nishiwaki trial [Nishiwaki et al. 2013], but not by our group.
As a limitation to our study; this is a short-term study with a small sample size.
In conclusion, both combinations reduced BP to the same extent. However, LA significantly reduced serum uric acid levels, whereas the combination of LH induced a nonsignificant increase in such levels.
LA in combination seems to be a good option for the treatment of hypertensive patients with gout, and also for patients with asymptomatic hyperuricemia.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declare that there is no conflict of interest.
