Abstract
Background:
The extent to which treatment with amlodipine/losartan may help patients and their doctors from Andean countries to reach their treatment goals remains unknown.
Methods:
We conducted a quasi-experimental, validation study in which patients with primary, uncomplicated hypertension received open-label, fixed combination therapy with amlodipine/losartan 5/50 mg or 5/100 mg according to their baseline blood pressure (BP). The primary outcome was echocardiographic reduction of left ventricular mass (LMV) after 6 months of treatment. Secondary outcomes were changes in BP, uricemia and albuminuria from baseline. We defined patients with adequately controlled hypertension as those with decreases in at least three out of these four variables.
Results:
From 109 eligible patients with hypertension, 99 started the protocol (70 under the low-dose combination). Echocardiography indices had mutual correlation and contributed independently to the primary outcome (Cronbach’s α index = 0.66). Intention-to-treat analysis showed that 60 patients [60.6%, 95% confidence interval (CI) 50.3–70.3] had reduced LVM. Echocardiographic changes were not associated with dose levels. Favorable changes in BP, uricemia and microabuminuria were seen in 77, 64 and 76 patients respectively. After 6 months there were 64 (95% CI 54–74%) patients with adequately controlled hypertension.
Conclusion:
Every two of three patients of this sample of Andean countries reached adequate control after 6 months of combined antihypertensive treatment with amlodipine/losartan.
Introduction
Hypertension is a common medical condition, and a well known risk factor for cardiovascular disease, that is modifiable with lifestyle changes and medications. This fact has promoted successful control initiatives both at societal and individual levels in multiple settings [Sliwa et al. 2011]. But despite the progress made so far, large gaps subsist in its diagnosis, awareness and control in high-, middle- and low-income countries [Bautista et al. 2002; Herrera et al. 2003; Egan et al. 2010].
Most patients with hypertension require medications, often combinations of two or more agents modifying different physiological pathways [Wrobel et al. 2011]. Combined treatments may be more efficacious and safe by reducing the dose of individual components. In general, starting combined therapies as well as escalating treatments allow for a faster and sustained reduction of blood pressure (BP) [Glynn et al. 2010; Fitzgerald, 2011; Brook and Weder, 2011].
Calcium channel blockers and renin–angiotensin system inhibitors have been shown as useful agents, suitable for a combined treatment for hypertension. However, most of this evidence comes from studies conducted in Europe, North America, or even South America, but rarely including Andean countries (i.e. Colombia, Venezuela, Ecuador, Peru and Bolivia). In the ASCOT-BPLA trial, the rate of cardiovascular events and mortality was significantly lower among patients treated with this combination over the comparator in the participating countries [Dahlöf et al. 2005]. Despite the potential for extrapolating this benefit to patients from Andean countries, it is still uncertain the proportion of patients from the region who, after a reasonable period of treatment (i.e. 3–6 months) reach therapeutic goals, adhering to the starting medication.
One of the goals of BP treatment is preventing left ventricular hypertrophy (LVH) and remodeling. The process is initiated by tissue injury, and mediated by inflammatory activity and hemodynamic changes. These are among the well known target organ consequences of uncontrolled hypertension, increasing further cardiovascular risk. One in three people with hypertension have LVH [Cuspidi et al. 2012; Ajayi et al. 2011; Cortés et al. 2012]. Controlling BP is associated with regression of this hypertrophy, thus reducing future cardiovascular events [Devereux et al. 2004; du Cailar et al. 2010].
This study sought to estimate the magnitude of change in BP and left ventricular mass (LVM) values, as well as the rates of adherence after initiation of combined treatment with amlodipine/losartan in patients from Andean countries.
Materials and methods
Study design
We designed a quasi-experimental, validation study to generate preliminary estimates of the 6-month change after a treatment with amlodipine/losartan in a number of predefined outcomes (see below). Study investigators screened eligible patients, asking for their consent to participate (by signing a form already approved by their institutional boards). Independent assessors, unaware of the study purpose, measured all study outcomes. Study sponsors were responsible for administering the database (and take responsibility for the integrity of the information), but had no role in this analysis, interpretation of the data, or writing this manuscript. The study was conducted in accordance with the Declaration of Helsinki.
Study population
Eligible patients were adults with established diagnosis of uncomplicated hypertension, living in one of the following Andean countries: Colombia, Venezuela, Ecuador and Peru. We excluded patients with secondary or malignant hypertension, severe obesity, gout, chronic kidney disease (serum creatinine over 1.5 mg/dl), history of (coronary or valve) heart disease, left ventricular dysfunction (ejection fraction below 40%), or cerebrovascular disease.
Interventions and variables
Study participants had a baseline assessment, including medical history, physical exam, and diagnostic workup (serum fasting glucose, lipids, uric acid, and creatinine levels; urinalysis; electrocardiogram and echocardiogram; and proteins and uric acid in 24 h urine samples). BP was the mean of three recordings, taken 3 min apart.
The study drugs were offered in accordance with BP levels, as follows: amlodipine/losartan 5/50 mg once a day, when BP was less than 160/100 mmHg; amlodipine/losartan 5/100 mg once a day, when BP was at least 160/100 mmHg. All participants were followed for a 28-week period to record study endpoints, with repeated BP measurements at weeks 6, 12, and 20. BP control (treatment goal) was defined as having a consistent value below 140/90 mmHg. After initial treatment allocation, the protocol allowed a stepwise dosage increase (to 5/100, or a third level with 10/100) when patients had not reached their BP goals by visits two or three respectively. Variables collected at the final visit were BP, serum uric acid, albumin in a 24 h urine sample, electrocardiogram, and echocardiogram.
Study endpoints
The primary outcome was the proportion of patients with a reduction in at least three of the following four echocardiographic indices, reflecting the response of the left ventricle to hypertension: LVM index (LVMI); posterior wall thickness (PWT); septal thickness (ST), and relative wall thickness (RWT). Our secondary outcomes were the mean before–after change for the above echocardiographic indices (both individually and as a pooled estimate of percentage of change); and the proportion of participants who were ‘adequately treated’, defined as the percentage of participants reaching at least three out of the following four goals for treatment: BP levels less than 140/90 mmHg; reduction of LVM (defined as any reduction in at least three out of the four aforementioned echocardiographic indices); any reduction in uricemia; or any reduction or stabilization in albuminuria. Estimation of the proportions set as study outcomes followed the intention-to-treat principle. That is, the denominator was the actual number of patients starting the protocol, regardless of the availability to provide outcome data during the follow-up visits.
Statistical analysis
We first cross-validated the database, using counts of missing data and correlation analysis among 10 randomly selected variables. Such examination did not trigger warning signals. We used SPSS software package version 17.0 for statistical analyses. We described clinical and diagnostic characteristics at baseline using means with standard deviations or counts with percentages for continuous or categorical variables respectively.
Prior to further analyses of outcome measures, we assessed the relationship among echocardiographic indices defining our primary outcome. We calculated Pearson’s correlation coefficients among these variables. In addition, we computed Cronbach’s α statistics to estimate the relative independence of these four indices to discriminate the regression of LVM after treatment. We predefined a value between 0.6 and 0.8 to be an acceptable measure of independent contribution to assess the response of the left ventricle.
We calculated the proportion [along with 95% confidence intervals (CIs)] of patients who, under different dosages, reached the primary and secondary outcomes of the treatment goals. Our a priori hypothesis was that treatment with amlodipine/losartan was appropriate (i.e. valid) for our target population when at least 50% of the participants reached their goals after 6 months of treatment. Assuming that we will find this proportion to be 66%, we estimated that inclusion of 100 patients provided 87% of statistical power to test the hypothesis of noninferiority (one-tailed, α level of 2.5%), allowing for up to 10% losses to follow up over the follow-up period.
Results
Patients and disposition
Of 114 patients assessed for inclusion, five did not meet our definition of hypertension and were excluded from the study protocol. In the first visit, 66.1% of the participants were given amlodipine/losartan 5/50 mg based on their BP levels (the remaining third received 5/100 mg). Ninety-nine of the 109 included patients (91%) provided outcome data after 6 months of treatment and were considered in the analysis (Figure 1).

Study flow chart.
Baseline characteristics of study population
Our population included participants living in all Andean countries (Ecuador 39; Perú 37; Venezuela 21 and Colombia 12), except Bolivia. Most (100) of our participants were mestizo (a mix of European, Aboriginal and African descent), whereas five were classified as white and four as black. LVH was present in 81–92% of participants, according to the echocardiographic criteria used (Table 1). Levels of total cholesterol, low-density lipoprotein cholesterol and triglycerides were above the upper limit in 43%, 33%, and 44% respectively. The glomerular filtration rate (GFR) was normal in 52%, while the remaining patients had values between 30 and 90 ml/min. Having a GFR between 30 and 60 ml/min was positively associated with having stage II hypertension (p < 0.015). Except for this parameter, there were no differences when comparing hypertension stages in our population.
Baseline characteristics of study participants.
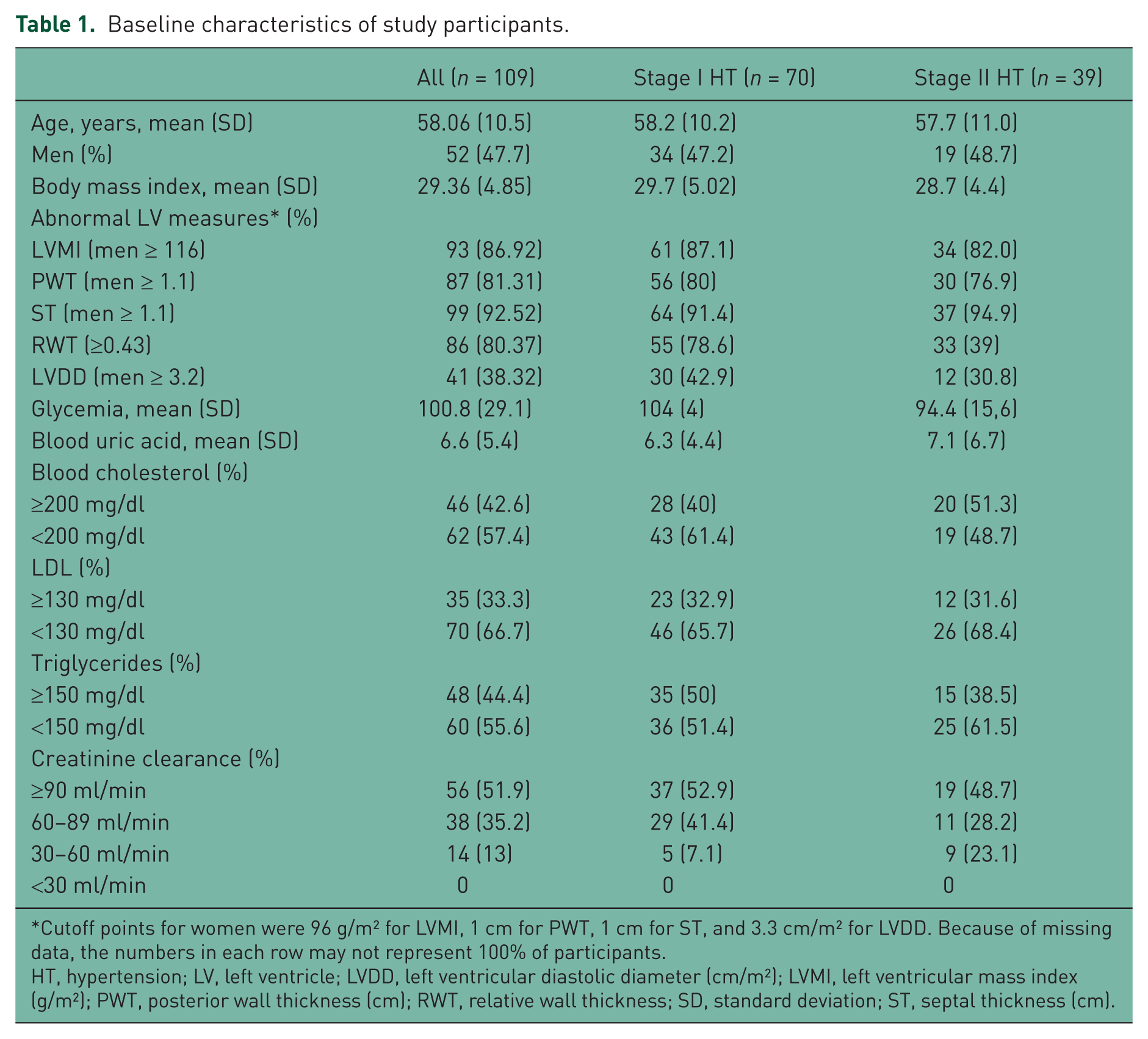
Cutoff points for women were 96 g/m2 for LVMI, 1 cm for PWT, 1 cm for ST, and 3.3 cm/m2 for LVDD. Because of missing data, the numbers in each row may not represent 100% of participants.
HT, hypertension; LV, left ventricle; LVDD, left ventricular diastolic diameter (cm/m2); LVMI, left ventricular mass index (g/m2); PWT, posterior wall thickness (cm); RWT, relative wall thickness; SD, standard deviation; ST, septal thickness (cm).
Echocardiographic findings
Pearson’s correlation coefficient among echocardiographic indices varied between 0.51 and 0.72, except for the relationship between RWT and LVMI (0.17) (Figure 2). The di

Correlation among echocardiographic indices. LVMI, left ventricular mass index; PWT, posterior wall thickness; RWT, relative wall thickness; ST, septal thickness.
Correlation among echocardiographic indices.

p > 0.05. Cronbach’s α = 0.66.
LVMI, left ventricular mass index; PWT, posterior wall thickness; RWT, relative wall thickness; ST, septal thickness.
Treatment goals
Sixty of the 99 patients (60.6%, 95% CI 50.3–70.3%) met our (primary outcome) definition of regression of LVM after 6 months of treatment, with no association with the treatment dose (p = 0.254) (Figure 3). Four of every five (77, 81.1%) of the study participants had BP levels below 140/90 at the final visit (no association with dosage). Levels of both uricemia and albuminuria dropped in 73.6% and 87.4% of the participants respectively (with a significantly higher reduction among those who received higher doses). Finally, 64 patients (95% CI 54–74%) met our secondary outcome, with no difference according to the dosage. There were significant reductions in three of the selected indices (LVMI, PWT, and ST, but not RWT), with a pooled mean change after 6 months of −11.3% (95% CI −10.6 to 11.8) (Figure 4).

Proportion of patients with desirable changes in biomarkers after 6 months of treatment by doses of study drug received. BP, blood pressure; LV, left ventricular.
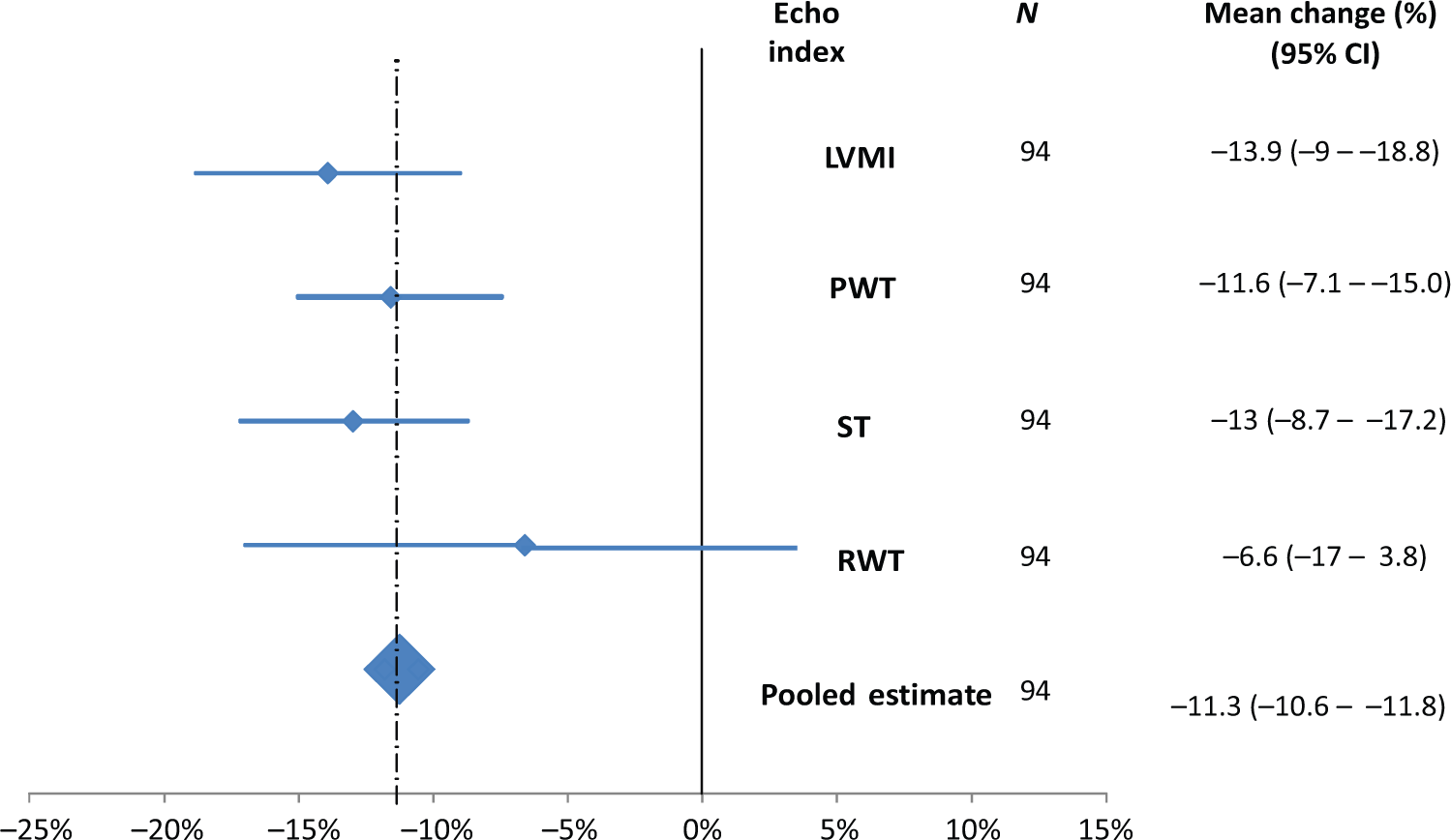
Mean changes in selected left ventricular indices by week 28 of treatment. CI, confidence interval; LVMI, left ventricular mass index; PWT, posterior wall thickness; RWT, relative wall thickness; ST, septal thickness.
Discussion
This approach to the effectiveness of a combined 6-month treatment with amlodipine/losartan suggests that nearly two of every three outpatients from Andean countries within the usual practice would be ‘adequately treated’. As most patients stayed on their treatment, we can consider this combination as a valid option for drug treatment of such patients.
The main strength of this study was the blind, independent assessment of a number of predefined outcomes, known as valid markers of response to treatment. To improve the robustness of our outcome measure, we used a composite of change of four indices (after evaluation of their independent value). We also sought consistency by defining adequacy of treatment as a reduction in at least three of the four predefined endpoints. An additional strength was to follow the intention-to-treat principle, assuming that participants not providing outcome data were not responsive to treatment. Weaknesses of this study include having a relatively small sample size in the absence of a formal sampling process. This raises the concern of selection bias, as the population may not represent the spectrum of outpatients with uncomplicated hypertension. However, the aim of this validation study was to give an initial estimate (through blind assessment) of the response to this treatment for hypertension. Under this consideration, we did not judge having such a formal sampling process to be necessary. Likewise, we thought that a randomized, controlled experiment was not the design required for the type of question posed.
We were encouraged by finding a significant reduction in LVM after a 6-month treatment period in our population. Such a reduction is not only in line with multiple sources of evidence in other populations, but is also in favor of a potential synergy of these agents. In a meta-analysis, Klingbeil and colleagues showed that combined therapy with calcium channel blockers and inhibitors of the renin–angiotensin system (such as amlodipine/losartan) was associated with higher reductions in LVM compared with β blockers and diuretics [Klingbeil et al. 2003]. The effects of these agents on LVM seem to have different origins. The comparison of amlodipine and losartan among patients with hypertension treated with combinations has shown similar reductions in BP, with a significantly higher reduction in LVM associated with losartan [Fogari et al. 2012; Martina et al. 1999]. It has been shown that this combination can reduce myocardial fibrosis as well as the cardiac cell volume. This potentially explains the reduction of LVM seen after treatment [Resl et al. 2011]. Our findings suggest, in the light of the available information, that a reduction in the biomarkers recorded in this study would be appropriate to document adequate control. Also, a 6-month period seems reasonable for patients and clinicians to judge that adequacy to decide on continuing or changing the therapy. As we did not have a control group, many other factors may contribute to the recorded outcomes (i.e. the ‘Hawthorne effect’ in uncontrolled experiments). However, the magnitude of the BP change recorded in the absence of cointerventions (e.g. other antihypertensive medications) makes this less likely.
Our findings did not, however, give support to the adequacy of this treatment on LVM in the mid or long term. But given that this combination has been effective in terms of reduction of major cardiac events [Dahlöf et al. 2005], short-term changes in our surrogate outcomes will likely remain over time in most patients. However, the extent to which patients from Andean countries will adhere to this combined treatment over time is unknown. Such uncertainties require further exploration with substantially more person-years of observation in future studies.
In addition to our main findings, we documented a reduction in the levels of microalbuminuria and uricemia. These biomarkers may be relevant for the treatment of patients with uncomplicated hypertension with low to moderate risk of cardiovascular events. The association between microalbuminuria and cardiovascular events is well known [Jolly et al. 2011; Wen et al. 2011]. Although uric acid is not a conventional risk factor itself, it has been associated with activation of the renin–angiotensin system, metabolic syndrome, endothelial dysfunction, and renal disease [Forman et al. 2009]. A reduction of levels in this biomarker among patients with hypertension has been associated with treatment with losartan, rather than other families of medications (such as renin–angiotensin inhibitors or calcium channel blockers). In the context of this study, having uricemia as a surrogate endpoint was useful to evaluate the consistency of the changes, along with the other variables. Further research will clarify whether antihypertensive medications reducing levels of uric acid may benefit patients with hypertension and hyperuricemia [Alderman and Aiyer, 2004; Feig et al. 2008].
In conclusion, most patients were responsive to 6 months of treatment with amlodipine/losartan in this outpatient population with uncomplicated hypertension from Andean countries. Therefore, this therapy may be one of the options when starting drug therapy. Stronger support for this option should come from larger studies measuring the extent of treatment goals reached among comparable groups receiving different doses.
Footnotes
Acknowledgements
The following investigators participated in the METAL study: I. Zuleta, M. Sánchez, S. Herrera, C. Chacón, W. Alarco, R.M. Azañero, J. López, E. Morales, A. Lairet, H. Acquatella, J. Valery, A.M. Granada, J.M. Corral, L.F. Leal, D.H. Prada.
Funding
Supported by Grupo Farma.
Conflict of interest statement
No potential conflict of interest relevant to this article was reported.
