Abstract
The objective of this review is to evaluate the role of fixed-dose triple-combination therapy for the management of hypertension. An assessment of clinical trials showed that half the patients with hypertension have uncontrolled blood pressure (BP), with underlying factors including therapeutic inertia and poor patient adherence. Many patients will require three antihypertensive agents to achieve BP goals, and current guidelines recommend combining drugs with complementary mechanisms of action. Three single-pill triple-combination treatments are available and each includes an agent affecting the renin-angiotensin-aldosterone pathway (either a direct renin inhibitor or an angiotensin II receptor blocker) in combination with a calcium channel blocker and diuretic. These triple-combination therapies consistently demonstrated significantly greater BP reduction relative to the component dual combinations, with BP reductions documented across a range of patient populations. Triple-combination treatments were well tolerated in all clinical trials reviewed. The use of single-pill, triple-combination antihypertensive therapy has been shown to be an effective, well-tolerated, and convenient treatment strategy that can help patients achieve BP control.
Keywords
Introduction
Uncontrolled blood pressure (BP) remains a major healthcare issue and individuals with high BP are at an increased risk of cardiovascular disease (CVD) morbidity and mortality [Egan et al. 2010; Heidenreich et al. 2011]. Data from the National Health and Nutrition Examination Survey 2007–2010 showed that an estimated 77.9 million adults ≥ 20 years of age have hypertension; of those with hypertension, 81.5% were aware of their condition, 74.9% were under current treatment, 52.5% had their BP controlled, and 47.5% did not have their BP controlled [Go et al. 2013]. The American Heart Association has forecasted that the economic burden of CVD attributable to hypertension will rise to approximately US$200 billion annually by 2030 [Heidenreich et al. 2011].
Effective BP reduction has been associated with reductions in the risk of cardiovascular and/or cerebrovascular events and related morbidity and mortality across a variety of patient populations [Lewington et al. 2002; Mancia et al. 2009; Staessen et al. 2005; Turnbull, 2003]. In fact, given the well-established benefits of BP control for CVD outcomes, the US Food and Drug Administration (FDA) has recently issued an industry guidance regarding antihypertensive drugs and recommends the following standardized language for the labeling of any such product, regardless of whether the antihypertensive agent has demonstrated CVD outcomes: ‘Lowering blood pressure reduces the risk of fatal and nonfatal cardiovascular events, primarily strokes and myocardial infarctions’ [FDA, 2011]. Furthermore, clinical trials such as the Valsartan Antihypertensive Long-term Use Evaluation (VALUE), Systolic Hypertension in Europe (Syst-Eur), and Avoiding Cardiovas-cular Events through Combination Therapy in Patients Living with Systolic Hypertension (ACCOMPLISH) have shown that earlier diagnosis and treatment of hypertension can improve outcomes such as myocardial infarction, stroke, and cardiac morbidity and mortality [Jamerson et al. 2007; Julius et al. 2004; Staessen et al. 2004].
The fact that multiple antihypertensive therapies are often required to achieve BP control has been consistently established by numerous clinical trials including the Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial (ALLHAT), the International Verapamil-Trandolapril Study (INVEST), the Study on Cognition and Prognosis in the Elderly (SCOPE), the Hypertension Optimal Treatment (HOT) study, the Modification of Diet in Renal Disease (MDRD), and the African American Study of Kidney Disease and Hypertension (AASK) [Cushman et al. 2002; Hansson et al. 1998; Jamerson, 2003; Klahr et al. 1994; Lithell et al. 2003; Pepine et al. 2003; Wright et al. 2002]. Thus, it is well accepted that the majority of patients with hypertension require combination therapy and, based on cumulative data from clinical trials, it has been estimated that at least 25% of patients will require triple-combination therapy to achieve BP control [Gradman, 2010].
A rationale approach to combination therapy in hypertension is the selection of antihypertensive drugs that have complementary mechanisms of action, for example, combining an agent targeting the renin-angiotensin-aldosterone system (RAAS) with a diuretic and a calcium channel blocker (CCB) [Mancia et al. 2009]. Such triple-combination treatments have demonstrated greater efficacy compared with the component dual combinations, as evidenced by greater proportions of patients on triple-combination therapy achieving BP control after only 2 weeks at the highest dose [Calhoun et al. 2009b; Oparil et al. 2010]. The focus of this review is to evaluate the role of fixed-dose triple-combination therapy in hypertension management.
Hypertension guidelines
In order to achieve BP control, current guidelines recommend stepwise treatment regimens, typically with more than one antihypertensive agent [Chobanian et al. 2003; Gradman et al. 2010; Mancia et al. 2009; NICE, 2011; Rosendorff et al. 2007; Whitworth, 2003]. The seventh report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure (JNC 7) states that when BP is >20/10 mmHg above goal, consideration should be given to initiating therapy with two agents [Chobanian et al. 2003]. Similarly, the International Society on Hypertension in Blacks recommends the use of combination therapy for patients with systolic BP (SBP) > 15 mmHg above goal and diastolic BP (DBP) > 10 mmHg above goal [Flack et al. 2010]. The practicality of initiating treatment with combination therapy was demonstrated by the Simplified Intervention to Control Hypertension (STITCH) study. The analyses were based on data from 2104 patients with uncontrolled hypertension, of whom 49% were already being treated at the time of baseline assessment. In this study, a simple step-care-based algorithm featuring a low-dose, fixed-dose combination regimen for initial treatment was readily implemented in family-practice settings and resulted in better BP control compared with more conventional guideline-based management [Feldman et al. 2009]. At practices assigned to STITCH care, BP target was achieved by 64.7% compared with 52.7% for those assigned to guideline care (between-group difference: 12.1%; 95% confidence interval [CI]: 1.5–22.4%; p = 0.026) [Feldman et al. 2009]. A recent study showed that initial combination therapy was associated with a significant risk reduction of cardiovascular events and more rapid achievement of target BP was found to be the main contributor to the estimated risk reduction [Gradman et al. 2013].
Barriers to effective BP control
Therapeutic inertia
The failure of healthcare providers to increase a patient’s antihypertensive dose or change regimens to achieve BP control has been described as clinical or therapeutic inertia and represents an important provider-related barrier to BP control [Heisler et al. 2008; Hyman et al. 2000; Nesbitt, 2010; Okonofua et al. 2006]. In one study that examined this phenomenon, physicians made changes to the antihypertensive medication regimen in only 13.1% of visits for which patients had elevated BP. Estimated elevations of 13.8 mmHg in SBP and 4.5 mmHg in DBP were attributed to the presence of therapeutic inertia compared with no therapeutic inertia [Okonofua et al. 2006]. A large retrospective cohort analysis conducted in Veterans Affairs healthcare facilities (n = 38,327) found that despite documented BP elevations (68,610 BP events; mean [standard deviation] SBP 151.7 [12.3] mmHg), antihypertensive therapy was only intensified 30% of the time [Heisler et al. 2008]. Among the potential reasons that have been suggested for therapeutic inertia are physicians: (a) overestimating BP control; (b) being satisfied with BP lowering regardless of goal attainment; (c) having misperceptions regarding the use of multiple agents including complexity and adverse event risk [Ofili, 2006; Rose et al. 2008].
Adherence
Many factors may contribute to poor adherence. In a survey of 1432 patients with hypertension, those who self-reported difficulty in taking their antihypertensive medication (n = 407) identified the following most common underlying reasons: ‘not remembering’ (32.4%); cost (22.6%); lack of insurance (22.4%); side effects (12.5%) [Vawter et al. 2008]. Comorbidities such as depression, anxiety, or migraine were also more prevalent among patients reporting difficulty with adherence to treatment. Poor adherence to antihypertensive therapy can contribute to failure to achieve BP control and thus may be associated with an elevated risk of cardiovascular events [Bailey et al. 2010; Bramley et al. 2006; Cherry et al. 2009; Dragomir et al. 2010; Fung et al. 2007; Ho et al. 2008]. A retrospective study of a state Medicaid population [Bailey et al. 2010] projected that if the medication refill adherence for US adults with hypertension was raised to ≥ 80%, approximately 200,000 lives would be saved over 5 years (this was based on the estimated 60% nonadherence rate of 68 million US adults with hypertension available at the time of the analysis; the estimated number of patients with hypertension has since increased to 77.9 million [Go et al. 2013]). Thus, strategies that can help improve patient adherence are essential.
There is an inverse relationship between adherence and the number of antihypertensive drugs in a regimen. It has been reported that as the number of drugs in the regimen increases, the proportion of adherent patients decreases [Fung et al. 2007]. In fact, fixed-dose dual-combination therapies have been reported to have higher rates of adherence compared with single-component combinations [Bangalore et al. 2007; Brixner et al. 2008; Dickson and Plauschinat, 2008; Gupta et al. 2010; Yang et al. 2010; Zeng et al. 2010]. In one study, the adjusted medication possession ratio (MPR) (the percentage of time a patient has access to medication) was estimated to be 72.8% for the fixed-dose combination cohort and 61.3% for the free-drug combination cohort, resulting in a between-cohort difference of 11.6% (difference or odds ratio, 11.6%; 95% CI, 11.4–11.7%) [Yang et al. 2010]. Meta-analyses comparing fixed-dose dual combinations versus the free-drug combinations have estimated that fixed-dose combinations improved adherence and persistence with therapy by 29% and reduced the risk of noncompliance by 24% relative to single-component combinations [Bangalore et al. 2007; Gupta et al. 2010]. Another recent analysis of 12 hypertension studies noted that, among each of the 7 studies that reported MPRs, adherence was significantly higher with single-pill combinations versus free-drug combinations. A meta-analysis of these data revealed that the MPR for single-pill combinations was 14% higher versus free-drug combinations among patients experienced with antihypertensive medications (an 8% higher MPR was reported among treatment-naïve patients) [Sherrill et al. 2011].
Adherence to antihypertensive medications has many potential benefits for patients. For example, a population-based study of medical and pharmacy claims showed that adherence was associated with a greater proportion of patients achieving BP control [Bramley et al. 2006]. Improved adherence to antihypertensive therapy may also result in fewer CVD-related hospitalizations and a reduced number of inpatient hospital days [Lynch et al. 2009; Pittman et al. 2010; Yang et al. 2010]. A potential benefit of improved adherence among employees with hypertension is fewer days absent from work [Lynch et al. 2009]. Ultimately, better adherence may translate to fewer patients with coronary artery disease, CVD, and chronic heart failure [Bailey et al. 2010; Dragomir et al. 2010].
Approved triple-combination treatments for hypertension
Overview of mechanisms of action
Antihypertensive drugs work through multiple pathways (Figure 1), and thus the combination of agents with complementary mechanisms has been put forth as a rational approach to achieving BP control [Chrysant, 2008; Elijovich and Laffer, 2009; Gradman, 2010; Sica, 2002, 2004; Wald et al. 2009]. Currently, three triple-combination therapies are available for the treatment of hypertension: Exforge HCT® (valsartan [VAL]/amlodipine [AML]/hydrochlorothiazide [HCTZ]) (Novartis Pharmaceuticals Corporation, East Hanover, NJ, USA); Tribenzor® (olmesartan medoxomil [OM]/AML/HCTZ) (Daiichi Sankyo, Inc., Parsippany, NJ, USA); Amturnide® (aliskiren [ALI]/AML/HCTZ) (Novartis Pharmaceuticals Corporation, East Hanover, NJ, USA) (Table 1). Each of these single-pill triple combinations contains either an angiotensin receptor blocker (ARB) or a direct renin inhibitor (DRI), combined with a CCB and a diuretic and thus capitalizes on the complementary mechanisms of action of these individual component therapies (Figure 2) [Mancia et al. 2009]. DRIs and ARBs target different aspects of the RAAS: DRIs reduce plasma-renin activity, resulting in reduced plasma levels of the potent vasoconstrictor angiotensin II (A-2), whereas ARBs directly inhibit A-2 through competitive displacement at the AT1 receptor [Barreras and Gurk-Turner, 2003; Burnier, 2001; Fogari and Zoppi, 2010]. CCBs elicit vasodilation, leading to BP reduction by binding to distinct sites at the L-type calcium channel and reducing the entry of extracellular calcium into the cell [Noll and Luscher, 1998; Richard, 2005]. This vasodilation activates both the RAAS and sympathetic nervous system and results in reflex vasoconstriction and tachycardia. However, addition of a RAAS-blocking agent attenuates the CCB-induced activation of the RAAS, resulting in additive antihypertensive efficacy [Oparil and Weber, 2009]. Thiazide diuretics have an unclear mechanism in hypertension, but are known to inhibit the renal Na+/Cl- cotransporter effecting sodium depletion and plasma volume reduction early on; however, with a longer duration of use, reduction in total peripheral resistance is likely to contribute to BP lowering [Birkenhager, 1990; Hjemdahl, 1984; Hughes, 2004]. Diuretics initially reduce intravascular volume and activate the RAAS, leading to vasoconstriction as well as salt and water retention. In the presence of a RAAS inhibitor, this counter-regulatory response is attenuated [Gradman et al. 2010]. Diuretic-induced activation of the RAAS increases substrate levels for angiotensin-converting enzyme (ACE) inhibitors and ARBs, converting low-renin patients to high-renin patients, ensuring consistent BP lowering [Epstein, 2010]. In addition to improved BP control, certain combinations may also be associated with improved tolerability, for example, the peripheral edema that occurs with a dihydropyridine CCB can be lessened by combining the CCB with an ARB and/or a diuretic [Gradman, 2010]. The cause of the edema is believed to be arteriolar dilation, resulting in an increased pressure gradient across capillary membranes in dependent portions of the body, which RAAS blockers are thought to counteract through venodilation. Furthermore, through the antisympathetic effects of ACE inhibitors and ARBs, RAAS inhibitors blunt the increase in heart rate that may accompany treatment with a dihydropyridine CCB [Gradman et al. 2010].
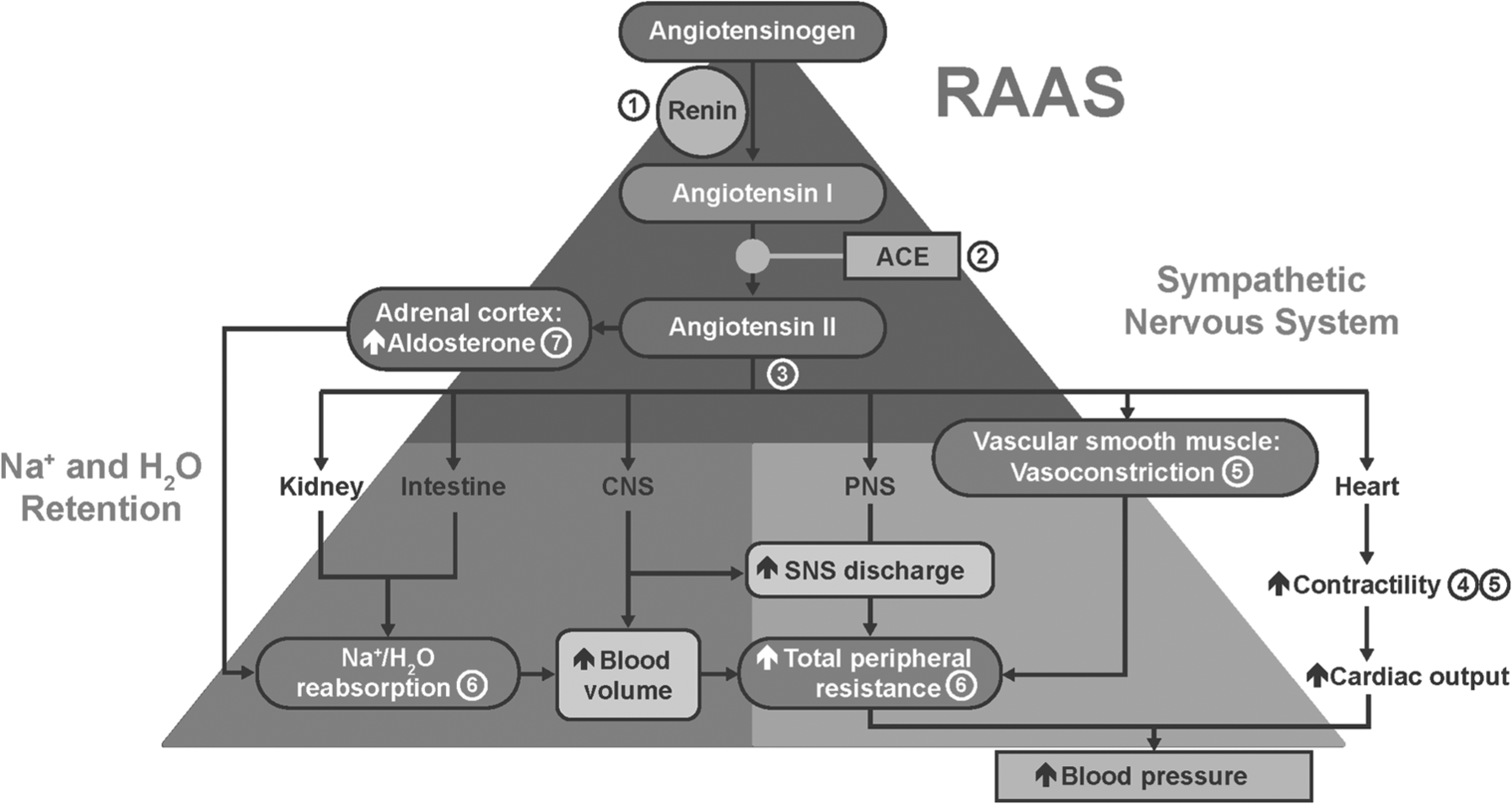
RAAS sites of action for major antihypertensive agents: 1 = direct renin inhibitors; 2 = ACE inhibitors; 3 = angiotensin receptor blockers; 4 = beta blockers; 5 = calcium channel blockers; 6 = diuretics; 7 = aldosterone antagonists. ACE, angiotensin-converting enzyme; CNS, central nervous system; PNS, peripheral nervous system; RAAS, renin–angiotensin–aldosterone system; SNS, sympathetic nervous system. Reproduced with kind permission from McGraw-Hill (Saseen and MacLaughlin, 2011).
Single-pill, fixed-dose, triple-combination therapies for hypertension: indication and usage.*

For prescribing information see Novartis Pharmaceuticals Corporation [2012a, 2012c]; Daiichi Sankyo, Inc. [2012b].
ALI, aliskiren; AML, amlodipine; ARB, angiotensin receptor blocker; BP, blood pressure; CCB, calcium channel blocker; DRI, direct renin inhibitor; HCTZ, hydrochlorothiazide; HTN, hypertension; OM, olmesartan; VAL, valsartan.

Complementary mechanism of combination antihypertensive therapy with an ARB or DRI, CCB, and a thiazide diuretic. ARB, angiotensin receptor blocker; AT1, angiotensin II receptor type 1; AT2, angiotensin II receptor type 2; BP, blood pressure; CCB, calcium channel blocker; DRI, direct renin inhibitor, MOA, mechanism of action; TPR, total peripheral resistance. *Exact mechanism of antihypertensive effect is not known. Reproduced with kind permission from JTE Multimedia (Oparil and Weber, 2009).
Drug interactions and pharmacokinetics
No drug interaction studies have been conducted with the three approved triple-combination treatments; however, potential drug interactions may be anticipated based on data available for the individual antihypertensive drug components (Table 2). Among the components, AML appears to be the most likely to be affected by cytochrome P450 enzymes. In particular, CYP3A4 inhibitors can increase AML concentrations, leading to increased risk of hypotension and/or edema [Pfizer Labs, 2011]. The antihypertensive effects of ALI, HCTZ, OM, and VAL may be attenuated with co-administration with nonsteroidal anti-inflammatory drugs (NSAIDs) [Lannett Company, Inc., 2007; Daiichi Sankyo, Inc., 2012a; Novartis Pharmaceuticals Corporation, 2012b, 2012d]. In addition, periodic renal monitoring is recommended in patients using NSAIDs with agents working through the RAAS pathway (ALI, OM, and VAL) [Daiichi Sankyo, Inc., 2012a; Novartis Pharmaceuticals Corporation, 2012b, 2012d].
Drug interactions and key pharmacokinetic/pharmacodynamic information for individual components of Exforge HCT, Tribenzor, and Amturnide.*

For prescribing information see Lannett Company, Inc. [2007]; Pfizer Labs [2011]; Daiichi Sankyo, Inc. [2012a]; Novartis Pharmaceuticals Corporation [2012b, 2012d].
This table includes the key or common drug interactions; this table does not include all the drug interactions for these agents.
ALI, aliskiren; AML, amlodipine; AUC, area under curve; Cmax, peak plasma concentration; COX-2 inhibitors, selective cyclooxygenase inhibitors; HCTZ, hydrochlorothiazide; IV, intravenous; NSAIDs, nonsteroidal anti-inflammatory drugs; OM, olmesartan; VAL, valsartan.
The FDA warns of possible risks when using ALI with ACE inhibitors and ARBs in patients with diabetes or kidney (renal) impairment. These drug combinations should not be used (i.e. are contraindicated) in patients with diabetes. In addition, a new warning is being added to avoid the use of these drug combinations in patients with kidney impairment. The prescribing information for ALI-containing drugs are being updated based on preliminary data from the Aliskiren Trial in Type 2 Diabetes Using Cardio-Renal Endpoints (ALTITUDE) [FDA, 2012].
In terms of pharmacokinetic parameters, the labeling for each of these single-pill triple combinations notes that the rate and extent of absorption of the components are the same as when administered as their individual dosage forms [Novartis Pharmaceuticals Corporation, 2012a, 2012c; Daiichi Sankyo, Inc., 2012b]. Key pharmacokinetic and pharmacodynamics parameters for the individual components are presented in Table 2.
Exforge HCT
The efficacy and safety of VAL/AML/HCTZ was evaluated in a pivotal, randomized, double-blind, placebo-controlled clinical trial [Calhoun et al. 2009b]. Patients with moderate to severe hypertension were randomized to treatment with VAL 320/AML 10/HCTZ 25 mg versus the component dual combinations (VAL 320/AML 10 mg, AML 10/HCTZ 25 mg, VAL 320/HCTZ 25 mg). Compliance was assessed by the investigator and/or study personnel at each visit using pill counts and information provided by the patient. VAL/AML/HCTZ significantly reduced seated SBP (SESBP) by 39.7 mmHg and seated DBP (SEDBP) by 24.7 mmHg (Table 3). SEBP reductions were significantly greater than those reported with each of the dual-combination treatments (all p < 0.0001). In addition, the proportion of patients achieving BP control at study endpoint was significantly greater with the triple combination (70.8%) compared with the dual combinations (44.8–54.1%). Most of the adverse events in the pivotal study of VAL/AML/HCTZ were of mild or moderate severity. Dizziness occurred more frequently with triple-combination therapy (7.7%) and VAL/HCTZ (7.0%) than with AML/VAL (2.3%) or AML/HCTZ (3.9%), and peripheral edema occurred less frequently with triple-combination therapy (4.5%) and VAL/HCTZ (0.9%) compared with AML/HCTZ (8.9%) or AML/VAL (8.5%) [Calhoun et al. 2009b].
BP reductions and goal attainment for VAL/AML/HCTZ, OM/AML/HCTZ, ALI/AML/HCTZ and component dual combinations for the total cohort.*

p < 0.0001, ‡ p < 0.001 versus component dual combinations.
ALI, aliskiren; AML, amlodipine; BP, blood pressure; HCTZ, hydrochlorothiazide; LS, least squares; OM, olmesartan; VAL, valsartan.
Secondary analyses of these data have shown that the benefits of BP lowering with triple therapy are observed early, after only 1 week of treatment with VAL 160/AML 5/HCTZ 12.5 mg, and that these early benefits are also seen in patients with severe systolic hypertension [Calhoun et al. 2009a]. More recently, the efficacy of VAL/AML/HCTZ was reported for a subgroup of patients from the pivotal study that had 24-h ambulatory BP monitoring (ABPM) at baseline and after 8 weeks of therapy [Lacourciere et al. 2011]. Reductions in ABP were greatest for patients on triple-combination treatment (p ≤ 0.01 versus dual-combination treatment), with a reduction in mean 24-h ambulatory SBP of 30.3 mmHg and DBP of 19.7 mmHg. These reductions were relatively uniform throughout the 24-h interval.
Tribenzor
The clinical efficacy and safety of OM/AML/HCTZ was evaluated in the Triple Therapy with Olmesartan Medoxomil, Amlodipine, and Hydrochlorothiazide in Hypertensive Patients Study (TRINITY) [Oparil et al. 2010]. This pivotal, randomized, double-blind, placebo-controlled trial evaluated OM 40/AML 10/HCTZ 25 mg versus the component dual combinations (OM 40/AML 10 mg; OM 40/HCTZ 25 mg; AML 10/HCTZ 25 mg) in patients with moderate to severe hypertension. Patient adherence was monitored by assessing the tablet count from drug packages at each visit. Adherence to study medication was similar across treatment groups, ranging from 98.0% to 98.5%. OM/AML/HCTZ reduced least squares (LS) mean SESBP by 37.1 mmHg and SEDBP by 21.8 mmHg (Table 3). This BP reduction was statistically significant compared with the dual-combination treatments (p < 0.001) at week 12. A significantly greater proportion of patients receiving triple-combination treatment achieved a BP target of < 140/90 mmHg (69.9%) compared with the dual combinations at week 12 (41.1–53.4%; p < 0.001 for all comparisons). The majority of treatment-emergent adverse events observed during the study were of mild or moderate severity. Nasopharyngitis (3.5%), muscle spasms (3.1%), hypotension (1.4%), and orthostatic hypotension (0.3%) occurred more frequently in the triple-combination treatment group compared with the dual-combination treatment groups. Incidences of dizziness in the OM 40/AML 10 mg, OM 40/HCTZ 25 mg, AML 10/HCTZ 25 mg, and OM 40/AML 10/HCTZ 25 mg groups were 4.9%, 10.0%, 3.1%, and 9.9%, respectively. The incidence of peripheral edema was 1.0% in the OM 40/HCTZ 25 mg group, compared with 7.0–8.3% in the other treatment groups in which AML was a component.
A subgroup of patients from the TRINITY study had 24-h ABPM at baseline and after 12 weeks of therapy [Izzo et al. 2011]. At week 12, OM 40/AML 10/HCTZ 25 mg resulted in a greater reduction in mean 24-h SBP and DBP (30.3/18.0 mmHg) compared with the three dual-combination regimens (OM 40/AML 10 mg: 23.5/13.9, OM 40/HCTZ 25 mg: 23.9/14.5; AML 10/HCTZ 25 mg: 18.5/10.7 mmHg; all p < 0.0001).
A prespecified subgroup analysis of Black and non-Black patients in the TRINITY study demonstrated that for each of the race subgroups, LS mean BP reductions at week 12 were significantly greater for the triple combination versus the dual combinations (p ≤ 0.0001) [Chrysant, 2012]. At week 12, BP goal attainment with the triple-combination treatment was comparable for Black patients (61.5%) and non-Black patients (65.5%), and significantly greater than that seen with the dual-combination treatments. Another subgroup analysis of the TRINITY study evaluated the efficacy and safety of OM/AML/HCTZ in patients with hypertension and diabetes [Chrysant et al. 2012]. Regardless of diabetes status, the triple combination resulted in significantly greater reductions in SEBP relative to the component dual combinations. Of note, although the goal BP in patients with diabetes was < 130/80 mmHg (10 mmHg lower than the goal in patients without diabetes), 41.1% of patients with hypertension and diabetes were able to achieve BP < 130/80 mmHg with OM/AML/HCTZ compared with 10.1–16.3% of diabetic patients taking the dual combinations (all p ≤ 0.0002).
The 40-week open-label extension of the TRINITY study demonstrated that long-term administration of OM/AML/HCTZ was associated with BP reductions comparable to those seen during the double-blind treatment period, thus demonstrating the durable antihypertensive effect of this triple combination. At the end of the study, the mean BP decreased from 168.6/100.7 mmHg (baseline BP at randomization) to 125.0–136.8/77.8–82.5 mmHg, depending on treatment [Kereiakes et al. 2012].
Amturnide
The efficacy and safety of ALI 300/AML 10/HCTZ 25 mg was compared with the component dual combinations (ALI 300/AML 10 mg; ALI 300/HCTZ 25 mg; AML 10/HCTZ 25 mg) in patients with moderate to severe hypertension [Lacourciere et al. 2012]. BP lowering achieved with the triple combination was significantly greater than with the component dual combinations. Treatment with the ALI/AML/HCTZ combination resulted in a LS mean SEBP reduction of 30.7/15.9 mmHg from baseline at week 4 and 37.9/20.6 mmHg at week 8 (p < 0.001 versus all dual-combination treatments) (Table 3). Clinically relevant SEBP reductions were evident within 2 weeks of treatment with the triple combination. In patients with severe hypertension (defined as SBP ≥ 180 mmHg at baseline), ALI/AML/HCTZ combination therapy resulted in a BP reduction of 49.5/22.5 mmHg at week 8 (p < 0.001 versus all dual combinations). Most adverse events were mild or moderate, with the overall incidence comparable in the four treatment groups: 33.4% (ALI/AML), 32.3% (ALI/HCTZ), 33.6% (AML/HCTZ), and 36.2% (ALI/AML/HCTZ). Peripheral edema was the most frequently reported adverse event in this study [Lacourciere et al. 2012]. A long-term, open-label study showed that ALI 300/AML 10/HCTZ 25 mg triple-combination therapy was well tolerated and efficacious for up to 54 weeks in patients with moderate to severe hypertension [Murray et al. 2012].
The Aliskiren Amlodipine HCTZ in Minority Patients with Stage 2 Hypertension (ASCENT) study evaluated the efficacy of ALI/AML/HCTZ versus the dual combination of ALI/AML in 412 self-identified minority patients (Black, Hispanic/Latino) [Ferdinand et al. 2011]. Although the ALI/AML dual combination resulted in effective BP reductions, the triple combination resulted in greater mean BP reductions (p < 0.0001 for SESBP and p = 0.0005 for SEDBP). Differences between the groups were seen by 2 weeks of treatment.
Potential place in therapy for single-pill triple combinations
Single-pill triple-combination therapy may be considered in patients who have uncontrolled hypertension and are taking two separate drugs, a two-drug combination, or three separate drugs, and are experiencing adherence issues [Elijovich and Laffer, 2009; Gradman, 2010]. Even among patients who have well-controlled BP, consolidating triple free-drug therapy into a single pill offers a regimen that is more convenient and perhaps more affordable in certain circumstances. If the cost of a single-pill triple-combination therapy represents a barrier to treatment for a particular patient, healthcare providers have an important role in raising awareness of or facilitating usage of available resources such as patient assistance or reimbursement programs. Perhaps the greatest clinical benefit of single-pill combinations relative to multiple pills is the potential for improved adherence and the resulting improved control of BP [Bangalore et al. 2007; Gradman et al. 2011; Gupta et al. 2010; Hess et al. 2008; Zeng et al. 2010]. Prospective data on adherence rates and outcomes for patients on single-pill triple combinations would help to better establish the place in therapy of these treatment options. In a recent study evaluating the impact of fixed-dose (two pill) versus loose-dose (three pill) triple-combination therapy, patients receiving fixed-dose therapy were more likely to be adherent (p < 0.001), and less likely to discontinue treatment (p < 0.001) across three cohorts (ARB, beta-blocker, and ACE-inhibitor cohorts). Fixed-dose therapy resulted in significantly lower adjusted risk of cardiovascular events (hazard ratio = 0.76, p = 0.005) in the beta-blocker cohort only, and total adjusted healthcare costs were significantly lower for fixed-dose therapy in the beta-blocker cohort only (cost ratio = 0.74 overall, p < 0.01 and 0.71 hypertension-attributable, p < 0.01) [Panjabi et al. 2013].
Summary
Combination therapy is a recognized mainstay of hypertension management and an estimated one out of every four patients will require three antihypertensive agents to achieve BP control [Gradman, 2010]. Unfortunately, the use of multiple pills can lead to poor patient adherence to therapy which, in turn, can compromise attainment of BP control [Fung et al. 2007; Ho et al. 2008]. Fixed-dose single-pill combination therapies have been associated with better patient adherence than single-component combinations [Bangalore et al. 2007; Brixner et al. 2008; Dickson and Plauschinat, 2008; Gupta et al. 2010; Yang et al. 2010; Zeng et al. 2010]. Newly available single-pill triple combinations offer patients a well-tolerated and convenient option that can improve adherence to therapy. Notably, all three currently available triple combinations have demonstrated significantly better BP lowering compared with the component dual combinations [Novartis Pharmaceuticals Corporation, 2012a; Calhoun et al. 2009b; Oparil et al. 2010].
Footnotes
Acknowledgements
Editorial support for this article was provided by Vrinda Mahajan, PharmD, of Peloton Advantage, LLC, Parsippany, NJ, USA and funded by Daiichi Sankyo, Inc, Parsippany, NJ, USA. The opinions expressed in the current article are those of the authors.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors report no conflict of interest.
