Abstract
Background:
The severity of coronary artery disease (CAD) is directly related to the quality of glucose control in diabetic patients. Additionally, mortality after an acute coronary syndrome is higher in patients with diabetes and it correlates to the level of glucose control. However, the role of higher gluconated hemoglobin in the process of coronary atherosclerosis and clinical outcome of acute myocardial infarction is unknown.
Objectives:
To evaluate the association of HbA1c level and severity of CAD and short-term outcomes of acute ST-elevation myocardial infarction (STEMI) in nondiabetics.
Methods:
A total of 290 nondiabetic patients with STEMI were prospectively enrolled following their admission. Patients were stratified into 2 groups based on the median percent of HbA1c (⩽5.8% ‘Low’ and >5.8% ‘High’). The severity of CAD based on the Califf scoring system, in-hospital mortality and morbidities of STEMI were compared between groups. Patients were followed for 1 year after discharge to assess readmission and mortality rate.
Results:
The severity score for CAD was significantly higher in the ‘High’ versus ‘Low’ HbA1c group (7.7 ± 2.7 and 5.5 ± 2.6, p = 0.001). A total of 15 patients died in both groups during the follow-up period. While in-hospital mortality was similar between the two groups, 12-month mortality was significantly higher in the ‘High’ group (7.7% versus 2.7%, p = 0.043). In addition, the rehospitalization rate within 1 year was 8.8% in the ‘Low’ group, which was significantly lower than 19.0% in the ‘High’ group (p = 0.016).
Conclusion:
Among nondiabetic patients presenting with STEMI, the severity of CAD was higher in those with HbA1c level >5.8%; 1-year mortality and hospital readmission rates were also higher in this group of patients.
Introduction
Patients with diabetes are at 3–4 times increased risk for cardiovascular mortality compared with nondiabetics [Preis et al. 2009]. In addition to the higher rate of acute ST-elevation myocardial infarction (STEMI) in diabetics, hyperglycemia is associated with poorer prognosis in these patients [Wahab et al. 2002; Wiviott et al. 2008; Li et al. 2011]. Although traditional cardiovascular risk factors including hypertension, dyslipidemia and obesity are more prevalent in diabetic patients, there are some evidence for an independent role of hyperglycemia on atherosclerosis [Gerrity et al. 2001; Renard et al. 2004].
Despite the fact that diabetes mellitus adds to the risk of coronary artery disease (CAD) independent of the serum glycemic level, uncontrolled glucose concentrations in plasma multiplies this risk several fold [Rubin et al. 2012]. Wong and colleagues showed that there is no glycemic threshold for retinopathy in diabetic patients [Wong et al. 2008]; similar reports are available indicating to lack of a threshold glycemic level for developing cardiovascular complications too [Diabetes Control and Complications Trial, 1996; Balkau et al. 1999; Shaw et al. 2000]. It is possible to consider this increased risk for people not defined as clinically overt diabetes based on their serum glycemic level and current criteria. Among nondiabetic adults attended in a second visit of the Atherosclerosis Risk In Communities (ARIC) study, higher HbA1c level was associated with higher cardiovascular disease and death [Selvin et al. 2010]. There are some other studies supporting the association between admission serum HbA1c level and increased long-term mortality of nondiabetic patients admitted with STEMI and a higher rate of CAD in these patients [Timmer et al. 2011, Pai et al. 2013].
To the best of our knowledge, this study is the first one evaluating the association of HbA1c with severity of CAD using the Califf scoring system in patients admitted with STEMI [Califf et al. 1985]. We have evaluated the association of admission levels of HbA1c with in-hospital and short-term outcome of nondiabetic patients with STEMI. We hypothesize that higher levels of HbA1c are associated with more severe CAD.
Methods
A total of 290 consecutive nondiabetic patients with STEMI admitted to the Madani Heart Center at Tabriz University of Medical Sciences, Tabriz, Iran, from May 2012 to April 2013 were enrolled in this analytical–prospective study. The ethics committee of the Tabriz University of Medical Sciences reviewed and approved the study protocol (ID: 5/4/3036), including the informed consent form for its ethical and scientific merit. The procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation with the Declaration of Helsinki 1975, revised Hong Kong 1989. After a thorough explanation of the study protocol to the patients or their next of kin, an informed consent was obtained from each patient.
The diagnosis of STEMI was based on typical rise and fall in cardiac biomarkers (CK-MB and cTnI), together with chest pain of at least 30 min duration or the ST-segment elevation ⩾1 mm in 2 or more contiguous limb leads and/or more than 2 mm in 2 or more precordial leads on the 12-lead electrocardiogram (ECG). Patients admitted more than 12 hours from symptoms onset, those with prior history of coronary artery bypass graft surgery (CABG), renal failure [estimated glomerular filtration rate (eGFR) <60 ml/min], documented diabetes and those with admission HbA1c ⩾6.5% or hemoglobin levels <12 gm/dl or prior history of hemoglobin disorders were excluded from the study.
Blood glucose concentration, HbA1c, and lipid profiles and cardiac enzymes were measured just after the arrival of the patients to the emergency room and recorded. Patients were stratified into two groups based on HbA1c levels at the time of admission. The median value of 5.8% was set as the cutoff point for this grouping. Those with HbA1c ⩽5.8% were allocated to the ‘Low’ group and those with HbA1c >5.8% were allocated to the ‘High’ group. In addition to the demographic information, Califf severity scores, left ventricular ejection fraction (LVEF), the occurrence of in-hospital complications, including Killip class of the patients and cardiac arrhythmias, were assessed and recorded. Patients were followed every 3 months for a period of 12 months after discharge from the hospital for readmission and mortality. Major adverse cardiac event (MACE) was used as a composite complication variable; it comprised any occurrence in death, myocardial re-infarction, recurrent ischemia, cardiogenic shock, or confirmed stroke with or without neurologic deficit.
We used the Califf scoring system to assess the severity of coronary artery involvement. For this purpose as previously described by Califf and colleagues [Califf et al. 1985], we divided the coronary system into six segments; proper left anterior descending artery (LAD); the first major septal branch; the major diagonal branch; proper left circumflex artery (LCX); major obtuse marginal branch; and posterior descending artery (PDA). In patients with a left coronary dominant system, the right coronary system was assigned a score of zero. A score of ‘2’ was given for the involvement of each segment with ⩾75% diameter narrowing and, for any mentioned segment distal to this diseased segment, a score of 2 was assigned.
Sample size determination and statistical analysis
The severity of the coronary disease as expressed by Califf scores was the primary endpoint of this study. Hospital readmission, in-hospital and 1-year mortalities were the secondary endpoints. A one grade increase in the Califf score as a clinically significant difference with a standard deviation of 2.0 would require a minimum of 63 patients in each HbA1c group in order to have a power of 80%.
The data collection form was completed and recorded in a Microsoft® Excel spreadsheet and transferred to SPSS version 17.0 (IBM, Chicago, IL, USA) for further analyses. All categorical data were analyzed by chi-square with Fisher’s exact test and presented as the actual raw number and percentage. Continuous data were analyzed by independent two-tailed t-tests and presented as the mean and standard deviation when they had a normal distribution. Otherwise, nonparametric tests were used and data were presented by median and 95% confidence interval (CI). Logistic regression with multivariate analyses was used for analysis of confounding variables and their effect on the outcome variables (in-hospital mortality). In addition, 1-year survival after the STEMI was analyzed with log-rank sum analysis and a Kaplan–Meier survival curve was plotted. Null hypotheses were rejected at an alpha error of 0.05 or less.
Results
Patient characteristics
A total of 290 patients with STEMI were studied. The mean age of the patients was 58.6 ± 13.2 years, with the maximum age of 85 years old and the minimum of 26 years old. Women made up 37.6% of the patients. All other demographic data of the patients are shown in Table 1. A total of 204 patients (70.3%) were treated with thrombolytic therapy, 56 cases (19.3%) underwent primary percutaneous coronary intervention (PCI) and the remaining 30 patients (10.4%) were medically treated and observed without any attempt to reperfusion (Table 1).
Demographic characteristics, comorbid disease and clinical coronary risk factors.
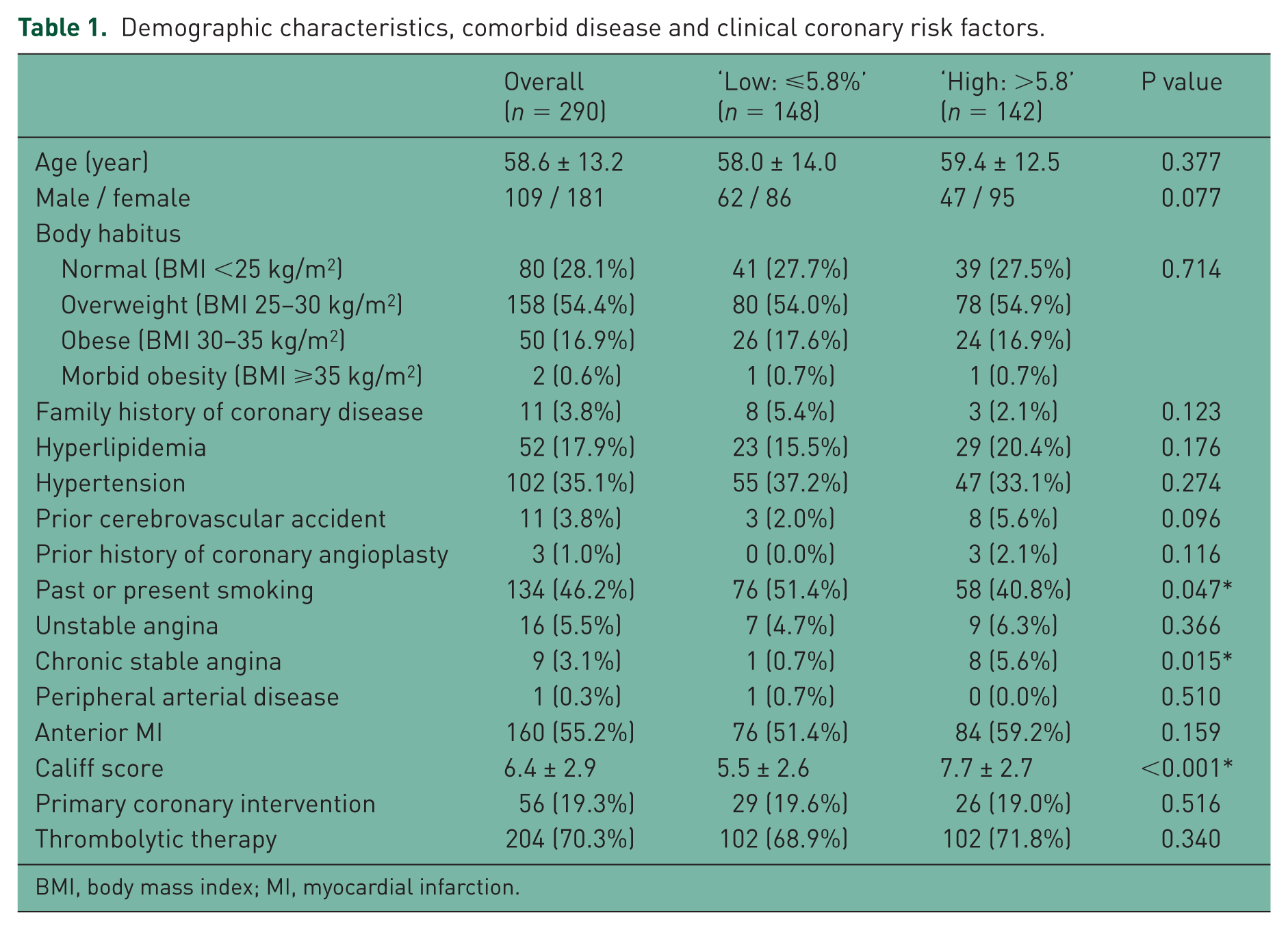
BMI, body mass index; MI, myocardial infarction.
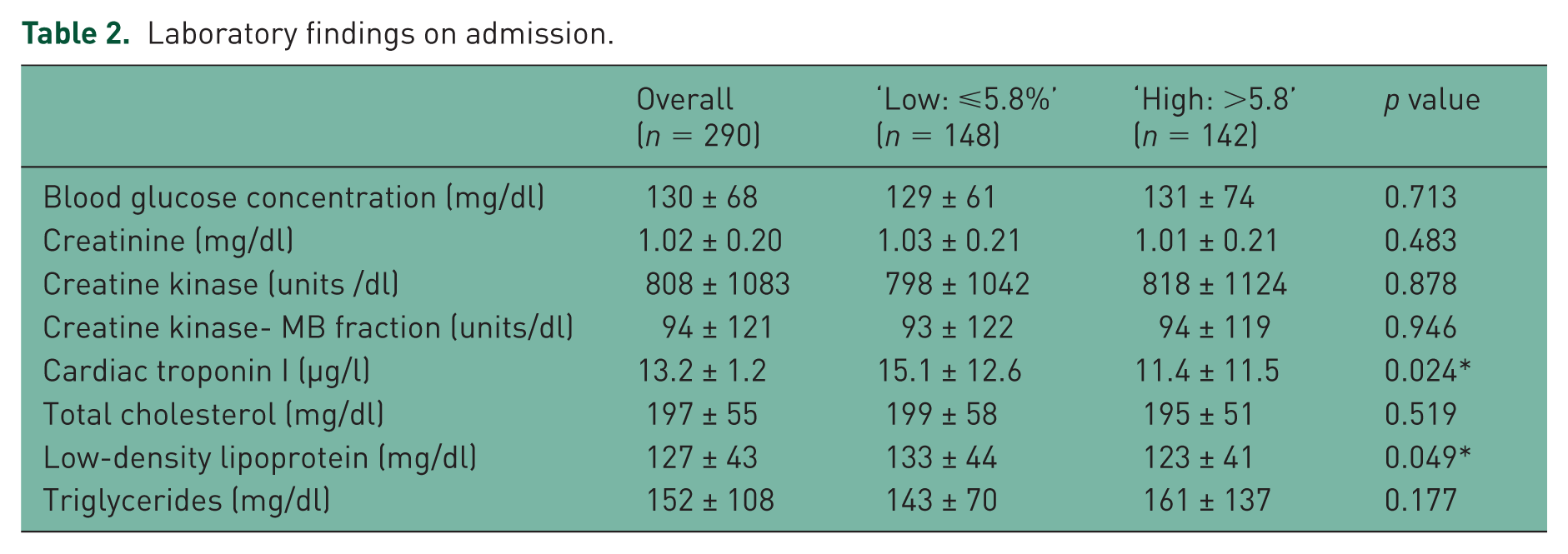
The median value for HbA1c was 5.8% with an interquartile range of 5.5–6.1%. We selected the median of 5.8% as the cutoff value to make our comparisons. There were 148 patients in the ‘Low’ group (HbA1C ⩽5.8%) and 142 patients in the ‘High’ group (HbA1C >5.8%). The admission blood glucose concentrations were 130 ± 68 mg/dl, ranging from 61 to 529 mg/dl. Other laboratory findings on admission are listed in Table 2.
Laboratory findings on admission.
Coronary angiography was performed in 258 patients (88.9%). The Califf score was significantly higher in the ‘High’ group compared with those in the ‘Low’ group (5.5 ± 2.6 versus 7.7 ± 2.7; p < 0.001). There was a significant correlation between the HbA1c levels and Califf score (r = 0.230; p = 0.007). Coronary angiographic findings and multivessel involvement based on HbA1c quartiles are shown in Figure 1. Patients with HbA1c >5.8% ‘High’ had twice more chance of having triple-vessel diseases [odds ratio (OR) = 2.21, 95% CI 1.34–3.65; p = 0.002]. Multivariate logistic regression analysis showed HbA1c >5.8% as an independent predictor of Califf scores >6 (OR = 3.17, 95% CI 1.79–5.69; p = 0.001) (Table 3).

Percentage of patients with 1-VD (single vessel disease) decreased with increasing quartile of hemoglobin A1c (HbA1c) while the percentage of patients with 3-VD (triple vessel disease) increased directly with the incremental quartiles of HbA1c. There was no difference in distribution of 2-VD (double vessel disease) over all four quartiles of HbA1c.
Results of multivariate regression analysis to predict Califf score >6 and hospital readmission.

95% CI, 95% confidence interval; HbA1c, hemoglobin A1c; HR, hazard ratio.
Hospital readmission rates
A total of 40 (13.8%) patients were readmitted to the hospital during the follow-up period, 27 patients (19.0%) in the ‘High’ group compared with 13 patients (8.8%) in the ‘Low’ group (p = 0.016). After excluding two patients who were admitted with exacerbated heart failure (one in each group), the remaining patients were readmitted to the hospital after their initial discharge with diagnosis of acute coronary syndrome or to perform coronary angiography for ongoing ischemic symptoms. The mean percentage of HbA1c in readmitted patients was 6.0 ± 0.3%, which was significantly higher than 5.7 ± 0.5% among those who were never readmitted to the hospital within 1-year follow up (p = 0.001). In univariate logistic regression, the ‘Low’ group demonstrated an approximately 2.5 times reduction in the risk of hospital readmission compared with the ‘High’ group [OR = 0.42, 95% CI 0.27–0.85; p = 0.017]. Multivariate logistic regression analysis showed HbA1c >5.8% as an independent predictor of readmission [OR = 2.55, 95% CI 1.25–5.22; p = 0.010] (Table 3).
In-hospital complications
Studied patients were hospitalized on average for 6.5 ± 3.4 days. A total of 4 patients (1.4%) died during hospitalization and the cause of death in all 4 patients was cardiac. An additional 11 patients (3.8%) died during the follow-up period; 9 deaths were due to cardiac causes and the remaining 2 were noncardiac. When the patients were grouped based on their HbA1c quartiles (Q1 = <5.5%; Q2 = 5.5–5.8%; Q3 = 5.8–6.1%; Q4 = >6.1), there was a stepwise increase in 1-year mortality in each quartile; 1-year mortality rates were 2.7%, 3.8%, 5.1% and 6.9% in the 1st through the 4th quartile of patients according to their HbA1c percentage (ordinal linearity p value = 0.031). It was found that 1-year survival was significantly better in the ‘Low’ group (p = 0.044) (Figure 2).

Kaplan–Meier analysis of 1-year survival showed that there was a significant reduction of 1-year survival in patients with hemoglobin A1c (HbA1c) >5.8%.
The frequency and the percentage of post STEMI complication during the hospitalization and 1-year follow-up period are listed in Table 4. There were 70 MACE during the hospitalization period. The prevalence of these events was similar between the ‘Low’ and the ‘High’ groups. Killip classes II and III as a measure of heart failure, recurrent ischemia and mitral valve regurgitation greater than Grade 2 were not significantly different between both groups. Less common complications including gastrointestinal bleeding were also not significantly different between two groups (Table 4). Additionally, 5 out of 148 patients (3.4%) in the ‘Low’ group and 8 out 142 patients (5.6%) in the ‘High’ group developed overt diabetes mellitus within 12 months after their initial admission. This difference was not statistically significant (p = 0.402).
Mortality and complications.

Discussion
In our study, HbA1c >5.8% in nondiabetic patients with STEMI was associated with severe CAD and multivessel involvement of the coronary arteries. Despite the similar occurrence of complications and mortality during the hospitalization period, 1-year survival was significantly decreased in patients with HbA1c >5.8. Among these patients, the rate of rehospitalization due to cardiovascular causes was also higher than those with HbA1c percentages below the median value. We believe this is in agreement with the higher CAD scores and presence of more triple-vessel disease in patients with higher HbA1c levels. Similarly, Cakmak and colleagues reported a significant correlation between HbA1c levels at admission and positive exercise test results after the 4-week follow-up period in a cohort of patients with and without known diabetes [Cakmak et al. 2008].
The International Expert Committee has recommended the use of HbA1c in diagnosing diabetes with a cutoff value of 6.5% [Gillett, 2009]. The advantages of HbA1c over the fast blood glucose are its lower intra-individual variability, assessment in nonfasting state and its superiority in monitoring blood glucose level [Selvin et al. 2007; American Diabetes Association, 2009]. Irrespective of its diagnostic role, recent studies have focused on its prognostic impact on predicting future occurrence of diabetes, CAD, cardiac and cerebrovascular death [Park et al. 1996; Preis et al. 2009; Selvin et al. 2010; Timmer et al. 2011; Pai et al. 2013]. Selvin and colleagues have suggested that glycosylated hemoglobin is superior to fasting blood glucose in predicting long-term risk of CAD in nondiabetics [Selvin et al. 2010]. Similar results are reported by Park and colleagues among nondiabetic women [Park et al. 1996]. In a cohort of European men, Khaw and colleagues reported that HbA1c correlates linearly to subsequent cardiovascular, as well as all-cause mortality [Khaw et al. 2001]. This correlation is maintained throughout the whole range of concentrations and even in the ranges used to define nondiabetic state. Based on the available evidence, the American Diabetes Association suggests that individuals with HbA1c concentrations of 5.7–6.4% should be informed of their increased risk for diabetes as well as for cardiovascular disease [American Diabetes Association, 2010].
The complexity and severity of the coronary disease was assessed by Califf score which was originally described by Robert Califf’s group in 1985 [Califf et al. 1985]. The scoring is based purely on angiographic findings (number of the diseased vessels and the degree of disease). The prognostic value of this system has been shown in predicting 5-year survival rate. In patients with a jeopardy score of 2, 97% of patients survive for 5 years; however, with each additional 2 scores, 5-year survival rates decrease (4 = 95%, 6 = 85%, 8 = 78%, 10 = 75% and 12 = 56%). This scoring system later evolved into more complex angiographic scoring systems such as SYNTAX and later SYNTAX II, and angioclinical scoring [veterans affair (VA) risk calculator and American College of Cardiology/American Heart Association (AHA/ACC) risk tools]. All scoring systems are strongly correlated with clinical outcomes such as MACE or major adverse cardiovascular and cerebrovascular events (MACCE). In all situations, the ACC/AHA high-risk criteria had the highest sensitivity, negative predictive value and negative likelihood ratio, and the Duke Treadmill Score had the highest specificity and positive predictive value. Califf scores correlated well with the extent of myocardium at risk as assessed by SPECT (R2 = 0.248, p < 0.0001) [Jaume Candell-Riera, 2001]. Although Califf’s jeopardy score has a modest predictive value of the clinical outcome compared with the other risk calculators, its simplicity and pure angiographic scoring makes it suitable to assess the severity of the CAD which was the main objective of this scoring system in our work.
We have previously established a role for activation of neutrophils in the peripheral blood of the patients after STEMI [Ghaffari et al. 2014]. Elevation of neutrophils in these patients is associated with increased frequency of pump failure and the incidence of cardiogenic shock following STEMI. Additionally, the likelihood for ventricular dysrhythmias is high in patients who had a higher count of neutrophils and white blood cells. The role of exuberant systemic inflammatory response to ischemia can directly affect both the contractile function and the relaxation of ischemic myocardium in the penumbra zone of the myocardium, and may promote the cell death extending beyond the nidus of the infarct. In a rat model of myocardial ischemia, we have previously shown that the excessive levels of tumor necrosis factor α advertently suppress myocardial contractility [Asgeri et al. 2014]. High serum levels of glucose whether in diabetic individuals or in nondiabetics are associated with variable levels of systemic inflammation and release of proinflammatory cytokines including tumor necrosis factor alpha [Gonzalez et al. 2014; Wongeakin et al. 2014]. We speculate that the association between higher blood levels of glycated hemoglobin with the severity of coronary disease and hospital readmission is related to an undergoing low level vascular inflammation with long-term promotion of atherogenesis.
The prognostic value of blood glucose level in the setting of acute myocardial infarction was tested for the first time in 1975 [Modan et al. 1975]. Kosiborod and colleagues, in a relatively large number of elderly patients with STEMI, revealed a linear relationship between admission blood glucose concentrations and short-term mortality. This association was stronger in those not diagnosed with diabetes. Less than 30% of these patients received reperfusion therapy either as thrombolytic therapy or angioplasty [Kosiborod et al. 2005]. In a cohort of 802 STEMI patients of whom 90% were treated with percutaneous coronary intervention (PCI), Ishihara and colleagues showed that blood glucose level is associated directly with 30-days mortality [Ishihara et al. 2007]. In the presence of known diabetes, the association between blood glucose levels and mortality extends up to 3 years [Ishihara et al. 2007]. In a more recent observational study of a cohort of STEMI patients treated with primary PCI, Timmer and colleagues showed that elevated HbA1c levels and admission glucose levels is associated with increases in both 1-year and long-term mortality [Timmer et al. 2011]. Only HbA1c is associated with long-term mortality, similar to our findings, while elevated glucose levels alone are associated with enzymatically defined larger infarct size.
The extent and the severity of CAD is another mechanism that may explain the association of diabetes or higher HbA1c level of nondiabetic patients with long-term outcome of patients with STEMI. Hong and colleagues reported a direct correlation between HbA1c levels and the severity of CAD based on the number of involved vessels in patients with stable angina [Hong et al. 2014]. In 299 patients with suspected ischemia undergoing diagnostic angiography, Ashraf and colleagues reported a significant association between Gensini scores and increasing HbA1c tertiles [Ashraf et al. 2013]. Our study is the first one performed in patients admitted with STEMI. We have used Califf scores as the assessment indices for the severity of CAD and have demonstrated that the underlying CAD is more severe based on higher scores in patients with higher glycosylated hemoglobin levels. Additionally, there is an association between the percentage of glycosylated hemoglobin and the number of diseased vessels.
Conclusion
Like many other studies that examine the role of HbA1c in prognostication of a poor outcome after STEMI, our study suffers from the lack of necessary power to detect any difference in hospital mortality, which fortunately has a low frequency. Multicenter prospective longitudinal studies enrolling large number of patients with relatively longer duration of follow-up period are required to address this issue. This study is also limited by the fact that all subjects have been enrolled from a single university center, which may result in sampling bias. However, we have demonstrated that there is a significant correlation between HbA1c and hospital readmission and the severity of coronary artery involvement, which may act as surrogates for a poorer long-term outcome. Therefore, we conclude that in nondiabetic patients with STEMI, higher HbA1c may suggest an extensive CAD and an increase in the probability of repeat hospitalization due to heart disease. The presence of factors such as swings in blood glucose levels may associate with ongoing atherosclerosis and the severity of coronary disease, and together in the long term, translate to a higher mortality.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declare no conflicts of interest in preparing this article.
