Abstract
Background
Primary percutaneous coronary intervention (PCI) has become the preferred reperfusion strategy over thrombolytic therapy in patients with stent thrombosis elevation myocardial infarction (STEMI) [Keeley et al. 2003]. Despite immediate reperfusion through an occluded epicardial coronary artery, myocardial perfusion still remains inadequate in 25–30% patients and results in a larger infarct size and increased mortality [van’t Hof et al. 1998]. The pathogenesis of microvascular injury resulting in the ‘no-reflow’ phenomenon is complex and multifactorial and is an important predictor of mortality, recurrent infarction and impaired ventricular function. Coronary microvascular dysfunction and ischemic reperfusion injury have been proposed as the potential mechanisms responsible for this inadequate myocardial perfusion (known as a ‘no-reflow phenomenon’) [Matsumura et al. 1998; Gottlieb et al. 1994]. Coronary microvascular dysfunction is related to distal embolization of plaque material and thrombus, microvascular spasm, myocardial edema, myocardial necrosis, leukocyte adhesion platelet aggregation all of which may contribute to the reperfusion injury [Kloner et al. 1991; Ambrosio and Tritto, 1999; Heusch et al. 2009].
Several pharmacological agents including adenosine have been studied in the prevention and treatment of the ‘no-reflow’ phenomenon [Neumann et al. 1998; De Luca et al. 2005; Hang et al. 2005; Taniyama et al. 1997; Ito et al. 1999; Ishii et al. 2005; Garratt et al. 1998; Mahaffey et al. 1993; Ross et al. 2005; Petronio et al. 2005; Barcin et al. 2004]. Previous studies comparing intravenous adenosine showed some promising results but intracoronary injection of medications has the advantage of allowing a high concentration of the drug near the distal coronary bed, with lower incidence of side effects [Petronio et al. 2005]. Also those studies were done in the era of thrombolytic therapy and their relevance in the contemporary era of primary PCI and advanced antiplatelet therapy is unknown. Clinical studies evaluating the role of the intracoronary adenosine in patients with acute myocardial infarction (AMI) undergoing primary PCI have yielded mixed results. Therefore, we performed a meta-analysis of these trials to evaluate the safety and efficacy of intracoronary adenosine administration in these patients.
Methods
We performed this review in accordance with the Quality of Reporting of Meta-analysis statement and the Consolidated Standards of Reporting Trials Group recommendations [Moher et al. 1999]. A protocol was prospectively developed, detailing the objectives, criteria for study selection and approach to assessing the study quality, primary outcome and methodology.
Literature search
We performed a computerized search to identify all relevant studies published in the English language untill May 2011 in EMBASE, CINAHL, PubMed and the Cochrane database. The key words used for the search were: acute myocardial infarction, ST elevation myocardial infarction, percutaneous coronary interventions, microvascular dysfunction, no-reflow phenomenon, adenosine and intracoronary adenosine.
Study selection
We reviewed all titles and abstracts from the results of our computerized search. We also went into the related links of all relevant articles. In addition to our computerized search, we manually reviewed the reference list of all retrieved articles to complete our search. The study selection process is outlined in Figure 1.
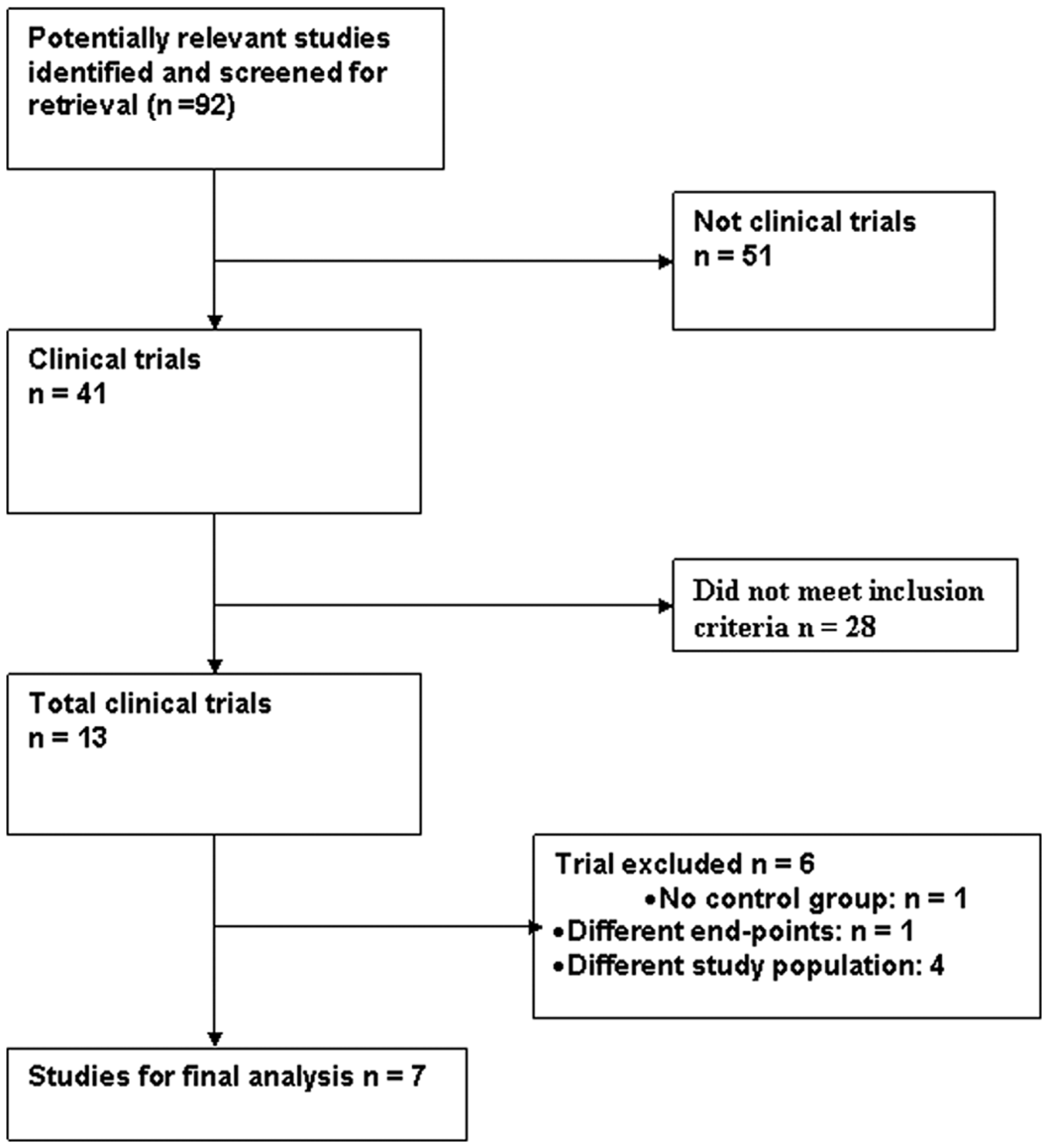
Study selection process.
Inclusion criteria
Studies had to meet all the following criteria to be included in the analysis:
(1) Prospective randomized controlled trial design.
(2) Include patients with AMI undergoing primary PCI
(3) Compare intracoronary adenosine with placebo group.
(4) Report at least one of the clinical, angiographic, cardiac imaging related, electrocardiography (EKG) related, cardiac biomarkers or safety-related outcomes, including all-cause mortality, cardiovascular (CV) mortality, heart failure (HF), major adverse cardiovascular event (MACE), thrombolysis in myocardial infarction (TIMI) grade III flow (TIMI 3 flow), myocardial blush grade (MBG) 3, mean difference in post-PCI ejection fraction (EF), post-procedure residual stent thrombosis (ST) segment elevation and ST segment resolutions (STRes), difference in peak creatine kinase (CK-MB) concentration, bradycardia, second degree atrioventricular block (AVB), ventricular tachycardia (VT), ventricular fibrillation (VF) and recurrence of chest pain (CP).
Exclusion criteria
Studies that did not meet the above criteria were excluded.
Data abstraction
After identifying all the relevant articles, we extracted characteristics of the study (author, year, design, duration, sample size, study patient population, clinical outcomes, angiographic outcomes, cardiac imaging related outcomes, EKG-related outcomes, cardiac enzymes comparison, safety endpoints and follow-up percentage). Two reviewers independently extracted data and assessed outcomes. The inter-rater agreement was 90%, and disagreements were resolved by consensus.
Quality assessment
All the trials reported adequate concealment of the randomized treatment sequence. In all studies, follow up was more than 90% complete.
Statistical analysis
The statistical analysis was performed using the Comprehensive Meta-Analysis software (Biostat, Englewood, NJ, USA). Heterogeneity of the studies was assessed for each outcome. Studies that were homogenous for an outcome were analyzed by the Mantel-Haenszel fixed effect model, while those studies that were heterogeneous for an outcome were analyzed by the random effect model. For each categorical variable, such as CV death or MACE, the common relative risk (RR) was, while for continuous variables, such as EF or CK-MB, the common mean differences between the adenosine and placebo groups were computed. A two-sided α error of less than 0.05 was considered to be statistically significant (p < 0.05).
Results
Literature search
A total of 92 articles were identified of which 41 were potentially relevant studies and screened for retrieval. After title and abstract screening, 28 studies were excluded and the remaining 13 studies were retrieved for a more detailed evaluation. Out of these 13 studies, 6 were excluded; out of these 6 excluded studies, 1 study had no control groups [Lim et al. 2004], 4 studies had different study population [Assali et al. 2000; Quintana et al. 2003; Kim et al. 2011; Pepine et al. 2010] and 1 study had different endpoints [Tsao et al. 2009]. Thus, a total of seven clinical trials were included in the final analysis [Marzilli et al. 2000; Claeys et al. 2004; Hendler et al. 2006; Stoel et al. 2008; Fokkema et al. 2009; Desmet et al. 2011; Grygier et al. 2011].
Endpoints
Table 1 lists the endpoint definitions used in the individual studies included in this meta-analysis. Out of seven trials, six [Marzilli et al. 2000; Claeys et al. 2004; Stoel et al. 2008; Fokkema et al. 2009; Desmet et al. 2011; Grygier et al. 2011] had clinical and EKG-related outcomes as their endpoints. Four trials had TIMI 3 flow as endpoints [Marzilli et al. 2000; Fokkema et al. 2009; Desmet et al. 2011; Grygier et al. 2011]. The angiographic outcome TIMI 3 flow was graded using a TIMI study criterion which was measured either at the end of the balloon dilation procedure [Marzilli et al. 2000; Grygier et al. 2011] or after primary PCI with stenting [Fokkema et al. 2009; Desmet et al. 2011]. Another angiographic outcome, MBG3, was defined as a normal myocardial blush or contrast density, comparable with that obtained during angiography of a non-infarct-related coronary artery territory [van’t Hof et al. 1998]. Three trials provided data on MBG3 outcomes [Fokkema et al. 2009; Desmet et al. 2011; Grygier et al. 2011]. Cardiac imaging related outcome (mean difference in post-PCI EF) which was assessed by either echocardiography [Hendler et al. 2006; Grygier et al. 2011] or magnetic resonance imaging [Desmet et al. 2011]. All clinical trials had safety events as one of their endpoints, except one trial [Hendler et al. 2006].
Endpoint definitions.

AMI, acute myocardial infarction; CV, cardiovascular; HF, heart failure; MACE, major adverse cardiovascular event; NA, not available; PCI, percutaneous coronary intervention; ST,
Overview of study and patient characteristics
The trials included in this meta-analysis consisted of a total of 1030 patients (placebo group, n = 570; treatment group, n = 460). Study design was similar in all seven trials: comparison between a placebo arm and an intracoronary adenosine arm. Intra coronary administration protocol was either as boluses or continuous infusion during primary PCI. The results of the meta-analysis are shown in Figures 2–7. Baseline characteristics of individual trials are given in Tables 2 and 3.
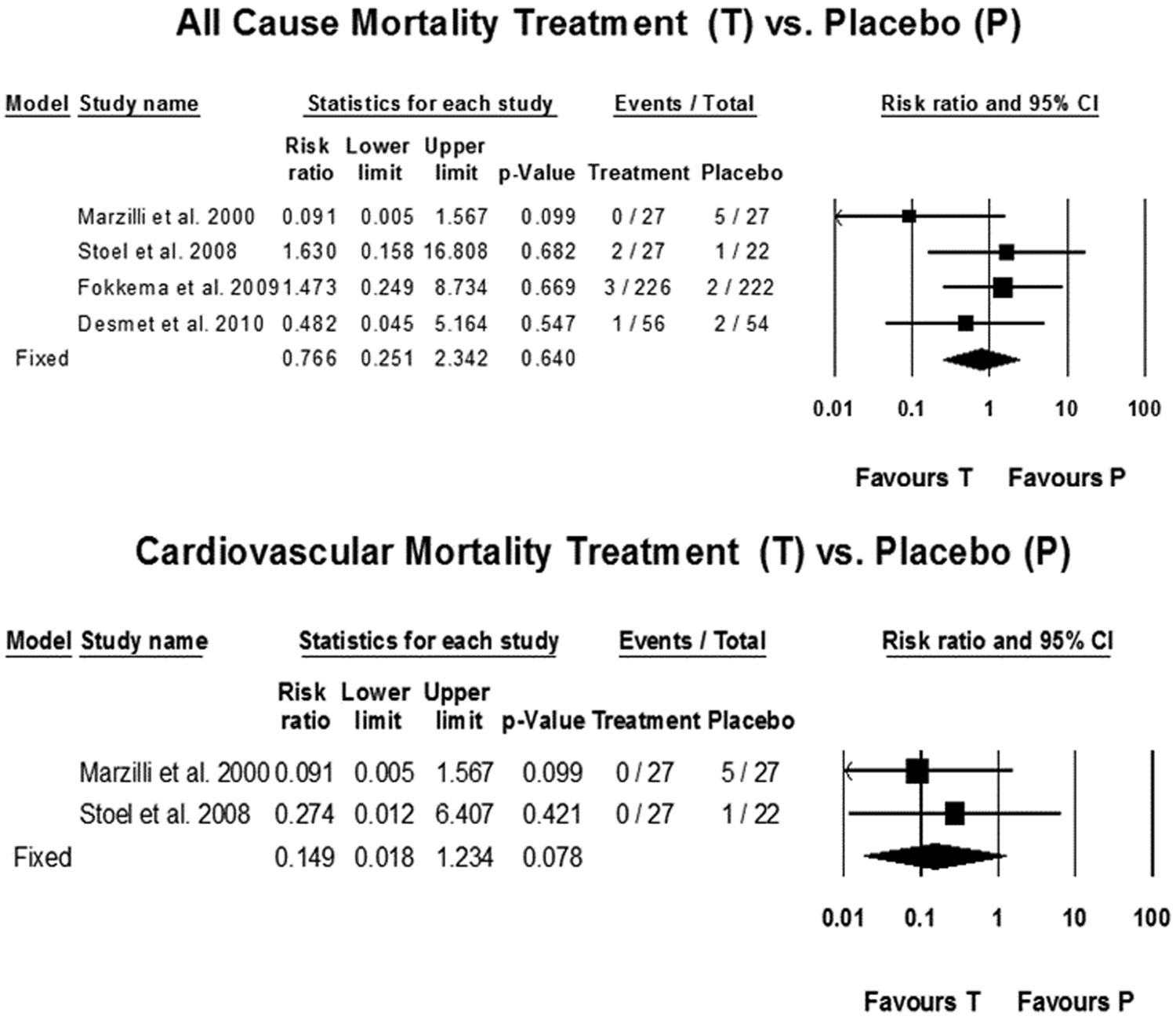
All-cause mortality (upper panel) and cardiovascular mortality (lower panel). CI, confidence interval.

Heart failure (upper panel) and major adverse cardiovascular event (lower panel). CI, confidence interval.

Stent thrombosis segment resolutions (STRes) (upper panel) and residual ST elevation (lower panel). CI, confidence interval.
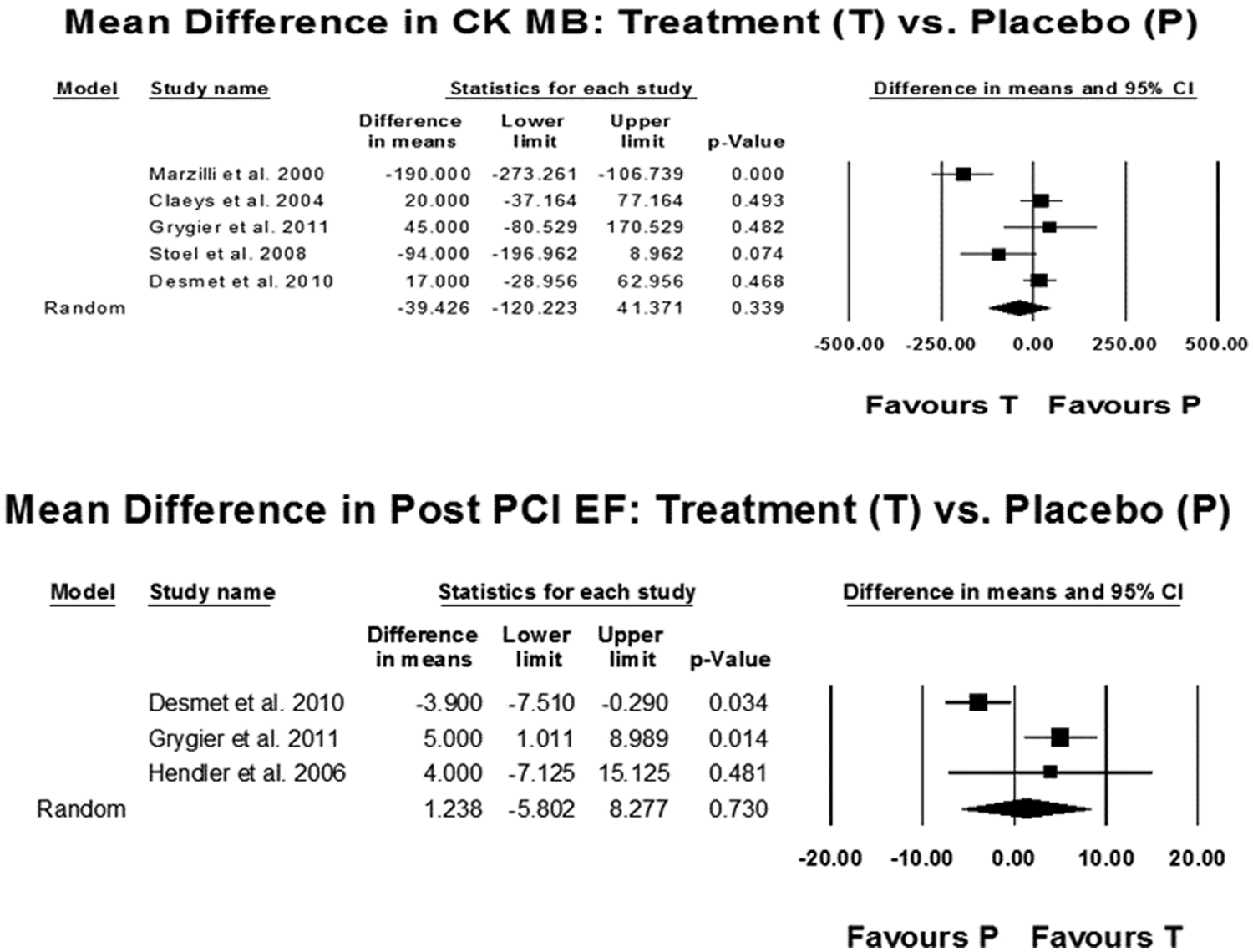
Cardiac biomarkers (upper panel) and mean difference in post percutaneous coronary intervention ejection fraction (PCI EF) (lower panel). CI, confidence interval; CK, creatine kinase.
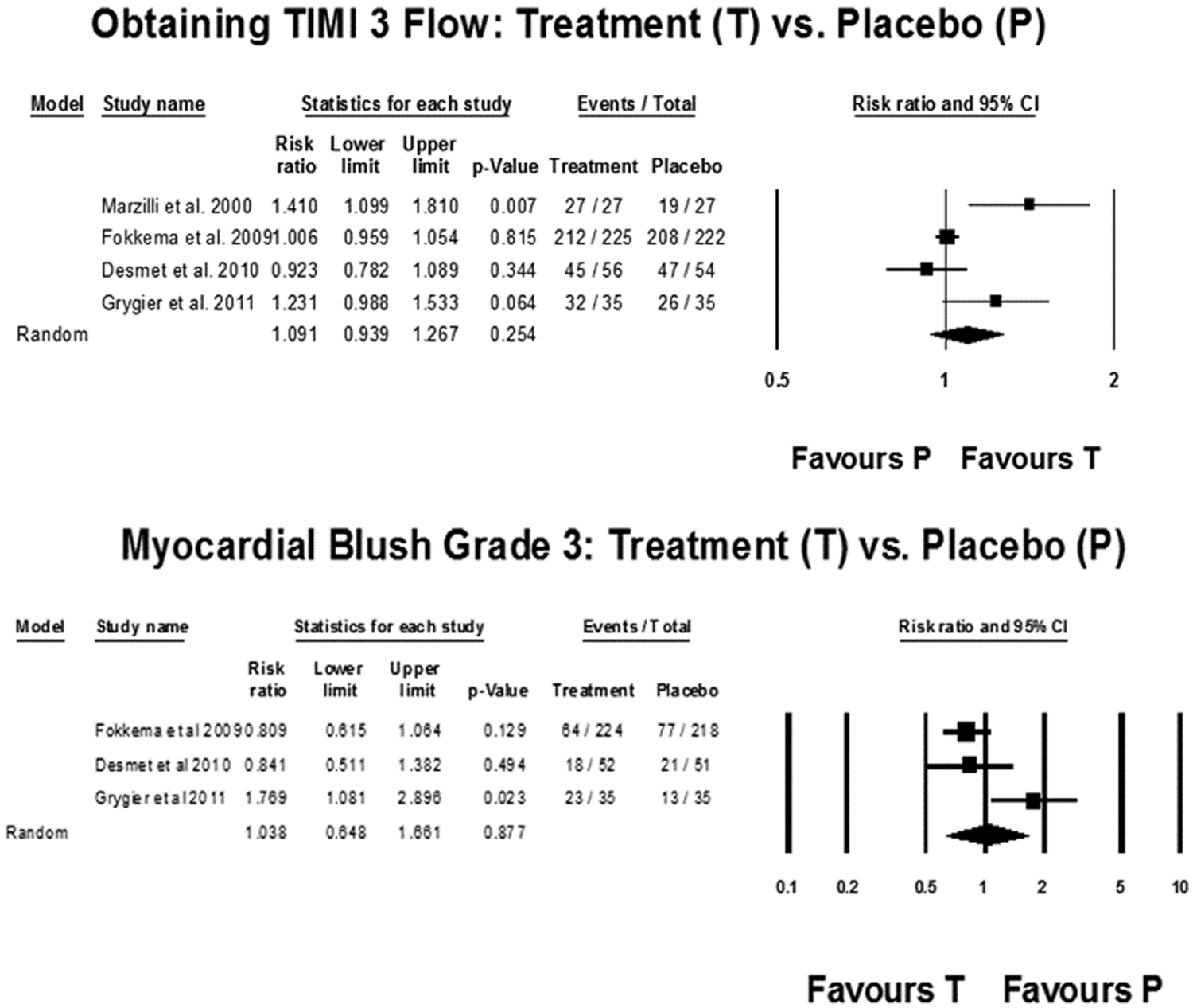
Thrombolysis in myocardial infarction (TIMI) grade 3 flow (upper panel) and myocardial blush grade 3 (lower panel). CI, confidence interval.

Safety analysis. AV, atrioventricular; CI, confidence interval; CP, chest pain; VF, ventricular fibrillation.
Study characteristics.

AMI, acute myocardial infarction; AVB, atrioventricular nodal block; CP, chest pain; CV, cardiovascular; EF, ejection fraction; HF, heart failure; MACE, major adverse cardiovascular event; MBG, myocardial blush grade; PCI, percutaneous coronary intervention; RCT, randomized control trial; ST, stent thrombosis; STEMI, elevation myocardial infarction; STRes, ST segment resolution; TIMI, thrombolysis in myocardial infarction; VF, ventricular fibrillation; VT, ventricular tachycardia; LCA, left coronary artery; RCA, right coronary artery.
Characteristics of study population.

Ant infarction, anterior infarction; DM, diabetes; HTN, hypertension; LAD, left anterior descending artery; MI, myocardial infarction, NA, not available.
Heterogeneity testing.
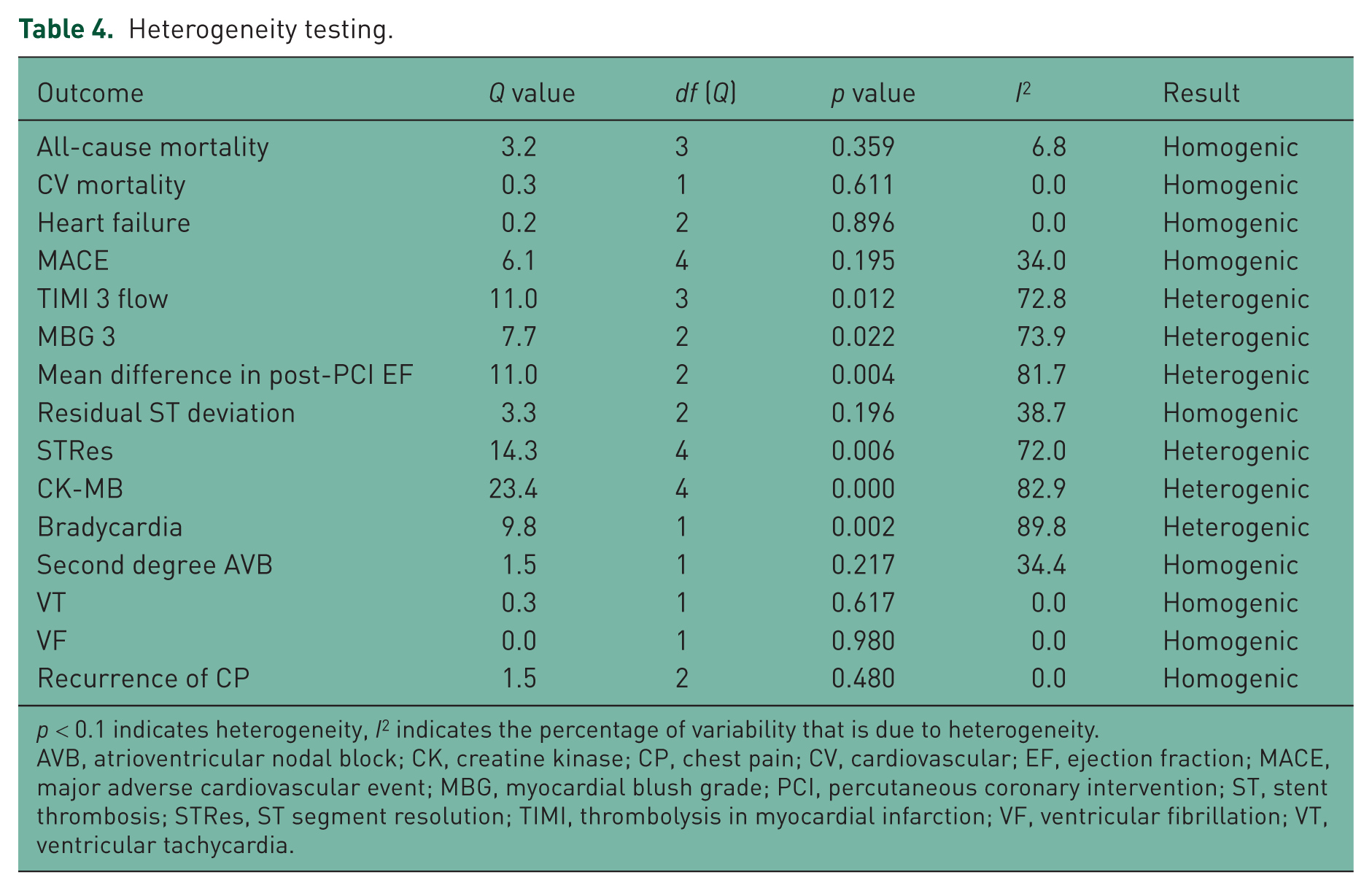
p < 0.1 indicates heterogeneity, I2 indicates the percentage of variability that is due to heterogeneity.
AVB, atrioventricular nodal block; CK, creatine kinase; CP, chest pain; CV, cardiovascular; EF, ejection fraction; MACE, major adverse cardiovascular event; MBG, myocardial blush grade; PCI, percutaneous coronary intervention; ST, stent thrombosis; STRes, ST segment resolution; TIMI, thrombolysis in myocardial infarction; VF, ventricular fibrillation; VT, ventricular tachycardia.
Clinical outcomes
There was no significant difference in the all-cause mortality [RR 0.77, 95% confidence interval (CI) 0.25–2.34; p = 0.64], or between the two groups (Figure 2). Proportionately more MACE were noted in the placebo group and there was a trend towards benefit with intracoronary adenosine therapy but it could not reach statistical significance [27 (6.85%) events in the adenosine group versus 48 (9.49%) in the placebo group with RR 0.64, CI 0.40–1.03; p = 0.06] (Figure 3). A similar trend was noted for HF events (RR 0.47, CI 0.19–1.12; p = 0.08), and CV mortality (RR 0.15, CI 0.02–1.23; p = 0.08) (Figures 2 and 3).
Electrocardiography and creatine kinase
A total of 225 (58.75%) subjects in the adenosine group had post-PCI STRes compared with 217 (43.23%) subjects in the placebo group. Analysis revealed significant post-PCI STRes (RR 1.39, CI 1.01–1.90; p = 0.04) and reduction in residual ST segment elevation (RR 0.82, CI 0.69–0.99; p = 0.04) in the adenosine group compared with the placebo group (Figure 4). However, mean difference in peak CK-MB concentration did not differ significantly between the two groups (mean difference –39.43, CI –120.223 to 41.371; p = 0.339) (Figure 5).
Post-percutaneous coronary intervention ejection fraction change
Intracoronary adenosine treatment did not lead to improvement in post-PCI EF (mean difference 1.238, CI –5.802 to 8.277; p = 0.730) compared with the placebo group (Figure 5).
Angiographic outcomes
Post-procedure TIMI 3 flow rate was similar between the two groups (RR 1.09, CI 0.94–1.27; p = 0.25) (Figure 4). Similar results were obtained for achievement of MBG3 with adenosine therapy (RR 1.04, CI 0.65–1.69; p = 0.88) (Figure 6).
Safety analysis
Overall there was no significant difference in CP events (RR 1.26, CI 0.55–2.86; p = 0.58), bradycardia (RR 2.19, CI 0.24–0.38; p = 0.49), VT (odds ratio 0.61, CI 0.08–4.90; p = 0.64) and VF (RR 0.49, CI 0.13–1.90; p = 0.30) between the two groups. However, the incidence of second degree AVB was significantly higher in the adenosine group (RR 7.88, CI 4.15–14.9; p < 0.01) compared with the placebo group (Figure 7).
Discussion
Angiographic evidence of impaired tissue perfusion, known as the ‘no-reflow’ or ‘slow-reflow’ phenomenon, is a serious complication associated with STEMI in patients undergoing primary PCI [Forman et al. 2006, 2008]. Various mechanisms implicated in the pathogenesis of the no-reflow phenomenon include oxygen-derived free radicals, distal embolization to micovascular bed, inflammatory marker mediated myocardial dysfunction and myocardial edema, localized neutrophil activation and platelet activity, amplification of the local release of serotonin and potent vasoconstrictor endothelin-1 by stent insertion, and angioplasty and activation of the sympathetic nervous and renin–angiotensin systems [Gech et al. 1996; Stone et al. 2005; Meldrum, 1998; Kerins et al. 1989; Leosco et al. 1999; Taylor et al. 2004; Gibson and Schomig, 2004]. Adenosine has been postulated to have strong vasodilator activity, anti-inflammatory, antiplatelet and postconditioning effect [Rosales et al. 2004; Headrick et al. 2003; Kloner and Rezkalla, 2006] which all can be of benefit in the prevention and treatment of the no-reflow phenomenon.
The results of the present meta-analysis show that in patients with STEMI, intracoronary adenosine therapy leads to improvement in post-PCI STRes. However, this did not translate into significant improvement in angiographic or clinical outcomes. There was a trend towards improvement in MACE (RR 0.64, CI 0.40–1.03; p = 0.06) but it could not reach statistical significance. A similar but less strong trend was also noted for HF events (RR 0.47, CI 0.19–1.12; p = 0.08) and CV mortality (RR 0.15, CI 0.02–1.23; p = 0.08). The reason for this disconnect may be due to small study populations, low event rates in individual trials, or short follow-up duration.
In animal models, it has been described that levels of adenosine may increase to levels producing maximal coronary arteriolar dilatation during ischemia [Rubio et al. 1969]. So it is possible that because of the high level of endogenous production of adenosine during myocardial ischemia, maximal vasodilatation is already present in patients with STEMI, and therefore, no further improvement is seen due to the absence of vasodilatory reserve during and after reperfusion by PCI. This may explain the absence of improvement in TIMI 3 flow with intracoronary adenosine as seen in the present meta-analysis. Moreover, the optimum dosing regimen for intracoronary adenosine is unknown. The difference in dosing regimen may be a critical factor in obtaining a good versus a neutral response due to the very short half life of this molecule. The amount of drug, timing of drug delivery, and the site of drug delivery are all critically important factors that need to be defined. Five studies in our analysis involved bolus injections [Marzilli et al. 2000; Hendler et al. 2006; Fokkema et al. 2009; Desmet et al. 2011; Grygier et al. 2011] while two involved infusion of adenosine [Claeys et al. 2004; Stoel et al. 2008]. Similarly the timing and site of drug delivery were different, which may have affected the results (Table 2).
Recently MBG has been demonstrated superior in predicting microcirculatory flow and tissue perfusion [Forman et al. 2006]. We did not find any beneficial effects of intracoronary adenosine on MBG. Our analysis of MBG3 included three clinical trials but only one out of these trials [Fokkema et al. 2009] allowed for manual thrombus aspiration while the study protocol of the other two trials [Desmet et al. 2011; Grygier et al. 2011] was designed before the era of thrombectomy and the distal protection device, which may decrease the distal emobilization responsible for poor MBG3. Thus it is possible that the contemporary treatment of patients with STEMI with thrombus aspiration and the administration of the glycoprotein IIb/IIIa inhibitor abciximab during primary PCI may result in an improvement in myocardial reperfusion [Svilaas et al. 2008; De Luca et al. 2005].
Postprocedural elevations of cardiac biomarkers like CK-MB and troponins are shown to be related to infarct size, late mortality and recurrent infarction [Antman et al. 1996; Cavallini et al. 2005]. In our analysis, the level of peak CK-MB was no different between the two arms, indicating no obvious benefit of intracoronary adenosine (Figure 5). This combined with the above findings can explain the lack of benefit seen in clinical outcomes. However, the only cardiac biomarker assessed was CK-MB because no data were available for troponins, which are more sensitive and specific markers.
The safety endpoint analysis demonstrated that intracoronary adenosine administration was associated with approximately a 17–18-fold higher rate of second degree AVB than placebo. However, simultaneously there was no increase in chest pain, bradycardia, VT or VF events. However, these were transient events due to the short half life of adenosine.
Adenosine is an important component of the interventional cardiologist's toolbox and plays a useful role in patients with slow flow or no reflow. Studying the effects of adenosine in these patients alone is likely to establish a benefit but may be a difficult study to do as few operators would be willing to randomize these patients to placebo. The trend towards MACE benefit that was seen in all patients with myocardial infarction should therefore not be minimized. In sufficient numbers this would likely have shown a benefit. Transient AVBs are expected with adenosine and should be appropriately minimized.
As with any meta-analysis, one of the limitations of our study is the difference in the definitions of the endpoints in the component trials. Baseline characteristics between two groups cannot be compared completely in most meta-analyses because of differences in the study protocols across the component trials. Also, there is a potential for publication bias but the trials in our analysis had different results which should reduce this potential risk.
Conclusion
Our results provide evidence that intracoronary adenosine may be a useful therapy as indicated by improvement in EKG findings. A trend towards improvement was noted in MACE and HF events but at present there are not enough data to reach strong conclusions on clinical outcomes. This also underscores the importance of conducting further long-term studies with larger numbers of patients to evaluate the clinical endpoints. Moreover, this meta-analysis highlights the gaps in the available literature about the optimum dose, timing and site of intracoronary adenosine administration which need further investigation.
Footnotes
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
The authors declare no conflicts of interest in preparing this article.
