Abstract
Background:
Coronary heart disease (CHD) is the number one cause of death in the US. The adipokine adiponectin has been studied intensively for presenting and inversed association with almost every stage of CHD. For instance, the evaluation of molecules capable of enhancing endogenous adiponectin expression is well justified. In this study, we investigated the effect of the vitamin D receptor activator (VDRA) paricalcitol and the angiotensin-converting enzyme inhibitor (ACEI) enalapril on adiponectin expression, lipid profiles, adenosine monophosphate activated protein kinase (AMPK) expression, monocyte chemo-attractant protein-1 (MCP-1), tumor necrosis factor-alpha (TNFα),cyclooxygenase-2 (COX-2), inducible nitric oxide synthase (iNOS), antioxidant capacity, CuZn-superoxide dismutase (CuZn-SOD), Mn-SOD, NADPH p22phox subunits, inducible nitric oxidesynthase (iNOS), endothelial marker eNOS, and 81 atherosclerosis-related genes in ApoE-deficient mice.
Method:
Seven-week-old ApoE-deficient mice were treated for 16 weeks as follows: Group 1, ApoE vehicle control (intraperitoneal [i.p.] 100 µl propylene glycol); Group 2, ApoE-paricalcitol (200 ng i.p., 3/week); Group 3, ApoE-Enalapril (30 mg/kg daily); Group 4, ApoE-paricalcitol + enalapril (described dosing); and Group 5, wild-type control (C57BLV).
Results:
All treated groups presented significant changes in circulating and cardiac adiponectin, cardiac cholesterol levels, AMPK, MCP-1, TNF-α, COX-2, iNOS, eNOS, CuZn-SOD, Mn-SOD and p22phox. There were 15 genes that differed in their expression, 5 of which are involved in cardioprotection and antithrombotic mechanisms: Bcl2a1a, Col3a1, Spp1 (upregulated), Itga2, and Vwf (downregulated).
Conclusion:
Together, our data presented a novel role for VDRA and ACEI in reducing factors associated with CHD that may lead to the discovery of new therapeutic venues.
Introduction
Coronary heart disease (CHD) is the number one cause of death in the US [Go et al. 2013]. Several studies have shown an inverse association between circulating adiponectin levels and the development of CHD at almost every stage [Nimitphong et al. 2009; Gannage-Yared et al. 2009; Trujillo et al. 2005; Lam et al. 2005]. This multimeric peptide hormone has been proposed as a key effector in insulin sensitivity and lipid metabolism, promoting weight loss and the reduction of triglycerides, even when combined with a high-fat diet [Tsatsanis et al. 2006; Ouchi et al. 2003; Yamauchi et al. 2001]. In homozygous Apo-lipoprotein E-deficient mice (ApoE-), adiponectin treatment (either globular or adenovirus-mediated) decreased the atherosclerotic-lesion formation [Ouchi et al. 2006; Hopkins et al. 2007; Okamoto et al. 2002]. Adiponectin protects against vascular disorders, as observed in a mouse model of spontaneous atherosclerosis [Yamauchi et al. 2003]. It also prevents almost every pathogenic event involved in atherosclerotic plaque formation by augmenting the endothelial nitric oxide (NO) production [Ouchi et al. 2003; Arita et al. 2002], reducing oxidative stress [Ouedraogo et al. 2006; Motoshima et al. 2004; Tao et al. 2007; Furukawa et al. 2004; Nakanishi et al. 2005], inhibiting endothelial cell activation [Kobashi et al. 2005; Tan et al. 2004], suppressing leukocyte-endothelium interaction [Ouedraogo et al. 2006; Ouedraogo et al. 2007; Joussen et al. 2004], inhibiting macrophage activation and foam cell formation [Park et al. 2007; Masaie et al. 2007; Peake et al. 2006; Kumada et al. 2004], regulating phagocytosis [Takemura et al. 2007; Saijo et al. 2005], antithrombotic activities [Shoji et al. 2006; Kato et al. 2006], and inhibiting smooth muscle proliferation [Okamoto et al. 2002; Hansmann et al. 2007; Wang et al. 2005; Kubota et al. 2002]. For instance, the identification of molecules that increase the production of endogenous adiponectin, and as a consequence its cardioprotective effects, would have a direct impact on the current strategies for preventing CHD. In this study, we investigated the effect of the vitamin D receptor activator (VDRA) paricalcitol and angiotensin-converting enzyme inhibitor (ACEI) enalapril in the production of adiponectin in an atherosclerosis mouse model and evaluated the downstream influence on lipids profile, macrophage chemo-attractant protein (MCP-1), tissue necrotic factor-alpha (TNF-α), cyclo-oxygenase 2 (COX-2), NADPH p22phox subunit, inducible nitric oxide synthase (iNOS), endothelial nitric oxide synthase (eNOS), superoxide dismutases (CuZn-SOD and Mn-SOD), adenosine monophosphate-activated protein kinase (AMPK), and 81 atherosclerosis-related markers in the heart of the atherosclerosis model of ApoE deficient mice.
Materials and methods
Chemicals
Enalapril was purchased from Sigma Chemical Company (St Louis, MO, USA) and VDRA (paricalcitol) was provided by Abbott Pharmaceuticals (Abbott Park, IL, USA). EnzyChrom Cholesterol Assay Kit (ECCH-100), EnzyChrom Triglyceride Assay Kit (ETGA-200), and EnzyChrom HDL and LDL/VLDL Assay Kit were purchased from BioAssay Systems (Hayward, CA, USA). Adiponectin kit was purchased from R&D Systems (Minneapolis, MN, USA). Antibodies were purchased from Santa Cruz Biotechnology, Santa Cruz, CA, USA and Abcam, Cambridge, MA, USA. RNA isolation kit was purchased from QIAGEN, Valencia, CA, USA.
Animals and treatments
Seven-week-old female mice weighing 23 ± 3 g were purchased from Taconic (Hudson, NY, USA). All animals were fed with NIH31 standard rodent diet and water
Following 16-week treatment, mice were euthanized with ether anesthesia and decapitated. Blood was collected in EDTA-coated vials and plasma was separated by centrifugation at 5000 rpm for 10 min. Hearts (
Biochemical analysis
We used EnzyChrom Cholesterol Assay Kit (ECCH-100), EnzyChrom Triglyceride Assay Kit (ETGA-200), and EnzyChrom HDL and LDL/VLDL Assay Kit from BioAssay Systems (Hayward, CA) to determine lipids in plasma, following the manufacturer’s instructions. Cardiac tissues (50.0 mg) were homogenized in 1.0 ml of 50 mM phosphate buffer containing 0.1 mM EDTA (pH 7.0) and were kept at −80°C until assayed. The antioxidant capacity was determined from cardiac homogenates using Cayman Antioxidant Assay (CAT#709001). The BioAssay Systems (Hayward, CA, USA) EnzyChrom AF Cholesterol Assay Kit (E2CH-100) was used (as specified by the manufacturer’s protocols) for determining cholesterol levels in the cardiac tissue. Circulating and tissue-specific (cardiac tissue) adiponectin levels were determined and expressed as ng/ml using R&D Systems’s (Minneapolis, MN, USA) Quantikine Mouse Adiponectin/Acpr30 enzyme-linked immunosorbent assay (ELISA) kit (MRP300), following the manufacturer’s instructions.
Western blot analysis
Protein extraction and Western blot analyses were performed as described previously [Husain et al. 2010]. Briefly, extracted proteins from tissues (40 µg) were resolved on 12.5% SDS polyacrylamide gel (SDS PAGE) resolving gel and a 5% stacking gel. Proteins were then electrotransferred onto nitrocellulose membranes at 120 volts for 1 h. After blocking in 5% nonfat powdered milk for 1 h, the membranes were treated with antibodies to MCP-1, TNF-α, COX-2, iNOS, eNOS, CuZn-SOD, Mn-SOD, p22phox and β-actin (1:1000 and 1: 5000) overnight at 4°C (Santa Cruz Biotechnology, Santa Cruz, CA, USA; Abcam, Cambridge, MA, USA). After washing the blot was incubated with horseradish peroxidase-conjugated secondary antibody IgG (1:5000 and 1:10000) for 1 h at room temperature (Santa Cruz Biotechnology, Santa Cruz, CA). The washed blot was then treated with SuperSignal West Pico chemiluminescent substrate (Pierce, Rockford, IL) for positive antibody reaction. Membranes were exposed to X-ray film (KODAK) for visualization and scanning of protein bands using Alpha Imager software.
Histological analysis
Tissues were fixed in 10% buffered formalin and then paraffin embedded. We used rabbit polyclonal antibodies against adiponectin (10 µg/ml), adiponectin receptor 1 (AR1) (1/100), and AMPK 1 (1/100) from Abcam plc. (Cambridge, MA, USA). Sections (10 µm) were labeled using the Vectastain ABC IgG peroxidase system and stained with an ImmPact DAB substrate kit (Vector Laboratories, Inc., Burlingame, CA, USA). Images were captured with a NIKON Eclipse N400 model and printed. The images were scored (0–5) in a blind fashion and then expressed as percentages as reported by evaluators in the following scale: 0, none; 1–2, weak; 3, moderate; 4–5, strong.
PCR arrays
RNA isolation from cardiac tissue stored in RNA later Stabilization Reagent was performed using RNA (QIAGEN, Valencia, CA). The RNA was quantified by spectrophotometry (Eppendorf BioPhotometer) and evaluated for integrity by electrophoresis. RT2 Profiler PCR Arrays (PAMM-038, SAbiosciences, Qiagen, Hercules CA) were run in a BioRad iCycler after reverse transcription and amplification, following the PCR Array protocol as provided by the manufacturer (SABiosciences, Hercules, CA). Analyses based on Ct values were determined by using PCR Array data analysis software (SABiosciences, Hercules, CA). The cut off to consider fold changes significant was 4.0.
Statistical analysis
The data were expressed as mean ± SEM and analyzed statistically using one-way analysis of variance (ANOVA). Two-way ANOVA was followed by
Results
Lipids profiles, adiponectin and antioxidant levels
The lipid profile included the circulatory levels of free glycerols, high-density lipoproteins (HDL), low-density lipoproteins (LDL), total lipids, cholesterol and triglycerides which are depicted in Table 1. The HDL levels were decreased in the group treated by the combination of paricalcitol and enalapril, and cholesterol levels were higher in groups that included paricalcitol as a treatment, either alone or combined with enalapril. All treatments were effective in reducing cholesterol levels in the heart by more than 50% (
Plasma lipid profile.

HDL, high-density lipoprotein; LDL, low-density lipoprotein
Adiponectin levels in plasma and adiponectin, cholesterol, and antioxidant (Trolox) levels in cardiac tissue.

Fold regulation; *consistent in all the treatments and wild-type control; †attributable to paricalcitol treatment; ‡attributable to enalapril treatment; §relevant to the scope of study and observed only in a treated group;
Apoe: apolipoprotein E; Bcl2a1a: B-cell leukemia/lymphoma 2-related protein A1a; Bcl2l1: Bcl2-like 1; Ccl2: chemokine (C-C motif) ligand 2; Ccr2: chemokine (C-C motif) receptor 2; Col3a1: collagen, type III, alpha 1; Eng: endoglin; Fn1: fibronectin 1; Icam1: intercellular adhesion molecule 1; Il1b: interleukin 1 beta; Itga2: integrin alpha 2; Mmp3: matrix metallopeptidase 3; Npy: neuropeptide Y; Ptgs1: prostaglandin-endoperoxide synthase 1; Sell: selectin, lymphocyte; Selp: selectin, platelet; Serpinb2: serine or cysteine) peptidase inhibitor, clade B, member 2; Spp1: secreted phosphoprotein 1; Thbs4: thrombospondin 4; Vwf: Von Willebrand factor homolog.
Western blot protein expressions
Cardiac NADPH oxidase subunit p22phox protein expression was increased in atherosclerotic mice compared with wild-type controls indicating an excess production of superoxide (Figure 1). Paricalcitol treatment decreased the subunit expression compared with ApoE-control. However, enalapril alone and in combination with paricalcitol protected against cardiac oxidative stress by downregulating the subunit p22phox protein expression of NADPH oxidase thus, lowering the superoxide production in the heart. There was an increase in cardiac mitochondrial Mn-SOD protein expression whereas cytosolic CuZn-SOD protein expression decreased in atherosclerotic mice compared with their normal counterparts (Figure 1). Paricalcitol and enalapril alone or in combination decreased the Mn-SOD induction and increased CuZn-SOD protein expression compared with ApoE-control.

Western analysis showing the effect of paricalcitol and enalapril, alone or in combination, on the expression of nitric oxide generating enzymes, inducible enzyme (iNOS), and constitutive endothelial nitric oxide synthase (eNOS), in the heart of mice. Protein expression was determined by Western analysis. The upper panel is a representative Western blot and the lower panel is the quantification of several analyses. The data are presented after normalization to β-Actin. Data are expressed as mean ± SEM,
The cardiac iNOS, MCP-1, TNF-α, and COX-2 protein expressions were increased whereas eNOS protein expression decreased in atherosclerotic mice compared to their normal counterparts in Group 5 (Figures 2 and 3) indicating the cardiac inflammatory response and oxidative endothelial injury in atherosclerosis. Paricalcitol and enalapril alone or in combination decreased the iNOS, MCP-1, TNF-α, and COX-2 induction compared with ApoE-control. Similarly, enalapril and paricalcitol alone and in combination protected against cardiac endothelial oxidative injury by enhancing eNOS protein expression compared with the ApoE-controls.

Western analysis showing the effect of paricalcitol and enalapril, alone or in combination, on superoxide generating enzyme NADPH oxidase subunit p22phox and superoxide scavenging enzymes Mn-SOD and CuZn-SOD protein expression in the heart of mice. Protein expression was determined by Western analysis. The upper panel is a representative Western blot and the lower panel is the quantification of several analyses. The data are presented after normalization to β-Actin. Data are expressed as mean ± SEM,
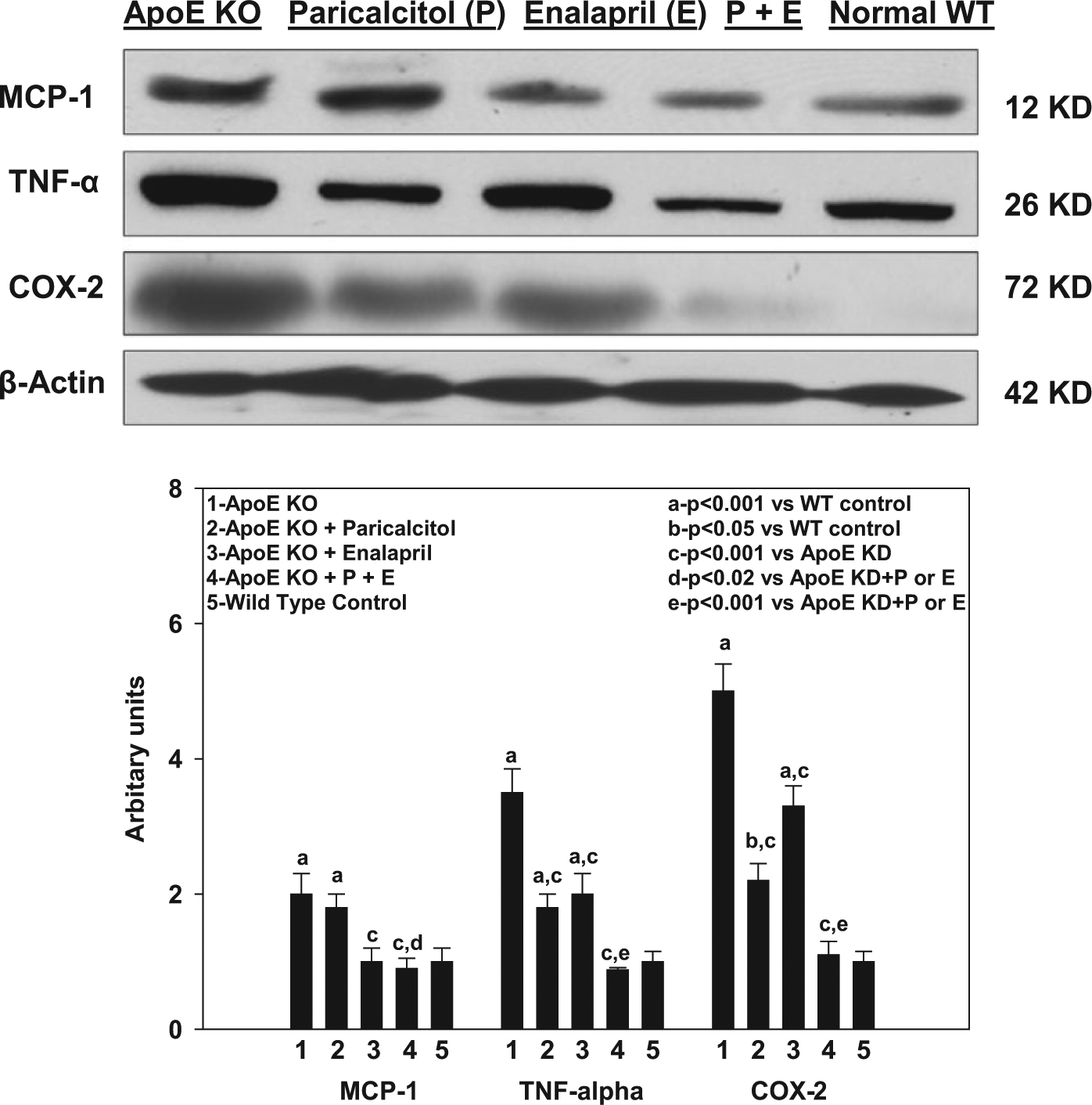
Western analysis showing the effect of paricalcitol and enalapril, alone or in combination, on macrophage chemo-attractant protein (MCP-1); tumor necrotic factor-alpha (TNF-α) and cyclooxygenase 2 (COX-2) protein expression in the heart of mice. Protein expression was determined by Western analysis. The upper panel is a representative Western blot and the lower panel is the quantification of several analyses. The data are presented after normalization to β-Actin. Data are expressed as mean ± SEM,
Histological analysis
We scored the staining intensity of AMPK, Adiponectin receptor 1 (AR1), and Adiponectin which are depicted in Figures 4–6. AMPK was more abundant in all treated groups compared with what was found in the ApoE-deficient control group (Figure 4). The two groups treated with paricalcitol had a greater degree of AMPK staining (followed by the enalapril-treated group) than did the wild-type control group. Compared with the ApoE-deficient control group, all other treatment groups tended toward having increased AR1s (Figure 5). In cardiac tissue, adiponectin immunostain scoring was noticeably increased in all of the treated groups (Figure 6) which was confirmed by the adiponectin measurement by ELISA (Table 2).

The effect of paricalcitol and enalapril, alone or in combination, on the percent immunostain intensities of AMP-Kinase in the heart of mice.

The effect of paricalcitol and enalapril, alone or in combination, on the percent immunostain intensities of adiponectin receptor 1 (AR1) in the heart of mice.

The effect of paricalcitol and enalapril, alone or in combination, on the percent immunostain intensities of adiponectin in the heart of mice.
PCR arrays of gene expressions
We analyzed the expression of 81 genes associated with atherosclerosis and lipid metabolism (either as risk factors or protectors). The fold-change in expression was determined by comparing the genetic expression of each group with that found in the ApoE- control group. As shown in Table 3, there was a difference in expression between the groups, and we focused our analyses on those genes that (1) are expressed in treated groups as well as in the CBL57 (nondiseased phenotype) and (2) are relevant to cardioprotection and the scope of this article. The information presented in Table 3 shows that there were significant changes in the expression of 15 genes.
Gene expression determined by real-time in comparison with ApoE-Control (no treatment).
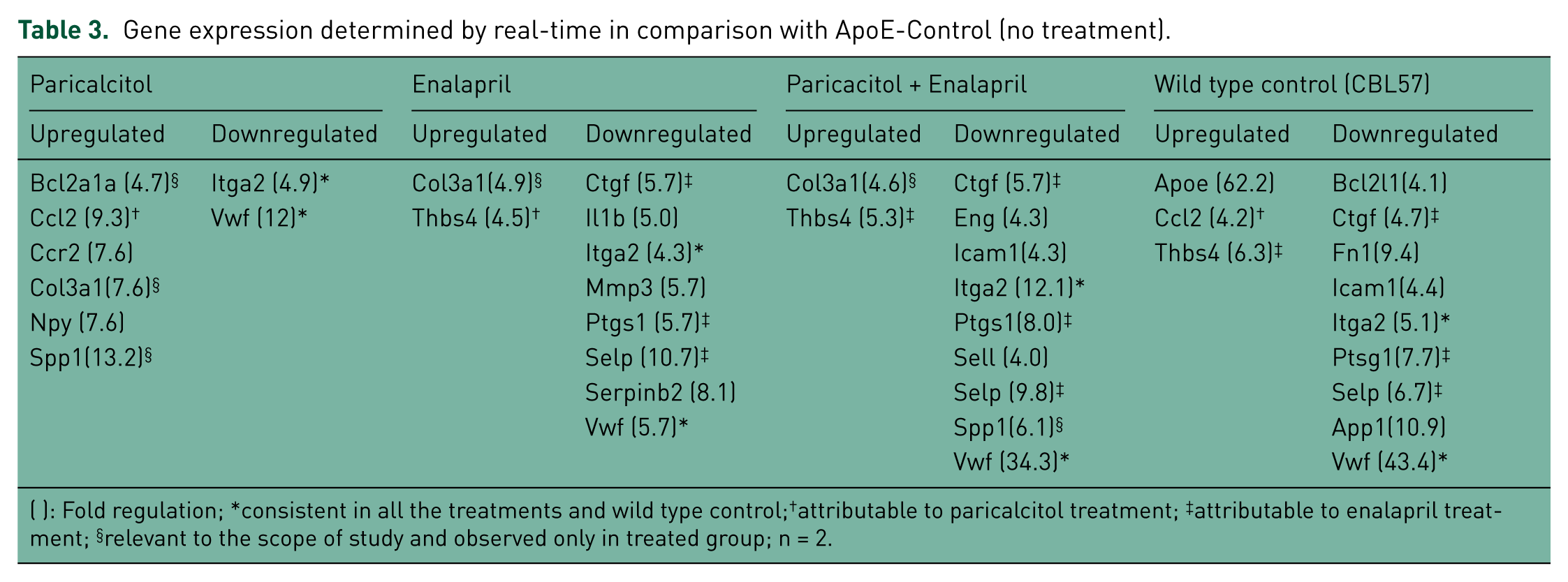
( ): Fold regulation; *consistent in all the treatments and wild type control;†attributable to paricalcitol treatment; ‡attributable to enalapril treatment; §relevant to the scope of study and observed only in treated group; n = 2.
Discussion
Vitamin D receptors are ubiquitous in nature and paricalcitol is a vitamin D receptor activator (VDRA) that has recently been recognized mainly for its cardiorenal protective effect in chronic kidney disease (CKD) [Finch et al. 2012; Nakamura et al. 2010; Husain et al. 2009]. Findings in CKD and the wider localization of VDRs in several tissues promoted our interest in studying the effects (unrelated to bone/calcium homeostasis) of vitamin D in organ systems. Similarly, enalapril is widely used to treat hypertension and has been well-recognized for its antioxidant/anti-inflammatory roles in several systems [Inserra et al. 2009; Finch et al. 2012; Husain et al. 2009]. Previously, our group reported that the combined therapy (paricalcitol and enalapril) prevented elevation of systolic blood pressure and affords greater protection against aortic inflammatory and oxidative injury in atherosclerosis than each monotherapy [Husain et al. 2009].
The resulted free glycerol, LDL, HDL, and total lipids levels in the ApoE groups, were consistent with the model phenotype [Plump et al. 1992; Zhang et al. 1992; Reddick et al.1994]. We also observed that groups that received paricalcitol treatment, either alone or combined with enalapril, showed higher circulating cholesterol levels. Hypercholesterolemia is a side effect of the chronic use and overdose of paricalcitol [National Kidney Foundation, 2003], which in this study consists of 200 ng, i.p., 100 µl injection of paricalcitol three times a week. However, aorta sections of these animals did not show signs of cholesterol accumulation or oxidation and, more importantly, there was a decrease of cholesterol levels at the cardiac tissue level. . All treatments were effective in reducing cholesterol levels in the heart by more than 50%, representing a protective factor for the development of cardiovascular complications.
The adiponectin levels in plasma and cardiac tissue increased significantly in all treated groups. This change in adiponectin levels was previously proposed in patients under enalapril treatment but was not supported, probably due to the limited sample size and study design, which included 70 participants with cardiac artery disease (CAD) and 25 controls (non-CAD) [Satoh et al. 2009]. Interestingly, the values obtained in circulating plasma and in the cardiac tissue, presented the same ratio of percentages: 34% in the group treated by paricalcitol, 73% in the group treated with enalapril, and 81% in the group treated with the combination of both drugs. This result suggests that circulating levels provide a good estimate about what the relative values are, at least at the cardiac site, when comparing therapeutic treatments. The combined treatment was more effective in increasing both levels: tissue and circulating. This finding is very important for supporting our hypothesis that paricalcitol and enalapril increase the expression of adiponectin endogenously and, consequently, the attributed downstream cardioprotective effects.
The building up of free radicals associated with the initiation and development of atherosclerosis and other CHDs is greatly influenced by the activation of the renin–angiotensin system (RAS) [Brewster et al. 2003; Weiss et al. 2001]. The ACEIs such as enalapril have previously been shown to inhibit the RAS and decrease the levels of toxic/reactive molecules at the renal and cardiac levels [Tardif et al. 2009; da Cunha et al. 2005]. The reduction of reactive species by antioxidant molecules is a crucial step in avoiding and decreasing the accumulation of oxidized products, which products promote atherosclerotic plaque formation and the chronic inflammatory response. The antioxidant capacity data obtained, provided an estimate of overall (cumulative effect) antioxidant capacity rather than measuring individual antioxidant components. The resulting antioxidant capacity in the cardiac tissue, increased significantly in all treated groups (paricalcitol 3×, enalapril 5.5×, and paricalcitol + enalapril 7.5×). The effect of enalapril on individual antioxidant components has being documented previously [Husain et al. 2010] and is consistent with our results. The combined therapy resulted in the most significant increase, providing evidence for yet another mechanism by which these medications confer cardioprotection.
The pattern observed (elevated iNOS and low eNOS) in the ApoE-controls is representative of oxidative and inflammatory processes. Under inflammatory conditions, iNOS produces NO [Michel and Feron, 1997]. Treatment with paricalcitol and enalapril alone or in combination ameliorated the alterations in both NO-generating enzymes in this model by switching the pattern to the other direction and, as a result, decreasing both the inflammation and the production of reactive oxygen species (ROS).
Superoxide dismutase is considered the first line of defense against the deleterious effects of ROS in the cells, and it scavenges superoxides. In terms of cytosolic CuZn-SOD, the low levels at the ApoE-control may be the result of inactivation and degradation for the excess of ROS [Salo et al. 1990]. The treated groups showed augmented expression of this antioxidant enzyme, but enalapril increased its levels even higher than the wild-type control. Hence, either treatment would be effective in the amelioration of inflammation and injury related to ROS, as previously reported [Wing et al. 2009].
TNF-α and MCP-1 are pro-inflammatory cytokines; COX-2 is a pro-inflammatory enzyme that has been shown to be elevated in patients with cardiovascular diseases [Brasier et al. 2010]. It is likely that these inflammatory mediators enhanced ROS production, causing oxidative injury to the cardiac tissue and leading to impaired function. In our study, MCP-1 expression was not detectably affected by paricalcitol treatment. In contrast, the enalapril and the combined treatments evidenced a decrease of this inflammatory marker. The TNF-α and Cox 2 protein levels showed marked changes in the group under the combined therapy. These results support previous findings from our group [Husain et al. 2010], indicating that the combination of paricalcitol with enalapril decreases inflammatory and cellular oxidative harm by affecting the expression of these molecules simultaneously. The obtained profile is congruent with the proposed cardioprotective effect of enalapril and paricalcitol as well as that of the combined therapy.
AMPK is a serine/threonine protein kinase that is involved in metabolism and plays a key role in monitoring and regulating energy and redox status [Zou et al. 2008]. Scoring from cardiac tissue for the presence of AMPK showed that in comparison with the ApoE- control there was a higher scoring for all treated groups and the wild type. This scoring was more evident in the group treated with the combination of paricalcitol and the enalapril. These results are consistent with the increase reported with the use of other therapeutic agents (statins, metformin, and rosiglitazone) [Wang et al. 2011]. The increase in AMPK is in accordance with the effect of adiponectin in this enzyme and may also explain part of the pathway responsible for the observed cardioprotective effect in the treated groups [Awazawa et al. 2011]. AMPK has been proposed as a candidate target for therapeutic intervention in the treatment of type 2 diabetes and metabolic syndrome [Zou et al. 2008] and, most recently, in the prevention of the progression of cardiac injury to cardiac failure [Beauloye et al. 2011]. The fact that paricalcitol, enalapril, and the combination of the two caused increased AMPK at the cardiac level provides new venues for studying this effect more deeply at the human population level.
The AR1 sections in all of the treated groups showed increases in the relative scoring for this nuclear protein. Although there exists some evidence suggesting the that enalapril affects the circulating levels of adiponectin [Santos et al. 2009], the direct evaluation of AR1 in the ApoE- model and in combination with paricalcitol at the cardiac level has not been stated previous to this work. The adiponectin levels in the cardiac tissue were increased in all treated groups and the analyses are informative in terms of the assessment of local effects and the attributed downstream cardioprotective effect, as we previously discussed in the sections above.
There were 15 genes that differed in expression, which expressions include the upregulation of Bcl2a1a, Col3a1, and Spp1 and the downregulation of Itga2 and Vwf. Bcl2a1a is an endogenous and cytokine-stimulated mediator of cytoprotection in a hyperoxic environment [He et al. 2005] and it has been shown to prevent cardiac hypertrophy and heart failure [Gupta et al. 2008]. Col 3 Type III collagen is crucial for collagen I fibrillogenesis and for normal cardiovascular development [Liu et al. 1997; Carter et al. 2009]. Spp1 increases the type III to type I collagen ratio seen in scarless fetal wounds and regulates collagen synthesis and accumulation after myocardial infarction [Cuttle et al. 2005]. Itga2 may affect ischemic stroke through the regulation of plasma lipid and lipoprotein levels [Wei et al. 2009; Matarin et al. 2008], and the Vwf product is a marker for endothelial cell damage [Horvath et al. 2004]. The expression of these genes is related to cardioprotective and antithrombotic activity.
Conclusions
We found significant changes in adiponectin (circulating and cardiac) and cholesterol levels in the heart, AMPK, inflammatory and oxidative stress markers, and genetic expression involved in cardiac protection after treating ApoE- mice with paricalcitol, enalapril, or the combination of the two. In the studied model, the observed cardioprotective effects are suggestive to be mediated by the modification of inflammation mediators, oxidative stress processes, lipids metabolism, and cardiac remodeling. Together, our data present a novel role for VDRA and ACEI, which role involves reducing factors associated with CHD that may lead to the discovery of new therapeutic venues. It provides new insights that open the field to future efforts for developing therapeutic agents with the capacity for inducing endogenous production of adiponectin and the expected downstream cardioprotective outcomes in humans.
Footnotes
Acknowledgements
We are grateful to J. Albelo, and S. Almodovar, PhD and the RCMI Publications Office (G12RR003050) for manuscript revision and to N. Rivera, G. Serrano, C. Irizarry, L. Colon, and F. Vazquez for technical support.
Funding
This research project was supported by funds from Abbott Pharmaceuticals, USA (grant number A07012).
Conflict of interest statement
The authors declare no conflict of interest in preparing this article.
