Abstract
Objective
To analyze the effect of 3% diquafosol sodium ophthalmic solution on tear film proinflammatory cytokine levels, ocular symptoms, and tear film stability in patients with dry eye disease.
Methods
This was an open-label, noncomparative, single-cohort, interventional, proof-of-concept study among patients with dry eye disease who were administered diquafosol eye drops six times a day for 4 weeks. Pretreatment and posttreatment tear samples were collected, and ocular surface disease index scores and fluorescein tear break-up time were measured. Interferon-gamma, interleukin-1 beta, interleukin-6, and tumor necrosis factor-alpha levels were measured in the tear samples using a cytokine magnetic bead panel. Independent associations of cytokine responses with sex, age, tear break-up time, and ocular surface disease index were determined.
Results
Thirty-one eyes (n = 31) in 31 patients with dry eye disease were included. At baseline, interleukin-6, tumor necrosis factor-alpha, and interferon-gamma were detected in all samples, whereas interleukin-1 beta was detected in 3% (1/31) of the samples. Baseline concentrations of interleukin-6, tumor necrosis factor-alpha, and interferon-gamma decreased by ≥25% in 58%, 51.6%, and 45.1% of the eyes, respectively, reaching significance for tumor necrosis factor-alpha (W = 101, z = 2.694, p = 0.006, two-tailed) and interleukin-6 (W = 134, z = 2.224, p = 0.026, two-tailed). The tear break-up time (from 3.95 to 4.65 s; p = 0.013) and ocular surface disease index scores (from 20.57 to 13.85; p = 0.005) significantly improved following treatment. Individual responses were found to be independent of age, sex, eye laterality, pretreatment tear break-up time, and ocular surface disease index scores based on the multivariate logistic regression.
Conclusion
Diquafosol reduced the levels of proinflammatory cytokines in the tears of patients with dry eye disease after 4 weeks of treatment, suggesting their possible role in modulating ocular surface inflammation. Larger prospective studies are recommended to validate these results.
Introduction
Dry eye disease (DED) is a common ocular condition, with population-based studies from Asia and Europe reporting prevalence rates ranging from 5% to 50%. 1 In an urban community in the Philippines, the prevalence was found to be 23%. 2 In 2017, the Asia Dry Eye Society consensus statement defined dry eye as a multifactorial disease characterized by tear film instability, resulting in various symptoms and/or visual impairment, potentially accompanied with ocular surface damage. 3 Inflammation plays a major role as an important risk factor and a consequence of DED. Understanding the relationship between inflammation and DED has led to the development of novel treatment options. 4
Tear film instability and hyperosmolarity initiate an inflammatory cascade that activates stress signaling pathways in the ocular surface epithelium and resident immune cells, causing de novo formation of innate inflammatory molecules and aggravation of dry eye symptoms.5,6 Hyperosmolar stress activates mitogen-activated protein kinases, resulting in downstream production of proinflammatory cytokines, chemokines, and matrix metalloproteinases as well as induction of apoptosis. The interplay of these mediators is complex and is known to further amplify the inflammatory cascade. 7 In a meta-analysis, patients with DED exhibited higher levels of interleukin (IL)-1β, IL-6, IL-8, IL-10, tumor necrosis factor-alpha (TNF-α), and interferon-gamma (IFN-γ) than controls. 8
Measuring cytokines in tears offers a valuable approach for quantifying inflammation in DED, with certain cytokines shown to correlate with DED severity. 6 Measurements of tear film cytokines before and 3 months after treatment with cyclosporine alone versus cyclosporine and methylprednisolone revealed decreased levels of IL-6 and IL-8 compared with those at baseline, with significantly greater reduction following the combination treatment group. 5 A comparison of three tear supplements on inflammatory biomarkers revealed that carboxymethylcellulose–glycerin–castor oil reduced cytokine levels more effectively than carboxymethylocellulose and hydroxypropyl guar. 9
Diquafosol, a P2Y2 receptor agonist, promotes tear fluid secretion from epithelial cells in the conjunctiva and mucin secretion from goblet cells.10,11 Evaluation of diquafosol on corneal epithelium in a rat model of dry eye demonstrated that after 28 days of treatment, the levels of intracellular reactive oxygen species, apoptosis, and inflammatory markers were reduced compared with those in untreated rats. 12 A significant reduction in proinflammatory cytokines (IL-1β and TNF-α) suggested that diquafosol inhibited the nuclear factor kappa-light-chain-enhancer of B cells (NF-κB) pathway and reduced the proinflammatory cytokine expression. In another DED mouse model, administration of diquafosol in the eyes for 2 weeks demonstrated improved fluorescein staining scores, mean tear break-up time (TBUT), rose Bengal staining, MUC5 mRNA expression, and goblet cell number after treatment. 13 Another study investigating the efficacy of diquafosol and cyclosporine A in patients with dry eye revealed significant reduction in pro-matrix metalloproteinase-9 (MMP-9) levels after 3 months of treatment; 14 however, these findings were inconsistent with those of a 2024 study, which reported no significant difference in MMP-9 levels following treatment with either diquafosol or rebamipide. 15 Real-world studies with up to 12 months of continuous treatment demonstrated that diquafosol is effective and well-tolerated. 16 In a recent 10-year follow-up report of patients compliant to the medication, sustained improvements were observed in total corneal staining, conjunctival staining (CS), and symptom scores, although no remarkable changes were noted in the TBUT, Schirmer scores, and central corneal staining scores. 17
To the best of our knowledge, only one other human study has investigated the effect of topical diquafosol on tear film cytokine levels, particularly IL-6, TNF-α, IL-1β, and IFN-γ, in DED. Quantifying its impact on inflammation in DED may help support hypotheses highlighting its immunomodulatory role in DED etiology. Therefore, we conducted a proof-of-concept study to determine the efficacy of 3% diquafosol sodium eye drops on the levels of tear film inflammatory markers, ocular surface disease index (OSDI) scores, and TBUT in patients with DED as well as identify independent variables associated with treatment response.
Methods
Study design and setting
This open-label, noncomparative, pretreatment, posttreatment proof-of-concept study was conducted from June 2021 to May 2022. Consecutive patients with DED were enrolled from the Ophthalmology Outpatient Department at St. Luke’s Medical Center-Quezon City.
The study adhered to the principles of the Declaration of Helsinki of 1975, as revised in 2024, as well as the Guidelines of the International Conference on Harmonization-Good Clinical Practice (ICH-GCP). Clinical protocols and documents were reviewed and approved by St. Luke’s Medical Center – Quezon City Institutional Ethics and Review Committee (SL-19208; April 21, 2021). Written informed consent was obtained from all enrolled patients.
Study population
Patients aged ≥18 years who were diagnosed with definite DED based on the Asia dry eye diagnostic criteria, including TBUT < 5 s and dry eye symptoms such as eye discomfort and visual disturbance, were eligible for this study. 3 Enrolled patients were de-identified and assigned codes. Subjective symptoms were assessed using the OSDI, with a cutoff value of >13.
The OSDI is proprietary, but now publicly available for clinical and research use (https://www.abbvieeyecare.com/content/dam/abbvieeyecare/pdf/office-resources/dry2.pdf). 1
Vital staining of the cornea and conjunctiva, hyperosmolarity test, and Schirmer test were not performed, as they were not considered essential for confirming a diagnosis of DED. Patients were excluded if they had other ocular conditions with symptoms similar to DED, wore contact lenses, used any eye drops, took systemic medications affecting tear production, had undergone ocular surgery within the past 12 months, had known hypersensitivity to diquafosol, or were unable to comply with the treatment regimen and follow-up. Only one eye of each patient was included in the study. The eye with the shorter TBUT was selected as the study eye; however, if both eyes had the same TBUT, the right eye was selected. The study’s reporting followed the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines, 18 as shown in the flowchart in Figure 1.

Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement 18 flow chart.
Study protocol
Demographic data and clinical symptoms were recorded, and participants completed the validated Filipino translation of the OSDI questionnaire. 19 All participants underwent basic ophthalmologic examinations, followed by fluorescein TBUT measurements using standard techniques. 20 All clinical examinations were performed by a single trained ophthalmologist.
Before initiating treatment, 1 µL of unstimulated tear fluid was collected by a single investigator from the study eye using a glass microcapillary tube. The standard collection site was the tear prism at the outer canthus of the lower lid of the study eye, with care taken to prevent additional tear reflex by avoiding contact with the conjunctiva as much as possible. Tubes with tear samples were de-identified and labeled with an assigned code, stored on ice in an insulated cooler, and immediately transported to a laboratory. Assessor masking was performed. A single research scientist processed all the samples, which were diluted (1:50), centrifuged, and stored at −80°C until analysis.
Patients were given a preweighed bottle of 3% diquafosol sodium (Diquas, Santen Pharmaceutical Co., Ltd., Japan) ophthalmic solution and were instructed to administer one drop six times daily in both eyes for 4 weeks. Patients were given verbal and written instructions regarding medication dosing schedules and enrolled in a daily short message service (SMS) reminder chain that alerted them 5 min before each scheduled dose. They were followed up weekly for adverse events, and diquafosol bottles were monitored for compliance.
OSDI surveys, TBUT measurements, and tear fluid sample collections from the same study eye were repeated after 4 weeks of treatment.
To reduce potential bias, the study implemented a standardized protocol for all the procedures, employed objective and quantitative outcome measures, ensured that the assistant analyzing the samples was masked to allocation, and conducted assessments at fixed follow-up intervals of 30 days.
Tear film cytokine analysis
IFN-γ, IL-1β, IL-6, and TNF-α levels were measured simultaneously in tear film samples using the MILLIPLEX® MAP human cytokine/chemokine magnetic bead panel (HCYTOMAG-60K; Merck Millipore, Germany). Samples (1 µL) were incubated at 4°C with antibody-coated capture beads for 18 h. Subsequently, the beads were washed, incubated with the detection antibody (biotin-labeled anti-human cytokine antibodies), and further incubated with streptavidin–phycoerythrin. The emitted mean fluorescence units were compared with the standard curves of human cytokines to determine cytokine concentrations. The minimum detectable concentrations (pg/mL) were as follows: IFN-γ, 0.8; IL-1β, 0.8; IL-6, 0.9; and TNF-α, 0.7. Samples with concentrations below the detection limits for each cytokine were excluded from the analysis. The median fluorescence intensity was analyzed via GraphPad Prism (9.5.0) using a 4-parameter, logistic curve-fitting method to calculate cytokine concentration. To obtain the final concentration, the calculated concentration was multiplied by the dilution factor.
Sample size estimation
Sample size was calculated a priori based on the proportion of responders to diquafosol eye drops, as determined by the tear film cytokine concentration. IL-1β, IL-2, IL-8, IL-17, IFN-γ, and TNF-α were used in the sample size calculations because they yielded the highest sample size. Assuming that 40% of patients responded to diquafosol eye drops for these cytokines, with a maximum allowable error of 10% and a reliability of 80%, a sample size of 23 participants was required. 9
Statistical analyses
Descriptive statistical analyses were performed to obtain the mean and SD values. Non-normal distribution was determined using the Shapiro–Wilk and Kolmogorov–Smirnov tests. The Wilcoxon signed-rank test was used for paired comparisons. Test statistics were approximated with continuity correction, and two-tailed p-values of <0.050 were considered statistically significant. After controlling for potential confounders, the presence of independent associations between individual cytokine responses and clinical parameters, including eye laterality, age, sex, pretreatment TBUT, and pretreatment OSDI score, was determined using multiple logistic regression analysis. A p-value of <0.050 was considered statistically significant.
Results
Thirty-one study eyes of 31 patients with DED were included, with a mean age of 48.2 years, comprising 9 (29%) males and 22 (71%) females. The mean baseline values were 3.95 s for TBUT and 20.6 for OSDI.
At baseline, IL-6, TNF-α, and IFN-γ were detected in all samples. IL-1β was detected in only 1 (3%) of the 31 samples and was therefore excluded from further cytokine analysis. A cytokine response, defined as a ≥25% reduction in pretreatment cytokine levels following diquafosol treatment, was observed for IFN-γ in 14 (45.1%) samples, IL-6 in 18 (58%) samples, and TNF-α in 16 (51.6%) samples. IFN-γ, IL-6, and TNF-α simultaneously decreased by ≥25% in 13 (42%) samples. The Wilcoxon signed-rank test demonstrated a significant difference between pre and posttreatment values for IL-6 (W = 134, z = 2.224, p = 0.026, two-tailed) and TNF-α (W = 101, z = 2.694, p = 0.006, two-tailed). (Figure 2 and Table 1).

Median cytokine concentration (pg/mL) for each cytokine before and after diquafosol treatment (n = 31 study eyes). Dots outside the interquartile range box indicate outliers.
Paired differences between concentrations of proinflammatory cytokines before and after treatment with diquafosol ophthalmic solution.

Wilcoxon signed-rank test
Results expressed as median and IQR
With continuity correction
Bold values indicate statistically significant results.
IQR: interquartile range; TNF-α: tumor necrosis factor-alpha; IL-6: interleukin-6; IFN-γ: interferon-gamma.
The TBUT and OSDI scores of all patients showed statistically significant changes after 1 month of diquafosol treatment. The TBUT improved from 3.95 to 4.65 s after treatment (p = 0.013), and the OSDI score improved from 20.57 to 13.85 (p = 0.005) (Figure 3).
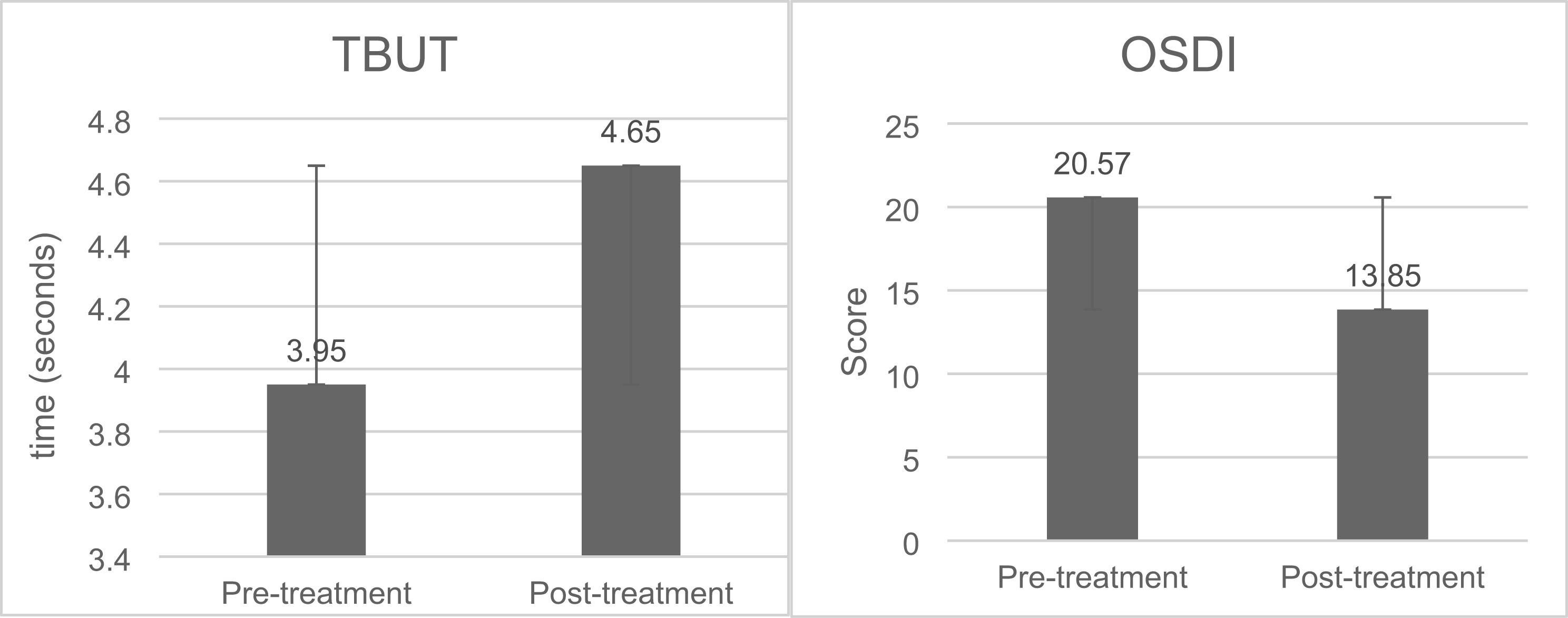
TBUT (left) and OSDI scores (right) before and after treatment. TBUT: tear break-up time; OSDI: ocular surface disease index.
Independent associations between cytokine responses, patient characteristics, and dry eye parameters were assessed after controlling for confounders. Multivariate logistic regression showed that individual responses to IFN-γ, IL-6, and TNF-α were independent of eye laterality, age, sex, TBUT, and OSDI scores (Tables 2 to 4).
Independent associations between clinical parameters and IFN-γ levels after treatment with diquafosol ophthalmic solution.

B: regression coefficient.
TBUT: tear break-up time; OSDI: ocular surface disease index; OR: odds ratio; CI: confidence interval; Sig: significance level; IFN-γ: interferon-gamma.
Independent associations between clinical parameters and TNF-α levels after treatment with diquafosol ophthalmic solution.
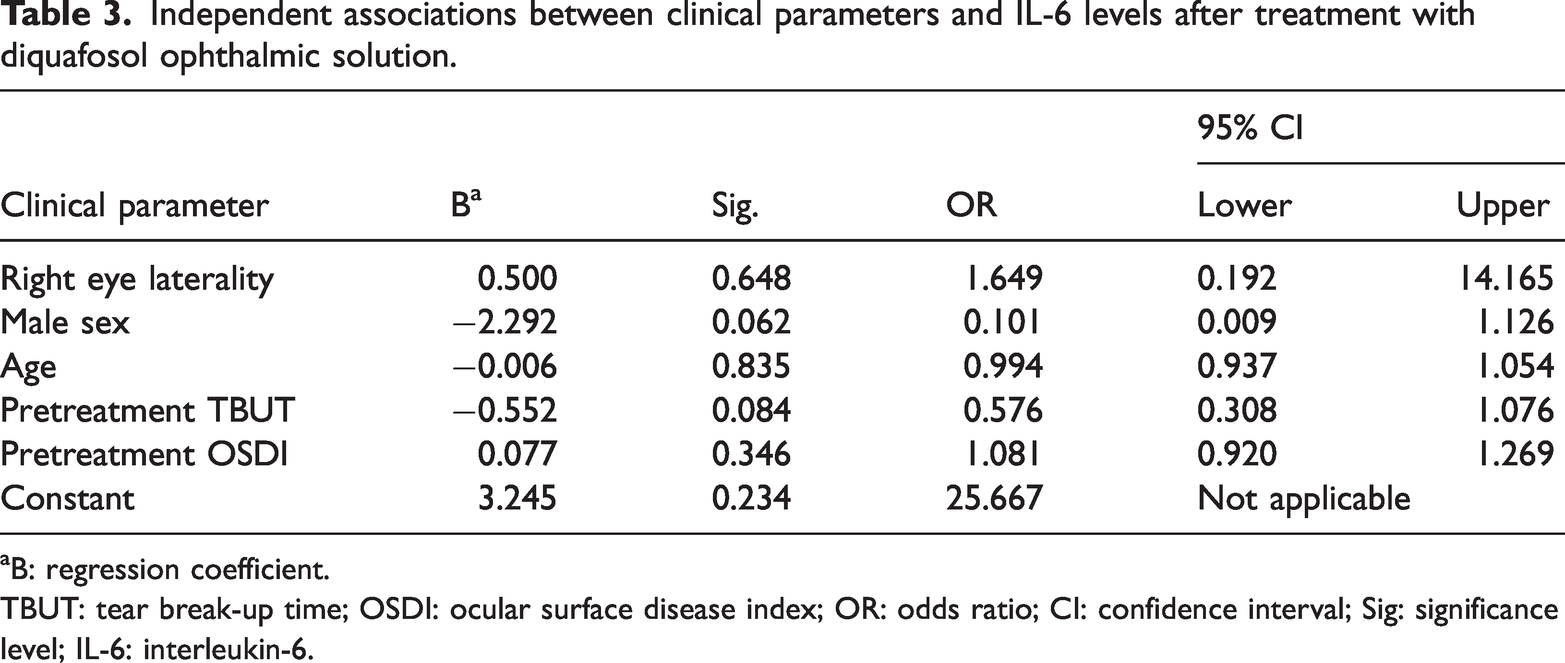
B: regression coefficient.
TBUT: tear break-up time; OSDI: ocular surface disease index; OR: odds ratio; CI: confidence interval; Sig: significance level; TNF- α: tumor necrosis factor-alpha.
Independent associations between clinical parameters and IL-6 levels after treatment with diquafosol ophthalmic solution.

B: regression coefficient.
TBUT: tear break-up time; OSDI: ocular surface disease index; OR: odds ratio; CI: confidence interval; Sig: significance level; IL-6: interleukin-6.
Discussion
This study investigated the effect of 3% diquafosol sodium ophthalmic solution on proinflammatory cytokine concentrations in patients with DED, particularly in those whose tear samples had the highest tear film concentrations of IFN-γ, IL-6, and TNF-α. This finding is consistent with those of previous studies, which reported a predominant presence of these three cytokines, in addition to IL-1β and IL-8, in patients with DED.21,22 In two other similar studies, apart from IL-1β, IL-6, and TNF-α, transforming growth factor-beta (TGF-β) was a consistently observed inflammatory marker.23,24 However, in the present study, IL-1β was the least detected (3%), precluding the assessment and interpretation of IL-1β concentrations for any treatment-induced changes.
We observed a statistically significant decrease in TNF-α levels after 1 month of treatment with diquafosol eye drops. A similar decrease in TNF-α and IL-6 levels following diquafosol treatment has been reported in patients after cataract surgery, compared with those in controls. 25 TNF-α is produced by stressed epithelial cells and may serve as an indicator of ocular surface stress. 22 Our data indicate a possible effect of diquafosol in reducing TNF-α levels; however, elucidating the associated mechanism is beyond the scope of this study. Based on prior in vitro findings, we speculate that inhibition of the NF-κB pathway, leading to decreased expression levels of TNF-α and IL-6 in human corneal epithelial cells, may underlie this effect. 26
In our study, a statistically insignificant proportion of patients demonstrated decreased IFN-γ levels following diquafosol treatment. Of interest is the putative effect of IFN-γ on goblet cells, wherein diquafosol primarily acts to increase mucin secretion. Elevated IFN-γ in tears reduced goblet cell density, increased epithelial apoptosis, and squamous metaplasia in one study. 27 Contrarily, another study reported an increase in goblet cell density, assessed using impression cytology, in a cohort of post-cataract surgery patients who received diquafosol sodium for 12 weeks. 28 Our results may reflect a positive effect of diquafosol on goblet cells; however, mechanistic studies are required to support this association.
IL-6 has been identified as a key marker in DED, as its levels are elevated in the tears and conjunctiva of affected patients. In a study examining the association between clinical parameters and proinflammatory cytokine concentrations in patients with evaporative-type DED, IL-6 levels were found to be positively associated with older age (66 ± 6 years in IL-6-positive patients vs. 53 ± 3 years in IL-6-negative patients). 24 Our DED cohort predominantly comprised younger patients (mean age: 48.2 years).
Our study eyes exhibited low IL-1β detection rates, which may be attributed to our cohort that primarily included patients with mild-to-moderate DED (OSDI scores, 13–32). A study evaluating conjunctival cytokine expression in tear fluid of patients with moderate dry eye reported the absence of IL-1β, suggesting that the cytokine does not play a modulatory role in moderate dry eye. 29 This observation is supported by another study assessing IL-1β, IL-6, and MMP-9 levels in the tears of patients with moderate DED, which reported a substantial increase only in pro-MMP-9 levels. 30 Recently, the Dry Eye Assessment and Management study also revealed that the severity of DED influences the profiles of tear cytokines. 31
Due to the paucity of data from human studies, we referred to studies on rat DED models and in vitro samples of human corneal endothelial cells (hCECs). An investigation of the anti-inflammatory effects of diquafosol on hCECs revealed a significant reduction in the IL-6 and TNF-α levels, 32 consistent with the findings of our study. A recent study investigating improvements in tear film parameters among post-cataract surgery patients randomized to either 3% diquafosol, cyclosporine A, or control similarly reported a significant decrease in IL-6 and TNF-α levels in the diquafosol and cyclosporine A groups, with no change observed in the control group. No differences were noted in epidermal growth hormone and TGF-β levels, the two other cytokines assessed. 25
We also analyzed potential associations between tear fluid proinflammatory cytokine levels and clinical parameters; however, we did not observe any association between the likelihood of a cytokine-reduction response and age, sex, TBUT, or OSDI scores for all detectable cytokines investigated. Similarly, another study reported no association between clinical variables and the levels of 15 cytokines and chemokines, including IL-6, IFN-γ, and TNF-α, with the exception of IL-8 and CXC motif chemokine ligand 8, in the tear samples of patients with DED. 24
We observed significant improvements in TBUT and OSDI scores following diquafosol treatment, consistent with multiple studies including systematic reviews reporting significant improvements in TBUT following diquafosol treatment for DED.33,34 Upon comparing the efficacy of diquafosol with sodium hyaluronate in patients with DED in China and Singapore, both groups demonstrated similar improvement in TBUT. 35 Comparable improvements in TBUT, OSDI, and corneal CS were observed in a randomized trial comparing a 4-week course of diquafosol versus rebamipide. 15
The efficacy of diquafosol has also been studied in specific patient groups, including postoperative patients, those with diabetic dry eyes, children with DED wearing orthokeratology lenses, 36 and patients with evaporative-type dry eyes. The TBUT and OSDI scores improved in patients who received diquafosol after cataract,37,38 vitrectomy surgery, 39 and small incision lenticule extraction 40 compared with those in controls. In a study comparing the effect of diquafosol with that of sodium hyaluronate in patients with diabetic dry eyes, noninvasive TBUT (NITBUT), CS, meibomian gland quality (MGQ), meibomian gland expressibility (MGE), and OSDI scores were found to be significantly improved in both groups (p < 0.050); however, an improvement in the tear film lipid layer (TFLL) was only observed in the diquafosol group. 41 Another study reported consistent findings in patients with diabetes and DED, showing additional improvements in the MMP-9 levels, corneal nerve fiber density, and conjunctival hyperemia redness scores after 8 weeks of treatment. 42 In patients with evaporative DED patients, a combination of diquafosol and intense pulsed light (IPL) therapy was more effective than IPL monotherapy and sham treatment in improving the NITBUT, TFLL, CS, MGQ, MGE, and OSDI score. 43
Although our study was significantly limited by its design because it was a nonrandomized, double-masked, vehicle-controlled, comparative clinical trial, the findings of our proof-of-concept study support the need for future randomized controlled trials, large prospective trial investigations, with longer treatment periods. We observed decreased levels of inflammatory biomarkers in response to diquafosol treatment, suggesting that longer treatment durations may be required to achieve more robust changes in cytokine levels. The low proportion of patients with DED with IL-1β in our study precluded the assessment of any diquafosol-induced changes in this cytokine. Further characterization of dry eye based on CS, hyperosmolarity, and Schirmer test results should be performed. Additionally, we recommend the evaluation of preservative-free diquafosol formulations. Although cytokine levels were measured objectively, confounding effects may have been introduced by the vehicle and/or preservatives in diquafosol eye drop formulations. Follow-up studies with larger cohorts may include subgroup analyses to classify patients according to DED type and severity. Finally, a wider array of cytokines associated with DED should be measured.
Conclusion
Administration of 3% topical diquafosol sodium reduced the IL-6 and TNF-α levels in the tears of patients with DED following 4 weeks of treatment, regardless of eye laterality, sex, age, or pretreatment TBUT and OSDI score. Therefore, 3% diquafosol sodium may effectively modulate ocular surface inflammation in dry eye and can be considered for inclusion in the treatment regimen for DED.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605251387924 - Supplemental material for The effect of diquafosol on tear film inflammatory markers in patients with dry eye: A noncomparative, open-label, proof-of-concept study
Supplemental material, sj-pdf-1-imr-10.1177_03000605251387924 for The effect of diquafosol on tear film inflammatory markers in patients with dry eye: A noncomparative, open-label, proof-of-concept study by Raymund V Tanchuling, Paula Vivian C Tan, Raymond Nelson C Regalado and Ruben Lim Bon Siong in Journal of International Medical Research
Supplemental Material
sj-pdf-2-imr-10.1177_03000605251387924 - Supplemental material for The effect of diquafosol on tear film inflammatory markers in patients with dry eye: A noncomparative, open-label, proof-of-concept study
Supplemental material, sj-pdf-2-imr-10.1177_03000605251387924 for The effect of diquafosol on tear film inflammatory markers in patients with dry eye: A noncomparative, open-label, proof-of-concept study by Raymund V Tanchuling, Paula Vivian C Tan, Raymond Nelson C Regalado and Ruben Lim Bon Siong in Journal of International Medical Research
Footnotes
Acknowledgments
The authors are grateful to the assistance provided by K-Anne C Apostol. and Aleczandria Esguerra, who served as research assistants, as well as Dr Macario Reandelar, who served as the statistician for this study.
Authors contributions
All authors contributed to the study design. R.T: conducted the study; R.T, P.T, and R. L. B. S: interpreted the data and wrote the manuscript; and R.T, R.R, and R. L. B. S: reviewed the manuscript.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Statement on AI use
The authors declare that artificial intelligence (AI) tools were not used for study conception, design, data collection, or writing of this manuscript.
Conflicts of interest
Ruben Lim Bon Siong is an advisory board member and speaker for Santen Pharmaceutical Co., Ltd. No other authors has relevant financial and non-financial competing interests.
Funding
This work was supported by the Research and Biotechnology Division, St. Luke’s Medical Center-Quezon City (grant number SL-19208). Santen Pharmaceutical Co., Ltd. funded the medical writing/editing services, which were provided by Dr Shawna Tan of Medical Writers Asia.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
