Abstract
Endothelin 1 (ET-1), a potent vasoconstrictive substance, was discovered in 1988 by Yanagisawa and colleagues, and since then, a quarter of a century has passed. Understanding the biology of ET-1 has rapidly developed by characterizing the components of its receptors and processing enzymes. Numerous studies have revealed not only physiological but also various pathophysiological roles of the ET system. At first, ET-1 was the attractive and promising target for the treatment of hypertension owing to its potent vasoconstrictive nature and a variety of ET receptor antagonists (ERAs) were studied. However, the clinical application to treat hypertension was disappointing because of the side effects, including liver toxicity and fluid retention. On the other hand, ERAs have been established as orphan drugs for the treatment of pulmonary arterial hypertension and improved the prognosis of patients. Furthermore, multipotency of the ET system in the pathogenesis of multiple diseases has led to the development of translational research not only in the field of hypertension but in a variety of fields. Furthermore, a range of studies are ongoing to apply ERAs to clinical situations. In this article, we review the pathophysiological roles of the ET system in hypertension and pulmonary hypertension and the potential of ET receptor antagonism for the treatment of these diseases.
Keywords
Introduction
A quarter of a century has passed since endothelin 1 (ET-1) was discovered in 1988, as a strong vasoconstrictive substance secreted from porcine aortic vascular endothelial cells [Yanagisawa et al. 1988]. Rapid developments in understanding of the ET system have been achieved by uncovering the components of the ET system, including related peptides [endothelin 2 (ET-2) and endothelin 3 (ET-3)] [Inoue et al. 1989], processing enzymes [endothelin converting enzyme 1 (ECE-1) and endothelin converting enzyme 2 (ECE-2)] [Xu et al. 1994; Emoto et al. 1995], and endothelin receptors [endothelin receptor A (ETA) and endothelin receptor B (ETB)] [Arai et al. 1990; Sakurai et al. 1990]. Numerous studies have elucidated not only the physiological roles of the ET system but also the pathophysiological roles in various diseases. Although the potent vasoconstrictive effect of ET-1 was the attractive and promising target for the treatment of hypertension and a variety of endothelin receptor antagonists (ERAs) were synthesized and analyzed, clinical application of ERAs for the treatment of hypertension has not been established because of their various side effects, including liver toxicity and fluid retention. However, ERAs have been established as orphan drugs for pulmonary arterial hypertension (PAH) [Galie et al. 2013] and systemic sclerosis-related digital ulcer [Matucci-Cerinic et al. 2011]. Because of its ubiquitous and pluripotent feature, the ET system might be involved in the pathogenesis of multiple diseases related to various types of organs and cells. Therefore, a range of studies are currently investigating the potential of ERAs in various diseases. Despite disappointing results in the field of hypertension, a better knowledge of the ET system might soon translate into clinical applications.
In this review, a PubMed search was performed using ‘Endothelin’ as a keyword. With milestone articles among 26,964 articles published between 1988 and 2014, we review the physiological action of the ET system and its pathophysiological roles in hypertension and pulmonary hypertension (PH), and focus on the current state of endothelin receptor antagonism in the treatment of these diseases.
Endothelin system
Endothelins
ET-1 is a 21-amino-acid peptide with two disulfide linkages, which has been purified from culture supernatant of porcine aortic endothelial cells (ECs) as a novel strong vasoconstrictive peptide [Yanagisawa et al. 1988]. Shortly after the discovery of ET-1, two structurally related peptides were identified as ET-2 and ET-3 [Inoue et al. 1989], the structures of which differ from that of ET-1 by 2 and 6 amino acids respectively. ET-1 is the predominant isoform of endothelins that mainly affects the cardiovascular system and is generated by ECs and other various types of cells, including epithelial cells, macrophages, fibroblasts, cardiomyocytes, and neurons [Kedzierski and Yanagisawa, 2001]. ET-2 is expressed mainly in epithelial cells of the upper and lower gastrointestinal tracts [Chang et al. 2013], and ET-3 is generated by brain neurons and epithelial cells in lung, intestinal tract, and renal tubules [Matsumoto et al. 1989]. Endothelins are synthesized from inactive precursors termed preproendothelins which are 212-amino-acid gene products. These proteins are processed to 38-amino-acid inactive intermediates, termed big endothelins by furin-like endopeptidase. Finally, big endothelins are cleaved at Trp21-Val22 to biologically active 21-amino-acid peptides by ECEs and other enzymes [Opgenorth et al. 1992]. ECEs are zinc metalloproteases that belong to the neutral endopeptidase superfamily, and consist of two family members, ECE-1 and ECE-2. ECE-1 is a membrane-bound metalloprotease which is expressed in a variety of cells, mainly in ECs [Xu et al. 1994], while ECE-2 is an intracellular processing enzyme that acts in acidic conditions and is expressed mainly in neurons [Emoto and Yanagisawa, 1995]. ECE-1 is the major isoform and the key regulator of the cardiovascular ET system.
Endothelin receptors
Endothelins act through two types of receptors, ETA and ETB, which are G-protein-coupled 7-transmembrane domain receptors [Arai et al. 1990; Sakurai et al. 1990; Davenport, 2002]. Each endothelin receptor activates intracellular signaling pathways, including increasing calcium influx, activation of phospholipase Cβ, phospholipase D, protein kinase C, Rho/Rho kinase pathway, and mitogen-activated protein kinases [Sakurai et al. 1990; Kasuya et al. 1992; Pollock et al. 1995; Douglas and Ohlstein, 1997; Gohla et al. 2000; Ivey et al. 2008; De Mey et al. 2011]. The affinity of ET-1 to ETA is 10 times higher than that of ET-2, and the affinity of ET-3 is much less than ET-1 or ET-2 [Arai et al. 1990], while the affinities to ETB are not different among ET-1, ET-2, and ET-3 [Sakurai et al. 1990]. Endothelin receptors are expressed in a diversity of cells, including ECs, vessel and airway smooth muscle cells (SMCs), cardiomyocytes, fibroblasts, renal mesangial and collecting-duct cells, hepatocytes, neurons, and macrophages [Hori et al. 1992; Kohan et al. 1992; Katwa et al. 1993; Takeda et al. 1994; Goldie et al. 1995; Kowala et al. 1995; Kedzierski and Yanagisawa, 2001]. Most cells express both ETA and ETB, except for ECs that express only ETB, and the effects of endothelins in each cell depend on receptor-coupled G proteins and their effectors. The actions of endothelins via endothelin receptors are mediated mainly at a autocrine/paracrine level, which is supported by the fact that ET-1 is secreted mainly abluminary in the vessel wall, and the plasma concentration of ET-1 is lower than the threshold level to activate endothelin receptors [Shichiri et al. 1991; Wagner et al. 1992; Battistini et al. 1993]. ETB mediates the clearance of endothelins by internalizing and degrading with them intracellularly [Dupuis et al. 1996]. The clearance of ET-1 is mainly performed by the pulmonary circulation and is rapid (the plasma half life of ET-1 is less than a minute) [Fukuroda et al. 1994; Dupuis et al. 1996]. It is assumed that a crosstalk exists between ETA and ETB and they might form functional heterodimers in some situations [Harada et al. 2002; Gregan et al. 2004]. These unique characteristics of endothelin receptors cause arguments on how to block the ET system with ERAs the most effectively in the treatment of pathological conditions.
Physiological roles of endothelins in cardiovascular system
Development
The ET system is closely related to the development of the cardiovascular system. The ET-1/ECE-1/ETA and ET-3/ECE-1/ETB pathways are involved in the development of neural crest-derived cells. In an animal model, genetic ablation of the ET-1/ECE-1/ETA pathway causes craniofacial and cardiovascular abnormality, including the absence or obstruction of large vessels, ventricular septal defect, and right-sided aortic arch, owing to impaired development of cephalic and cardiac neural crest cells [Kurihara et al. 1994; Clouthier et al. 1998; Yanagisawa et al. 1998]. In mice lacking both ECE-1 and ECE-2, severe cardiac abnormalities including atrioventricular valve and endocardial cushion malformation are also seen [Yanagisawa et al. 2000]. These animals are embryonic lethal or die shortly after birth. In contrast, the ET-3/ECE-1/ETB pathway is involved in the development of vagal and trunk neural crests. ET-3-, ECE-1-, and ETB-deficient animals suffer from aganglionic megacolon and coat color spotting and die within 1 month of birth [Baynash et al. 1994; Hosoda et al. 1994; Yanagisawa et al. 1998]. For these reasons, difficulties have arisen to reveal the physiological and pathophysiological roles of each component in mice, and therefore, studies with conditional gene-targeted mice have been undertaken.
Vascular endothelin system
Owing to the most potent vasoconstrictive capacity of ET-1, the ET system is recognized as one of the main modulators of vascular homeostasis. ET-1 is the predominant isoform of endothelins produced by the vessel wall, mainly by ECs. Up to 80% of ET-1 is secreted via the basolateral side of ECs [Wagner et al. 1992]; the local ET-1 concentration is thus 100 times higher than the plasma level, where it is rapidly cleared from the circulation [Howard et al. 1992; Battistini et al. 1993]. Shear stress on ECs is the essential stimulus to regulate ET-1 synthesis, which shows early upregulation followed by a sustained suppression [Malek et al. 1999]. The synthesis of ET-1 is also upregulated by norepinephrine, angiotensin II, insulin, tumor necrosis factor α, transforming growth factor β, and hypoxia [Yanagisawa et al. 1988; Bodi et al. 1995; Kedzierski and Yanagisawa, 2001], and downregulated by nitric oxide, atrial natriuretic peptide, and prostacyclin [Boulanger and Luscher, 1990; Saijonmaa et al. 1990; Prins et al. 1994].
In the vessel wall, ET-1 secretion is mediated by two secretory pathways: the constitutive pathway and the regulated secretory pathway [Ozaka et al. 1997; Russell and Davenport, 1999; Khimji and Rockey, 2010]. The constitutive pathway that is a common pathway to secreted proteins, proteoglycans, and lysosomal enzymes mediates continuous secretion of ET-1 through the endoplasmic reticulum–Golgi complex and secretory vesicles. ET-1 secretion by the constitutive pathway is suggested to be polarized to the basolateral membrane, and to be responsible for the maintenance of basal vascular tone through ETA on the surface of vascular SMCs (VSMCs), and ETB on the surface of ECs [Yoshimoto et al. 1991; Wagner et al. 1992; Russell and Davenport, 1999; Khimji and Rockey, 2010]. The regulated secretory pathway is mediated by Weilbel–Palade bodies which are cytoplasmic organelles of ECs, and store substances related to coagulation, including von Willebrand factor [Weibel, 2012]. The secretion of ET-1 by the regulated secretory pathway is modified by stimulations like hypothermia, mechanical stretch, histamine, and thrombin [Khimji and Rockey, 2010].
Intravenous ET-1 injection leads to a biphasic effect on blood pressure, which features a short decrease followed by a long-term increase [Wright and Fozard, 1988]. Vasoconstictive action of ET-1 is mainly mediated through ETA on VSMCs (Figure 1). ET-1–ETA interaction increases intracellular calcium, leading to the phosphorylation and activation of myosin light chain (MLC), which causes cellular contraction and vasoconstriction [Wirth et al. 2008]. ET-1–ETA interaction is also thought to mediate vasoconstriction through the calcium-independent Rho-kinase pathway [Miao et al. 2002]. In pathophysiological conditions, the expression of ETB on VSMCs is increased and ET-1–ETB interaction also promotes vasoconstriction [Chester et al. 1989]. However, vasodilatation by ET-1 is mediated through ETB on ECs. ET-1–ETB interaction stimulates the phosphorylation of endothelial nitric oxide (NO) synthase and NO production through the activation of the protein kinase B/Akt pathway [Liu et al. 2003]. The increased production of NO in ECs leads to an increase in cyclic guanine monophosphate in VSMCs, which causes vasodilatation through reduction of intracellular calcium, and inhibition of phosphorylation of MLC. Furthermore, ET-1–ETB interaction also stimulates the production of prostacyclin and promotes vasodilatation [Gratton et al. 1995].

Vascular endothelin system and representative endothelin receptor antagonists that were examined in clinical trials for hypertension and pulmonary hypertension. ECE-1, endothelin converting enzyme-1; ETA, endothelin receptor A; ETB, endothelin receptor B; ET-1, endothelin-1; NO, nitric oxide; PGI2, prostaglandin I2.
Endothelin receptor antagonism in hypertension
Endothelin system in the pathogenesis of hypertension
Due to the potent vasoconstrictive nature and the potential to maintain basal vascular tone, ET-1 was suggested to be strongly involved in the pathogenesis of hypertension, which was supported by some clinical observations. Patients with malignant hemangioendothelioma presented with highly increased plasma ET-1 levels and severe hypertension, which were normalized by the resection of the tumors [Yokokawa et al. 1991]. Patients with salt-sensitive hypertension and African American patients with hypertension also have elevated plasma ET-1 levels [Ergul et al. 1996; Ferri et al. 1997]. Furthermore, in patients who are normotensive, a high level of plasma ET-1 is related to the development of hypertension [Kumagae et al. 2010]. Having in mind the autocrine/paracrine characteristics of ET-1 action, it is not surprising though that plasma ET-1 levels in patients with essential hypertension do not usually correlate with blood pressure [Hirai et al. 2004]. The expression of ET-1 in resistant arteries, however, is elevated in moderate to severe essential hypertension [Schiffrin et al. 1997]. In addition, plasma ET-1 levels are related to subclinical renal function [Hirai et al. 2004]. These findings suggest that high plasma ET-1 levels in these patients appear to be the result of an overflow of local ET-1, and the local ET system might be essential in the development of hypertension and hypertensive organ damage.
The roles of the ET system in the pathogenesis of hypertension have been revealed by numerous studies using genetically modified animals, although animals that are deficient for either one of the main components of the ET system die at birth or early in life, as described above. ET-1 heterozygous knockout mice (ET-1+/−) that are morphologically normal (in contrast to ET-1−/− mice) paradoxically have an elevated blood pressure [Kurihara et al. 1994]. A systemic decrease in ET-1 did not lead to lower blood pressure and this unexpected phenomenon was explained later in animals genetically modified in specific cell types. Kisanuki and colleagues reported that endothelial cell specific ET-1 knockout mice showed reduced blood pressure without changes in the expression of ETA or ETB [Kisanuki et al. 2010]. They also demonstrated that systemic heterozygous ET-1 knockout mice (ET-1dlox/+) had normal blood pressure. In this study, they concluded that vascular endothelial ET-1 had an essential role in the maintenance of basal vascular tone, and a half dosage of ET-1 was sufficient to maintain it. These facts are supported by some studies using receptor knockout mice. ETA heterozygous knockout mice (ETA+/−) demonstrated normal blood pressure but decreased hypertensive response to ET-1 [Berthiaume et al. 2000], while SMC-specific ETA knockout mice showed decreased hypertensive response to ET-1 and decreased blood pressure [Stuart et al. 2013; Donato et al. 2014]. However, systemic ET-1 overexpressing mice did not show elevated blood pressure, while EC-specific ET-1 overexpressing mice developed hypertension [Hocher et al. 1997; Leung et al. 2011]. From these results, one may conclude that endothelial ET-1 is responsible for the pathogenesis of hypertension by regulating vascular tone, but nonendothelial ET-1 might have a lowering effect on blood pressure.
The ETB action towards blood pressure control is not simple. Genetic disruption and pharmacological blockade of ETB increase blood pressure, which suggests a depressor effect of ET-1 [Ohuchi et al. 1999]. However, endothelial ETB knockout mice do not develop high blood pressure but endothelial dysfunction characterized by decreased acetylcholine-induced vasodilatation [Bagnall et al. 2006]. Furthermore, these mice have higher plasma ET-1 levels caused by the decreased clearance of ET-1, but these changes were not pharmacologically sufficient to change the vessel tone. ETB thus appears to play a minor role in the control of vascular tone in hypertension. Gariepy and colleagues reported that ETB-deficient rats developed salt-sensitive hypertension, which suggests that ET-1 effects on the kidney through the ETB receptor might be relevant [Gariepy et al. 2000]. This experiment sheds light on the fact that the kidney is the other main organ for controlling blood pressure and is involved in the pathogenesis of hypertension along with the vasculature [Bolivar, 2013]. Further studies with cell-specific genetically modified animals revealed the detailed roles of the ET system in the kidney. Collecting duct-specific ET-1 knockout mice also demonstrated salt-sensitive hypertension [Ahn et al. 2004], which was caused by decreased ETB action through enhancing epithelial Na channel activity that leads to sodium retention [Ge et al. 2006; Bugaj et al. 2012]. The disruption of ETA in collecting duct in mice also revealed the role of ETA in salt-induced hypertension in addition to that of ETB. These mice further developed fluid retention [Ge et al. 2008; Stuart et al. 2013]. The ET system is thus tightly and intricately involved in the pathogenesis of hypertension, not only through the regulation of vascular tone but most importantly by the control of sodium retention, which might account for the main undesired side effects of ERAs.
Clinical application of endothelin receptor antagonists
ERAs have been developed in order to pharmacologically target hypertension in the clinical setting. With the development of orally active ERAs, a long-term blood pressure lowering effect of ERAs was demonstrated [Clozel et al. 1993; Li and Schiffrin, 1995]. A clinical trial for the treatment of hypertension was initially reported in 1998 (Table 1), and demonstrated that a high dose of dual ETA/B antagonist, bosentan (2000 mg/day), reduced blood pressure in a similar fashion to angiotensin-converting enzyme inhibitor, enalapril, at a dose of 20 mg/day [Krum et al. 1998]. This report also demonstrated bosentan-induced side effects including headache, flushing, leg edema, and liver injury. In 2002, another clinical trial showed the efficacy of darusentan, a selective ETA antagonist, without major side effects [Nakov et al. 2002]. However, ERAs have not been further developed because of their limited efficacy and the relatively high incidence of side effects like fluid retention and liver toxicity. Recently, the efficacy of darusentan in patients with treatment-resistant hypertension who took three or more antihypertensive drugs was reported [Weber et al. 2009]. In those patients, darusentan provided additional reduction in blood pressure. Fluid retention was well managed with diuretics. Furthermore, in a larger study, additional antihypertensive effect of darusentan was demonstrated in patients with treatment-resistant hypertension in whom it was more effective than guanfacine to reduce 24 h ambulatory blood pressure [Bakris et al. 2010]. Taken together, these results suggest that the pharmacological effects of ERAs on hypertension are specific compared with other established drugs, and in treatment-resistant hypertension, endothelin receptor antagonism could be a reasonable therapy. This assumption is supported by the findings of an elevated plasma ET-1 concentration in patients with severe hypertension and American African patients who are often treatment resistant.
Characteristics of the main clinical trials of endothelin receptor antagonists in patients with essential hypertension.

ESRD, end-stage renal disease; sCr, serum creatinine concentration.
The other therapeutic target of ERAs could be hypertension-induced end organ damage, especially chronic kidney disease. Plasma ET-1 levels are known to correlate with serum creatinine levels in healthy subjects and in patients with chronic kidney disease caused by hypertension, diabetes mellitus, and polycystic renal disease [Deray et al. 1992; Hirai et al. 2004]. ET-1 transgenic mice developed glomerulosclerosis, interstitial fibrosis without hypertension, and ERAs attenuated renal injury induced by hypertension [Hocher et al. 1997; Kassab et al. 1998]. From these findings, we can conclude that the ET system might be involved in the pathogenesis of hypertension-induced renal injury. The ASCEND study demonstrated that avosentan, an ETA receptor antagonist, reduced proteinuria in patients with type 2 diabetes, approximately 90% of whom had hypertension [Mann et al. 2010]. In the future, organ protection against hypertension might be a target of ERAs beyond their pressure-lowering effects [Anggrahini et al. 2009; Widyantoro et al. 2010; Vignon-Zellweger et al. 2012; Heiden et al. 2014]. Further long-term clinical trials are needed to develop ERAs as therapeutic agents in hypertension.
Endothelin receptor antagonism in pulmonary hypertension
Endothelin system in the pathogenesis of pulmonary arterial hypertension
PAH is a progressive intractable disease characterized by elevated pulmonary vascular resistance, right heart failure, and finally death. Classically, the pathophysiology of PAH has been considered the result of vasoconstriction and increased shear stress of pulmonary vessels [Rubin, 1993]. Like in the case of systemic hypertension, the ET system was considered to be involved in the pathogenesis of PAH because of the vasoconstrictive nature of ET-1. Based on the findings that patients with PH had higher plasma and tissue ET-1 levels with increased ET-1 gene expression [Stewart et al. 1991; Giaid et al. 1993], the roles of the ET system in PH have been further investigated using animal models, and the efficacy of ERAs have been demonstrated [Rubin, 2012].
Clinical application of endothelin receptor antagonists
Among ERAs, the dual ETA/ETB antagonist bosentan was the first approved by the US Food and Drug Administration (FDA) for the treatment of PAH with World Health Organization functional class (WHO-fc) III and IV. This was achieved after a first pilot randomized controlled trial (RCT) [Channick et al. 2001] (Table 2) while intravenous epoprostenol was the only available drug in the treatment of PAH at the time. A larger clinical trial, BREATHE-1, also demonstrated the efficacy of bosentan with the improvement in WHO-fc, exercise capacity, and time to clinical worsening after 16-week treatment [Rubin et al. 2002]. The efficacy of bosentan in PAH has been further confirmed by additional clinical trials of combination therapy with epoprostenol [Humbert et al. 2004], for Eisenmenger syndrome [Galie et al. 2006], and for less functionally impaired patients [Galie et al. 2008b], with improved exercise capacity and hemodynamics. In open label trials, improvement of hemodynamics and exercise capacity was also observed in children with PAH and PAH associated with human immunodeficiency virus infection [Barst et al. 2003; Sitbon et al. 2004]. Long-term effects of bosentan have been demonstrated by open-label trials following RCTs with an estimated survival rate at 1 and 2 years of 96% and 89%, respectively [McLaughlin, 2006] compared with the predicted survival of 69% and 57%, estimated using a National Institutes of Health equation [D’Alonzo et al. 1991]. With regards to the side effects of bosentan, liver injury occurred in 10% of the patients. This effect was dose dependent, as observed in the clinical trial for essential hypertension, and was reversible after dose reduction or discontinuation [Rubin et al. 2002]. At present, the recommended therapeutic dose of bosentan is 250 mg/day since liver injury was more frequent and severe with 500 mg/day. Bosentan has thus been established as a first-line therapy in patients with WHO-Fc II and III PAH [Galie et al. 2013].
Characteristics of the main clinical trials of endothelin receptor antagonists in patients with pulmonary hypertension.
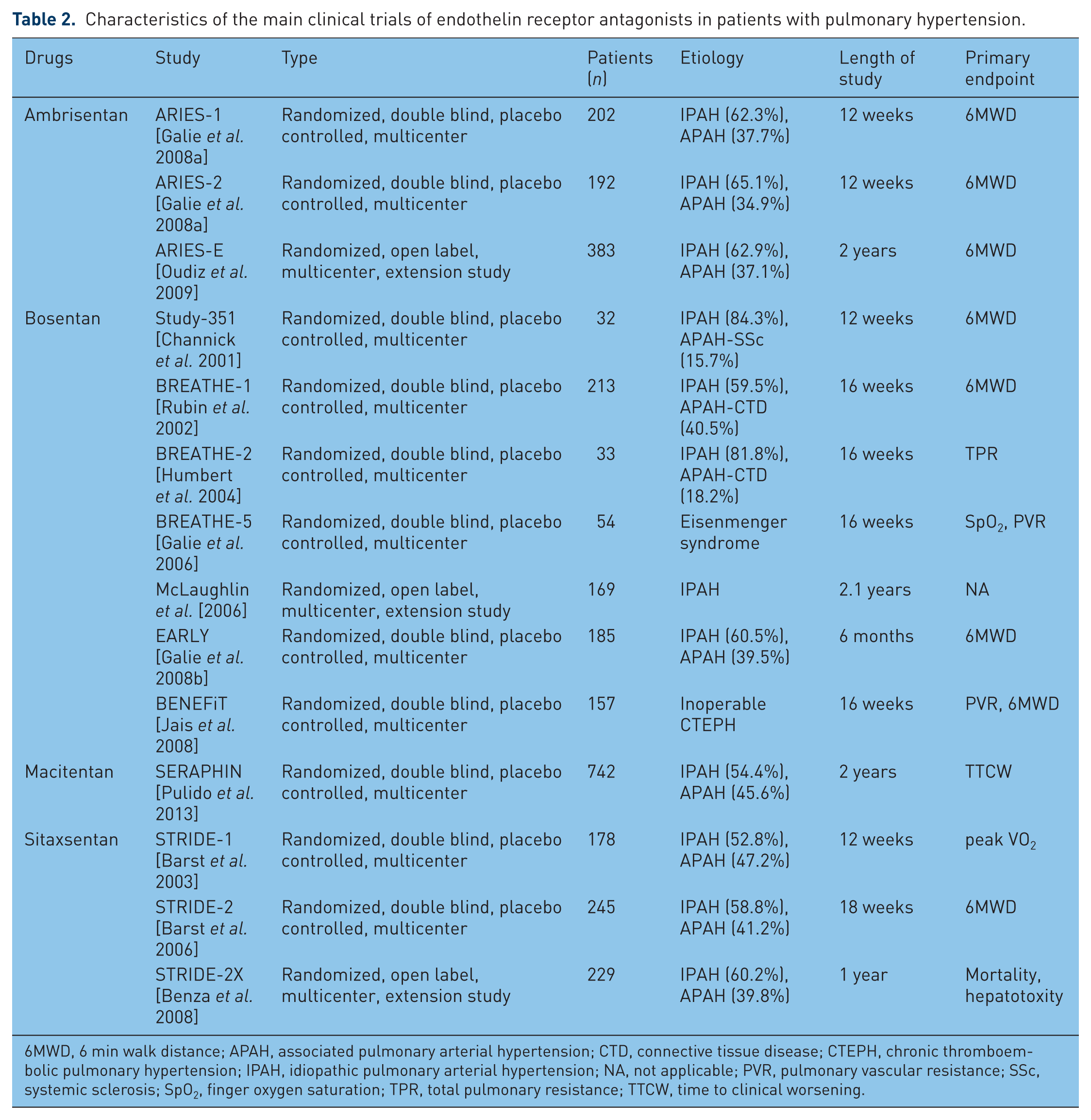
6MWD, 6 min walk distance; APAH, associated pulmonary arterial hypertension; CTD, connective tissue disease; CTEPH, chronic thromboembolic pulmonary hypertension; IPAH, idiopathic pulmonary arterial hypertension; NA, not applicable; PVR, pulmonary vascular resistance; SSc, systemic sclerosis; SpO2, finger oxygen saturation; TPR, total pulmonary resistance; TTCW, time to clinical worsening.
The efficacy and safety of the selective ETA receptor antagonist sitaxsentan was also evaluated in a small open-label trial including children and adults with WHO-fc II, III or IV PAH, which showed improvement in exercise capacity and hemodynamics [Barst et al. 2002]. Larger RCTs and a following long-term open-label trial also demonstrated the efficacy of sitaxsentan [Barst et al. 2004; Langleben et al. 2004] and it was approved mainly in Europe. However, liver injury became the main problem, and although its incidence was lower (up to 5%) compared with bosentan [Barst et al. 2006], cases of death due to severe liver toxicity [Lavelle et al. 2009] led to the withdrawal of sitaxsentan from the market.
Ambrisentan was the second ETA selective antagonist that was established as first-line therapy in patients with WHO-fc II and III disease [Galie et al. 2013]. Randomized controlled 12-week trials in patients with PAH demonstrated significant improvement in hemodynamics, exercise capacity, and time to clinical worsening [Galie et al. 2008a]. A long-term open-label trial following the RCTs also demonstrated sustained improvements in exercise capacity and time to clinical worsening after 2 years of ambrisentan treatment [Oudiz et al. 2009]. Furthermore, ambrisentan was well tolerated, and the incidence of liver injury induced by ambrisentan was less than 2%. Peripheral edema also occurred but remained mild to moderate and did not require discontinuation of the drug.
Recently, a new ERA was developed for the treatment of PAH. Macitentan, a dual ETA/ETB antagonist, has been shown to significantly reduce morbidity and mortality in patients with PAH with WHO-fc II, III, or IV disease with minimum side effects in a long-term RCT, which was the largest event-driven study in PAH [Pulido et al. 2013]. The incidence of macitentan-induced liver injury was comparable to that induced by placebo. In September 2013, macitentan was approved by the FDA followed by European administrations and has become first-line treatment for patients with WHO-fc II or III PAH [Galie et al. 2013]. Macitentan has a 40- and 2000-fold increased affinity for tissue, with a longer half life compared with bosentan and ambrisentan respectively [Iglarz et al. 2008]. The new pathophysiological concept of PAH involves the proliferation of apoptosis-resistant ECs, VSMCs, and fibroblasts in the pulmonary vasculature caused by abnormal response to vascular cell injury. Genetic predisposition, inflammation, and dysregulation of cellular metabolism are suggested to be responsible for disease progression [Rai et al. 2008; Rabinovitch, 2012]. Currently available therapies cannot reverse the pathophysiological changes efficiently. Strong inhibition of the ET system by macitentan might be a robust therapy against the pathogenesis of PAH.
Endothelin receptor antagonism in other pulmonary vessel disease
Left heart disease (LHD) is the most common cause of PH [Vachiery et al. 2013]. Although several RCTs have been conducted to clarify the efficacy of ERAs in severe systolic heart failure, the results obtained from almost all studies were disappointing owing to no effect or early termination caused by ERA-induced liver injury or fluid retention [Kalra et al. 2002; Luscher et al. 2002; Anand et al. 2004; Packer et al. 2005]. No clinical trial for PH related to heart failure with preserved ejection fraction has been performed. In PH due to LHD, the efficacy of ERAs remains unknown.
Chronic lung disease (CLD) is also a common cause of PH, and the prognosis of CLD with PH is worse than without PH [Seeger et al. 2013]. At this time, there is no clinical study targeting CLD- PH and there is no consensus about the potential of ERAs. There are several clinical trials that studied the efficacy of ERAs in idiopathic pulmonary fibrosis (IPF) in anticipation of the antifibrotic effect of ERAs; however, they failed to show improvement [King et al. 2011]. Ambrisentan has been reported to even deteriorate the clinical status in patients with IPF [Raghu et al. 2013].
Chronic thromboembolic pulmonary hypertension (CTEPH) is PH due to stenosis and obstruction of pulmonary artery with nonresolving organized thromboemboli. Pulmonary endarterectomy is the only therapy that cures this pathology, but up to 40% of patients cannot undergo surgery [Kim et al. 2013]. A clinical trial for these patients demonstrated that bosentan reduced pulmonary vascular resistance, but failed to improve exercise capacity [Jais et al. 2008]. More RCTs are needed to clarify the role of ERAs in CTEPH.
The selectivity of endothelin receptor antagonists
Whether receptor selectivity of ERAs matters is an issue which has not been satisfactorily resolved. In systemic hypertension, ETB antagonism in addition to ETA antagonism can lead to greater vasodilatation [Cardillo et al. 1999], but induces sodium retention as noted above. Recent clinical trials mainly studied the efficacy of ETA selective antagonists. In the treatment of hypertension, ETA selective antagonism seems to be reasonable. In PAH, the selectivity of receptor antagonism also remains an open question. From the perspective of the pathophysiology of PAH, which features not only vasoconstriction but also abnormal cell proliferation, robust blockade of the complete ET system seems to be needed. In order to block the ET system sufficiently, both ETA and ETB antagonism would be needed because cell growth might be mediated by both types of receptors [Davie et al. 2002]. In addition, each receptor probably interacts and heterodimerizes with another and, in this context, dual ERAs like macitentan might be eligible. At present, there is no study that directly compares the efficacy of selective ETA antagonists with dual ETA/ETB antagonists. Further trials are needed to reveal the most suitable endothelin receptor antagonism.
Future directions of endothelin receptor antagonism
In this quarter of a century since ET-1 was discovered, understanding of the ET system has rapidly developed, produced numerous studies, and finally led to clinical application that can improve the prognosis of patients as a result of translational research. The research is now developing in hypertension and pulmonary hypertension, as well as other diseases. In systemic sclerosis-related digital ulcer, bosentan therapy has been established. Diabetic nephropathy would be the next promising target. A large RCT demonstrated that avosentan, a selective ETA antagonist, could reduce albuminuria [Mann et al. 2010], and this effect seems to be independent of the renin angiotensin system [Kohan et al. 2011; Andress et al. 2012]. Furthermore, cancer and vasospasm after subarachnoid hemorrhage can be the next targets of ERAs [Macdonald et al. 2012; Nelson et al. 2012].
Another therapeutic intervention to block the ET system has emerged. ECE-1 is not only the key regulator of ET production but also a vasopeptidase that can hydrolyze several vasoactive peptides, including atrial natriuretic peptide and bradykinin [Johnson et al. 1999], which promote vasodilatation and organ protection. In a recent clinical trial, the ECE and neutral endopeptidase inhibitor daglutril improved blood pressure control in patients with hypertension and type 2 diabetes [Parvanova et al. 2013]. ECE-1 inhibitors could be a plausible therapeutic drug in cardiovascular diseases [Emoto et al. 2005; Miyagawa et al. 2010; Hartopo et al. 2013]. Thus, extended understanding of endothelin’s physiology could lead to further clinical applications in a variety of diseases mediated by the ET system.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declare no conflicts of interest in preparing this article.
