Abstract
Background:
Continuous long-term treatment with nitrates may cause nitrate tolerance. Nebivolol is a highly selective beta1-adrenergic antagonist with additional nitric oxide (NO)-mediated vasodilatory effects. However, there have been no investigations into whether or not the long-term administration of nebivolol might cause nitrate tolerance.
Methods:
We performed a randomized, double-blind, placebo-controlled, cross-over study in 16 healthy men. Subjects received 5 mg nebivolol or placebo once daily for 8 days in random order divided by a drug-free interval of 2 weeks. Forearm blood flow (FBF) was measured by venous occlusion plethysmography 3 h after oral intake of the first and last doses of nebivolol and placebo, respectively. FBF was measured again following the intravenous administration of 4 μg nitroglycerin/kg body weight/min for 5 min.
Results:
Following 8 days of continuous intake of placebo, nitroglycerin increased FBF by 54% (
Conclusions:
These findings indicate no evidence of nitrate tolerance caused by long-term administration of nebivolol. On the contrary, long-term intake of nebivolol increases rather than decreases the NO-mediated vasodilating effects.
Introduction
There is an ongoing discussion on nitrate tolerance and the mechanisms that it might be caused by [Kiowski, 1991; Elkayam, 1991; Stoschitzky et al. 1995; Parker and Parker, 1998; Ignarro et al. 2002; Gori and Parker, 2002; Münzel et al. 2005; Fox et al. 2006; Jansen et al. 2006; Gutterman, 2007; Dejam et al. 2007; Wenzel et al. 2007; Wölkart et al. 2008; Sayed et al. 2008; Yamauchi et al. 2008; Schmidt et al. 2008; Dudek et al. 2010; Münzel et al. 2011; Fekete et al. 2013], and a recent review further concludes that prolonged exposure to organic nitrates not only induces nitrate tolerance
Nebivolol is a third-generation beta1-selective beta-blocker with additional nitric oxide (NO)-mediated vasodilatory effects, which have been shown
In order to address these unsolved issues we performed a randomized, double-blind, placebo-controlled, cross-over study in 16 healthy men. Subjects received either nebivolol or placebo for 8 days, each separated by a drug-free interval of 2 weeks. Vasodilating effects were measured by venous occlusion plethysmography. In addition, effects on heart rate and blood pressure were measured. All these investigations were performed following administration of the first as well as the respective last doses following 8 days of chronic intake of nebivolol or placebo.
Methods
The study was performed in 16 healthy men according to a randomized, double-blind, placebo-controlled, cross-over protocol. Subjects received single oral doses of 5 mg nebivolol or placebo every morning, i.e. once daily, for 8 days, separated by a drug-free interval of 2 weeks.
Investigations were performed immediately before as well as 3 h after intake of the respective first dose (day 1) and eighth dose (day 8) of nebivolol and placebo in order to investigate acute and long-term effects.
Haemodynamic effects
Heart rate (by electrocardiography monitoring) and blood pressure (by sphygmomanometry) were measured at rest before the intake of the respective first doses (baseline) and repeated 3 h following intake of the respective first doses on day 1 as well as 3 h after intake of the respective last doses on day 8.
NO-mediated effects on forearm blood flow
Forearm blood flow (FBF) was measured by venous occlusion plethysmography on the right arm as described previously [Kiowski et al. 1994]. Briefly, a mercury-in-silastic strain gauge was placed at the upper third of the forearm, which rested comfortably on a support slightly above the level of the heart. The strain gauge was coupled to an electronically calibrated plethysmograph (Hokanson EC-5R, Issaquah, WA, USA). Venous occlusion was achieved by a blood-pressure cuff applied proximal to the elbow and inflated to 40 mmHg by a rapid cuff inflator (Hokanson E20). The hand was excluded from the circulation by inflating a paediatric blood-pressure cuff placed around the wrist to 50 mmHg above systolic pressure 1 min prior to and during the FBF measurement in order to eliminate the unpredictable influence of arteriovenous shunts in the hand. The mean values of 4–6 recordings obtained within 1 min were taken for statistical evaluation. Results of FBF are given as ml/min/100 ml of forearm tissue.
This investigation mentioned above was performed before the intake of the first dose of either drug and repeated 3 h later on the first day (day 1), as well as immediately before and 3 h after the intake of either drug on the eighth day (day 8). Herewith, baseline values, effects following intake of a single dose, as well as effects after long-term administration of the drugs were determined.
On day 8, this investigation was followed by a period of recovery of 15 min, then subjects received intravenous infusions of 4 μg nitroglycerin/min/kg body weight over 5 min, and blood flow was measured again according to the protocol mentioned above.
Statistical methods
Results are given as means ± standard deviation. Significances of differences within groups were calculated by repeated measures ANOVA (repeated measures ANOVA on ranks when applicable),
Results
NO-derived vasodilation
Neither oral intake of nebivolol nor placebo caused any significant effect on FBF. However, intravenous administration of nitroglycerin markedly increased FBF. Following 8 days of continuous intake of placebo, nitroglycerin increased FBF by 54% (
Forearm blood flow (ml/min/100 ml forearm tissue)*.

Nebivolol and placebo
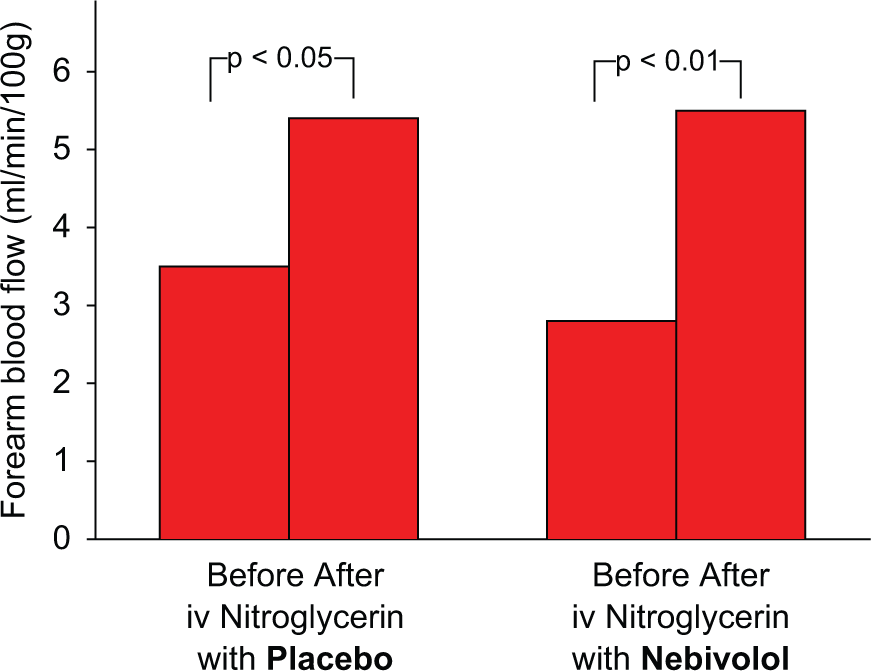
Effects of 4 mg intravenous nitroglycerin / minute / kg body weight over five minutes on FBF following one week of continuous oral intake of Placebo (+54%, left panel) or 5 mg Nebivolol once daily (+96%, right panel), with the increase following Nebivolol being more pronounced (p < 0.05) than that following Placebo.
Haemodynamic effects
Compared with placebo, heart rate was decreased by 11% 3 h following intake of the respective first doses of nebivolol (day 1) and by 8% 3 h following intake of the respective last doses of nebivolol (day 8); systolic blood pressure was decreased by 5% 3 h following intake of the respective first doses of nebivolol (day 1) and by 4% 3 h following intake of the respective last doses of nebivolol (day 8); the rate pressure product (RPP), i.e. the product of heart rate × systolic blood pressure, was decreased by 16% 3 h following intake of the respective first doses of nebivolol (day 1) and by 11% 3 h following intake of the respective last doses of nebivolol (day 8); all these effects were statistically significant (
Discussion
It has been shown and discussed repeatedly that long-term administration of nitrates may cause nitrate tolerance (as mentioned above). However, our findings indicate no evidence of nitrate tolerance caused by long-term intake of nebivolol. On the contrary, they show that long-term administration of nebivolol appears to increase rather than decrease the vasodilating effects of nitroglycerin. This might, at least in part, be caused by the antioxidant effects of nebivolol [Khan et al. 2013].
Although the increase of FBF following 1 week of oral intake of nebivolol compared with baseline was significantly more pronounced than with placebo (
In addition, our data confirm that nebivolol, a third-generation beta1-selective beta-blocker with additional NO-mediated vasodilatory effects, significantly decreases both heart rate and systolic blood pressure and, particularly, the RPP.
In summary, our data show no evidence that long-term intake of nebivolol might cause nitrate tolerance. On the contrary, long-term administration of nebivolol appears to increase rather than decrease the NO-mediated vasodilating effects.
Footnotes
Acknowledgements
The authors wish to thank Menarini SA, Florence, Italy for supporting this study.
Funding
The research was supported by Menarini SA, Florence, Italy, and the declaration was accepted by the Ethical Committee of the Medical University of Graz, Austria.
Conflict of interest statement
The authors declare no conflicts of interest in preparing this article.
