Abstract
Background:
We hypothesized that the combination of nebivolol and lifestyle modification would reduce large artery stiffness in middle-aged and older hypertensive adults more than either intervention alone.
Methods:
To address this, 45 men and women (age 40–75 years) with stage I hypertension were randomized to receive either nebivolol (NB; forced titration to 10 mg OD; n = 15; age 57.2 ± 11.4 years; body mass index [BMI] 30.8 ± 5.8 kg/m2), lifestyle modification (LM; 5–10% weight loss via calorie restriction and physical activity; n = 15; age 52.7 ± 8.5 years; BMI 33.9 ± 7.2 kg/m2) or nebivolol plus lifestyle modification (NBLM; n = 15; age 58.9 ± 9.4 years; BMI 32.5 ± 4.9 kg/m2) for 12 weeks. β-stiffness index, a blood-pressure-independent measure of arterial stiffness, and arterial compliance were measured via high-resolution ultrasound and tonometry at baseline and after the 12-week intervention. There was no difference between groups in age, body weight or composition, blood pressure, or in β-stiffness index or arterial compliance at baseline (all p > 0.05).
Results:
Following the 12-week intervention, body weight decreased ~5% (p < 0.05) in the LM and NBLM groups but did not change from baseline in the NB group (p > 0.05). Supine brachial and carotid systolic and diastolic blood pressure declined following treatment in each of the groups (p < 0.05). However, the magnitude of reduction was not different (p < 0.05) between groups. β-stiffness index declined (–2.03 ± 0.60, –1.87 ± 0.83 and −2.51 ± 0.90 U) and arterial compliance increased similarly (both p > 0.05) in the NB, LM and NBLM groups, respectively.
Conclusion:
In summary, our findings indicate that the combination of nebivolol and lifestyle modification reduced large artery stiffness to a similar degree as either intervention alone in middle-aged and older hypertensive adults.
Introduction
Advancing age [Avolio et al. 1983; Vaitkevicius et al. 1993], obesity [Sutton-Tyrrell et al. 2001; Wildman et al. 2003], and hypertension [Liao et al. 1999] are associated with stiffening of the large elastic arteries in the cardiothoracic region. Arterial stiffness is an independent predictor of coronary events [Boutouyrie et al. 2002], stroke [Laurent et al. 2003], and all-cause mortality [Laurent et al. 2001] in individuals with essential hypertension.
Numerous antihypertensive drugs have been efficacious in reducing arterial stiffness and, as such, may contribute to improved outcomes in hypertensive patients. Beta-blockers are frequently prescribed to individuals with hypertension and are efficacious in reducing brachial arterial pressure. However, the impact of on large artery stiffness is more variable and less favorable compared with other classes of antihypertensive drugs [Dhakam et al. 2008; Mackenzie et al. 2009; Williams et al. 2006].
Nebivolol, a third-generation beta-blocker, has been reported to augment nitric oxide bioavailability [Maffei and Lembo, 2009; Vanhoutte and Gao, 2013] and improve flow-mediated dilation [Simova et al. 2009] in patients with hypertension. The results of previous studies also suggest that nebivolol is efficacious in reducing arterial stiffness in hypertensive adults [Agabiti-Rosei et al. 2009; Dhakam et al. 2008; Soanker et al. 2012] although these reports are based on indirect, blood pressure (BP)-dependent indices. Whether nebivolol reduces large artery stiffness independent of reductions in BP is unclear.
Lifestyle modification is the first line of treatment for hypertension. Regular aerobic exercise [Tanaka et al. 2000], weight loss [Cooper et al. 2012; Dengo et al. 2010; Hughes et al. 2012], and sodium restriction [Gates et al. 2004; Seals et al. 2001], all components of comprehensive lifestyle modification, have been shown to reduce large artery stiffness. However, whether nebivolol and lifestyle modification reduce arterial stiffness more than either intervention alone in hypertensive adults is not known. The purpose of the present study was to test this hypothesis.
Methods
Subjects
A total of 45 men (n = 21) and women (n = 24) aged 40–75 volunteered and were determined to be eligible for the study from the 104 individuals who were screened. Subjects were hypertensive (BP ≥ 140 and/or 90 mmHg and <160 and/or 100 mmHg) and free from overt chronic disease. The subjects were not taking any medications that could affect variables of interest at the time of the study. Subjects receiving antihypertensive therapy prior to the study discontinued their medications for 2 week before baseline testing. All subjects were weight stable (±2 kg) and sedentary (<30 min/week of low-intensity physical activity) for at least 6 months prior to joining the study. The Virginia Polytechnic and State University Institutional Review Board approved all study protocols. The nature, purpose, risks, and benefits were explained to each subject prior to obtaining informed consent.
Experimental design and protocol
Following the completion of baseline testing, subjects were randomly assigned to nebivolol (NB; n = 15), lifestyle modification (LM; n = 15), or the combination of nebivolol and lifestyle modification (NBLM; n = 15) groups. Subjects randomized to NB or NBLM groups began with 5 mg/day of nebivolol and increased to 10 mg/day if brachial BP was >120/80 mmHg during the first 2 weeks of therapy. Nebivolol was provided bimonthly with a random quantify of extra tablets, and compliance was assessed by pill count. The participants in the NB group were instructed to maintain their current body weight, habitual dietary intake, and physical activity level. Subjects randomized to the LM and NBLM groups received weekly lifestyle counseling by a registered dietitian to ensure adequate progress and compliance. Sample menus, 14 days of meal plans, and grocery shopping lists were provided to each individual. Individuals in the LM and NBLM groups were instructed to reduce their daily calorie intake by 500–1000 calories and to perform a minimum of 150 min/week of moderate-intensity physical activity or 3000 steps/day above baseline levels. The diet plan conformed to the DASH dietary guidelines emphasizing low-fat dairy products, fruits, and vegetables and contained 55% calories as carbohydrates, 30% calories as fat, and 15% calories as protein [Chobanian et al. 2003]. The sodium consumption goal was 1500 mg/day for all subjects in the LM and NBLM groups. All measurements were performed in the Virginia Tech Human Integrative Physiology Laboratory between 0700 to 1100 hrs after a 12-hour fast. Every participant refrained from caffeinated products for 24-hour and exercise for 48-hours prior to each testing session. All subjects were free of acute illness or infections for at least 1-week prior to each testing day.
Measurements
Body weight was measured to the nearest 0.1 kg using a digital scale (Model 5002, Scale-Tronix, Inc.) at baseline and every week throughout the 12-week intervention. Height was measured to the nearest 0.1 cm using a stadiometer. Body composition was measured via dual-energy X-ray absorptiometry (DEXA; Lunar Prodigy Advance, GE Medical Systems, software version 8.10e).
Resting arterial pressure measurements in the right brachial artery were performed every week throughout the treatment period. Subjects were instructed to avoid consuming caffeinated products 12 hours prior to the visit. The recordings were made in quiet and comfortable conditions, strictly conforming to the American Heart Association guidelines [Pickering et al. 2005]. Subjects rested in a seated position for 10 minutes prior to the automated sphygmomanometry (Pilot model 9200, Colin Instruments Corp.). Measurements were performed every 3 minutes until BP stability (±6 mmHg difference for both systolic and diastolic BP among three sequential measurements) was achieved.
To determine habitual dietary intake and compliance, all participants completed a detailed 4-day diet record (consecutive 3 weekdays and 1 weekend day) at baseline and week-12 of the intervention. Total calorie, macronutrient, and micronutrient intake were analyzed using the Nutrition Data System for Research (NSD-R 6.0, University of Minnesota) software. Daily step counts were measured using pedometers (Accusplit Eagle 120XL) which were returned every 2 weeks to assess compliance with the daily physical activity instructions.
Plasma lipid and lipoprotein concentrations were quantified using conventional methods in a commercial laboratory. Plasma glucose concentrations were determined with a YSI Stat Plus glucose analyzer (model 2300, Yellow Springs Instruments). High-sensitivity C-reactive protein (hsCRP), tumor necrosis factor-α (TNF-α), and interleukin-6 (IL-6) plasma concentrations were measured by enzyme-linked immunosorbent assay (ELISA; R&D Systems, Minneapolis, MN). Oxidized low-density lipoprotein (LDL) and plasma insulin concentrations were measured with commercially available ELISA kits (ALPCO Diagnostics, Salem, NH and Diagnostic System Laboratories, Webster, TX, respectively). Insulin sensitivity was estimated by the homeostasis model assessment (HOMA) approach. The HOMA index was calculated by the product of plasma blood glucose and insulin divided by 22.5.
β-stiffness index was measured using an ultrasound unit (Sonos 7500, Philips Medical Systems) equipped with a high-resolution linear array transducer (3–11 MHz) and applanation tonometry to acquire carotid artery waveforms (NIHem, Cardiovascular Engineering, Inc.). After 20 minutes of quiet rest in a supine position, longitudinal B-mode images of the left common carotid artery diameter 1–2 cm proximal to the carotid bulb were obtained over the course of 15 consecutive cardiac cycles. The transducer was placed at a 90° angle directly over the artery for clear visibility of the near and far walls. The images were stored on optical disks for offline quantification of systolic and diastolic carotid artery diameters using commercially available software (Vascular Research Tools 5, Medical Imaging Applications, LLC). Brachial BP (within ±6 mmHg for both systolic BP [SBP] and diastolic BP [DBP] among three sequential measurements) was measured via an automated sphygmomanometer (NIHem, Cardiovascular Engineering, Inc.) and used to calibrate peak and trough single-averaged waveforms. β-stiffness index was calculated as: β = ln(P1/P0)/((D1 – D0)/D0), where D0 represents the minimal diameter recorded during diastole, D1 represents the maximal diameter recorded during systole, P0 represents the pressure measured during diastole, and P1 represents the pressure measured during systole.
Statistical analysis
Repeated measures analysis of variance was used to examine the between and within groups differences in arterial stiffness and other dependent variables from baseline to follow-up testing. Post hoc comparisons were performed utilizing the Tukey procedure. The present study was not designed or powered to detect gender differences in the primary outcome. As such, we pooled the data for males and females for each treatment group. Pearson’s product moment correlations were used to explore the relationship among key variables of interest. Data are expressed as means ± standard error (SE). The significance level was set a priori at p < 0.05.
Results
Subject characteristics at baseline and following the intervention are shown in Table 1. There were no baseline differences in age, weight, body mass index (BMI), body fat percentage, fat mass, and fat free mass between groups (all p > 0.05). Supine resting heart rate, brachial SBP and DBP, and carotid SBP and DBP was not significantly different between groups. In addition, baseline daily physical activity (steps) was not different in the LM and NBLM groups (p > 0.05). The numbers of premenopausal and postmenopausal women in each group were 1/7, 3/5, and 2/5 in the NB, LM, and NBLM groups, respectively. There were two postmenopausal women receiving hormone replacement therapy in the NB group and one premenopausal woman taking oral contraceptives in the NBLM group.
Subject characteristics at baseline (PRE) and after the intervention (POST).
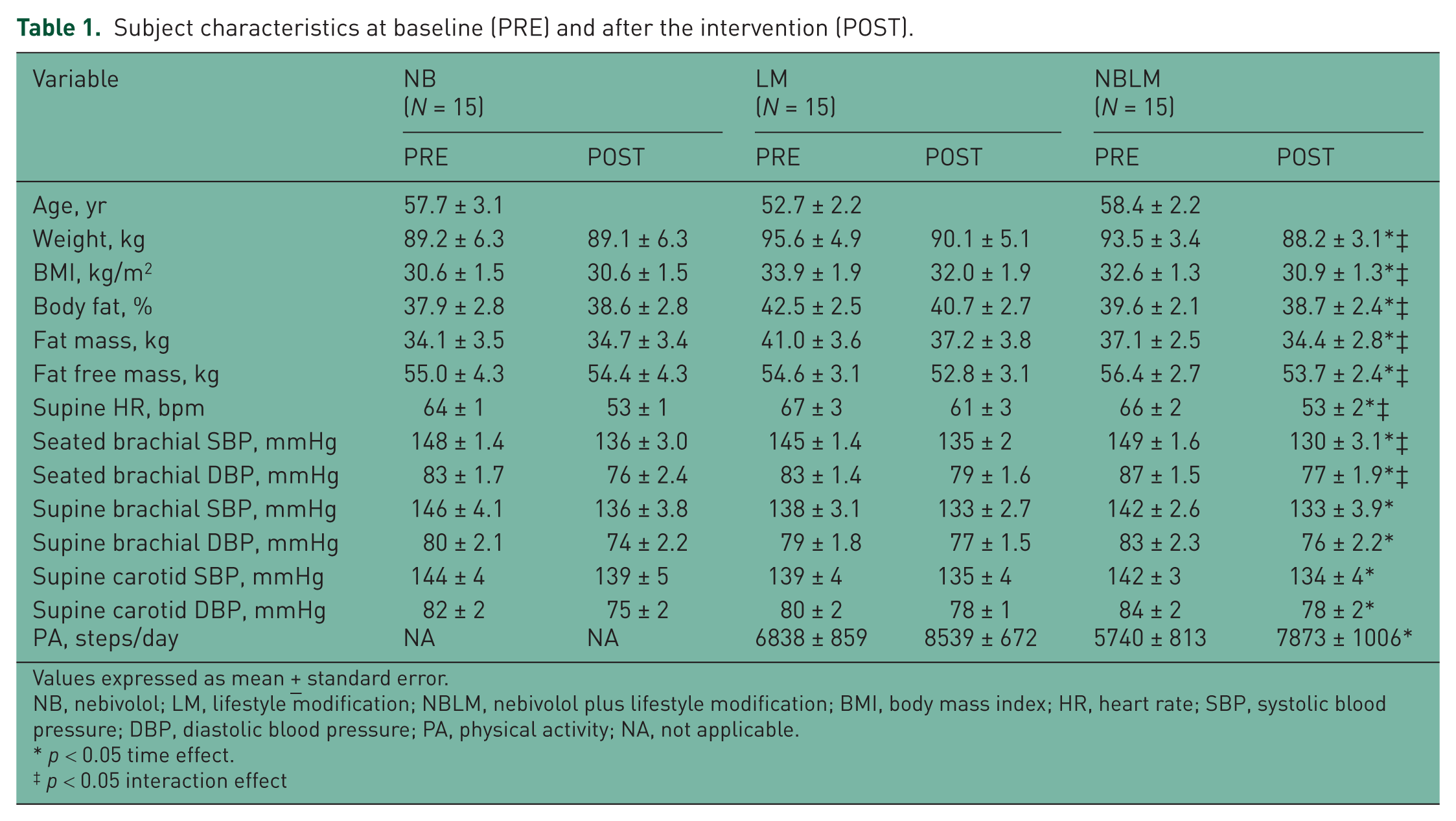
Values expressed as mean ± standard error.
NB, nebivolol; LM, lifestyle modification; NBLM, nebivolol plus lifestyle modification; BMI, body mass index; HR, heart rate; SBP, systolic blood pressure; DBP, diastolic blood pressure; PA, physical activity; NA, not applicable.
p < 0.05 time effect.
p < 0.05 interaction effect
Following the 12-week intervention, there was a significant decrease in body weight (–5.55 ± 0.59 and −5.35 ± 0.97 kg), corresponding to −6.1% and −5.6% of initial body weight in the LM and NBLM groups, respectively. BMI (–1.92 ± 0.20 and −1.77 ± 0.34 kg/m2), body fat percentage (–1.83 ± 0.32 and −0.87 ± 0.59%), fat mass (–3.79 ± 0.36 and −2.67 ± 0.72 kg), and fat free mass (–1.76 ± 0.35 and −2.69 ± 0.62 kg) decreased in the LM and NBLM groups, respectively. There was a significant increase in step count (1701 ± 550 and 2032 ± 669 steps/day) in the LM and NBLM groups, respectively. However, there were no changes in body weight, BMI, fat mass, and fat free mass (all p > 0.05) in the NB group following the 12-week intervention. Supine resting heart rate decreased (–5 ± 2, –10 ± 1, and −12 ± 2 bpm) in the LM, NB, and NBLM groups, respectively. Supine brachial SBP and DBP decreased (–9 ± 3/–6 ± 2, –5 ± 2/–2 ± 1 and −9 ± 3/–7 ± 2 mmHg; p < 0.05) in the NB, LM, and NBLM groups, respectively. In addition, supine carotid SBP and DBP decreased (–5 ± 4/–7 ± 2, –4 ± 2/–2 ± 1, and −7 ± 4/–7 ± 2 mmHg; all p < 0.05) in the NB, LM, and NBLM groups, respectively. The magnitude of change was not significant between groups.
Compliance to nebivolol was 98.2% overall with no individual missing more than three daily doses. A total of 5 subjects (2 from the NBLM group) remained on the 5 mg/day dose while 25 participants had their dosages increased to 10 mg/day after the initial 2 weeks for the remainder of the study. There was no significant difference in dosage received or compliance in the NB and NBLM group.
Habitual dietary intake before and after the intervention are shown in Table 2. Baseline carbohydrate intake (percentage of the total) was higher (p > 0.05) in the NBLM group compared with the LM group. There were no other baseline differences in macronutrient or micronutrient intake (all p > 0.05).
Dietary Intake at baseline (PRE) and following the intervention (POST).
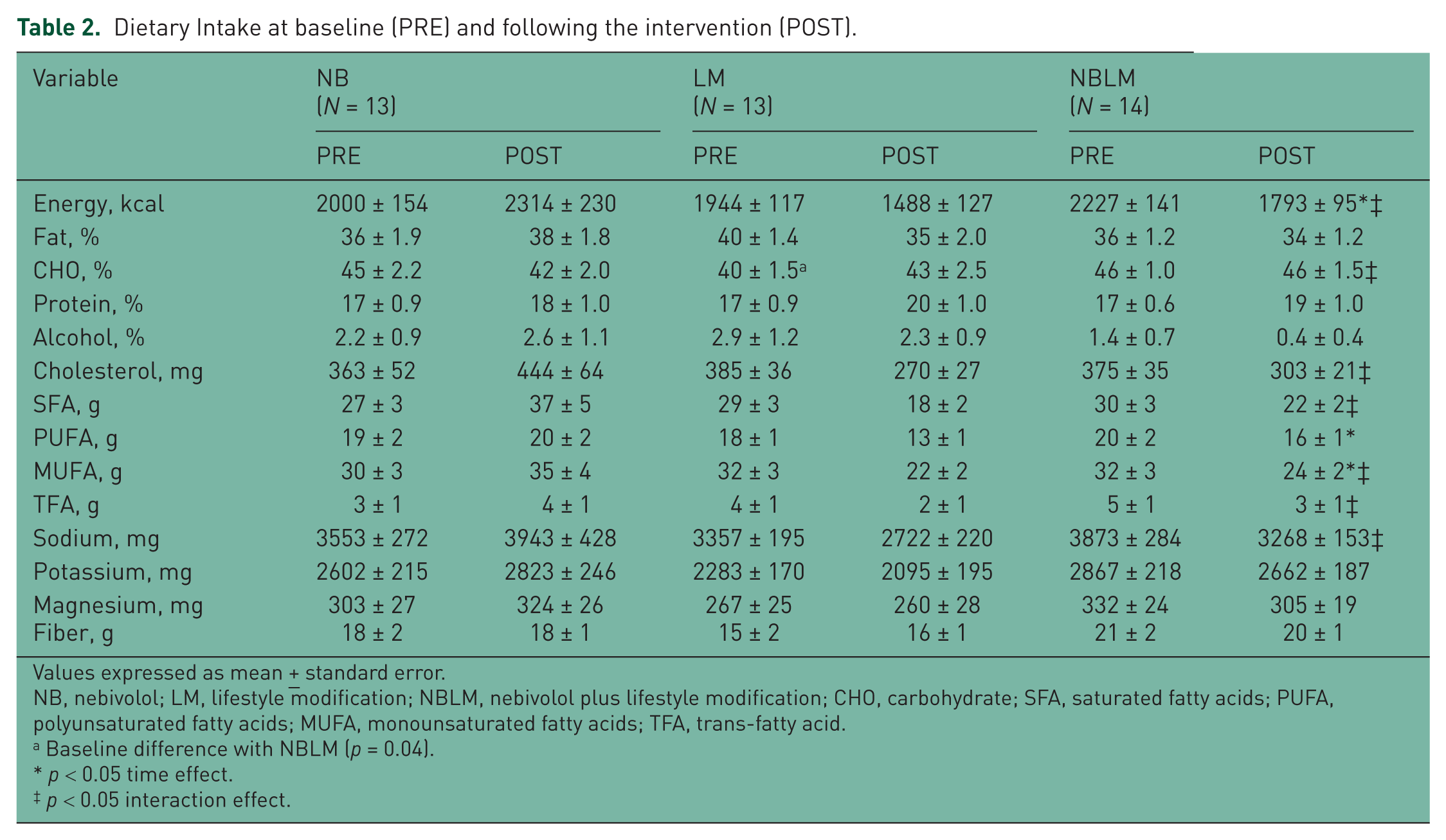
Values expressed as mean ± standard error.
NB, nebivolol; LM, lifestyle modification; NBLM, nebivolol plus lifestyle modification; CHO, carbohydrate; SFA, saturated fatty acids; PUFA, polyunsaturated fatty acids; MUFA, monounsaturated fatty acids; TFA, trans-fatty acid.
Baseline difference with NBLM (p = 0.04).
p < 0.05 time effect.
p < 0.05 interaction effect.
Following the 12-week intervention, the total calorie intake declined (–442 ± 144 and –399 ± 132 kcal; p < 0.05) in the LM and NBLM groups, respectively. Total calorie intake did not change (p > 0.05) in the NB group during the intervention. There were no changes in macronutrient composition in the three groups following the treatment period (all p > 0.05). Cholesterol intake decreased (–107 ± 47 and −47 ± 47 g; p < 0.05) in the LM and NBLM groups, respectively, but increased (40 ± 46 g; p < 0.05) in the NB group. Saturated fatty acid intake decreased significantly (–11 ± 3 and −7 ± 3 g) in the LM and NBLM groups, respectively, while it increased (10 ± 4 g) in the NB group. Monounsaturated fatty acid consumption decreased (–10 ± 3 and −8 ± 3 g; p < 0.05) in the LM and NBLM groups, respectively. Trans-fatty acids intake decreased (–2 ± 1, 1 ± 1, and −2 ± 1 g; p = 0.04) in the LM, NB, and NBLM groups, respectively. Daily sodium intake changed (373 ± 306, –516 ± 230 and −577 ± 303 g; p = 0.049) over the treatment period in the NB, LM, and NBLM groups, respectively. There were no other differences in dietary intake between groups or following the intervention.
Circulating metabolic and cardiovascular disease risk factors before and after the intervention are shown in Table 3. There were no baseline metabolic and cardiovascular disease risk factor differences (all p > 0.05).
Cardiometabolic risk factors at baseline (PRE) and following the intervention (POST).

Values expressed as ± standard error of mean.
NB, nebivolol; LM, lifestyle modification; NBLM, nebivolol plus lifestyle modification; HDL, high-density lipoprotein cholesterol; LDL, low-density lipoprotein cholesterol; HOMA, Homeostatic Model Assessment; hsCRP, high-sensitivity C-reactive protein; IL-6, interleukin 6; TNF-α, tumor necrosis factor alpha.
p < 0.05 time effect.
p < 0.05 interaction effect.
Total cholesterol, plasma high-density lipoprotein (HDL) cholesterol, triglycerides, and blood glucose concentrations declined during the treatment period (all p < 0.05). However, there were no significant differences in the magnitude of change between the groups. Following the intervention, HOMA index decreased (–0.36 ± 0.33, –1.98 ± 1.43, and −2.12 ± 0.73; p = 0.04) and oxidized LDL changed (–2.5 ± 6.2, 4.7 ± 4.2, and −13.4 ± 5.1 ng/dl; p = 0.048) in the NB, LM, and NBLM groups, respectively. LDL cholesterol, insulin, and other markers of inflammation did not change following the 12-week intervention (all p > 0.05).
β-stiffness and compliance before and after the intervention are shown in Table 4. No baseline differences in stiffness variables were observed in the groups (all p > 0.05). β-stiffness index decreased (–2.03 ± 0.60, –1.87 ± 0.83, and −2.51 ± 0.90 U; p < 0.01) (Figure 1, upper panel) and arterial compliance increased (0.140 ± 0.040, 0.200 ± 0.060, and 0.180 ± 0.060 mm2/mmHg × 10–1; p = 0.02) in the NB, LM, and NBLM groups, respectively (Figure 1, lower panel). The magnitude of changes in β-stiffness index and arterial compliance were not different between groups.
β-stiffness and compliance before (PRE) and following intervention (POST).

Values expressed as ± standard error of mean.
NB, nebivolol; LM, lifestyle modification; NBLM, nebivolol plus lifestyle modification; β-SI, β stiffness index; AC, arterial compliance.
p < 0.05 time effect.
p < 0.05 interaction effect.

Change in β-Stiffness Index (upper panel) and Arterial Compliance (lower panel) with Nebivolol (NB), Lifestyle Modification (LM), and Nebivolol + Lifestyle Modification (NBLM). Values are mean ± SE.
There were no significant correlates of the change in β-stiffness and compliance following the interventions.
Discussion
The major finding of the present study was the combination of nebivolol and lifestyle modification reduced large arterial stiffness and central BP to a similar degree as either intervention alone in middle-aged and older adult hypertensives. However, the combination of nebivolol and lifestyle modification were associated with greater improvement in insulin sensitivity and reductions in oxidized LDL compared with the individual interventions. There was no obvious added benefit of the combined intervention on inflammatory biomarkers.
Reductions in arterial stiffness with nebivolol [Agabiti-Rosei et al. 2009; Dhakam et al. 2008; Soanker et al. 2012] and lifestyle modification [Cooper et al. 2012; Hughes et al. 2012] have been reported previously. Our findings extend previous observations and indicate that the combination of the two interventions are not additive or synergistic in reducing larger artery stiffness, i.e., β-stiffness index. The reductions in β-stiffness with the respective interventions in the present study suggests that these reductions in large artery stiffness were, at least in part, independent of BP lowering.
Despite significant reductions, large artery stiffness remained considerably elevated in the present population compared with healthy younger (i.e. 18–40 years) individuals previously studied in our laboratory [Orr et al. 2009]. As such, there would appear to be significant ‘residual risk’ which remains following the treatment period in each of the groups studied. Future studies will be needed to determine whether large artery stiffness can be reduced beyond that achieved in the present study.
In contrast to earlier generation β-blockers [Jacob et al. 1996], nebivolol has been reported to have a more favorable metabolic profile [Ayers et al. 2012; Bell et al. 2009; Wojciechowski and Papademetriou, 2008]. Nebivolol therapy also appears to reduce markers of oxidative stress [Serg et al. 2012]. In the present study, the combination of nebivolol and lifestyle modification appeared to improve HOMA-IR and reduce oxidized LDL more than the singular interventions. Given the small or absent impact of the lifestyle intervention it would appear that nebivolol might be exerting a large influence on these outcomes. However, future studies will be necessary to confirm this observation.
There are some limitations of our study that should be considered. First, our sample size was small, and the age range of our subjects was confined to 40–75 years. Consequently, we are not able to generalize these results beyond this group. Second, our study was not powered to detect gender and ethnic/racial differences in arterial destiffening. Whether the combination of nebivolol and lifestyle modification might be more efficacious in reducing large arterial stiffness in men and women or different racial/ethnic groups is unknown. Finally, the intervention period was limited to 12 weeks in duration. Whether the reductions in large artery stiffness are sustained over a longer period is not known.
In summary, our findings indicate that the combination of nebivolol and lifestyle modification reduce large artery stiffness to a similar degree as either singular intervention. Future studies are needed to determine if large artery stiffness can be reduced beyond what has been achieved with interventions studied to date.
Footnotes
Acknowledgements
We are grateful to our participants for their time, effort, and commitment to the study.
Funding
This study was supported by Forest Laboratories, Inc. (K.P.D.).
Conflict of interest statement
The authors declare no conflicts of interest in preparing this article.
