Abstract
Biomarkers are being increasingly used in the study of cardiovascular disease because they provide readily quantifiable surrogate endpoints and allow accurate assessment of the effects of therapy on particular pathological processes. However, in order to be useful, biomarkers must be relevant, predictable, accurate, and reproducible. There is compelling evidence from large-scale clinical trials that inhibitors of the renin–angiotensin system [angiotensin-converting enzyme inhibitors and angiotensin type II receptor blockers (ARBs)] and calcium channel blockers (CCBs) may have beneficial effects beyond blood pressure control in the treatment of hypertension. Biomarkers are expected to provide further insight into these beneficial effects and allow for quantitative assessment. This review summarizes the published clinical evidence on the effects of various antihypertensive drugs, particularly ARBs (e.g. losartan and olmesartan medoxomil) and CCBs (e.g. amlodipine), alone and in combination with other agents (e.g. hydrochlorothiazide), on central aortic pressure and the biomarkers high-sensitivity C-reactive protein (hsCRP), adiponectin, cystatin C, homeostasis model assessment of insulin resistance (HOMA-IR), procollagen, tumor necrosis factor-α, and interleukin-6. Of these biomarkers, the benefits of antihypertensive therapy on hsCRP, adiponectin, and HOMA-IR reflect a potential for quantifiable long-term vascular benefits.
Keywords
Introduction
Vascular dysfunction and disease are a leading cause of morbidity and mortality in the United States [Lloyd-Jones et al. 2010]. It is estimated that approximately 80 million US adults (more than one in three) have one or more types of cardiovascular (CV) disease, including hypertension, atherosclerosis, and congestive heart failure, and CV disease has been estimated to account for 34.3% of all deaths in the United States in 2006 [Lloyd-Jones et al. 2010]. Biomarkers are playing an increasing role in the study of CV disease as we attempt to define their function in improving clinical management and outcomes [Maisel, 2009; Packard and Libby, 2008]. By definition, biomarkers are objectively measured indicators of biological processes, and to be of use, they must be relevant, predictable, accurate, and reproducible [Maisel, 2009].
There are many types of biomarkers, and they serve a wide range of purposes in both qualitative and quantitative analysis of disease states. They may be used in the following:
Risk assessment or assessment of the rate of progression of disease. Diagnosis or differential diagnosis. Investigation of disease mechanism(s); selection of the most appropriate intervention for an individual patient (patient stratification). Determining prognosis, and overall morbidity and mortality of the disease state; and monitoring the efficacy of treatment (as a surrogate endpoint for long-term morbidity or mortality outcome).
The benefits of surrogate endpoints (which form the focus of this review) over clinical outcomes in studies and clinical practice (i.e. accuracy, timeliness, specificity, and sensitivity) allow for prompt diagnosis and more timely initiation of appropriate treatment, improved monitoring, and treatment augmentation [Vasan, 2006]. Surrogate endpoint biomarkers of vascular disease are diverse. This review will focus on the following eight biomarkers, which have been used as predictors of vascular outcome in patients with hypertension and those with metabolic syndrome: central aortic pressure (CAP), high-sensitivity C-reactive protein (hsCRP), adiponectin, cystatin C, homeostasis model assessment of insulin resistance (HOMA-IR), procollagen, tumor necrosis factor-α (TNF-α), and interleukin (IL)-6.
Some of the benefits from inhibiting the renin–angiotensin system (RAS) with angiotensin type II receptor blockers (ARBs) and angiotensin-converting enzyme (ACE) inhibitors have been shown to be independent of blood pressure (BP) reduction [Lambers Heerspink et al. 2009; Novo et al. 2009]. Therefore, surrogate biomarkers may provide a viable means of measuring the response to treatment. Anti-inflammatory, antiatherogenic, and/or improved metabolic homeostasis independent of BP lowering seen with some antihypertensives may benefit high-risk patient populations or those who do not achieve adequate BP control. These include ethnic groups such as African Americans, patients with obesity, and patients with renal disease, metabolic syndrome, diabetes mellitus, and/or existing vascular disease.
Improvement in inflammatory and other biomarkers has been reported with ARBs and ACE inhibitors in patients with obesity and metabolic syndrome [Nagamia et al. 2007; Sola et al. 2005] and in patients with hypertension with and without type 2 diabetes mellitus (T2DM) [Derosa et al. 2010; Persson et al. 2006]. Similarly, calcium channel blockers (CCBs) have been shown to improve markers of inflammation in patients with hypertension [Komoda et al. 2010; Shurtz-Swirski et al. 2006], while β-blockers such as nebivolol were shown to modify markers of inflammation and obesity in African Americans with obesity and hypertension [Merchant et al. 2010]. As a class, ARBs are known to have anti-inflammatory properties, which may contribute to their pharmacological effects. Biomarker studies in patients with hypertension have demonstrated the effect of ARBs on inflammatory and other biomarkers [Del Fiorentino et al. 2009; Sola et al. 2005], including CAP [Karalliedde et al. 2008; Polonia et al. 2003], hsCRP [Del Fiorentino et al. 2009; Futai et al. 2009; Bloch, 2007; Ridker et al. 2006], adiponectin [Moriuchi et al. 2007], cystatin [Watanabe et al. 2006a; Schepke et al. 2001], HOMA-IR [De Luis et al. 2010; Derosa et al. 2007; Usui et al. 2007], procollagen [Kawamura et al. 2010; Muller-Brunotte et al. 2007; Christensen et al. 2006], TNF-α [Schulz and Heusch, 2009; Fogari et al. 2006; Fujita et al. 2006], and IL-6 [Pai et al. 2004; Cesari et al. 2003; Luc et al. 2003].
This review summarizes the role of biomarkers as surrogate endpoints in the treatment of patients with hypertension and discusses the evidence for the effects of ARBs and other antihypertensive drugs on biomarkers and their correlation with clinical efficacy. The source material for this review was derived from a MEDLINE literature search, performed from 1999 to July 2010, to identify published studies investigating the use of selective antihypertensive agents using at least one of the eight previously mentioned biomarkers. The agents specified in the search were amlodipine, olmesartan medoxomil, combination amlodipine plus olmesartan medoxomil, losartan, hydrochlorothiazide (HCTZ), and combination losartan plus HCTZ.
Role of biomarkers and mechanisms in specific patient groups
Central aortic pressure
While brachial BP is easily measured and has been shown to be predictive of CV morbidity and mortality, it is an imperfect surrogate measure of CAP. Peripheral systolic BP (SBP) measured at the brachial artery does not accurately reflect CAP, particularly in youths, as a result of peripheral amplification of the pulse pressure wave [Nelson et al. 2010; Williams and Lacy, 2009; Wang et al. 2008]. This amplification difference decreases with increasing age or increasing arterial stiffness. Central aortic systolic pressure or pulse pressure has been shown to be a powerful and more robust predictor of end organ damage and CV mortality than brachial BP, irrespective of whether the central pressures were derived from noninvasive pulse wave analysis or measured directly during routine catheterization [Williams and Lacy, 2009]. Although techniques have been developed to a stage where direct noninvasive measurement of CAP could be readily applied to routine clinical practice, the studies conducted to date assessing the predictive value of CAP have been relatively small. Thus, additional data from large interventional studies of clinical outcomes and larger-scale longitudinal epidemiological studies of clinical outcomes are required to confirm the utility of CAP as a predictor of vascular risk before it becomes part of routine clinical practice [Nelson et al. 2010; Williams and Lacy, 2009; Wang et al. 2008].
Noninvasive assessment of the central pulse pressure waveform is performed by applanation tonometry, which involves applying a pressure sensor (tonometer) with mild pressure over the radial or carotid artery. The recorded waveform is then used to algorithmically derive central pressure indices from a peripheral brachial BP measurement [Nelson et al. 2010]. In addition, aortic pulse wave velocity (PWV), which is usually recorded between the carotid and femoral arteries, is used to determine vessel distensibility; the velocity of the pulse wave increases with decreasing vessel distensibility (increased arterial stiffness) [Wang et al. 2008]. Increased PWV (arterial stiffness) appears to be associated with endothelial dysfunction, inflammation, and atherosclerosis, and has been shown to be an independent predictor of coronary events and CV mortality [Wang et al. 2008]. Hence, arterial stiffness is an emerging biomarker for therapeutic interventions.
High-sensitivity C-reactive protein
C-reactive protein (CRP), an acute phase reactant predominantly produced in the liver in response to IL-6, IL-1β, and TNF-α, is a marker of the general inflammatory response. Epidemiological studies have established that individuals with higher levels of CRP, as determined with the high-sensitivity assay, have increased CV risk [Paoletti et al. 2004]. The nature of the relationship between hsCRP and CV disease is unclear, but hsCRP provides a useful measure of risk and the effects of interventions [Black et al. 2004; Paoletti et al. 2004].
Studies have shown a continuous independent association between serum CRP levels and elevated BP [Bautista et al. 2004]. In elderly people who are normotensive, higher baseline CRP levels were associated with a higher incidence of new-onset hypertension after 2 years [Dauphinot et al. 2009]. Moreover, in apparently healthy adults representative of the US population, a 10-mmHg increase in pulse pressure was associated with significant increases of 12–15% in the odds of having an elevated CRP level, independent of SBP or diastolic BP (DBP), or demographic factors [Abramson et al. 2002]. There are even suggestions that hsCRP may be a better marker of coronary artery disease (CAD) than low-density lipoprotein cholesterol (LDL-C) [Packard and Libby, 2008; Genest, 2004]. Evidence suggests that inhibition of the RAS with certain agents, particularly ARBs and ACE inhibitors, may improve CV outcomes by reducing vascular inflammation and remodeling independently of BP reductions [Savoia and Schiffrin, 2007].
Adiponectin
Adiponectin is one of the adipocyte-derived hormones that has profound anti-inflammatory and antiatherogenic properties, which is also thought to play an important role in the modulation of glucose and lipid metabolism [Montecucco and Mach, 2009; Packard and Libby, 2008]. Reduced adiponectin levels have been noted in men, people with obesity, and patients with hypertension, CAD, or T2DM [Karthikeyan and Lip, 2007]; they have been shown to be predictive of CAD and myocardial infarction (MI) [Shargorodsky et al. 2009]. Animal studies have shown that increased adiponectin levels are protective against atherosclerosis, while clinical studies with antihypertensive drugs, including ARBs, ACE inhibitors, and CCBs, have associated improvements in BP and insulin resistance with increased adiponectin levels [Karthikeyan and Lip, 2007; Makita et al. 2007]. In people with obesity, serum adiponectin levels were inversely associated with intima-media thickness, a surrogate measure of subclinical atherosclerosis, and positively associated with arterial compliance [Shargorodsky et al. 2009]. The mechanism behind the beneficial effects of adiponectin is uncertain; one hypothesis suggests that adiponectin increases nitric oxide activity, thereby inhibiting platelet activation, while another hypothesis suggests it suppresses monocyte activation [Karthikeyan and Lip, 2007].
Cystatin C
The serum cystatin C level directly correlates with the glomerular filtration rate (GFR) and is produced constantly, independent of muscle mass, age, or sex. It is therefore an easily obtained biomarker for renal dysfunction that may be more reliable than measurement of creatinine levels in certain patient populations, particularly in children [Massey, 2004; Laterza et al. 2002; Ylinen et al. 1999]. However, there are concerns over the cost of the immunoassay, intraindividual variability, and its sensitivity in patients after a transplant or its suitability in patients with cancer, in whom cystatin C production may vary [Laterza et al. 2002]. Notably, a study in patients after heart transplantation found that cystatin C was superior to creatinine as a prognostic indicator of early renal dysfunction during 4 years of follow up [Kniepeiss et al. 2010].
Cystatin C is a predictor of CV morbidity and mortality, and it has been suggested that this association may be independent of renal function [Mena et al. 2010; Prats et al. 2010]. In one study, cystatin C, but not creatinine or GFR, was closely associated with left ventricular (LV) mass in patients with hypertension, suggesting utility as a marker for cardiac hypertrophy [Prats et al. 2010].
Homeostasis model assessment of insulin resistance
HOMA-IR is a mathematical model prediction that provides an accurate quantitative assessment of insulin resistance [Matthews et al. 1985], which is associated with hypertension, obesity, and diabetes, and an increased risk of CAD [Cooper-Dehoff et al. 2009].
Many CV drugs adversely affect glucose and lipid homeostasis, and insulin resistance is an important mediator of these adverse effects on glucose metabolism [Cooper-Dehoff et al. 2009]. Direct RAS inhibitors (ARBs and ACE inhibitors) and some other antihypertensives provide beneficial effects in terms of glucose homeostasis [Cooper-Dehoff et al. 2009].
Procollagen
Collagen fractions in the extracellular matrix are intimately involved in the atherosclerotic process and the vascular remodeling that occurs in CV disease [Szmigielski et al. 2006]. There is evidence that altered collagen metabolism (e.g. elevated serum levels of tissue inhibitor of metalloproteinase-1) is associated with hypertension [Szmigielski et al. 2006], and that plasma markers of collagen metabolism are positively correlated with arterial stiffness measured by PWV in patients with hypertension and LV hypertrophy [Ishikawa et al. 2005].
Therefore, measurement of serum procollagen fractions as indicators of myocardial fibrosis may be useful in the clinical assessment of CV risk [López et al. 2005].
Tumor necrosis factor-α
TNF-α is a marker of inflammation and is believed to promote the development of insulin resistance and hyperinsulinemia, and thereby affect BP [Fogari et al. 2006]. TNF-α is released from mast cells and macrophages in the myocardial endothelium during acute myocardial ischemia, and from cardiomyocytes during persistent ischemia. The released TNF-α contributes to ischemic and/or reperfusion injury and is believed to contribute to cardiac contractile dysfunction after MI via a local inflammatory reaction [Schulz and Heusch, 2009]. Surprisingly, low levels of TNF-α may be beneficial and display a cardioprotective effect, reducing infarct size [Schulz and Heusch, 2009]. TNF-α is also believed to play a role in the development of atherosclerosis by upregulating cell surface receptors for advanced glycation end products that promote the release of inflammatory mediators in the endothelium [Fujita et al. 2006]. The differential effects are possibly related to which of the two receptor types (TNF-R1 or TNF-R2) the TNF-α molecule interacts with.
Interleukin-6
IL-6 is an inflammatory cytokine that, along with TNF-α, is one of the main inducers of acute phase reactants, such as CRP. It has been positively correlated with CV risk. For instance, in elderly people without known CV disease, serum levels of IL-6 were significantly associated with CAD, stroke, and congestive heart failure events, and to a greater extent than CRP or TNF-α levels [Cesari et al. 2003]. Similarly, in older men without CAD, IL-6 was found to be more discriminating than CRP and fibrinogen in predicting a first coronary artery ischemic event, being associated with MI/coronary death but not CAD endpoints (angina) [Luc et al. 2003]. However, not all studies have found strong correlations between IL-6 and CAD [Pai et al. 2004].
Clinical evidence for impact of antihypertensive treatment on biomarkers
Summary of clinical studies of antihypertensive treatment utilizing the biomarker central aortic pressure (CAP) as a surrogate endpoint.

ABP, ambulatory BP; ACEI, angiotensin-converting enzyme inhibitor; AIx, augmentation index (the proportional increase in systolic pressure to which the left ventricle is subjected due to the reflected wave from the periphery); AML, amlodipine; Ao-PWV, aortic pulse wave velocity; ASCOT, Anglo-Scandinavian Cardiac Outcomes Trial; ATE, atenolol; AZ, azelnidipine; BB, β-blocker; BIS, bisoprolol; BNP, brain natriuretic peptide; BP, blood pressure; CADP, central aortic diastolic pressure; CAFE, Conduit Artery Function Evaluation study; CAPP, central aortic pulse pressure; CASP, central aortic systolic pressures; CCB, calcium channel blocker; CV, cardiovascular; FOS, fosinopril; HCTZ, hydrochlorothiazide; HTN, hypertension; IDP, indapamide; ISH, isolated systolic HTN; LIS, lisinopril; LOS, losartan; OM, olmesartan medoxomil; PBO, placebo; PER, perindopril; PP, pulse pressure; PWA, pulse wave analysis; PWV, pulse wave velocity; RAAT, radial artery applanation tonometry; SBP, systolic blood pressure; SeBP, seated BP; T2DM, type 2 diabetes mellitus; TR, time for transmission of the reflected wave; VAL, valsartan.
Summary of clinical studies of antihypertensive treatment utilizing the biomarker high-sensitivity C-reactive protein (hsCRP) as a surrogate endpoint.

AIx, augmentation index (the proportional increase in systolic pressure to which the left ventricle is subjected due to the reflected wave from the periphery); AML, amlodipine; ARB, angiotensin type II receptor blocker; ATO, atorvastatin; AZ, azelnidipine; BP, blood pressure; CAD, coronary artery disease; CAN, candesartan; CKD, chronic kidney disease; CRP, C-reactive protein; CV, cardiovascular; DMA, dimethylarginine; ENA, enalapril; EUTOPIA, European Trial on Olmesartan and Pravastatin in Inflammation and Atherosclerosis; HCTZ, hydrochlorothiazide; HDL-C, high-density lipoprotein cholesterol; HOMA-IR, homeostasis model assessment of insulin resistance; HTN, hypertension; ICAM-1, inter-cellular adhesion molecule 1; IL, Interleukin; LDL-C, low-density lipoprotein cholesterol; LOS, losartan; LV, left ventricle; LVMI, left ventricular mass index; MARIMBA, Manidipine versus Amlodipine in Reducing Insulin Resistance in Metabolic Syndrome Patients; MetS, metabolic syndrome; OLAS, the effects of olmesartan/amlodipine on metabolic and inflammation mediators in hypertensive patients with metabolic syndrome; OM, olmesartan medoxomil; PBO, placebo; PRA, pravastatin; RAM, ramipril; RAS, renin–angiotensin system; SBP, systolic BP; SIM, simvastatin; T2DM, type 2 diabetes mellitus; TCM, trichlormethiazide; TNF, tumor necrosis factor; VAL, valsartan; VCAM-1, vascular cell adhesion molecule 1; YKL-40, chitinase-3-1-protein.
Summary of clinical studies of antihypertensive treatment utilizing the biomarker adiponectin as a surrogate endpoint.

ACR, albumin:creatinine ratio; AML, amlodipine; ANOVA, analysis of variance; ARB, angiotensin type II receptor blocker; ATE, atenolol; AT-HEC, after treatment hyperinsulinemic euglycemic clamp; AZ, azelnidipine; BMI, body mass index; BP, blood pressure; CAN, candesartan; CCB, calcium channel blocker; CETP, cholesteryl ester transfer protein; CKD, chronic kidney disease; CV, cardiovascular; DBP, diastolic BP; eGFR, estimated glomerular filtration rate; ELISA, enzyme-linked immunosorbent assay; ENA, enalapril; FPI, fasting plasma insulin; HCTZ, hydrochlorothiazide; HDL-C, high-density lipoprotein cholesterol; HEC, hyperinsulinemic euglycemic clamp; HOMA-IR, homeostasis model assessment of insulin resistance; hsCRP, high-sensitivity C-reactive protein; HTN, hypertension; IDP, indapamide; IR, insulin resistance; LDL-C, low-density lipoprotein cholesterol; LOS, losartan; MARIMBA, Manidipine versus Amlodipine in Reducing Insulin Resistance in Metabolic Syndrome Patients; MET, metoprolol; MetS, metabolic syndrome; MW, molecular weight; OM, olmesartan medoxomil; PAI-1, plasminogen activator inhibitor-1; RAM, ramipril; RIA, radioimmunoassay; RSG, rosiglitazone; SBP, systolic BP; SCD-1, stearoyl-CoA desaturase 1; SIM, simvastatin; T1DM, type 1 diabetes mellitus; T2DM, type 2 diabetes mellitus; TC, total cholesterol; TEL, telmisartan; TG, triglyceride; TNF, tumor necrosis factor; VAL, valsartan.
Summary of clinical studies of antihypertensive treatment utilizing the biomarker cystatin c as a surrogate endpoint.
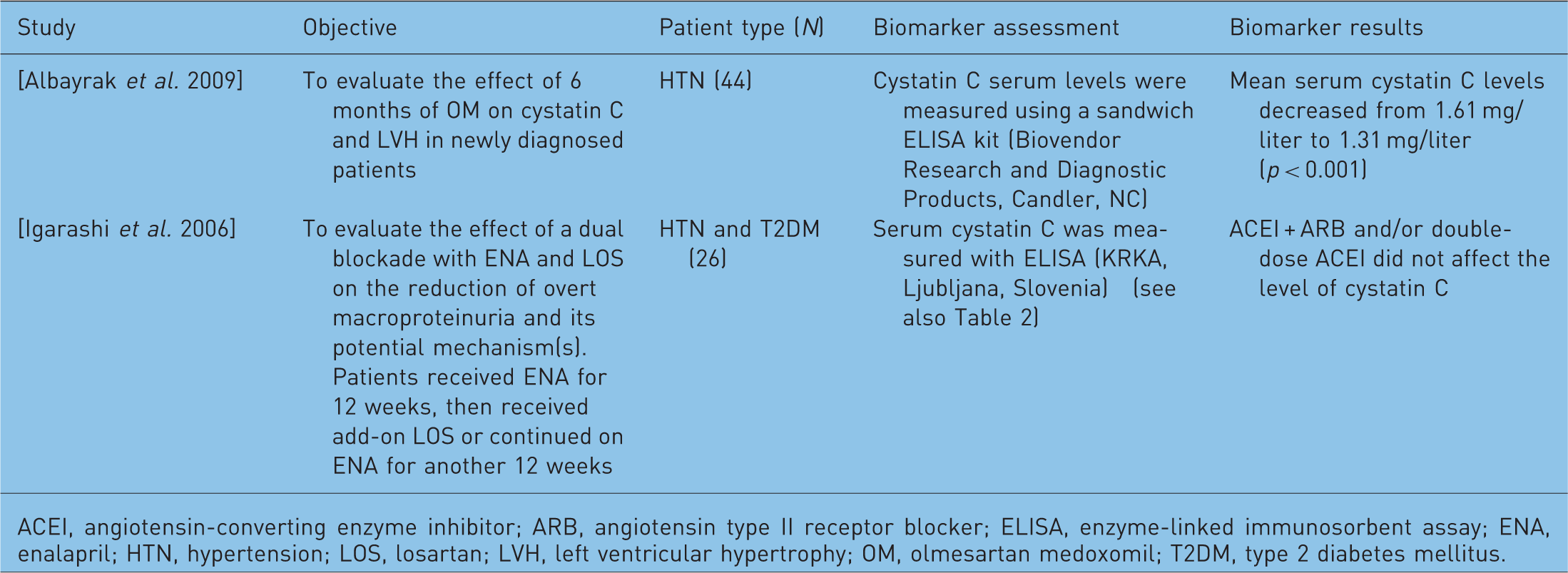
ACEI, angiotensin-converting enzyme inhibitor; ARB, angiotensin type II receptor blocker; ELISA, enzyme-linked immunosorbent assay; ENA, enalapril; HTN, hypertension; LOS, losartan; LVH, left ventricular hypertrophy; OM, olmesartan medoxomil; T2DM, type 2 diabetes mellitus.
Summary of clinical studies of antihypertensive treatment utilizing the homeostasis model assessment of insulin resistance (HOMA-IR) as a surrogate biomarker endpoint.

ACR, albumin:creatinine ratio; ALT, alanine aminotransferase; AML, amlodipine; ARB, angiotensin type II receptor blocker; ATO, atorvastatin; AZ, azelnidipine; BP, blood pressure; CAN, candesartan; CCB, calcium channel blocker; CHC, chronic hepatitis C; CIGMA, continuous infusion of glucose with model assessment; CKD, chronic kidney disease; CRP, C-reactive protein; FPG, fasting plasma glucose; FPI, fasting plasma insulin; HbA1c, glycosylated hemoglobin; HCTZ, hydrochlorothiazide; HDL-C, high-density lipoprotein cholesterol; hsCRP, high-sensitivity C-reactive protein; HTN, hypertension; IDP, indapamide; IFG, impaired fasting glucose; IGF-I, insulin-like growth factor-I; IGFBP-3, insulin-like growth factor binding protein-3; IL, interleukin; IR, insulin resistance; LDL-C, low-density lipoprotein cholesterol; LIS, lisinopril; LOS, losartan; LVH, left ventricular hypertrophy; LVMI, left ventricular mass index; MARIMBA, Manidipine versus Amlodipine in Reducing Insulin Resistance in Metabolic Syndrome Patients; MET, metoprolol; MetS, metabolic syndrome; NAFLD, nonalcoholic fatty liver disease; OM, olmesartan medoxomil; RAM, ramipril; RSG, rosiglitazone; TC, total cholesterol; TEL, telmisartan; TG, triglyceride; TNF, tumor necrosis factor; VAL, valsartan; YKL-40, chitinase-3-1-protein.
Summary of clinical studies of antihypertensive treatment utilizing the biomarker procollagen as a surrogate endpoint.

AML, amlodipine; BP, blood pressure; CAN, candesartan; CITP, collagen type 1 carboxy-terminal telopeptide; CV, cardiovascular; CVF, collagen volume fraction; DBP, diastolic BP; ESRD, end-stage renal disease; HTN, hypertension; IBS, ultrasonic integrated backscatter; LOS, losartan; LVH, left ventricular hypertrophy; LVM, left ventricular mass; PBO, placebo; PICP, procollagen type I carboxyterminal propeptide; PIIINP, procollagen type III aminoterminal propeptide; PIIIP, procollagen type III; PINP, procollagen type I aminoterminal propeptide; PIP, procollagen type 1; RAM, ramipril; RIA, radioimmunoassay; RP, Reynaud’s phenomenon; SBP, systolic BP; SSc, systemic sclerosis; TGF, circulating transforming growth factor; VAL, valsartan.
Summary of clinical studies of antihypertensive treatment utilizing the biomarker tumor necrosis factor-α (TNF-α) as a surrogate endpoint.

ACR, albumin:creatinine ratio; AML, amlodipine; ARB, angiotensin type II receptor blocker; ATO, atorvastatin; BMI, body mass index; BP, blood pressure; CCB, calcium channel blocker; CKD, chronic kidney disease; CV, cardiovascular; DBP, diastolic BP; ELISA, enzyme-linked immunosorbent assay; HCTZ, hydrochlorothiazide; HDL-C, high-density lipoprotein cholesterol; HOMA-IR, homeostasis model assessment of insulin resistance; hsCRP, high-sensitivity C-reactive protein; HTN, hypertension; IR, insulin resistance; LDL-C, low-density lipoprotein cholesterol; LOS, losartan; MARIMBA, Manidipine versus Amlodipine in Reducing Insulin Resistance in Metabolic Syndrome Patients; MetS, metabolic syndrome; mo, OM, olmesartan medoxomil; PAI-1, plasminogen activator inhibitor-1; PRA, pravastatin; SBP, systolic BP; T2DM, type 2 diabetes mellitus; TC, total cholesterol; TEL, telmisartan; TG, triglyceride; VAL, valsartan.
Summary of clinical studies of antihypertensive treatment utilizing the biomarker interleukin-6 (IL-6) as a surrogate endpoint.
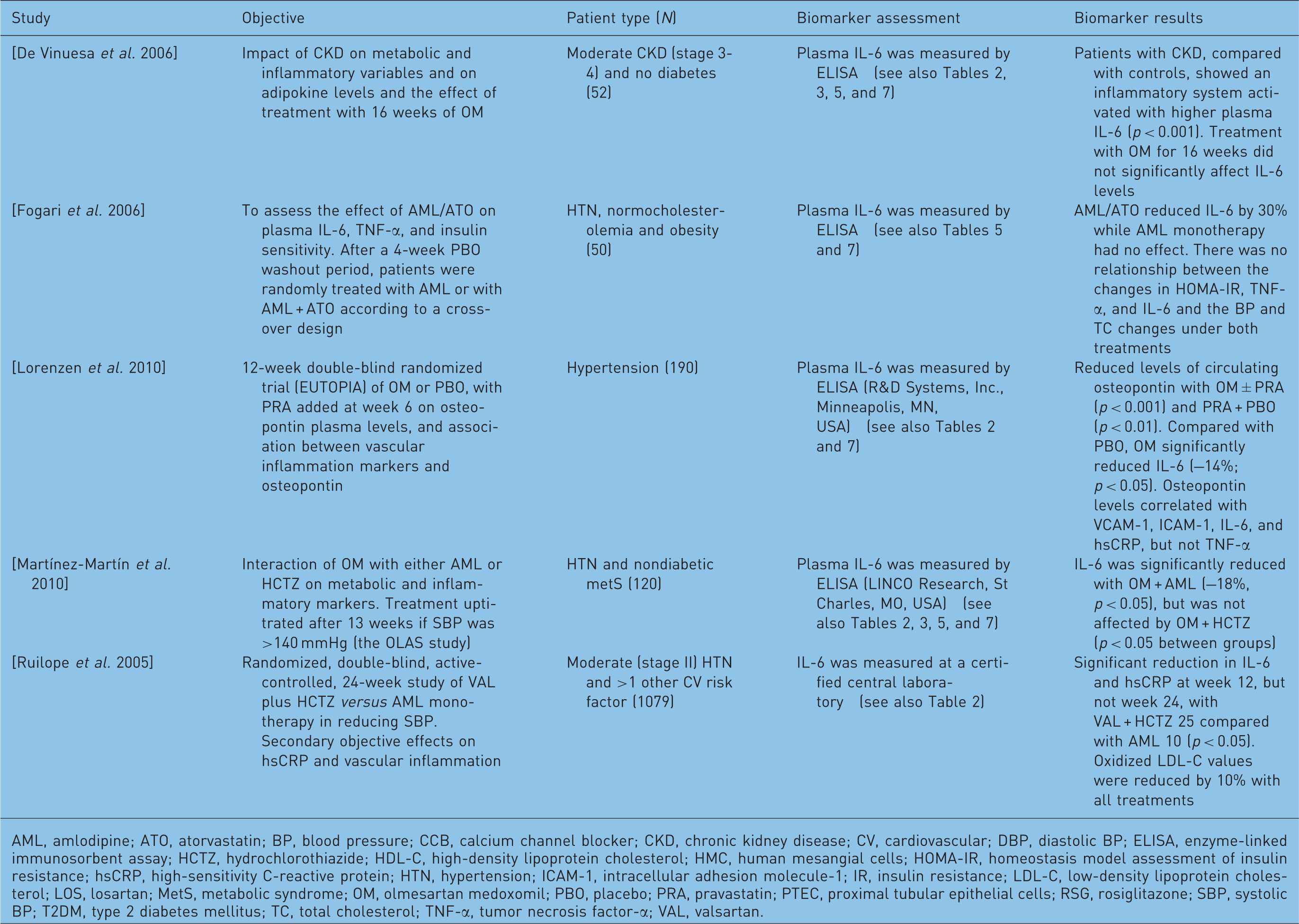
AML, amlodipine; ATO, atorvastatin; BP, blood pressure; CCB, calcium channel blocker; CKD, chronic kidney disease; CV, cardiovascular; DBP, diastolic BP; ELISA, enzyme-linked immunosorbent assay; HCTZ, hydrochlorothiazide; HDL-C, high-density lipoprotein cholesterol; HMC, human mesangial cells; HOMA-IR, homeostasis model assessment of insulin resistance; hsCRP, high-sensitivity C-reactive protein; HTN, hypertension; ICAM-1, intracellular adhesion molecule-1; IR, insulin resistance; LDL-C, low-density lipoprotein cholesterol; LOS, losartan; MetS, metabolic syndrome; OM, olmesartan medoxomil; PBO, placebo; PRA, pravastatin; PTEC, proximal tubular epithelial cells; RSG, rosiglitazone; SBP, systolic BP; T2DM, type 2 diabetes mellitus; TC, total cholesterol; TNF-α, tumor necrosis factor-α; VAL, valsartan.
Central aortic pressure
Of the various antihypertensive drug classes, RAS inhibitors (ARBs and ACE inhibitors) and CCBs generally appear to have greater effects on CAP than β-blockers and thiazide diuretics (Table 1). Despite similar brachial BP reductions, the combination of amlodipine plus perindopril was associated with greater reductions in CAP than atenolol plus a thiazide diuretic [Williams et al. 2006]. Lisinopril also significantly reduced central SBP, central pulse pressure, and the augmentation index, while bisoprolol only significantly lowered central DBP and actually increased the augmentation index [Neal et al. 2004]. Another study showed significant reductions in both brachial and central BP reductions with different drug classes in the following order: CCBs > diuretics (HCTZ) > ACE inhibitors [Morgan et al. 2004]. β-blockers did not significantly lower peripheral or central BP.
Similar brachial BP and CAP reductions were achieved with valsartan plus HCTZ versus amlodipine; however, valsartan plus HCTZ provided a greater reduction in arterial stiffness (estimated by aortic PWV) [Karalliedde et al. 2008]. Reductions in central SBP were greater with fosinopril plus HCTZ than with indapamide or amlodipine; this correlated with 24-h and night-time SBP reductions, but not with seated cuff SBP [Ferguson et al. 2008].
High-sensitivity C-reactive protein
Several antihypertensive drug classes, such as ARBs, ACE inhibitors, and CCBs, lower serum hsCRP in addition to BP, indicating a reduction in the inflammatory processes involved in the progression of atherosclerosis (Table 2). ARBs, in particular, seem to have a strong depressor effect on this marker of inflammation. Patients with chronic kidney disease (CKD), who have higher baseline levels of inflammation than controls with normal renal function, displayed significant reductions in hsCRP and brachial BP with olmesartan medoxomil treatment [De Vinuesa et al. 2006]. In a small study of 10 patients with mild to moderate hypertension, olmesartan medoxomil did not reduce BP significantly, but did produce significant reductions in hsCRP and appeared to improve myocardial function independent of BP lowering [Futai et al. 2009]. In patients without diabetes but with hypertension and the metabolic syndrome, olmesartan medoxomil plus amlodipine and olmesartan medoxomil plus HCTZ effectively reduced BP and CRP with no differences between groups. However, olmesartan plus amlodipine produced greater reductions in all other inflammatory markers [Martínez-Martín et al. 2010].
In a study comparing the CCB azelnidipine or the thiazide diuretic trichlormethiazide added to an ARB, the ARB plus azelnidipine combination produced significantly greater reductions in hsCRP than the ARB plus thiazide combination; this reduction mirrored the BP-lowering effects [Doi et al. 2010]. Similar data were shown in a 4-month crossover study comparing olmesartan medoxomil plus azelnidipine or trichlormethiazide [Ishimitsu et al. 2009]. Comparison of 12 weeks of combination therapy with enalapril plus add-on losartan with higher dose enalapril monotherapy showed a significant reduction in hsCRP with combination therapy, but not with high-dose enalapril alone; BP reductions were significant and similar in both groups [Igarashi et al. 2006].
Evidence suggests that ARBs may differ in their anti-inflammatory effects. For instance, in patients with CAD, olmesartan medoxomil and valsartan both produced significant reductions in BP, but only olmesartan medoxomil induced a significant reduction in hsCRP [Sugihara et al. 2009]. Studies comparing the hsCRP-lowering effects of ARBs and CCBs have shown variable results. One study found no difference in hsCRP reductions after 8 weeks of therapy with losartan or amlodipine regimens [Aksnes et al. 2007]. The effects on hsCRP and other inflammatory markers did not explain the greater improvements in insulin sensitivity seen with ARBs over CCBs. However, in patients with hypertension and other CV risk factors, therapy with valsartan plus HCTZ was significantly more effective than amlodipine in reducing hsCRP. These biomarker results correlated with BP reductions [Ruilope et al. 2005]. hsCRP improvement did not correlate with endothelial function in a study comparing candesartan with amlodipine; both treatments significantly improved endothelial function (assessed by changes in forearm blood flow in reactive hyperemia), whereas significant reductions in hsCRP levels were seen only with candesartan and not amlodipine therapy [Tomiyama et al. 2007]. The study investigators concluded that the anti-inflammatory effects observed with candesartan may be related to observed improvement in insulin sensitivity. In a study of patients with CAD, treatment with irbesartan did not lower hsCRP levels [Navalkar et al. 2001]. The lack of effect of irbesartan may have been due to low levels of hsCRP at study baseline. Patients were also receiving statin and aspirin therapy, which lower levels of this marker.
As with ARBs, CCBs seem to differ in their ability to reduce inflammatory markers. In patients with hypertension and the metabolic syndrome, similar significant reductions in hsCRP and BP were seen with manidipine and amlodipine, but these data did not correlate with changes in other biomarkers, such as adiponectin, HOMA-IR, and TNF-α, which showed greater improvements with manidipine than with amlodipine [Martínez Martín, 2009]. In a different study in patients with arterial hypertension and insulin resistance who were already receiving at least two antihypertensive agents, neither moxonidine nor amlodipine showed significant changes in hsCRP, whereas both treatments resulted in significant BP lowering [Masajtis-Zagajewska et al. 2010].
Adding an HMG-CoA reductase inhibitor (atorvastatin) to amlodipine therapy produced significantly greater reductions in BP and hsCRP than was seen with amlodipine alone [Ge et al. 2008], but losartan plus simvastatin achieved similar reductions in hsCRP compared with losartan or simvastatin alone [Koh et al. 2004]. BP reductions were significantly greater with losartan or losartan plus simvastatin than with simvastatin alone.
These observations suggest that RAS antagonists such as ARBs and ACE inhibitors have a significant anti-inflammatory effect, and there may be variations within these classes. As diseases such as diabetes mellitus and atherosclerosis are inflammatory processes, the clinical benefits seen with these classes of antihypertensives may be a combination of the suppression of inflammation and the reduction of BP.
Adiponectin
Studies assessing the effect of the selected antihypertensive drugs on the serum levels of adiponectin are shown in Table 3. Antihypertensive agents do not uniformly influence metabolic parameters in patients with hypertension. In a comparison of telmisartan and irbesartan in patients with obesity, insulin resistance, and hypertension, increases were significantly greater with telmisartan, although both treatments resulted in significant increases in adiponectin levels [Negro et al. 2006]. Adiponectin changes correlated inversely with changes in BP for telmisartan, but not for irbesartan. The investigators speculate that the differences between the two agents may be partly due to partial peroxisome proliferator-activated receptor-γ (PPAR-γ) agonist activity exhibited by telmisartan, although a study by Kintscher and colleagues in 14,200 patients confirmed that irbesartan also activates PPAR-γ [Kintscher et al. 2007]. Despite similar reductions in BP, olmesartan medoxomil plus amlodipine produced significant increases in adiponectin levels in patients with hypertension and the metabolic syndrome, whereas olmesartan medoxomil plus HCTZ did not [Martínez-Martín et al. 2010]. The increase in adiponectin correlated with a lower risk for developing T2DM and paralleled reductions (improvements) in HOMA-IR index and fasting plasma insulin levels.
In patients without diabetes but with proteinuria, treatment with losartan plus HCTZ reduced BP, proteinuria, and LDL-C, and increased adiponectin, but the change in adiponectin correlated with adverse reductions in high-density lipoprotein cholesterol (HDL-C) levels [Krikken et al. 2009]. In a crossover study that investigated possible factors to explain improvements in insulin sensitivity with ARB therapy compared with CCB therapy in patients with hypertension, no between-group differences were noted for increases in adiponectin levels or reductions in BP after 8 weeks of therapy with losartan or amlodipine [Aksnes et al. 2007]. In contrast, although both telmisartan and amlodipine increased adiponectin levels in patients with hypertension and T2DM, the increases were higher with telmisartan than with amlodipine [Negro et al. 2006]. Both groups showed a similar significant decrease in BP. Similarly, in patients with prediabetes, losartan produced greater increases in adiponectin than a CCB, whereas BP reduction was similar and significant in both groups [Nishimura et al. 2008].
ARBs and statins have additive effects on adiponectin. Losartan plus simvastatin or losartan alone resulted in significantly greater increases in adiponectin levels from baseline than with simvastatin alone [Koh et al. 2004]. This correlated with BP reductions, which were greater with losartan or losartan plus simvastatin than with simvastatin alone. A correlation was also observed with LDL reductions, which were greater with simvastatin or simvastatin with losartan relative to losartan alone.
There were no changes in adiponectin levels with the aldosterone blocker spironolactone or the CCB amlodipine in patients with diabetic nephropathy or in controls. However, spironolactone, but not amlodipine, increased adiponectin in a subgroup of patients with poor baseline glycemic control [glycosylated hemoglobin (HbA1c) ≥ 8%]. A significant decrease in SBP, but not DBP, was observed in both treatment groups [Matsumoto et al. 2006]. This link between the rennin–angiotensin cascade and aldosterone would suggest a possible mechanism by which spironolactone provides an increased level of adiponectin in hyperglycemia.
In a comparison of enalapril, metoprolol, amlodipine, and indapamide, no changes in adiponectin level were seen with enalapril, amlodipine, or metoprolol, whereas a reduction in adiponectin was seen with indapamide. This reduction in adiponectin with the thiazide-like diuretic correlated with increased insulin resistance [Piecha et al. 2007]. In a comparison of metoprolol, amlodipine, ramipril, doxazosin, and valsartan in patients with hypertension and the metabolic syndrome, both ramipril and valsartan resulted in significantly higher increases in adiponectin than the other regimens; adiponectin levels inversely correlated with SBP [Yilmaz et al. 2007].
In a small study in patients with hypertension, ramipril, candesartan, and amlodipine were associated with greater increases in adiponectin levels while thiazide and atenolol were associated with a decrease in adiponectin. There were no correlations with BP lowering, which was greatest with atenolol, amlodipine, and candesartan therapies than with ramipril (p < 0.05) [Koh et al. 2010].
Unlike the situation in patients with hypertension, obesity, or diabetes, in whom adiponectin levels are reportedly reduced, the levels of adiponectin are raised in patients with renal disease when compared with healthy controls. Thus, in patients with renal disease, a positive correlation between adiponectin and insulin resistance is seen, and increased adiponectin levels are associated with increased all-cause and CV mortality (the opposite of that seen in patients with obesity or those with T2DM without renal disease). Paradoxically, short-term losartan therapy in patients with T2DM nephropathy was associated with a significant decrease in adiponectin levels compared with amlodipine therapy [Guo et al. 2009].
Adiponectin, secreted by fat cells, regulates the insulin response and has a favorable effect on glucose and lipid metabolism. Insulin resistance is a hallmark for the progression of vascular disease. The quantitative changes in adiponectin provide insight into how antihypertensive agents such as ARBs may be effective in attenuating or reversing the pathogenesis of atherosclerosis and diabetes mellitus.
Cystatin C
There were two studies from the MEDLINE literature search that assessed the effect of the selected antihypertensive drugs on serum cystatin C levels (Table 4). In one study, a significant decrease in cystatin C with olmesartan medoxomil therapy correlated with improvements in BP, LV mass index, and LV hypertrophy at 6 months [Albayrak et al. 2009]. However, another study found no significant decrease in cystatin C with enalapril/losartan combination therapy or with high-dose enalapril, despite significant reductions in BP [Igarashi et al. 2006].
The use of cystatin C as an early marker for CKD may be helpful in longitudinal follow-up analyses. The findings in the above studies are preliminary but suggest that BP reduction may be associated with lower cystatin C levels. It is too early to determine whether inhibition of the RAS (in the form of ACE inhibitors or ARBs) may have an effect on cystatin C that is superior to other antihypertensive drugs.
Homeostasis model assessment of insulin resistance
HOMA-IR is a model and calculation to determine quantification of insulin resistance. Antihypertensive drugs appear to have differing effects on insulin resistance, with ARBs foremost among those improving insulin sensitivity, although considerable variability has been observed and not all ARBs may be equal in this regard (Table 5). RAS inhibitors generally have greater effects on glucose homeostasis than CCBs, which are usually considered to have neutral effects.
In patients with hypertension, a significantly greater reduction in HOMA-IR was seen with losartan/amlodipine therapy than with high-dose amlodipine [Aksnes et al. 2007]. In contrast to previous results, both losartan and telmisartan had neutral effects on insulin resistance in 42 patients with hypertension and the metabolic syndrome, with no significant reductions in HOMA-IR in either group; BP reductions were similar for both ARBs [Bahadir et al. 2007].
Patients with CKD and no diabetes had a high prevalence of insulin resistance, metabolic syndrome, and chronic inflammation. Treatment with olmesartan medoxomil for 16 weeks was associated with a significant reduction from baseline in HOMA-IR, along with reductions in markers of inflammation (see Tables 2 and 3) [De Vinuesa et al. 2006]. Losartan therapy was associated with improvements in fasting plasma insulin and HOMA-IR in patients with T2DM nephropathy, in parallel with reductions in adiponectin levels [Guo et al. 2009]. Both olmesartan medoxomil and telmisartan were shown to improve HOMA-IR in patients with nonalcoholic fatty liver disease and chronic hepatitis C, conditions with a greater incidence of insulin resistance than other liver diseases [Enjoji et al. 2008].
In a study investigating the effect of combination therapy with amlodipine plus olmesartan medoxomil on HOMA-IR in patients with hypertension and the metabolic syndrome, HOMA-IR was significantly reduced with olmesartan medoxomil/amlodipine (p < 0.01), whereas no significant changes were seen with olmesartan medoxomil/HCTZ. The reductions in the HOMA-IR index strongly correlated (p < 0.001) with the increases in adiponectin level in the group treated with olmesartan medoxomil/amlodipine [Martínez-Martín et al. 2010].
In a crossover study of amlodipine with or without atorvastatin therapy in patients with obesity and hypertension, and normal lipid profiles, combination amlodipine/atorvastatin therapy produced a significantly greater reduction in HOMA-IR than amlodipine monotherapy; there was no correlation with BP reduction with either treatment [Fogari et al. 2006]. In patients with the metabolic syndrome and no diabetes, manidipine, but not amlodipine, significantly reduced HOMA-IR [Martínez Martín, 2009].
In patients with hypertension and insulin resistance, neither moxonidine nor amlodipine produced changes in HOMA-IR. Both treatments significantly lowered BP and increased HDL-C, but only moxonidine reduced serum triglycerides. Neither drug affected serum CRP levels [Masajtis-Zagajewska et al. 2010].
In patients with T2DM nephropathy, losartan, but not amlodipine, reduced HOMA-IR from baseline, but the between-group difference was not significant. However, other parameters of glucose metabolism (e.g. fasting blood glucose, HbA1c, and insulin sensitivity) were improved significantly more with losartan than with amlodipine [Jin and Pan, 2007]. In patients with hypertension and T2DM, telmisartan resulted in greater improvements in HOMA-IR than amlodipine [Negro et al. 2006].
Similar results with losartan and amlodipine were seen in patients with prediabetes, with greater improvements in HOMA-IR with losartan than with amlodipine; the two agents resulted in similar BP reductions [Nishimura et al. 2008].
In a study in patients with hypertension, both candesartan and amlodipine significantly improved endothelial function, but significant decreases in HOMA-IR and CRP were only observed with candesartan [Tomiyama et al. 2007].
In a comparison of losartan and amlodipine in Japanese patients with hypertension, with or without diabetes, losartan provided greater increases in adiponectin than amlodipine. These increases correlated with HOMA-IR changes [Watanabe et al. 2006b].
In agreement with the adiponectin results discussed earlier, indapamide treatment increased HOMA-IR in patients with hypertension, whereas no changes in HOMA-IR were seen with enalapril, metoprolol, or amlodipine [Piecha et al. 2007]. In patients with hypertension and the metabolic syndrome, doxazosin, amlodipine, ramipril, and valsartan produced significant reductions in HOMA-IR, whereas no changes were seen with metoprolol [Yilmaz et al. 2007].
Insulin resistance is a central force in the pathogenesis of vascular diseases, and HOMA-IR provides a reasonable assessment of the quantification of insulin resistance. Several long-term clinical studies have demonstrated the clinical benefit of ARBs in diabetic kidney disease, both in late stage (losartan in the Effects of Losartan on Renal and Cardiovascular Outcomes in Patients with Type 2 Diabetes and Nephropathy (RENAAL) study and irbesartan in the Irbesartan Diabetic Nephropathy Trial (IDNT) study) and early stage (irbesartan in the Irbesartan and Microalbuminuria (IRMA-2) study). The role of HOMA-IR may be beneficial in clinical practice, and quantitative and longitudinal analysis could provide long-term follow-up of disease management.
Procollagen
Several studies have assessed the effect of antihypertensive agents on procollagen fractions as a marker of atherogenesis and vascular remodeling (Table 6). Valsartan and ramipril, but not amlodipine, were associated with reductions in procollagen; despite similar BP lowering, valsartan and ramipril were more effective than amlodipine in preventing new episodes of atrial fibrillation [Fogari et al. 2008].
Another study showed a significant difference in procollagen type I carboxy-terminal peptide (PICP) lowering between candesartan and amlodipine. Although BP control was similar, 24-h SBP was significantly lower and LV mass index significantly decreased with amlodipine (p < 0.05), while the effect of ARBs on procollagen indicate that they protect against CV fibrosis and renal injury [Ishimitsu et al. 2005].
There were no differences in procollagen markers with losartan- or atenolol-based regimens after the first year of treatment; changes in PICP during the first year of treatment were related to subsequent changes in LV mass index after 2 and 3 years of treatment (r = 0.28 and 0.29, both p < 0.05) in patients randomized to losartan, but not atenolol [Christensen et al. 2006]. Losartan-related reduction in procollagen was shown to be greater in patients with higher baseline levels (those with hypertension and severe myocardial fibrosis) [Diez et al. 2002] and was significantly associated with symptom improvement [Dziadzio et al. 1999].
Tumor necrosis factor-α and interleukin-6
The effects of antihypertensive drugs on the inflammatory biomarker TNF-α have been somewhat variable (Table 7). In patients with hypertension, olmesartan medoxomil reduced TNF-α levels in one study [Lorenzen et al. 2010], but in another study in Japanese patients, neither losartan nor the CCB amlodipine significantly affected TNF-α levels [Watanabe et al. 2006b]. Amlodipine was effective in reducing TNF-α in one study, but was significantly more effective when combined with atorvastatin [Fogari et al. 2006]. Another study found no difference between losartan and amlodipine in TNF-α levels after treatment, but the investigators did not appear to perform baseline assessments in order to determine if either drug reduced TNF-α from baseline levels [Aksnes et al. 2007].
Losartan therapy significantly reduced TNF-α in patients with hypertension and T2DM [Park et al. 2006]. However, olmesartan medoxomil combined with HCTZ had no effect on TNF-α in patients with hypertension and the metabolic syndrome (without diabetes), but when olmesartan medoxomil was combined with amlodipine, the combination did significantly reduce TNF-α levels [Martínez-Martín et al. 2010]. Interestingly, amlodipine alone was shown in another study to have no effect on TNF-α levels in patients with hypertension and the metabolic syndrome, whereas manidipine monotherapy was effective in lowering TNF-α [Martínez Martín, 2009]. Olmesartan medoxomil had no effect on TNF-α levels in patients with stage 3 or 4 CKD [De Vinuesa et al. 2006], and TNF-α was unaffected by amlodipine or spironolactone in patients with diabetic nephropathy [Matsumoto et al. 2006].
Studies investigating the effect of antihypertensive drugs on IL-6 levels are summarized in Table 8. Olmesartan medoxomil reduced IL-6 levels in one study in patients with hypertension [Lorenzen et al. 2010], but had no effect in patients with stage 3 or 4 CKD [De Vinuesa et al. 2006]. Olmesartan medoxomil was ineffective when combined with HCTZ in patients with hypertension and the metabolic syndrome, but was effective in these patients when combined with amlodipine [Martínez-Martín et al. 2010]. Valsartan combined with HCTZ was more effective than amlodipine alone in reducing IL-6 [Ruilope et al. 2005].
These cytokines are rather nonspecific for quantification of inflammation; however, these studies do reflect the general state of inflammation in the vasculature. Clinical studies that measure the level of the cytokines demonstrate variable results. Multiple studies with antihypertensives indicate a general reduction in the levels of cytokines, suggesting a decrease in vascular inflammation. In context with the clinical situation and other risk factors, the measurement of these biomarkers may be useful.
Conclusions
It can be expected that biomarkers will continue to play an increasing role in the management of CV disease. Their importance or significance is likely to increase in direct proportion to the growth in our knowledge of disease pathophysiology and the mechanisms of drug action. The use of biomarkers does, however, depend upon the markers being accurate, relevant to the purpose, easy to measure, and consistently reproducible.
There is a wealth of evidence for improvement of validated biomarkers of vascular disease with most classes of antihypertensive treatment in a range of high-risk patient populations. These include people with obesity, patients with diabetes, patients with renal disease and/or metabolic syndrome, existing vascular disease, and African American patients. Benefits have also been observed in those with normal BP, but with other CV risk factors. There is some evidence to suggest that at least part of the effect seen with some antihypertensives on these biomarkers may be independent of BP reduction. Different drugs may have quite different effects on biomarkers, despite very similar or equivalent effects on BP. However, with other drugs, the changes in certain biomarkers appear to parallel changes in BP. In addition, there appear to be clear associations between certain biomarkers, such as HOMA-IR and adiponectin, and the manner in which they are affected by certain antihypertensive drugs.
There is particularly compelling evidence that RAS inhibitors (ACE inhibitors and ARBs) and CCBs may have beneficial effects beyond BP control, making them particularly attractive for either monotherapy or combination therapy. In contrast, other drugs, such as the thiazide diuretic HCTZ, appear to counter the beneficial effects on biomarkers normally observed with ARBs when they are used in combination.
Of the biomarkers selected for review in this paper, the benefits of antihypertensive therapy on hsCRP, adiponectin, and HOMA-IR reflect a potential for quantifiable long-term vascular benefits. However, more evidence is required to elucidate the mechanisms involved and understand the variability and apparent anomalies observed. In addition, more information about any differences between specific antihypertensive agents within the same class is needed. More evidence is required to determine the relevance of improvements observed with antihypertensive therapy on CAP, cystatin C, procollagen, TNF-α, and IL-6 to a reduction in the risk of subsequent vascular events.
For example, among the antihypertensive agents discussed in this review, the effects on biomarkers of olmesartan medoxomil and amlodipine are being investigated in phase 3 studies. Effect of Olmesartan Medoxomil on Vascular Markers in Hypertensive Patients with Metabolic Syndrome (VAMOS) is a 6-week comparison of the effects of low- and high-dose olmesartan medoxomil with amlodipine on the level of the inflammatory biomarker hsCRP in patients (estimated enrollment, n = 60) with hypertension and the metabolic syndrome [ClinicalTrials.gov identifier: NCT00891267]. The Arterial Stiffness in Humans (ArtStiff) study is a long-term, placebo-controlled assessment over 1 year that is investigating the effect of olmesartan medoxomil therapy on arterial stiffness measured by carotid-femoral PWV in patients (projected enrollment, n = 318) with hypertension and the metabolic syndrome [ClinicalTrials.gov identifier: NCT00676845].
Further research is required to determine the extent to which these antihypertensive-related improvements in biomarkers contribute to the overall clinical outcome achieved in tandem with other CV risk reduction strategies and interventions. In addition, long-term studies with biomarkers are also required to show whether biomarkers correlate with long-term clinical outcomes.
Footnotes
Acknowledgements
We thank Robert Schupp, PharmD and Ray Hill, of inScience Communications, a Wolters Kluwer business, who provided medical writing support funded by Daiichi Sankyo, Inc.
Funding
Dr Khan has received financial support from Daiichi Sankyo, Inc. in the form of an unrestricted grant for investigator-initiated studies.
Conflict of interest statement
Dr Khan has received financial support from Daiichi Sankyo, Inc. in the form of fees as a scientific advisory board member and in the receipt of an unrestricted grant for investigator-initiated studies.
