Abstract
Pulmonary hypertension (PH) due to left heart disease is classified as group II according to the Dana Point classification, which includes left ventricular systolic and/or diastolic left heart failure, and left-sided valvular disease. PH due to left heart disease is the most common cause and when present, especially with right ventricular dysfunction, is associated with a worse prognosis. Left heart disease with secondary PH is associated with increased left atrial pressure, which causes a passive increase in pulmonary pressure. Passive PH could be superimposed by an active protective, and in some patients by an ‘out of proportion’, elevated precapillary pulmonary vasoconstriction and vascular remodelling which leads to greater or lesser further increase of the pulmonary artery pressure. In this review, epidemiological and pathophysiologic mechanisms for the development of group II PH are summarized. The conflicting data about the haemodynamic and possible parameters to diagnose passive versus reactive and ‘out of proportion’ PH are presented. The different therapeutic concepts, along with novel treatment strategies, are reviewed in detail and critically discussed regarding their effectiveness and safety.
Keywords
Introduction
Pulmonary hypertension (PH), any condition with a mean pulmonary pressure ≥25 mmHg at rest, is currently classified into five different groups. International meetings on PH in the past have focused predominantly on pulmonary arterial hypertension (PAH; Group I), with the result that enormous progress has been achieved in the pathophysiology and therapy of PAH patients. The 4th World Symposium was the first to assign a working group to address in detail the so-called non-PAH forms of PH, that is, those forms of PH encountered in patients with left heart disease (Group II), with chronic obstructive disease (COPD) and interstitial lung disease (Group III), venous thromboembolism (Group IV) and other conditions (Group V) [Galie et al. 2009; Hoeper et al. 2009].
In this review, we focus on PH due to left heart disease as the perhaps most common PH scenario. We present an overview on the pathophysiology, natural history, clinical signs and treatment of PH in left heart disease.
PH due to left heart disease as a postcapillary PH
The elevated pulmonary vascular resistance in PH patients of groups I, III, IV and V originates from the precapillary regions of the pulmonary vascular system. Different pathological entities are found within the distal pulmonary arteries, including intimal proliferative, fibrotic and thrombotic lesions, as well as inflammatory infiltrates. Pulmonary veins are predominantly unaffected [Hoeper et al. 2009; Simonneau et al. 2004]. According to the haemodynamic definitions of PH, such patients are characterized by a mean pulmonary arterial pressure (PAP) ≥25 mmHg with classically normal left ventricular filling and left atrial pressures [pulmonary capillary wedge pressure (PCWP) ≤15 mmHg], resulting in an elevated transpulmonary gradient [TPG = PAPmean – mean pulmonary wedge pressure (PCWPmean) (Figure 1).

Haemodynamic definition of pulmonary hypertension (ESC guidelines) [Galie et al. 2009].
In contrast, PH due to left heart disease has been defined as a postcapillary form of PH. In the original nomenclature from 1998, this form of PH had been classified as secondary PH or ‘pulmonary venous hypertension’ to indicate that the elevation of the PAP is solely the consequence of increased pressure within the left atrium and pulmonary veins (PWP ≥15 mmHg) and of a low pressure transpulmonary gradient within normal range (5–9 mmHg) [Guazzi and Arena, 2010]. A reduction in PAPmean is strongly dependent on a PCWP reduction, which implies that left atrial pressure largely determines the pulmonary artery pressure in left heart disease. In the case of a normal range TPG, no component of the PH is derived from abnormalities intrinsic to the pulmonary arterial bed. Therefore, left-sided PH in this stage has been defined as a passive PH [Galie et al. 2009; Guazzi and Arena, 2010; Simonneau et al. 2004].
Passive versus reactive PH due to left heart disease
Haemodynamic studies from patients with left heart disease confirm the high correlation between increased and elevated left atrial pressure (PCWP) and the resulting mean pulmonary arterial pressure [Drazner et al. 1999; Ilsar et al. 2010]. However, a wide scattering of values along the correlation line has been appreciated. We have observed in 120 patients with advanced systolic heart failure that the same PCWP may have very different TPGs, resulting in different PAP [H. Schroetter A. Schmeisser, R. Strasser and R. Braun-Dulleaus. Personal communication (unpublished results)] (Figure 2). This implies that, in the case of elevated TPGs, a reactive precapillary component of pressure elevation is added to the passive transmission of the left atrial pressure. At this stage of a reactive PH, both functional and structural abnormalities of the pulmonary arterial bed are presumed [Galie et al. 2009; Hoeper et al. 2009].

Correlation of TPG with PCWPmean in patients with advanced systolic heart failure.
Functional and structural pathology in reactive PH
The endothelium-mediated local control of vasomotility in healthy individuals is primarily based on a balanced release of nitric oxide (NO) and endothelin-1 (ET-1) [Budhiraja et al. 2004]. The imbalance of these two opposing systems is suggested in patients with left heart disease and PH resulting in an increased resistive pulmonary arterial tone [Giaid et al. 1993; Giaid and Saleh, 1995]. Human and animal studies demonstrated a lack of NO synthesis in response to mechanical or receptor-mediated stimulation in lung venous capillaries and small arterioles in group II PH [Cooper et al. 1998]. In addition, recent data from a rat model of congestive heart failure could identify an impaired endothelial Ca2+ signalling and NO synthesis caused by an extensive cytoskeletal reorganization in lung endothelial cells [Kerem et al. 2010]. In contrast, increased endothelial cell expression and elevated plasma levels of the very potent vasoconstrictor ET-1 and its increased lung endothelial cell expression have been shown in experimental models and human patients with heart failure [Cody et al. 1992].
Furthermore, an elevated pulmonary precapillary tone as a functional abnormality in a large number of patients with left heart disease is also associated with a morphological remodelling of the distal pulmonary arteries, the capillaries, the venous bed and the alveoles. Acutely or chronically increased left atrial pressure may lead to alveolar stress failure, a process that disrupts the functional properties of the alveolar capillaries. While acute stress failure is a reversible phenomenon, the alveolar capillary membrane undergoes a remodelling process during chronic capillary pressure elevation that eventually is not reversible [Guazzi and Arena, 2010].
In chronic PH, a superimposition of additional factors other than mechanical stress, such as neurohormonal, cytotoxic, hypoxic and genetic factors, further injure lung capillaries and alveolar spaces, which triggers a process of alveolar remodelling that ultimately leads to gas exchange impairment [Budhiraja et al. 2004; De Vita et al. 2002; Guazzi et al. 2002; Guazzi and Arena, 2010]. Enlarged and thickened pulmonary veins, dilatation of pulmonary capillaries, enlarged lymphatic vessels, muscularization, and perivascular fibrosis of the arterioles are morphological changes of this type of PH [Chen et al. 2012; Delgado et al. 2005; Guazzi et al. 2002; Yin et al. 2011].
The degree of change in vessel structure and vascular resistance varies widely, which implies that the natural evolution of PH is very different across the spectrum of patients with group II PH. The most severe histological changes have been reported in patients with mitral stenosis with substantially increased systolic and diastolic left atrial pressure over a long time period [Tandon and Kasturi, 1975; Tryka et al. 1985]. In contrast, the main changes found in patients with systolic heart failure were discrete medial hypertrophy of the distal arterioles (<600 µm) and only mild or no vascular intimal fibrosis was detected [Delgado et al. 2005]. Vasodilator testing of systolic heart failure patients supports these findings as at least a partially preserved vascular responsiveness of the pulmonary arterioles was demonstrated [Braun et al. 2007].
In our own experience of over 400 cases of provocative vasodilator testing (Iloprost ± sildenafil), a nonexistence of pulmonary vasoreactivity or ‘fixed’ PH could be found in <10% of patients with PH due to systolic heart failure. In these nonresponsive cases, irreversible obstructive remodelling was suspected. The International Society for Heart and Lung Transplantation (ISHLT) has defined unresponsive ‘fixed’ PH in patients with systolic heart failure awaiting heart transplantation as when the systolic PAP remains >50 mmHg, the pulmonary vascular resistance (PVR) >5 WU (Wood Units) and the TPG >16 mmHg after pharmacological testing [Mehra et al. 2006]. However, even this condition may be reversible, as effective unloading of the left heart by left ventricular (LV) assist devices has been shown to induce significant regressions of these functional and structural precapillary remodelling processes, allowing a lower risk heart transplantation at a later time point [Martin et al. 2004; Zimpfer et al. 2007].
Haemodynamic definitions of reactive versus passive PH due to left heart disease (LHD)
Transpulmonary gradient
For the first time, the European Society of Cardiology (ESC) Guidelines for the Diagnosis and Treatment of Pulmonary Hypertension differentiates between passive and reactive PH due to left heart disease according to very specific transpulmonary gradient (TPG) cutoff values. The authors selected a TPG limit of >12 mmHg as reactive PH and ≤12 mmHg as passive PH [Galie et al. 2009] (Figure 3).

Differentiation in passive versus reactive ‘out of proportion’ pulmonary hypertension according to ESC guidelines [Galie et al. 2009].
However, this arbitrarily selected cutoff lacks prospectively designed studies for precise pathophysiological, clinical and prognostic discrimination. In addition, the reactive PH was also referred to as ‘out of proportion’ PH by the same authors [Galie et al. 2009], which means that every elevation of PAmean with a TPG >12 mmHg is considered ‘out of proportion’ to the underlying cause of left heart disease. Such nomenclature completely ignores the very complex function of the pulmonary vascular bed to regulate the interplay between the often very differently affected right and left ventricular function. A dynamically changing TPG/PVR could regulate the afterload sensitive ejection of the right ventricle in order to adjust the ventricular preload of a diseased left heart and to protect it from a volume overload. In addition, defining a TPG cutoff contradicts the physiology of the pulmonary vascular bed. The gradient between PAmean and PCWP is sensitive to changes in cardiac output and both the recruitment and distension of the pulmonary vessels, which decrease the upstream transmission of left atrial pressure. Furthermore, pulmonary blood flow is pulsatile, with systolic and mean PAP and, logically, also the TPG determined by stroke volume and arterial compliance [Naeije et al. 2012].
Diastolic pressure gradient
This discussion shows that, in practice, the TPG may overdiagnose or underdiagnose pulmonary vascular disease in left heart failure. Naeije and colleagues supposed in their very smart review that this could be avoided to a large extent by the use of the gradient between diastolic PAP and PCWP, or ‘diastolic pressure gradient’ (DPG) [Naeije et al. 2012]. The theory behind this is that pulmonary flow reaches a maximum during systole and is inappreciable at the end of diastole. Accordingly, diastolic PAP in a normal pulmonary circulation is approximately equal to the pressure in left atrium (LA), more specifically, the mean pressure during the atrial systole (or ventricular diastolic phase). In a small haemodynamic study in healthy volunteers, an upper limit of 5 mmHg was established for the normal DPG [Harvey et al. 1971]. Values above 5 mmHg in combination with signs of elevated left atrial pressure would indicate an ‘out of proportion’ PH in patients with PH due to left heart disease.
The advantage of using DPG instead of TPG lies in the reduced sensitivity to changes in the cardiac output, stroke volume and absolute values of atrial pressure [Naeije et al. 2012]. However, these are hypotheses, and new pharmacological and haemodynamic clinical studies are needed to clarify these considerations.
Pulmonary vascular resistance
Recently, different authors proposed that a PVR >3 WU versus ≤3 WU could differentiate between reactive and passive PH [Aronson et al. 2011; Khush et al. 2009]. However, PVR is influenced by two variables: TPG and cardiac output. In particular, the determination of the right cardiac output by thermodilution or by the Fick method is often incorrect and unreliable. An example of this is that a combination of a highly normal TPG of 10 mmHg and a very low cardiac output (CO) of 3 l/min would also result in a PVR (PVR = TPG/CO) >3 WU, which does not really indicate an ‘out of proportion’ PH.
Determinants of PH in left heart disease
PH due to left ventricular systolic or diastolic dysfunction
The hallmark of PH in left heart disease is elevated left atrial (LA) and pulmonary venous pressure. Elevated LA pressure in patients with left ventricular dysfunction is predominantly the result of backward transmitted elevated left ventricular filling pressure (LVEDP) [Enriquez-Sarano et al. 1997; Kerem et al. 2010]. Increased LV filling pressure is associated with dysfunctional early diastolic relaxation and late filling compliance in patients with primary diastolic heart failure (heart failure with preserved ejection fraction, HFPEF). In systolic heart failure patients (HF with reduced EF, HFREF), heightened filling pressure is the mixed consequence of a right shift of the Frank Starling curve to a higher LVEDP and, in most cases, to an additional diastolic dysfunction. Owing to the open mitral valve, an elevated left ventricular filling pressure determines the LA pressure with an equalizing of pressure during the late ventricular diastole (communicating holes). The left atrium is filled (atrial diastole) by the pulmonary venous blood during the left ventricular systole. The resulting left atrial filling pressure (v-wave), in turn, is determined mainly by the compliance of the left atrium. That means that the diastolic LA pressure, as a direct reflection of the LVEDP, is added by the atrial filling pressure (v-wave) during the cardiac cycle (for example in Figure 5 below). The impact of LA stiffness for the generation of the v-wave is supported by the data of Kurt and colleagues [Kurt et al. 2009]. They showed that the LA stiffness is tightly related to the elevated pulmonary pressure in patients with both HFREF and HFPEF.
In addition, some patients have increased LA pressure due to atrial and ventricular interdependence mediated by a constrained pericardial sack relative to an enlarged right atrium and/or right ventricle.
Different grades of accompanying mitral regurgitations have been described, especially in patients with systolic heart failure, and also in patients with HFPEF [Marechaux et al. 2011; Rossi et al. 2011]. In such cases, the systolic regurgitation fraction of the diseased mitral valve is added to the systolic LA filling volume. Depending on the LA stiffness, this results in further increases of the left atrial filling pressure (v-wave) and the consecutive pulmonary pressure.
PH due to valvular disease
Mitral valve regurgitation increases the v-wave of the LA pressure curve very directly depending on the regurgitation volume, LA compliance and LVEDP [Barbieri et al. 2011; Saraiva et al. 2009].
In turn, mitral valve stenosis obstructs the diastolic emptying of the LA. Elevated LA pressure (a-wave) during the diastole can be observed. The additional systolic filling results in an elevated systolic pressure (v-wave) because of the end-diastolic volume overload of the LA [Tryka et al. 1985].
In contrast to the direct effects of mitral valve disease on pulmonary pressure elevation, the development of pulmonary hypertension in aortic valve disorders is predominantly not mediated by the diseased valve itself. Over time, aortic valve stenosis as well as aortic valve insufficiency leads to progressive left ventricular systolic and diastolic dysfunction, reflected by LVEDP. The latter and often accompanying mitral valve insufficiencies are the main causes of PH in aortic valve disease [Kapoor et al. 2008; Malouf et al. 2002; Varadarajan and Pai, 2012].
In conclusion, the development of PH due to systolic/diastolic myocardial dysfunction and/or valvular disease is dependent on an elevated left ventricular filling pressure, mitral valve disease, impaired compliance of the left atrium, and a variable precapillary transpulmonary pressure gradient (Figure 4).

Proposed mechanisms for the development of pulmonary hypertension in valvular heart disease.
Epidemiology and prognosis of PH in left heart disease
PH owing to left heart disease is a common phenomenon that appears to be highly relevant for the patient’s outcome [Barbieri et al. 2011; Bursi et al. 2012; Damy et al. 2010; Malouf et al. 2002; Szwejkowski et al. 2012; Varadarajan and Pai, 2012]. However, the development, severity and prognostic relevance are variable and are dependent on the pathophysiology of the different underlying left heart diseases (Table 1).
Classification of PH due to left heart disease.

CMP: cardiomyopathy
Heart failure with preserved ejection fraction (HFPEF)
The prevalence of diastolic heart failure (HFPEF) is in up to 25% of the predominantly elderly population [Kuznetsova et al. 2009; Tribouilloy et al. 2008]. Most HFPEF patients have elevated pulmonary arterial pressure because of an increased left ventricular filling pressure. Recent data from Olmsted County, Minnesota, USA, confirm that 83% of patients with HFPEF (n = 224) develop pulmonary hypertension [pulmonary artery systolic pressure (PASP) >35 mmHg]. In comparison, only 8% of patients with sole arterial hypertension were detected to have PH [Lam et al. 2009b]. PASP elevation was highly predictive for survival in HFPEF.
Although the increased left ventricular filling pressure (PCWP, LVEDP) contributes to PH, it does not fully explain the severity of PH in HFPEF, suggesting that a component of precapilliary pulmonary pressure elevation (TPG) also contributes. However, the PCWP values were estimated only by echocardiography in which the ratio of early transmitral flow velocity (E) to early mitral annular diastolic velocity (e′, tissue doppler) was used. Up to now, this method is not likely to replace the right heart catheter, especially to determine the resulting transpulmonary gradient precisely [Bhella et al. 2011]. Haemodynamic studies are necessary to recognize the true extent of the precapilliary component of PH in HFPEF.
Heart failure with reduced EF (HFREF)
Epidemiological data and observational studies have shown that at least two-thirds of patients with severe systolic heart failure develop PH [Cappola et al. 2002; Ghio et al. 2001]. PH in HFREF is associated with an increased mortality, hospitalization rate and need for cardiac transplantation [Abramson et al. 1992; Cappola et al. 2002]. Mean systemic pressure and mean pulmonary artery pressure in patients with newly diagnosed cardiomyopathy (n = 1134) have emerged as important haemodynamic predictors of death [Cappola et al. 2002]. As in the case of PAH, chronic elevation of PASP results in permanent right ventricular afterload stress, which may ultimately lead to progressive right heart failure [de Groote et al. 2012; Haddad et al. 2008a, 2008b]. Ghio and colleagues [Ghio et al. 2001] demonstrated that the presence of right heart dysfunction in patients with HFREF was associated with a twofold higher mortality than in isolated LV failure.
Passive versus ‘out of proportion’ PH
To the best of the authors’ knowledge, no randomized prospective studies have been published targeting the prognostic value of passive versus reactive PH in patients with PH due to left heart disease. Two retrospective analyses carried out in patients with recently decompensated systolic heart failure examined the prognostic implications of PH subtypes. The subtype definitions of PH were based on a PVR cutoff of >3 WU (reactive PH) versus ≤3 WU (passive PH) [Aronson et al. 2011; Khush et al. 2009]. While Khush and colleagues [Khush et al. 2009] found that the PVR-based PH subtypes of the ESCAPE (Evaluation Study of Congestive Heart Failure and Pulmonary Artery Catheterization Effectiveness) trial were not associated with a different six-month survival, analysis of the VMAC (Vasodilation in the Management of Acute Congestive Heart Failure) trial data showed a twofold higher six-month mortality in patients with a PVR >3 WU [Aronson et al. 2011].
To interpret these partly conflicting results, we have to remember that PVR is a calculated parameter, namely the quotient of TPG and cardiac output. Therefore, a high PVR may result from an elevated TPG and/or a lowered cardiac output. Hence, a normal TPG (e.g. 9 mmHg) in combination with a very low cardiac output (e.g. 3.0 l/min) would also result in a high PVR of 3 WU, reflecting the compromised LV function but not the intrinsic abnormalities of the pulmonary vascular tree. Indeed, reactive PH patients within the VMAC trial were characterized by significantly higher left ventricular filling pressures and lower stroke volumes than passive PH patients. This implicates their worse LV function in comparison with the passive PH group, which has certainly influenced the worse prognosis too [Aronson et al. 2011].
PH in left-sided valvular disease
The prevalence of PH in left-sided valvular disease correlates with the severity of the valve disease, the concomitant diastolic dysfunction of the left atrium and ventricle, and clinical symptoms [Bonow et al. 2006, 2008].
Mitral stenosis is the classical disease associated with PH in left heart disease. The prevalence of PH varies depending on the severity of the valvular disease and may be as common as 75% [Bonow et al. 2006; Hart et al. 2010]. In contrast, mild, moderate or moderate-to-severe mitral valve regurgitation is associated with PH pressure >40 mmHg in only 14%, 26% and 44% of affected patients, respectively [Parker et al. 2010]. However, it was shown recently that exercise-induced PH [systolic pulmonary arterial pressure (PAP) >60 mmHg] was common in patients with asymptomatic moderate mitral regurgitation ith a prevalence of more than 46% (compared with 15% of PH at rest) [Magne et al. 2010]. Notably, the severity of mitral regurgitation did not correlate with the systolic PAP. In the study by Saraiva and colleagues [Saraiva et al. 2009], the only independent echocardiographic predictor of PH was the E/A ratio reflecting the LV diastolic dysfunction. This suggests that LA volume overload may be balanced by LA compliance and/or pulmonary vascular capacitance and resistance even when chronic mitral regurgitation is severe.
Compared with mitral stenosis, the prevalence of PH in patients with aortic stenosis is lower (approximately 30%) [Faggiano et al. 2000; Johnson et al. 1988; Malouf et al. 2002]. Although the risk of perioperative and postoperative heart failure and mortality is increased in these patients, only aortic valve replacement may improve the extremely limited prognosis [Malouf et al. 2002; Zuern et al. 2012]. In patients with mild, moderate and severe aortic regurgitation, a PH with a pulmonary systolic pressure >40 mmHg was found in 26%, 28% and 18%, respectively [Parker et al. 2010].
Even though PH constitutes a risk factor for valvular surgery, the surgical correction of significant valvular disease results in a considerable reduction of PH in most cases [Vahanian et al. 2007].
Right ventricular function and PH due to left heart disease
The normal right ventricle (RV) delivers the same stroke volume as the left ventricle, but with one-fifth of its mass and wall thickness, and only 20% of the intrinsic contractility of the left heart [Haddad et al. 2008a, 2008b]. In addition, due to complex crescent-like geometry and myocyte arrangement, the right ventricle is very well-suited to accommodate an increase in volume load, but is extremely afterload sensitive [Anderson et al. 2009; Haddad et al. 2008b; Nielsen et al. 2009]. The main adaptive mechanism that enables the right ventricle to maintain an adequate cardiac output is the development of hypertrophy, which can reach an extensive degree especially in the case of chronic mitral valve stenosis. Although this pattern of RV remodelling is often tolerated for many years, it may progress to chamber dilatation, functional tricuspid regurgitation and frank RV failure [Champion et al. 2009]. Wall dilatation especially in combination with increased RV pressure leads to an increased wall stress, which may enhance myocardial oxygen demand, thus provoking ischemia and further RV dysfunction.
In the case of PH due to left heart disease, more different and often combined reasons than only afterload mismatch are responsible for the development of right-sided ventricular dysfunction. Mainly, chronic reductions of the intrinsic contractility of the right ventricular muscle by the same cardiomyopathic process involving the left ventricle, such as myocardial infarction with RV participation, biventricular dilative cardiomyopathy, and biventricular arrhytmogenic right ventricular dysplasia (ARVD), often accompanied by functional tricuspid regurgitation and ventricular interactions, can aggravate the RV dysfunction in comparison to a singular RV afterload increase. Biventricular distributed infiltrative and/or hypertrophic cardiomyopathies can lead to pronounced diastolic LV and RV dysfunction and failure. In addition, Tedford and colleagues [Tedford et al. 2012] recently explained that afterload mismatch in PH due to left heart disease is not the same as in precapilliary pulmonary artery hypertension. They found that the compliance of the pulmonary vascular bed in group II PH is lower at any given PVR in comparison with pulmonary artery hypertension. Higher PCWP appears to enhance net right ventricular afterload by elevating pulsatile load and this may contribute to the further decline of right ventricular function.
The occurrence of right ventricular dysfunction and frank RV failure are undesirable in group II PH, because of the further compromised ventricular forward output and the facilitation of oedema formation by increasing the right ventricular and consecutive atrial pressure [Guazzi and Arena, 2010; Guazzi and Borlaug, 2012]. This in itself carries additional multiorgan toxicity, such as interstitial fluid accumulation in the lungs and pleura spaces by compromised lymphatic drainage, hepatic and splanchnic congestion with impaired gut absorption, development of ascites and translocation of gut microbes, and consecutive release of endotoxins with subsequent inflammatory states in heart failure [Guazzi and Borlaug, 2012; Niebauer et al. 1999]. In summary, progressive RV failure in group II PH aggravates the left heart disease by triggering different pathophysiological mechanisms that worsen not only the clinical symptoms, but also the prognosis of these patients.
Different observational studies have shown that right ventricular dysfunction and failure in patients with PH due to systolic left heart failure is an important predictor for a worse prognosis [de Groote et al. 2012; Ghio et al. 2001]. In addition, retrospective analysis of data from BEST (Beta-Blocker Evaluation in Survival Trial) highlighted that an impaired right ventricular ejection fraction (RVEF) is associated with lower survival in patients with HFREF. An RVEF especially of <20% was an independent predictor of mortality and hospitalization for HFREF patients [Meyer et al. 2010].
Diagnosis
The diagnostic approach of PH due to left heart disease basically corresponds to PAH. It always has the following three goals:
to objectify the presence of PH;
to differentiate between precapilliary (group I, III, IV, V) or postcapilliary (group II) PH;
in the case of precapilliary PH, to identify group I or IV diseases in which specific pharmacological or operative therapies, respectively, are indicated and may improve the mostly worse prognosis [Galie et al. 2009].
The initial diagnostic workup includes a careful physical examination, chest radiography, electrocardiography and doppler echocardiography. A right heart catheterization is essential to differentiate between PH due to left heart disease (postcapilliary) and precapilliary PH, to measure pulmonary haemodynamics of valvular heart disease, or to evaluate haemodynamics prior to heart transplantation [Rosenkranz et al. 2011a, Rosenkranz et al. 2011b].
Common symptoms of patients with PH due to left heart disease are orthopnea, paroxysmal nocturnal dyspnea and exertional dyspnea [Guazzi and Arena, 2010; Guazzi and Borlaug, 2012]. Patients with left heart failure display a number of ventilatory abnormalities that are probably related to pulmonary vasculature. In patients with left heart failure, an impaired exercise ventilator efficiency ratio of ventilation to carbon dioxide uptake (VE/Vco2) slope is observed, which is related to pulmonary vasoconstriction, RV dysfunction [Di Salvo et al. 1995; Reindl et al. 1998] and adverse clinical events [Kleber et al. 2000; Lewis et al. 2008]. The incidence of sleep apnoea as well as oscillatory ventilator patterns during exercise are related to impaired pulmonary vascular haemodynamics, heart minute volume and increased left ventricular filling pressure [Guazzi et al. 2005, 2012a, 2012b; Solin et al. 1999]. Chest radiography may show pulmonary vascular congestion, pleural effusion or pulmonary oedema, and will often reveal cardiomegaly in the case of systolic heart failure. Electrocardiographic signs favouring group II PH include mostly LV hypertrophy, left atrial enlargement or atrial fibrillation [Guazzi and Borlaug, 2012]. Clinical signs of right heart failure are variable and are more common in advanced stages of biventricular heart failure. In addition, they involve a clinical exercise limitation, fatigue and evidence of specific venous congestion and secondary fluid retention (e.g. peripheral oedema, anasarca or ascites).
Echocardiography
Echocardiography plays a key role in the initial diagnosis of PH. Typically, transthoracic echocardiography is the primary modality chosen because it provides detailed anatomical and functional information about both ventricles, cardiac valves, pulmonary artery pressure, inferior vena cave width and pericardium [Howlett et al. 2009; Lindqvist et al. 2008]. With the use of transoesophageal echo, a more objective assessment of the severity of mitral and aortic regurgitation can be reached.
The PASP can be estimated from the peak tricuspid regurgitation (TR) jet velocity, using the simplified Bernoulli equation and combining this value with an estimate of the right atrial (RA) pressure: PASP = 4(V2) + RA pressure, where V is the peak velocity (metres per second) of the TR jet, and RA pressure is estimated from the inferior vena cava diameter and its respiratory changes [Rudski et al. 2010]. Normal resting values are usually defined as a peak TR gradient of ≤2.8 to 2.9 m/s or a peak systolic pressure of 35–36 mmHg, assuming a RA pressure of 3–5 mmHg [Badesch et al. 2009]. This value may increase with age and increasing body surface area [Lam et al. 2009a; McQuillan et al. 2001]. Determination of PASP by the sum of peak RV–RA gradient and RA pressure has been established and proven by multiple studies, but more recent reports have questioned the accuracy of this relationship, particularly at higher PAP [Fisher et al. 2009; Yock and Popp, 1984]. Sources of additional errors include overestimation of RA pressure and the reliance of inadequate Doppler signals. Despite the high usefulness of PASP estimation in clinical praxis, echocardiography is not able to locate with certainty the origin of pulmonary pressure elevation, namely precapillary or postcapilliary, or to differentiate group II PH in reactive or passive PH. At least, in the context of different clinical and echocardiographic signs, some prediction can be reached for distinguishing PAH from PH due to left heart disease [Rudski et al. 2010].
Echocardiography in PH due to heart failure with reduced EF (PH-HFREF)
PH in combination with reduced ejection fraction in dilated left ventricles, including different grades of functional mitral regurgitation, are typical signs of a correct diagnosis. Notably, in patients with concomitant right heart failure and severe tricuspid regurgitation, the Doppler envelope may be cut off because of an early equalization of RV and RA pressure, and the simplified Bernoulli equation may underestimate the RV–RA gradient and the pulmonary resistance [Rudski et al. 2010]
Echocardiogaphy in PH due to heart failure with preserved EF (PH-HFPEF)
Patient demographics provide perhaps the greatest predictive value for the differentiation of PH-HFPEF from PAH. Key risk factors for HFPEF or comorbidities are metabolic syndrome including systemic hypertension, obesity, diabetes mellitus, atrial fibrillation, older age and chronic obstructive lung disease [Aurigemma and Gaasch, 2004; Ho et al. 2012]. Echocardiographic findings suggestive of PH-HFPEF include the combination of preserved ejection function with left atrial dilatation, left ventricular hypertrophy, abnormal Doppler estimates of mitral and pulmonary venous flow velocity, and mitral tissue Doppler velocities. The ratio of early transmittal flow velocity (E) to tissue Doppler early diastolic (e′) mitral annular velocity (E/e′ ratio) has been widely accepted as a noninvasive measure of left ventricular filling pressure [Arques et al. 2007; Dokainish et al. 2010; Nagueh et al. 1997]. However, a number of recent studies have questioned the value of this tool to determine the degree of left ventricular diastolic dysfunction and to unambiguously distinguish between PAH and PH due to left heart disease [Bhella et al. 2011; Mullens et al. 2009].
Right heart catheterization
The European Society of Cardiology and the European Respiratory Society (ERS) guidelines do not provide a clear recommendation regarding the indication for right heart catheterization in patients with suspected group II PH. Indeed, only the careful and precise invasive measurement of the mean pulmonary pressure (≥25 mmHg) and left ventricular filling pressure (PCWP ≤15 mmHg versus >15 mmHg) may diagnose PH and is able to differentiate between precapillary and postcapilliary PH. The same applies to the evaluation of passive versus reactive PH regarding the height of TPG and/or PVR and/or diastolic pulmonary pressure gradient (see above in the section, ‘Haemodynamic definitions of reactive versus passive PH due to LHD’). Therefore, right heart catheterization should be considered in patients in whom a differentiated diagnosis of PH would alter management [e.g. PAH, chronic thromboembolic PH (CTEPH), valvular heart disease] and in patients with systolic left heart failure being evaluated for heart transplantation.
If right heart catheterization is performed, all parameters that are important to fully characterize pulmonary haemodynamics have to be measured, including a detailed oxymetry for the determination of cardiac output and to avoid overlooking shunt defects. Figure 5 shows a haemodynamic example of a patient with systolic heart failure and PH, in which conflicting results were provided regarding the differentiation between passive versus reactive ‘out of proportion’ PH) [Galie et al. 2009; Rosenkranz et al. 2011a].

Right heart catheter measurement.
It has to be realized that using PCWP as a central variable for diagnosis of group II PH could be associated with different problems. The actual pressure measurement of PCWP may be (pseudo)normal as a result of aggressive diuresis potentially leading to misdiagnosis and mistherapy of the PH subtype [Haddad et al. 2011; Hoeper et al. 2009]. A volume challenge (e.g. 500–1000 ml normal saline over 5–10 min) or exercise haemodynamics have been proposed to identify or exclude LV dysfunction and consecutive dynamic PH [Grunig et al. 2011; Kovacs et al. 2009; Rosenkranz et al. 2011a, 2011b].
In heart transplant candidates, the careful determination of the TPG (PAmean – PCWP) and cardiac output are especially important because the persistent increase of the PVR >2.5 WU and/or TPG >15 is associated with an up to threefold rate of postoperative right heart failure and mortality [Chang et al. 2005; de Groote et al. 2008; Mehra et al. 2006].
Diagnosis of right ventricular dysfunction
Right ventricular systolic dysfunction and failure in patients with PH due to left heart and valvular disease is an important predictor of a worse prognosis, but the analysis of right ventricular systolic function remains a difficult task. As the RV is positioned close to the sternum and is also very complex in its geometric shape and wall motion pattern, assessment by two-dimensional (2D) echocardiography is limited [Rudski et al. 2010]. In addition, there is a paucity of ultrasound studies providing normal reference values of right heart size and function. Radionuclide angiography and magnetic resonance imaging (MRI) are considered as the gold standard for the determination of right ventricle ejection function (RVEF) [de Groote et al. 2012; Kind et al. 2010; Meyer et al. 2010]. Alternative echocardiographic methods for the estimation of RV systolic function are fractional area change (FAC), myocardial performance index (MPI), tricuspid annular plane systolic excursion (TAPSE), or tissue Doppler peak systolic tricuspid annular velocity (STr) [Lindqvist et al. 2008; Rudski et al. 2010]. These parameters are simple to collect, reproducible and useful for risk stratification, at least for patients with systolic heart failure. Recently, in a retrospective analysis of 527 patients with stable systolic heart failure, best cutoff values for a longer cardiac survival were 37% RVEF (radionuclide angiography), 9.7 cm/s STr and 18.5 mm TAPSE determined by echo [de Groote et al. 2012].
More sophisticated techniques, such as MRI flow measurements, the determination of myocardial acceleration during isovolumetric contraction, regional RV strain and strain rate by echo, or the invasive evaluation of pressure volume loop by conductance catheters, are reserved for specific clinical and research applications in experienced laboratories.
Management of PH due to left heart disease
PH is a strong prognostic factor in patients with left heart disease. However, no specific therapy for the treatment of PH due to left heart disease has been established to date. What are the causes of this therapeutic dilemma?
To answer this question, we have to remember the determinants of PH in left heart disease to identify the therapeutic targets:
Pulmonary venous transmission of elevated left atrial pressure, induced by mitral valve disease and/or increased left ventricular filling pressure as the predominant postcapilliary part
The precapilliary generated pulmonary vascular resistance elevation, currently measured as elevated TPG, diastolic PAP gradient or PVR.
The management seems clear for the postcapilliary part of PH. In fact, several studies have shown that PH usually regresses after the optimization of heart failure of reduced or preserved ejection function or correction of valvular heart disease [Braunwald et al. 1965; Guazzi and Borlaug, 2012; Levine et al. 1989; Rosario et al. 1998; Zimpfer et al. 2007]. Application of established heart failure therapies, such as renin angiotensin aldosteron system (RAAS) inhibitors, ß-blockers, ivabradin and diuretics, especially in patients with left heart failure may improve PH through reduction of filling pressure and functional mitral insufficiency [Rosario et al. 1998]. Cardiac resynchronization therapy (CRT) in patients with systolic heart failure and wide QRS complex within the electrocardiogram may improve pulmonary haemodynamics, such as PAP, and PVR and RV remodelling [Bleeker et al. 2005]. However, further studies are required regarding the mechanisms, because these benefits seem to be predominantly mediated by the resynchronization of LV function, including improvements of both systolic and diastolic function [lowering LVEDP and left atrial pressure (LAP)/PCWP] and the reduction of functional mitral insufficiency (lowering LAP/PCWP).
The existence of moderate or severe functional mitral valve insufficiency in patients with advanced systolic heart failure deteriorates the quality of life, exercise capacity and prognosis. The reduction of the concomitant mitral insufficiency by newer interventional techniques, such as mitral clip therapy, has been shown to improve the exercise capacity and quality of life of those patients affected [Auricchio et al. 2011; Gaemperli et al. 2012; Rudolph et al. 2011; Whitlow et al. 2012]. The clinical benefits are mainly suspected by lowering the postcapilliary pressure elevation (LAP/PCWP), which results in a decrease of PAP and right ventricular afterload [Franzen et al. 2011].
In summary, optimizing heart failure regimens (and corrective valve surgery in primary valve disease) represents the cornerstone of the treatment of PH due to left heart disease. However, none of these therapeutic options, which may reduce the PH globally, have been systematically evaluated regarding the specific effects on pulmonary vascular tone, compliance, resistance, or structure (Figure 6).

Therapeutic management of pulmonary hypertension due to left heart disease (ESC guidelines) [Galie et al. 2009].
What are the data about the supposed second therapeutic target: the precapilliary derived pulmonary arterial resistance elevation? It is interesting that therapeutic concepts to modulate, very specifically, the precapilliary pulmonary vascular resistance in PH due to left heart disease have not really been established. Various authors, including the ESC Guidelines for the Diagnosis and Treatment of Pulmonary Hypertension have proposed that ‘out of proportion’ PH in these patients could be a therapeutic target [Galie et al. 2009; Guazzi and Arena, 2010; Guazzi and Borlaug, 2012; Rosenkranz et al. 2011b]. The transpulmonary gradient >12 mmHg has been recommended for the detection of intrinsic pulmonary vascular precapilliary disease and ‘out of proportion’ PH in these patients. As we have pointed out, this value is arbitrary, because the TPG is a highly dynamic parameter. How much pulmonary hypertension secondary to advanced left heart failure results is the passive consequence on an increase in LA pressure can be observed after LV assist system implantations. Several different groups have shown that mechanical LV unloading is able to reverse previously suspected ‘fixed’ precapilliary TPG or PVR, allowing candidacy for heart transplantation [Beyersdorf et al. 2010; Etz et al. 2007; Mikus et al. 2011a, 2011b]. If the proposed DPG is superior to the TPG or PVR for the diagnosis of frank ‘out of proportion’ has yet to be proven [Naeije et al. 2012].
In conclusion, we do not yet really know which degree of precapilliary pressure elevation is ‘out of proportion’ and which is basically protective against volume overload of the diseased left ventricle. In addition, we do not yet have a reliable parameter to differentiate the passive from the ‘out of proportion’ situation.
Are there alternative strategies to this dilemma? We suggest abandoning the unilateral view on the precapilliary pressure gradient in favour of a more interactive and integrated approach between pulmonary vascular blood flow and left atrial/ventricular filling behaviour. In this context two approaches could be envisaged:
Approach 1: Develop a test procedure
This approach allows a very selective lowering of the elevated precapilliary pressure gradient in a very predictable and gradual manner.
In the case of protective passive PH, the filling pressure would significantly increase because of an inadequate volume overload of the LA and LV. The proposed therapy of such protective passive PH would solely include procedures that unload the LV, such as vasodilators, diuretics or LV assist devices.
In the ‘out of proportion’ situation, gradual lowering of the precapilliary pressure gradient in a (high) normal range should not result in a left ventricular and left atrial volume overload. The filling pressure should increase. A therapy that selectively targets the precapilliary pulmonary vascular disease could be considered in these cases. However, because of co-existing left ventricular dysfunction, a balanced decrease of the pulmonary and systemic resistance would be preferable (Figure 7).
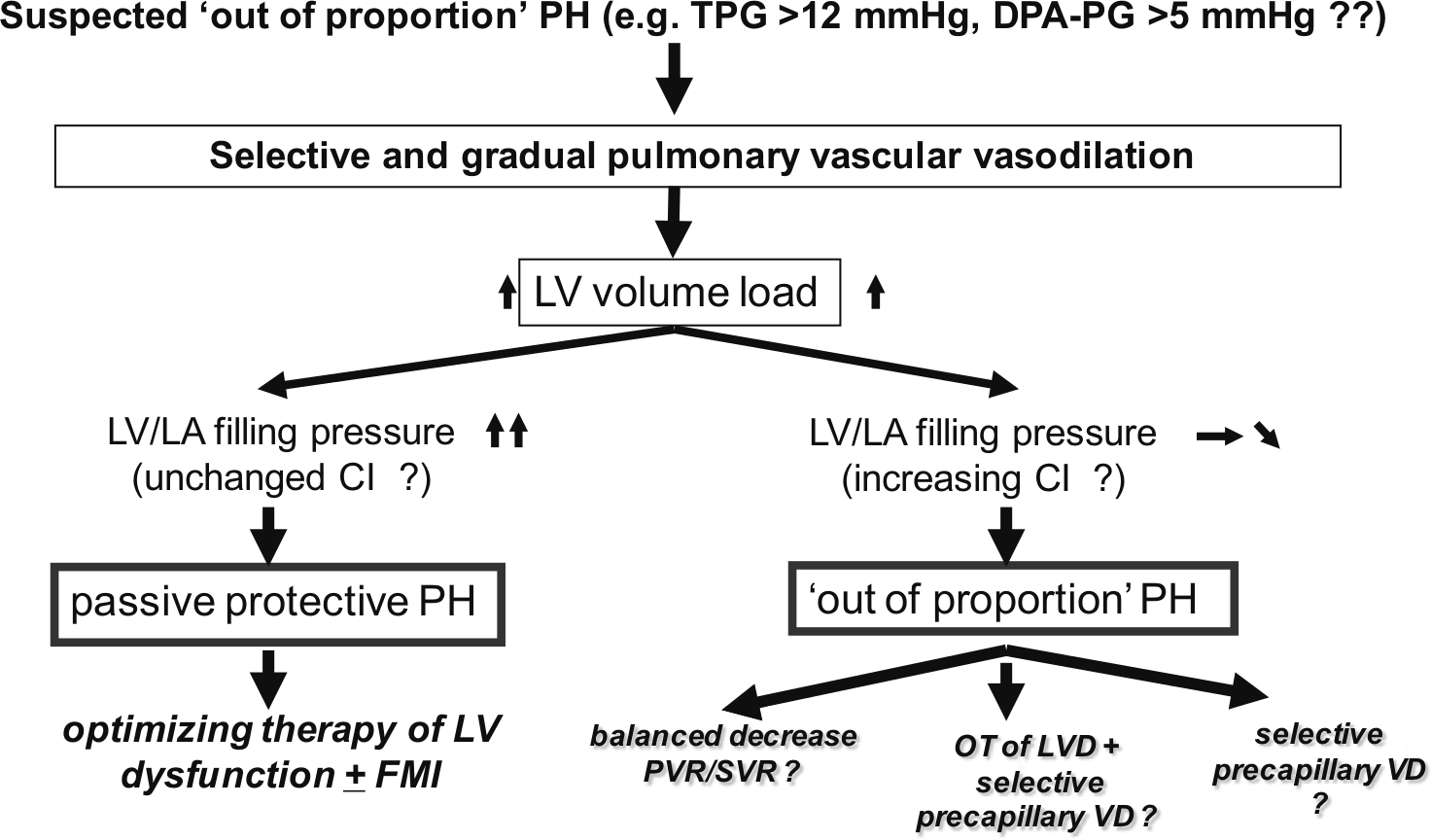
Hypothesis for alternative definition of passive protective versus ‘out of proportion’ pulmonary hypertension in left ventricular disease (TPG >12 mmHg) and proposed resulting therapy strategies.
Additional haemodynamic parameters, such as cardiac index and right ventricular filling pressure, have to be measured and critically evaluated during such a test procedure. It seems to be very useful to analyse not only filling pressure, but also the intrinsic diastolic function of the diseased ventricle, left-side pressure–volume loops (conductance catheter).
Recently, Naeije and colleagues [Naeije et al. 2012] proposed DPG-derived diagnostic algorithms (≤5 mmHg versus >5 mmHg) to distinguish between passive upstream transmission of left atrial pressure and increased PVR due to pulmonary vasoconstriction and/or pulmonary vascular structural changes. The goal of this algorithm is solely to identify precapilliary pulmonary vascular disease in different disorders such as heart failure, sepsis, hypervolaemia and hypovolaemia. However, the reaction of the diseased left ventricle on selective pulmonary vasodilatation is not integrated with this approach. From our point of view, not only is the isolated understanding of the pulmonary vascular pathology crucial for the evaluation of new and promising options to manage PH due to left heart disease, but also the development of a concept that integrates the (patho)physiology of the pulmonary vascular bed with the upstream and downstream right and left ventricular function and interaction (Figure 7).
Approach 2: Develop and use only therapeutics
The second approach would be to develop and use only therapeutics which lower the pulmonary vascular resistance and simultaneously unload the left ventricle in a very balanced manner.
Above a normal TPG (about 5–9 mmHg), a further differentiation in reactive protective PH or ‘out of proportion’ PH would not be necessary because of the simultaneous decrease of PVR and systemic vascular resistance (SVR). Different medications, originally approved in PAH (group I), with additional vasodilating effects on the systemic circulation have been examined in patients with systolic heart failure during the past few years. However, the results were very disappointing regarding both endothelin antagonists (bosentan, tezosentant, darusentan) and intravenous prostaglandin application (Flolan). In the Flolan International Randomized Survival Trial, the use of low dose epoprostenol was associated with excess mortality, perhaps because of the cyclic adenosine monophosphate (cAMP) dependent positive inotropic effects of the substance [Califf et al. 1997; Shan et al. 2012]. The endothelin receptor blockade in patients with systolic heart failure was associated with only neutral effects at best [Anand et al. 2004; Kalra et al. 2002]. It should be noted, however, that PH was not a mandatory inclusion criteria in all these studies.
The results of a number of pilot trials that examined the effects of a third class of PH-specific drugs, the PDE-5 inhibitors, are more promising. At this time, only sildenafil has been studied in patients with PH due to both systolic and diastolic heart failure. Both acute and chronic trials in patients with PH due to systolic heart failure have shown that sildenafil improves exercise capacity, ventilation efficiency, breathing patterns including exercise oscillatory breathing, and quality of life. These effects may be due to the beneficial effects on pulmonary vascular tone and RV function, reduction in PCWP, and direct modulation of the peripheral skeletal muscle ergoreceptor overstimulation [Lewis et al. 2007a, 2007b]. In addition, sildenafil may improve alveolar-capillary membrane conductance and gas exchange, suggesting an important role for the cyclic guanosine monophosphate (cGMP) downstream pathway in the protection of endothelial permeability [Guazzi et al. 2007, 2008; Yin et al. 2011]. Recently, the effects of a long-term treatment of one year with sildenafil were investigated in a randomized trial of 45 patients with PH and systolic heart failure [Guazzi et al. 2004]. In the sildenafil group, only LV ejection fraction and early diastolic tissue Doppler velocities (E′) significantly increased and a reverse remodelling of the left atrial volume index and LV mass was observed at six months and one year. In another pilot study in patients with PH and HFPEF, long-term therapy with sildenafil resulted in an improvement of LV diastolic function and cardiac index, as well as a marked reduction of RV dilatation and PAP [Guazzi et al. 2011]. Thus, the very promising haemodynamic short- and long-term effects point towards a balanced haemodynamic profile of sildenafil, lowering both the pulmonary and systemic vascular resistance [Katz et al. 2000; Lepore et al. 2005; Lindman et al. 2012].
Despite the encouraging findings, prospective multicentre controlled trials are urgently needed to better define the safety, tolerability and effectiveness of PDE-5 inhibitors in PH due to left ventricular disease. Such studies are especially necessary in the context of the disturbing results of recently published experimental studies. Nagendran and colleagues [Nagendran et al. 2007] and later Shan and colleagues [Shan et al. 2012] found that, in contrast to normal healthy right ventricles, PDE-5 was markedly upregulated in hypertrophied human right heart specimens. Interestingly, PDE-5 inhibition in a rat model of RV hypertrophy and also in human hypertrophied RV specimens resulted in an increase of intrinsic right ventricular inotropy. One explanation could be the suppressed protein kinase G activity in RV hypertrophy, so that the PDE-5 inhibitor induced cGMP increase could lead to inhibition of the cGMP sensitive PDE-3 with an increase of myocardial cAMP levels [Nagendran et al. 2007]. It should be noted, however, that markedly increased PDE-5 immuno-reactivity has also been detected in left ventricular cardiomyocytes of patients with dilated and ischemic cardiomyopathy [Pokreisz et al. 2009]. These findings have important implications because all therapies which lead to increased myocardial cAMP levels, such as PDE-3 inhibitors, epoprostenol and different catecholamines, were associated with excess mortality in patients with systolic heart failure [Felker and O’Connor, 2001a, 2001b].
In view of these data, European guidelines do not recommend treatments with targeted PAH drugs in patients with left heart failure and PH [Galie et al. 2009]. Instead, the main goal is to establish and maintain a guideline-oriented treatment of the underlying heart failure. Within the specific recommendations of the German Cologne Consensus Conference 2011, it was pointed out that in rare and justified exceptions in which a considerable precapilliary component is the key aspect of the disease and the clinical symptoms, PAH therapy may be considered after the patient has been informed in detail. If patients may be eligible for targeted therapy, the primary objective is the enrolment of patients in ongoing clinical trials. Large expert centres and patient associations (in Germany: pulmonale Hypertonie e.v.) will provide information on this [Rosenkranz et al. 2011b]. The uncritical use of targeted PAH drugs outside of the exceptional cases mentioned above is strongly discouraged.
Conclusion
PH due to left heart disease is the most common cause of PH and, when present, especially with right ventricular dysfunction, is associated with a worse prognosis. The most important determinants of PH due to left heart disease are elevated left ventricular filling pressures, mitral regurgitation and a lowered compliance of the left atrium. In all cases, the resulting elevated left atrial filling pressure causes a passive increase in pulmonary pressure. Passive PH could be superimposed by an active protective and, in some patients, by an ‘out of proportion’ elevated precapilliary pulmonary vasoconstriction and vascular remodelling which lead to a greater or lesser further increase of the PAP and consecutive right ventricular afterload.
Before considering a therapeutic intervention of PH due to left ventricular systolic or diastolic dysfunction, a reliable haemodynamic test or imagine method is needed to differentiate the ‘out of proportion’ PH from the reactive protective form. This seems necessary because the same pulmonary-selective vasodilative therapy would lead to very useful haemodynamic and clinical results in the frank ‘out of proportion’ situation, but possibly to left ventricular volume overload and consecutive pulmonary congestion in the reactive protective form. However, neither the proposed cutoff values of TPG nor PVR are suitable for discriminating between these two PH forms, because both are sensitive to flow, pulmonary vascular recruiting and compliance. Whether the flow insensitive gradient (DPG) between diastolic PAP and PCWP is more suitable for this purpose has to be demonstrated in future clinical investigations. In addition, not only is isolated understanding of its pulmonary vascular pathology crucial for the evaluation of new and promising options to manage PH due to left heart disease, but also the development of a concept that integrates the (patho)physiology of the pulmonary vascular bed with the upstream and downstream right and left ventricular function and interaction.
Footnotes
Authors’ note
Alexander Schmeisser and Hagen Schroetter contributed equally to this paper.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
A. Schmeisser: Remunerations for lectures for Actelion, Pfizer, BeyerSchering, and GSK. H. Schroetter: Remunerations for lectures for Lilly, Pfizer. R. Braun-Dulleaus: Remunerations for lectures for Novartis, BeyerSchering.
