Abstract
Exercise testing is an established noninvasive tool in cardiology used to diagnose and guide treatment in individuals with suspected or confirmed coronary artery disease. Owing to the wealth of information it provides, exercise testing is also being utilized to evaluate prehypertensive stages, characterize hypertension, assess tolerance to exercise and the efficacy of antihypertensive therapies, and predict target organ damage and cardiovascular risk. The literature on exercise tolerance tests is relatively limited since these studies are difficult to conduct although they represent a valuable test for evaluating the benefits of antihypertensive therapies beyond their blood-pressure-lowering efficacy at rest or during exercise. Such a setting can be immensely useful for the evaluation and for the differentiation of treatments, especially in patients with evident rises in systolic blood pressure and with concomitant diseases, who are at higher risk of stroke. Exercise-induced increase in systolic blood pressure from rest to peak exercise should therefore be used as the primary efficacy variable. There is growing evidence that central pressure is a better predictor for cardiovascular risk than peripheral blood pressure, since this variable takes into account the overall effect of vascular aging and increased arterial stiffness that age and other concomitant diseases may induce. It is also important to include central aortic blood pressure and biomarkers of hypertension and cardiac disease in the overall assessment.
Introduction
Exercise testing is being increasingly recognized as a useful tool for cardiovascular risk prediction of asymptomatic patients with normal or high normal blood pressure (BP). Data from the Framingham Offspring Study establish that an excessive rise in diastolic BP (DBP) during exercise and systolic BP (SBP) during the recovery period are related to increased risk of cardiovascular events, and also predictive of incident hypertension in a multivariable model that includes exercise duration and peak exercise [Flack et al. 1996; Hansson et al. 1992; Mundal et al. 1994; Singh et al. 1999]. In normotensive individuals, a systolic response during exercise (SBP ≥ 210 mmHg) is associated with increased left ventricular (LV) mass and risk of LV hypertrophy (odds ratio 3.16) [Molina et al. 1999]. This has been corroborated by results from the Framingham Group, where SBP of 150 mmHg at exercise levels of 5 metabolic equivalents (METs) or a change in SBP from rest to exercise levels of 5 METs was found to be a strong predictor of LV hypertrophy [Kokkinos et al. 2007]. Because LV hypertrophy is an independent risk factor for cardiovascular morbidity and mortality, this may partly predict the risk of systemic hypertension in these individuals [Bikkina et al. 1994; Koren et al. 1991; Levy et al. 1990].
An exaggerated response to exercise in patients with high normal BP may also be associated with some systolic dysfunction, which occurs before the development of LV hypertrophy [Mottram et al. 2004]. This condition may represent early hypertensive heart disease. These and several other studies add to the growing evidence of the predictive value of responses of exercise testing for the assessment of functional cardiovascular capabilities. Follow up of approximately 2000 middle-aged subjects over 16 years has correlated SBP early rise and pulse pressure during exercise with cardiovascular mortality [Mundal et al. 1994]. In addition, in hypertensive patients, baseline BP values are considerably higher than normal, exposing subjects to a continuous high cardiovascular stress. BP increases further during routine active life, making exercise testing an interesting model to assess the protective potential of antihypertensive treatment.
Several clinical studies using exercise tests have shown that antihypertensive treatments significantly blunt the SBP at all stages during exercise. However, the differences observed between drugs are limited, often so by the power of the studies [Czendlik et al. 1997; Duprez et al. 2007; Little et al. 2004; Rangemark et al. 1996; Sullivan et al. 1989; Warner et al. 1999; Williams et al. 2012]. Thus, these results should be considered indicative of a trend, requiring verification by more judicious and affirmative methods which can evaluate treatments under conditions of exercise-induced stress.
Exercise-induced increase in SBP, in addition to the stimulation of the sympathetic nervous system and consequently the increase in plasma catecholamines may also be partly mediated by the stimulation of the renin–angiotensin–aldosterone system (RAAS) system, due to angiotensin (Ang) II, the systemic levels of which increase during exercise [Aldigier et al. 1993] Ang II also impairs LV relaxation and increases LV diastolic pressures [Cheng et al. 1996]. Therefore, it may be relevant to evaluate the relative effects of RAAS blockers in an exercise setting. Angiotensin receptor blockers (ARBs), angiotensin-converting enzyme (ACE) inhibitors, and direct renin inhibitors (DRIs) form the mainstay of antihypertensive therapy blocking the RAAS pathway. Exercise-induced changes in BP have been used to compare the ARBs, candesartan, valsartan, and losartan with other antihypertensive agents. Candesartan was equally effective as verapamil (a calcium-channel blocker) [Little et al. 2004] and significantly more effective than ramipril (an ACE inhibitor) in blunting the exercise-induced increase in SBP [Ketelhut and Franz, 2006]. The SBP increases were larger after treatment with valsartan than with atenolol (the β-blocker) or the combination of atenolol and valsartan [Czendlik et al. 1997]. However, valsartan [Duprez et al. 2007] and losartan [Warner et al. 1999] were more effective than placebo in blunting the post-exercise increase in SBP.
The ACE inhibitor lisinopril was found to be as effective as atenolol in reducing the exercise-induced increase in BP [Rangemark et al. 1996]. Similarly, the ACE inhibitor fosinopril and the β-blocker propanolol were equally effective in reducing post-exercise BP [Sullivan et al. 1989]. A comparison of two different strategies of inhibiting the RAAS system between aliskiren and valsartan, direct renin inhibition and AT1 receptor antagonism, has provided similar results [Williams et al. 2012]. In almost all studies, treatment with antihypertensive agents including, but not limited to, enalapril, fosinopril, aliskiren and atenolol resulted in significantly decreased BP at rest and during exercise and alleviated the exaggerated BP response to exercise in patients with hypertension [Sullivan et al. 1988; Williams et al. 2012; Yamauchi et al. 1989]. Accordingly, RAAS blockers are often the first-line antihypertensive agents in physically active patients, especially those with concomitant metabolic risk factors such as diabetes or dyslipidemia [Chobanian et al. 2003].
Another important criterion to evaluate antihypertensive drugs is their duration of action since compliance with drug therapy is an issue for most therapies. Because studies have shown an inverse relationship between the number of daily doses and compliance with drug therapy, there is a need for effective antihypertensive agents with a potential for sustained efficacy over a 24 h dosing period, and beyond [Burnier, 2006; Claxton et al. 2001; Saini et al. 2009]. Even with once-daily doses, patients often miss their doses inadvertently or intentionally. Thus, sustained antihypertensive efficacy beyond 24 h has the potential for reducing the risk of cardiovascular morbidity and mortality associated with BP fluctuations over time in relation to missed doses [Elliott, 2004]. The benefit of medium-intensity physical exercise has been shown also for hypertensive subjects [Chanudet et al. 2006]. As medicated hypertensive subjects with a variable level of compliance to their medication exercise, the effect of one missed dose of medication was assessed during exercise showing that some drugs are protecting patients better than others against the sudden rise in BP, and that this is dependent on their longer duration of their antihypertensive effect as measured by biomarker kinetic during exercise [Düsing et al. 2011; Williams et al. 2012]. Combining missed dose and ‘exercise test’ in studies may allow the identification of drugs that provide sustained blunting of exercise-induced SBP elevation, amplifying differences between treatments as compared with resting conditions.
Recent guidelines recommend that management of hypertension should be tailored to an individual patient’s profile based on estimates of absolute cardiovascular risk [Mancia et al. 2007, 2009]. This risk is largely associated with the presence or absence of concomitant risk factors (including lipid profile, smoking habits, and the presence or absence of diabetes) and existing evidence for target organ damage (electrocardiography, renal damage). Antihypertensive therapy should aid the long-term objective of reducing morbidity and mortality associated with progression of cardiovascular diseases. These recommendations are especially relevant for patients >50 years of age, who constitute ~70% of the overall hypertensive population.
Hypertension has historically been evaluated based on the systolic and diastolic components of BP. However, several reports, pioneered by results from the Framingham study, emphasize the importance of controlling SBP, especially in older patients [Kannel, 2000]. Elevated SBP is regarded as an independent predictor of cardiovascular outcome, superior to DBP, especially in older patients. SBP rises as a result of arteriosclerosis, which develops with age, and results in progressive reduction in elasticity of the aorta the other large arteries. DBP is a function of peripheral resistance, which increases until the age of ~50 years, and then declines [Williams et al. 2008]. Consequently, as age progresses, there is a gradual shift from DBP to SBP and then to pulse pressure as a predictor of cardiovascular risk [Franklin et al. 2001; Sever, 1999]. This suggests that prognosis of hypertension would be improved by identifying treatments while focusing on SBP rather than on DBP [Hozawa et al. 2000]. It may be relevant to evaluate hypertensive treatments based on their effect on both sitting and standing BP, at rest and under conditions of stress. There is now enough evidence to support that central measurements of SBP and pulse pressure reflect cardiac afterload and perfusion. Also, a lack of normal differentiation of central aortic and brachial SBP indicates impending cardiovascular disease [McEniery et al. 2008]. This is confirmed by results from the Conduit Artery Function Evaluation Study (CAFÉ), where it was observed that antihypertensive drugs have differential effects on central and peripheral pulse pressures, which translates into different outcomes [Williams et al. 2006]. Despite this, most studies for evaluating antihypertensive drugs still rely only on brachial BP measurements. The consequences of arterial stiffening and vasculature compliance potential play a role in buffering the sudden rises in BP during exercise, and its value has been observed in the course of years [Lund-Johansen, 1991]. Thus, the evaluation of antihypertensive drugs including measurement of peripheral SBP and central aortic systolic pressure (CASP) warrants further investigations. Further to the development of devices for the noninvasive measurement of CASP, once calibrated with the brachial BP, they can be used for routine CASP as well as 24 h ambulatory BP measurements, in both routine clinical practice and in clinical trials. Practically, during exercise, the two arms will be used for simultaneous measurement of peripheral BP and CASP to obtain simultaneous measurements including peak exercise [Düsing et al. 2011; Williams et al. 2011].
With improved understanding of complex physiological mechanisms, it is evident that abnormalities in multiple biological pathways antedate the onset of hypertension and cardiovascular diseases. Accordingly, various biomarkers have been identified and are being validated as surrogate indicators of an individual’s physiology. Of these, adrenaline, plasma renin activity (PRA), and L-NMMA are of specific cardiovascular interest. The rise in BP during exercise in response to the increased demand for blood flow to muscle which is achieved via an increase in cardiac output and redistribution of blood flow via activation of the sympathetic nervous system and RAAS [Aldigier et al. 1993; Fallo, 1993]. During exercise, β-type natriuretic peptide (BNP) and asymmetric dimethyl-arginine (ADMA), despite being relevant biomarkers and surrogate endpoints in other cardiovascular pathologies, have been shown to play a marginal or nonexistent role [Campbell et al. 2011; Kjaer et al. 2004; Martinelli et al. 2010; Williams et al. 2012]. Captopril resulted in a profound reduction in Ang II levels at rest and during exercise, which did not translate to any significant effect on activity of the sympathetic nervous system in patients with essential hypertension. Adrenaline and noradrenaline levels remained unchanged during the study [Manhem et al. 1981].
Stress in essential hypertension causes increases in plasma adrenaline (epinephrine) accompanied by sodium retention and enhanced vascular reactivity [Strobel et al. 1994; Tuck, 1986]. Endogenous plasma adrenaline concentrations in resting adults are normally less than 10 ng/l, but may increase by 10-fold during exercise and by 50-fold or more during times of stress. Persistently elevated levels of adrenaline are associated with hypertension and predict a poor prognosis in these patients. However, the adrenaline levels vary over a 24 h period, which necessitates constant monitoring of levels for guiding clinical opinion and also present a wide variability between patients [Elam and Grassi, 2000]. Nevertheless, physiological increments of circulating adrenaline concentration increase PRA by an extra-renal β-receptor mechanism, which provides an alternate valid measure amenable to routine use in clinical trials [Johnson et al. 1979]. PRA is a measure of the bioactivity of renin, the first enzyme of the RAAS cascade. Elevations in PRA have been associated with increased risk of cardiovascular events [Alderman et al. 1997].
The classical endocrine effects of natriuretic peptides are to modulate fluid and electrolyte balance and vascular smooth muscle tone. These are complemented by autocrine and paracrine actions, which include regulation of coronary blood flow. BNP is a member of the natriuretic peptide family, and is a potent vasodilator with mitogenic, hypertrophic and pro-inflammatory properties that is upregulated in hypertensive diseases. An association between BNP increase and an abnormal increase in SBP in response to exercise has been reported in patients with asymptomatic aortic stenosis [Van Pelt et al. 2008]. The role of BNP during exercise has been studied in overt hypertensive patients before, during and after submaximal exercise, and in the recovery period. No changes were observed within each exercise test, before or after antihypertensive treatment [Williams et al. 2012].
Assessing endothelial function represents a way to measure the status of the compliance capacity of the arterial tree, and this is why it could be have been logical to test for markers of endothelial function. For assessing endothelial function during exercise, several biomarkers have been used, including ADMA and nitrates [Allen et al. 2006; Hambrecht et al. 1998]. ADMA is a derivative of L-arg and a regulator of the L-arginine nitric oxide (NO) synthesis pathway but the study of its kinetic at rest, during and after exercise found no changes, both before and after treatment [Williams et al. 2012].
Exercise testing as a tool for drug development in hypertension
The exercise stress test is a validated procedure for evaluation of the cardiopulmonary status. International guidelines recommend studies to determine the efficacy of an antihypertensive treatment, and exercise testing studies should follow such recommendations [Offerhaus, 1979]. When evaluating the efficacy of antihypertensive drugs in the controlled environment of a clinical study in the exercise setting, a minimum of 4 to a maximum of 12 weeks is what is most likely to be a standardized approach for such a test. Owing to the variability of response to exercise, it is required that the study participants should include a homogeneous representative group of hypertensive patients. Entry criteria should take into account the stress conditions and consequently increased SBP that patients might be exposed to, with foreseeable SBP level of up to 230–250 mmHg; therefore concomitant diseases allowed in the study should be carefully selected in the patients’ best interest (Table 1) [Fletcher et al. 2001; Williams et al. 2010]. In addition, it is well known that exercising causes long-term effects in sedentary patients, with exercise repetition leading to the so-called ‘training effect’ [Fletcher et al. 1996]. This represents a confounding factor that can be avoided only to a certain extent, by enrolling, if possible, patients confident in exercising only, and excluding patients starting different dietary regimens or weight-loss programs.
Basic inclusion and exclusion criteria for a study evaluating antihypertensive drugs by exercise testing.

AHA, American Heart Association; msDBP, mean sitting diastolic blood pressure; msSBP, mean sitting systolic blood pressure; NYHA, New York Heart Association.
In order to correctly evaluate a drug’s protective potential, eligible patients should ideally then enter a washout/placebo run-in period, which could also be flexible according to BP levels and the patients’ prior antihypertensive treatment [Sweeney et al. 1989]. In our group, when referring patient’s to such procedures for an assessment, that most likely is of little interest for the patient and more for the scientific community, use of a home BP monitoring device, with or without telemonitoring capabilities is recommended, in order to ensure safe withdrawal from previous antihypertensive treatments and also bring to notice any untoward rise in BP (≥180/110 mmHg) during the study period [Fletcher et al. 2001].
These study participants should perform a treadmill exercise test according to a standard protocol, and for referencing the most used is the Bruce protocol, under clinical supervision, so as to reach 85% of their age-predicted heart rate according to the Bruce exercise protocol described in Table 2 [Fletcher et al. 2001]. Those who qualify can be deemed eligible for study participation, and be randomized or allocated to treatment as per the intended study protocol and evaluated for SBP rise from rest to peak exercise during the exercise test. Additional assessments for sitting and standing BP, pulse waveforms, CASP, and biomarker should be recorded for the eligible patients, to enable valid analyses of the effects of study treatment [Williams et al. 2006, 2010, 2011]. Key potential assessments that we recommend while using this protocol are tabulated in Table 3.
The Bruce exercise testing protocol.
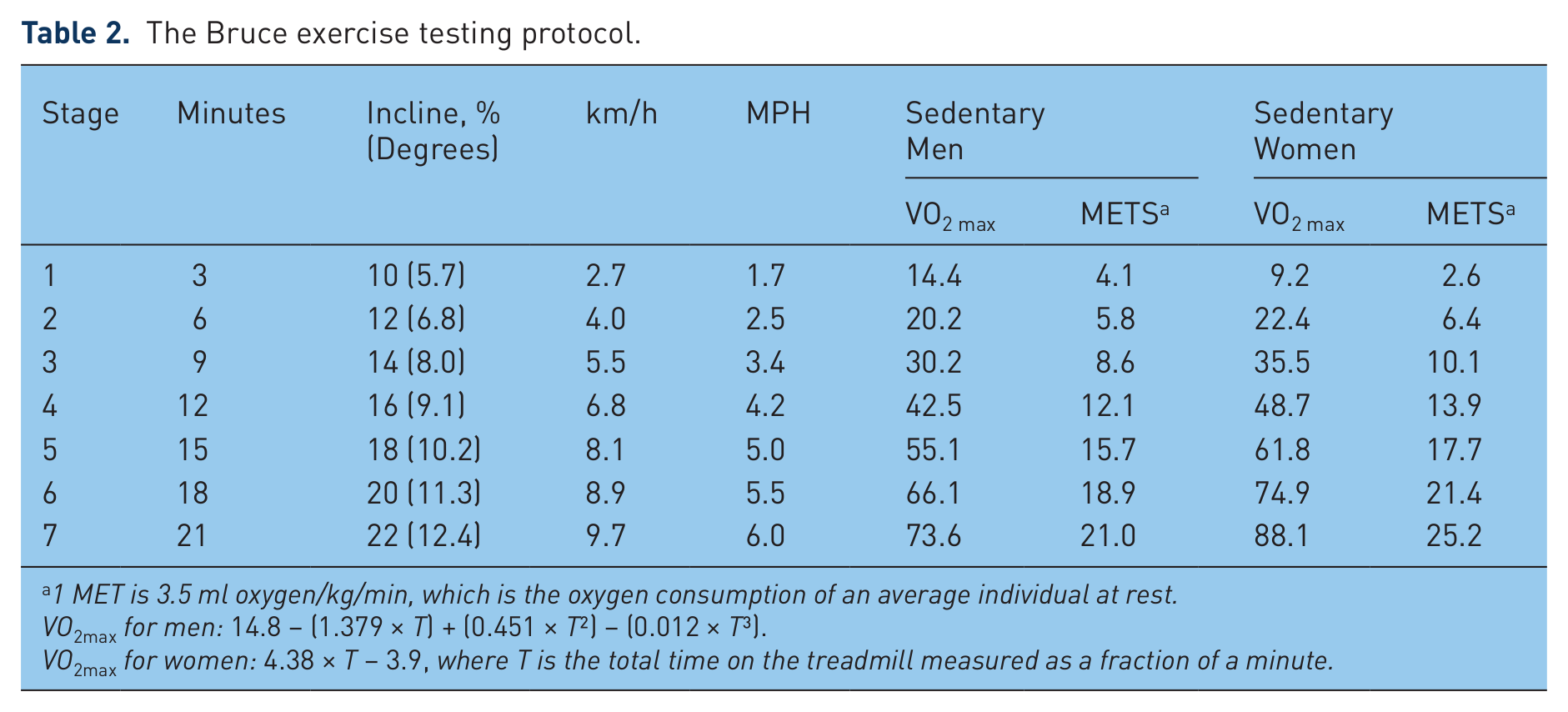
1 MET is 3.5 ml oxygen/kg/min, which is the oxygen consumption of an average individual at rest.
VO2max for men: 14.8 – (1.379 × T) + (0.451 × T2) − (0.012 × T3).
VO2max for women: 4.38 × T − 3.9, where T is the total time on the treadmill measured as a fraction of a minute.
Key assessments during the study period.

Index of SBP increase is the difference between peak SBP during exercise and at rest, divided by METs developed.
Augmentation Index (AIx) represents the timing of pressure wave reflection in relation to left ventricular systolic pressure. It is calculated as the ratio of the pressure difference between the shoulder of the wave and peak systolic pressure (δP) and the pulse pressure (PP) according to the formula: AIx = (δP/PP) × 100.
PP is the difference between SBP and DBP in mmHg.
It is recommended to initiate patients on a low dose of the drug or drug combination, followed by force-titration to the final treatment dose. Although we encourage proactive alarms to be raised by retained participants based on results of home BP measurement, we also recommend reasonably spaced, scheduled intermittent visits during the treatment period to ensure the safety of patients [Baguet and Mallion, 2002; Celis et al. 2005]. Patients with mean sitting SBP (msSBP) ≥180 mmHg or msDBP ≥110 mmHg at any time during the study period should be discontinued from the study. The end of the study period would warrant another round of a complete set of investigations and assessments, to compare change in functional capability from baseline to end of the study period [Fletcher et al. 2001]. Further, as BP variability over time contributes significantly to the overall outcome of any antihypertensive regimen, it might add more value to evaluate ambulatory BP measurements over a period of 24 h, and also to evaluate the effect of antihypertensive treatment after a missed dose [Dusing et al. 2012; Kabakci et al. 2010].
To ensure the safety of the study participants, it is necessary to check specific contraindications to exercise testing before commencing the test (Table 4). Such recommendations are in line with the practice guidelines specified by the American College of Cardiology/American Heart Association, to ensure that patients who are at increased risk due to exercise are excluded or discontinued from the study [Gibbons et al. 2002]. Furthermore, continuous ECG monitoring is necessary during the exercise test. A standard 12-lead ECG should be performed at screening, baseline, and at the end of the study or premature discontinuation visit with a validated device, to record sitting and standing resting ECGs. A short period of ECG recording during hyperventilation is also valuable for identifying changes resulting from hyperventilation rather than from coronary ischemia. During the exercise test, the ECG device should provide a continuous record of the heart rate, and a 12-lead ECG should be recorded intermittently [Gibbons et al. 2002].
General contraindications to exercise testing.

Relative contraindications can be superseded if the benefits of exercise outweigh the risks.
In the absence of definitive evidence, the committee suggests systolic blood pressure of >200 mmHg and/or diastolic blood pressure of >110 mmHg.
There are limitations that should be carefully considered when designing an exercise test study. Trained athletes are known for their cardiovascular fitness. However, hypertension and cardiovascular disease are reported in these individuals as well, albeit with lower incidence. Moreover, the ‘training effect’ as a result of a formal exercise schedule is also seen, and varies between individuals. Since these patients differ in their ECG and cardiovascular responses from untrained individuals, interpretation of their exercise test results requires special attention. This requires that normal and abnormal responses are determined according to gender, age, and physical fitness of individuals. Previous studies have shown that the relative heart rate, defined by the percentage of heart rate change from rest to peak activity, can normalize individual differences and provide more accurate estimates of exercise intensity in both athletes and nonathletes [Karvonen and Vuorimaa, 1988]. It is also acknowledged that the BP and heart rate responses during the post-exercise recovery phase provide valuable additional information regarding the cardiovascular status of individuals. Therefore, it is important to evaluate the SBP response to a graded exercise test based on relative heart rate increases, not only at rest and during exercise, but also at defined time points during the recovery phase. This approach can allow a comparison of individuals with varying fitness levels based on gender, age, and physical training [Miyai et al. 2002].
In conclusion, the exercise stress test may represent a unique tool to assess the effective protection potential of drugs against important increase in SBP in patients with hypertension while exercising. The study design elements proposed have the potential to establish exercise testing as a valid tool to assess the efficacy of antihypertensive treatments during stress conditions. Isolated systolic hypertension is common in elderly patients, especially in those with comorbidities such as chronic kidney disease and diabetes [Cheng et al. 2008; Os et al. 2006]. It is also associated with increased risk of incident heart failure and stroke in ambulatory older adults [Ekundayo et al. 2009]. Uncontrolled hypertension not only results in disease progression, but also significantly increases the risk of cardiovascular morbidity and mortality in these patients [Iyer et al. 2010]. However, most recommendations in comorbid patients are based on extrapolations from results in hypertensive patients without concomitant diseases, post hoc analysis, or prespecified subgroup analyses in these patient populations. The study design outlined in this article offers a valid methodology for evaluating the cardiovascular protective potential of antihypertensive therapy. It will be worthwhile to evaluate the potential benefits of therapy specifically in these subgroups of patients. The assessments and endpoints outlined here show promise for providing wide clinical relevance to patient outcomes in the future.
Footnotes
Acknowledgements
All authors participated in the development and writing of the paper, and approved the final manuscript for publication. The authors take full responsibility for the content of the paper and thank medical writers Shruti Agarwal (Novartis) for medical writing support and Dr Madhavi Dokku (Novartis) for editorial assistance, collation and incorporation of comments from all authors.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
Dr Patrick Brunel and Dr Fabio Baschiera were involved in the writing of this review; they are employees of Novartis and are therefore eligible for Novartis stock and stock options.
