Abstract
Objective:
This study examined comorbidity prevalence and general medication use among individuals with atrial fibrillation in the United States to convey a more comprehensive picture of their total disease burden.
Methods:
This was a retrospective, observational evaluation of responses to the 2009 wave of the annual Internet-based National Health and Wellness survey, which collects health data including epidemiologic data and information on medical treatment from a representative nationwide sample of adults in the United States. Responses were assessed to determine three measures of comorbidity: mean number of comorbidities, CHADS2 score reflecting stroke risk (0–6 points; low risk: 0; moderate risk: 1; high risk: ≥2), and scores on the Charlson Comorbidity Index, which is a measure of general comorbidity reflecting presence of a wide range of comorbidities.
Results:
Of the overall sample, 1297 participants reported having been diagnosed with atrial fibrillation. Almost all (98%) of the predominantly male (65.1%) and older (≥65 years of age, 65.7%) population with atrial fibrillation had at least one additional comorbidity, and 90% had cardiovascular comorbidities. On the Charlson Comorbidity Index, 44.9% of the respondents had scores of 1–2 and 20.5% had scores of 3 or higher. High risk for stroke, demonstrated by a CHADS2 score of at least 2, was present in 45% and moderate risk (CHADS2 score 1) in 36%. Of the respondents with atrial fibrillation, 71% reported current use of medication to manage the condition, but only 48% of individuals at high risk for stroke reported use of anticoagulation therapy. Of those who reported having common risk factors for stroke, the majority reported receiving prescription therapy for these conditions.
Conclusions:
The health burden carried by patients often extends far beyond atrial fibrillation. Physicians should carefully consider comorbidities and concomitant medications when managing patients with atrial fibrillation.
Keywords
Background
Atrial fibrillation (AF) is a common condition worldwide. It affects more than 2 million in the USA, and the most recent estimates of US prevalence are closer to 3 million [Naccarelli et al. 2009; Go et al. 2001]. As the most common sustained cardiac arrhythmia, AF contributes substantially to health burden, largely because of its association with an increased risk of stroke. Because prevalence increases with age, AF also predominantly affects an older population, three-quarters of which are older than 65 years. As a result, AF often adds to an already high health burden [Go et al. 2001]. Comorbidities are common in AF, affecting the majority of patients [Wu et al. 2005; Holstenson et al. 2011].
AF often develops subsequent to and in parallel with other conditions. It is therefore critical to understand the total disease burden associated with AF to improve treatment and health outcomes. Many common AF comorbidities increase the patient’s risk of stroke, and prevalence data for these have been reported. In patients from the National Registry of Atrial Fibrillation (NRAF), who were aged 65–95 years, had been discharged from hospitalization with an AF diagnosis, and were not receiving warfarin, congestive heart failure and hypertension were each seen in 56%, diabetes in 23%, and prior stroke or transient ischemic attack (TIA) in 25% [Gage et al. 2001]. Though large, international studies in AF populations have reported the prevalence of comorbidities in AF populations, relatively few studies have reported comorbidities in US AF populations, and the data generally suffer from datedness, lack of generalizability, study design limitations, or a combination of these factors [Wu et al. 2005; Benjamin et al. 1994; AFFIRM Investigators, 2002; Reiffel et al. 2010; Reynolds et al. 2010]. Furthermore, these studies typically restrict comorbidity data presented on stroke risk factors and other cardiovascular comorbidities, and data on general comorbidities are lacking.
The range of conditions reported in the literature is, in most cases, narrow and cannot convey the extent of comorbidity in patients with AF. This study was undertaken to examine the prevalence of a wide range of comorbidities, as well as general medication use, among patients with AF in the USA, to convey a more comprehensive picture of the total disease burden carried by patients with AF.
Methods
Study design
This was a retrospective, observational evaluation of responses to the 2009 wave of the annual National Health and Wellness Survey (NHWS), designed, managed, and administered by Kantar Health. The NHWS collects data on epidemiology, medical treatment, and health behaviors and outcomes. The survey is carried out annually over the Internet via a self-administered questionnaire. Respondents form a nationwide sample of adults (aged ≥ 18 years) who participate in a web-based consumer panel; all response data are stratified by gender, age, and race/ethnicity. As a result of this stratification, the demographic composition of the 2009 US NHWS sample is comparable to that of the US adult population as described in the March 2008 Current Population Survey of the US Census Bureau. All subjects provided informed consent, and the study was approved by the Essex Institutional Review Board (Lebanon, NJ).
Members of a consumer panel who had been recruited with opt-in emails, coregistration with panel partners, e-newsletter campaigns, and banner placements were approached to participate in the NHWS. Members of the consumer panel had explicitly agreed to join the panel, and each had registered with a unique email address. They had also completed a registration profile giving detailed demographic information.
Study measures
For this analysis, participant reports of demographic and socioeconomic characteristics, diagnosed diseases, and medication use were evaluated. For an assessment of respondents’ overall burden of comorbidity, the mean number of comorbidities, CHADS2 score, and the Charlson Comorbidity Index (CCI) were evaluated.
Participant responses to the NHWS were used to determine each respondent’s total number of comorbidities. Respondents’ reports of physician-diagnosed organ system comorbidities were also captured.
The CHADS2 score (0–6 points; low risk: 0; moderate risk: 1; high risk: ≥2) was calculated from participants’ self-reports of risk factors: congestive heart failure diagnosis, hypertension diagnosis, age at least 75 years, diabetes mellitus diagnosis, and report of prior stroke or TIA. Each participant was assigned one point for each of the factors, with the exception of prior stroke or TIA, which led to the assignment of two points.
Comorbidity burden was assessed with the CCI, which evaluates the presence of the following conditions: human immunodeficiency virus (HIV) or acquired immune deficiency syndrome (AIDS), metastatic tumor, lymphoma, leukemia, any tumor, moderate or severe renal disease, hemiplegia, diabetes, mild liver disease, ulcer disease, connective tissue disease, chronic pulmonary disease, dementia, cerebrovascular disease, peripheral vascular disease, myocardial infarction, and congestive heart failure. The CCI score is the sum of the weighted value of these conditions, so that higher total index scores indicate greater comorbid burden. Though the original CCI incorporated diabetes with end organ damage and moderate or severe liver disease into the score, this analysis used the adjusted CCI, which excludes these factors.
Statistical analysis
For categorical variables, data are reported in frequencies and percentages; continuous variables are reported in means and standard deviations.
Results
Demographics and patient characteristics
The sample used in this study was derived from 75,000 participants included in the 2009 wave of the NHWS. A total of 1297 participants (1.7%) from the overall NHWS sample reported having been diagnosed with AF and were included in the analyses.
Participants were predominantly men (65.1%) with a mean age of 64.9 years; 93.7% had health insurance (Table 1). The majority of patients were older: 65.7% were at least 65 years of age and 18.4% were at least 75 years of age. A large majority of participants were white (92.8%), and a majority were not working (74.2%). Approximately half of the participants were college educated (47.3%), and almost as many had annual household incomes of US$50,000 or more (44.4%). A total of 13.4% of respondents smoked, 56.0% engaged in vigorous exercise for at least 20 min once per month, and 59.8% drank alcohol. Almost half were obese (43.2%). The mean time since diagnosis of AF was 9.0 years.
Demographics and patient characteristics.

Source of insurance: Medicare, 72%; private, 42%; other, 23%. Participants may have been covered with more than one type of insurance. AF, atrial fibrillation; CCI, Charlson Comorbidity Index; SD, standard deviation.
Prevalence of comorbidities in patients with atrial fibrillation
The most common comorbidities were other cardiovascular conditions (90%), followed by urologic conditions (62%), pain-related conditions (61%), respiratory conditions (42%), including chronic obstructive pulmonary disease in 11% and asthma in 10%, and gastrointestinal conditions (41%) (Figure 1). The most prevalent cardiometabolic comorbidities were hypertension (66%), hyperlipidemia (57%), arrhythmia (other than AF) (37%), diabetes (29%), and history of myocardial infarction (21%). For conditions that comprise the CHADS2 score, 19% reported congestive heart failure, 13% reported stroke, and 9% reported TIA.

Prevalence of comorbidities by organ system in patients with atrial fibrillation.
The distribution of numbers of comorbidities, CHADS2 scores, and CCI scores indicated a substantial comorbidity burden among patients with AF (Figure 2). Almost all respondents (98%) had at least one comorbidity in addition to AF (Table 2). Approximately 63% of respondents had four or more comorbidities in addition to AF, while half had five or more comorbidities. As a measure of general comorbidity burden, the CCI indicated that 35% of respondents had scores of 0, nearly half (45%) had scores of 1–2, 15% had scores of 3–4, 3% had scores of 5–6, and 2% had scores of at least 7 (Figure 2). As assessed with CHADS2 score, high risk for stroke (CHADS2 score ≥ 2) was seen in almost half (45%) of respondents and moderate risk (CHADS2 score, 1) in 36%. The mean (± standard deviation) CHADS2 score was 1.59 (± 1.1).

Distribution of Charlson Comorbidity Index (CCI) scores and CHADS2 scores.
Number of diagnosed comorbidities in addition to atrial fibrillation.

Ever diagnosed.
Prevalence of medication use in patients with atrial fibrillation
Of participants reporting a diagnosis of AF, 71% reported current use of a prescription medication for AF, and 43% reported use of an anticoagulant (warfarin/Coumadin (Bristol-Myers Squibb, Princeton, New Jersey), or heparin). Of those at high risk for stroke (CHADS2 score ≥2), only 48% reported use of an anticoagulant. Rates of prescription medication use among those reporting diagnoses of common stroke risk factors (hypertension, diabetes mellitus, myocardial infarction, congestive heart failure, and prior stroke) are reported in Figure 3. High rates of prescription medication use were also reported among those reporting other conditions. Of 57% reporting a diagnosis of hyperlipidemia, 50% were using a prescription medication; of 37% diagnosed with an arrhythmia, 29% were using a prescription medication. Of those at moderate risk for stroke (CHADS2 score = 1), less than half reported anticoagulant use, as did approximately half of those at high risk for stroke (CHADS2 score ≥ 2) (Figure 4).
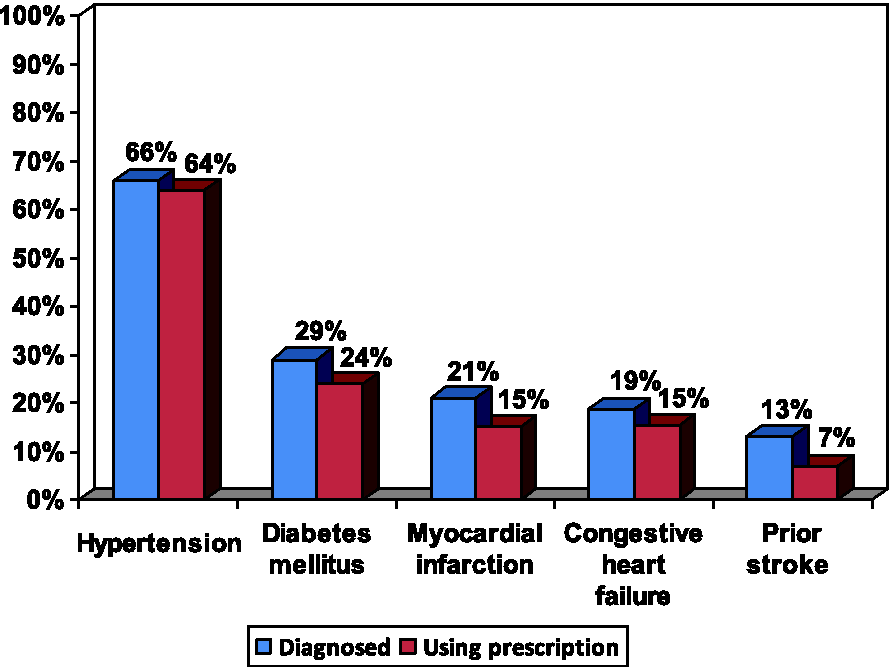
Prescription medication usage in patients with atrial fibrillation by stroke risk factor.
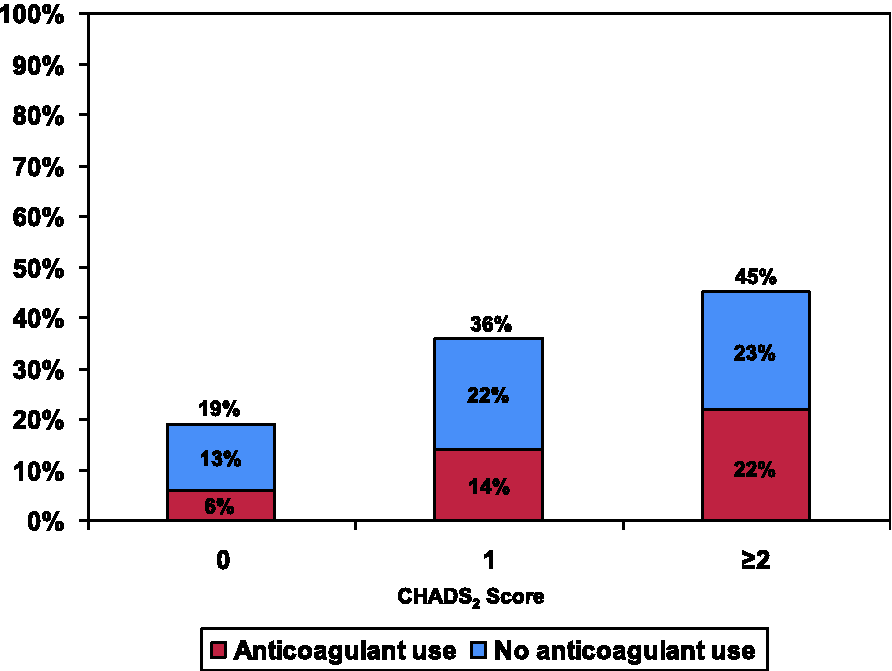
Anticoagulant medication usage in patients with atrial fibrillation by CHADS2 score.
Discussion
The study population of individuals with AF showed a high level of comorbidity burden. Almost 98% of NHWS respondents reported at least one comorbidity in addition to AF, while almost two-thirds reported at least four comorbidities. The comorbidity level was also high when assessed by CHADS2 score, a marker of stroke risk; almost half had scores of at least 2. General comorbidity (e.g. HIV/AIDS, cancer) as assessed by CCI also showed high levels; 45% of patients had CCI scores of 1–2, while 21% had scores of 3 or higher. Though outcomes associated with CCI in the AF population have not been identified, the general impact of this comorbidity level has been evaluated in a previously studied population with ischemic heart disease, among whom CCI scores of 1–2 were associated with a 10% mortality rate, more than three times as high as the mortality rate found in those with CCI scores of 0 [D’Hoore et al. 1996]. Scores of 3–4, demonstrated in 15% of the NHWS respondents, were associated with a mortality rate of 19%; scores of 5 and higher, found in 5% of NHWS respondents, were associated with mortality rates exceeding 30%.
The finding of multiple comorbidities in an older population is common [van den Bussche et al. 2011; Wolff et al. 2002]. Rates of 65–73% have been reported for two or more conditions; for three or more conditions, reported rates are 43–62%. The percentage of NHWS respondents reporting two or more conditions in addition to AF (91%) is well above the highest reported percentage having three total comorbidities found in general populations of older people. Likewise, the percentage reporting three or more comorbidities in addition to AF (78%) tops the 24–49% reported to have four total comorbidities in general populations of older people. The number of comorbidities identified in an AF population has not previously been reported, but these comparisons with data from general populations demonstrate the comparatively high rate of comorbidities in this population. Furthermore, the number of comorbidities typically increases with age in older populations [Wolff et al. 2002; Uijen and van de Lisdonk, 2008]. This group of NHWS respondents, with a mean age of 65 years, was relatively young for an older population, and for an AF population.
Many of the conditions identified in this population have been identified in other studies reporting comorbidities in patients with AF. The rates of most of these conditions, as identified in the NHWS, are generally similar to rates found in the NRAF participants. One exception is congestive heart failure, which at 56% was more common in the NRAF than in the NHWS cohort (19%), perhaps due to the younger age of NHWS respondents (mean age: NHWS, 65 years; NRAF, 81 years). In the Framingham cohort, among participants who developed AF (mean age: men, 72 years; women, 75 years), congestive heart failure was found in 20.6% of men and 26.0% of women; corresponding rates for other conditions were 44.1% and 51.7% for hypertension, 16.3% and 15.5% for diabetes, and 25.5% and 13.0% for myocardial infarction [Benjamin et al. 1994]. These rates are also all relatively similar to those identified by the NHWS, with a mean age of only 65 years.
Additional noninterventional studies provide varying levels of support for the theory that age is an important influence on comorbidity levels. The mean age was more similar to the age of the NHWS population in the The Fibrillation Registry Assessing Costs, Therapies, Adverse events and Lifestyle (FRACTAL) registry, whose North American population (US 80%) had a mean age of 66 years [Reynolds et al. 2010]. Baseline prevalence of most conditions reported in the newly diagnosed patients was typically lower than the rates in NHWS respondents: hypertension, 48% (versus 66% in NHWS), diabetes, 11% (versus 29%), and previous myocardial infarction, 14% (versus 21%); only congestive heart failure, at 23% (versus 19%), was slightly higher. The criterion for a new diagnosis of AF in the FRACTAL registry may have had some influence on the rates of comorbidities. In contrast, a database study by Wu and colleagues that was performed in a privately insured US AF population under 65 years of age found that prevalence was relatively low (<5%) for specific risk factors such as heart failure, myocardial infarction, and stroke; hypertension prevalence was not reported [Wu et al. 2005]. These study findings in a comparatively young population provide support for an effect of age on rates of cardiovascular comorbidities.
Although cardiovascular comorbidities are commonly found in patients with AF, they do not represent the total comorbidity burden of any patient population. The Wu study included data on comorbidities affecting other systems. Though providing a broader range of concomitant conditions than is reported in other registries and database studies, the analysis grouped data into system classes such as ‘systemic conditions’ and ‘associated general symptoms’ that limit the ability to determine the nature and extent of the comorbidity burden. Presentation differences also limit the ability to compare NHWS data with data from the FRACTAL registry [Reynolds et al. 2010]. Rates of specific respiratory comorbidities (chronic obstructive pulmonary disease 9%; asthma 6%) in FRACTAL were similar to those reported in the NHWS survey (chronic obstructive pulmonary disease 11%; asthma 10%).
Large clinical trials have also identified comorbidities in AF populations, again focusing on stroke risk factors and cardiovascular comorbidities. However, these studies are typically designed to present data on patients with specific risk levels. The Atrial Fibrillation Follow-up Investigation of Rhythm Management (AFFIRM) investigators evaluated comorbidities in a population enrolled from US and Canadian sites between 1995 and 1999, which was limited to patients with at least one risk factor other than AF for stroke or death [AFFIRM Investigators, 2002]. The participants, not surprisingly, showed unusually high comorbidity levels, with 71% having hypertension. The rate of hypertension in another study limited to patients with AF and at least one stroke risk factor, ACTIVE W, was similarly high, at 82–83% [ACTIVE Writing Group of the ACTIVE Investigators et al. 2005]. The rate of heart failure, another stroke risk factor, was also high at 30–31%. Rates of other comorbidities were more in line with the findings of the NHWS: 17–18% had a history of myocardial infarction, and 21% had diabetes. The rate of history of stroke or TIA (15%) was only slightly higher than the NHWS-identified rate (13%), despite the criteria for selection of the ACTIVE W population.
Therefore, the NHWS data cannot easily be compared with figures found in the literature. There is a paucity of recent data in US populations with AF. Figures (in patients recruited in 2003–2004) indicate high rates of many comorbidities but because the population was selected for an elevated risk of stroke, it is not comparable to a general AF population [ACTIVE Writing Group of the ACTIVE Investigators et al. 2005]. Similar limitations affect many other available studies, whether registries, databases, or clinical trials. The focus of participant criteria limits the ability to duplicate the real-world environment. For example, the impact on comorbidity rates of restricting participants to those not given warfarin, applied in the NRAF, is unclear. The data restrictions in the available literature increase the value of the NHWS survey data. However, it is important to note that the comparatively young survey population cannot closely approximate a general AF population.
Despite their relative youth, the NHWS population showed a substantial comorbidity burden both in cardiovascular and noncardiovascular comorbidity. A large majority (90%) of participants with AF in this analysis had cardiovascular comorbidities, many of which are risk factors for development of AF. Previous myocardial infarction was reported in 21% and physician-diagnosed hypertension in 66%. Forty-three percent were obese and conditions related to obesity were also common, with hyperlipidemia reported in more than half and diabetes in almost one-third.
Many of the reported comorbidities are also risk factors for stroke in AF. Congestive heart failure was reported in 19%, hypertension in 66%, diabetes in 29%, and prior stroke or TIA in 13%. Eighteen percent of the population were aged 75 years and over, which is an additional risk factor. Anticoagulation is recommended in the American College of Cardiology/American Heart Association guidelines for all high-risk patients with AF and some moderate-risk patients with AF who can safely receive anticoagulant therapy [Goldstein et al. 2011]. Despite the stroke risk factors identified in the NHWS respondents and a median CHADS2 of almost 2, only 43% of the population reported receiving anticoagulants, and the 48% rate of anticoagulation among survey participants with CHADS2 scores indicating high risk of stroke contrasts with the 55% found by Goto and colleagues in a corresponding population [Goto et al. 2008]. Likewise, among the subset of patients with AF with a CHADS2 score of 0, 32% reported receiving anticoagulant therapy, while a 45% rate of anticoagulation at this risk level was reported by Goto and colleagues. Though other researchers have identified inadequate levels of oral anticoagulant use in higher-risk patients, they have typically not been this low, with rates ranging from 46% to 64% in AF populations consisting of patients entirely or predominantly showing at least one risk factor [Lakshminarayan et al. 2006; Waldo et al. 2005; Abel-Latif et al. 2005]. Among the NHWS respondents, the reasons why anticoagulation therapy was not being used were not identified, but it is possible that comorbidities and their associated treatments may have interfered with AF treatment selection.
The comorbidities found were often treated with medications, potentially complicating the patients’ pharmacotherapy. The majority (71%) of patients were taking medication for AF. Most of the patients diagnosed with the most common comorbidities, that is, hypertension, hyperlipidemia, arrhythmia other than AF, and diabetes, were taking medication for the condition, including almost all of those diagnosed with hypertension. Drug–drug interactions and adverse drug reactions are frequent in this setting.
Collection of data on comorbidities that were not stroke risk factors allowed identification of additional conditions that may add to the overall burden of AF in a patient population, while complicating AF treatment by incurring the need for concomitant medications. Urologic conditions were reported in a majority of the patient population (62%), as were pain conditions (61%). As with numbers of comorbidities, the comorbidity patterns in this AF population to some degree reflect those identified in general populations of older people. Hypertension, hyperlipidemia, and diabetes have all been reported as among the most common conditions in Medicare populations, while pain conditions such as low back pain and osteoarthritis are also common [van den Bussche et al. 2011; Schneider et al. 2009].
The study findings are limited by the self-reported nature of the collected data. Recall bias may have introduced additional error into the observed associations. In addition, the use of a self-reported Internet tool excludes less computer-literate individuals and could potentially have added bias to the study results. Furthermore, the relatively young age of the respondents may limit the generalizability of results to the general AF population.
Conclusions
In this study, the majority of patients with AF surveyed reported a high comorbidity burden, reflected in the number of comorbidities, CHADS2 score of stroke risk, and CCI, an index of general comorbidity. A substantial majority of patients with AF also had other cardiovascular diagnoses such as hypertension, congestive heart failure, or myocardial infarction. Prevalence of stroke risk factors (congestive heart failure, hypertension, age ≥ 75 years, diabetes, prior stroke) was also high in this population, despite the group’s relative youth and activity level. Despite the level of stroke risk reflected in the CHADS2 score, only 43% of the population reported receiving the anticoagulant therapy recommended in guidelines. The comorbidity burden extended into several other organ systems (e.g. urologic, respiratory, and gastrointestinal), and many of these comorbidities were being actively treated. The healthcare burden of patients with AF often extends far beyond AF. Physicians should carefully consider comorbidities and concomitant medications when managing patients with AF.
Footnotes
Acknowledgements
The authors thank Tania Vila, PharmD, and Elizabeth Villeponteaux, MA, from PPD, Inc, for writing assistance.
Funding
This work was supported by Janssen Scientific Affairs, LLC.
Conflict of interest statement
JCL, SHM, JRS, AAP, and WWN are employees of Janssen Scientific Affairs and have received stock or stock options from Johnson and Johnson. HJG and MdD are employees of Kantar Health, to whom Janssen Scientific Affairs provided consulting fees for work on the design of this study, analysis of data, and review of the manuscript.
