Abstract
Keywords
Introduction
The study of the pathophysiological mechanisms of poisoning and molecular characterization of toxins from Bothrops jararaca venom, resulted in two major relevant scientific contributions: the discovery of the potent vasodilator, bradykinin (Bk) [Rocha et al. 1949], and peptides that display Bk-potentiating activity [Ferreira, 1965; Ferreira et al. 1970a] and inhibition of somatic angiotensin I-converting enzyme (ACE) [Ferreira et al. 1970b]. These peptides were not only useful in validating ACE as a target to treat human hypertension, but also as model for captopril development, the first commercial ACE inhibitor [Ondetti and Cushman, 1981; Ondetti et al. 1977].
Later, plenty of bradykinin potentiating peptide (BPP) sequences from crude venom and tissues of B. jararaca were identified using different approaches, such as functional assays followed by mass spectroscopy and molecular biology tools [Hayashi et al. 2003; Ianzer et al. 2004; Ondetti et al. 1977]. Typically, BPPs found in the B. jararaca venom [Ferreira et al. 1970a; Ianzer et al. 2004] and brain [Hayashi et al. 2003] are oligopeptides of 5 to 14 amino acid residues characterized by pyroglutamyl (<Glu) residue at the N-terminus and a proline residue at the C-terminus positions. Most of these peptides share common features, such as many proline residues in the middle of its structure and tripeptide Ile–Pro–Pro at the C-terminal portion. However, there are exceptions for short BPPs, such as BPP-5a (<Glu–Lys–Trp–Ala–Pro) [Ianzer et al. 2004].
In vitro assays showed that BPPs are able to distinguish between the two catalytic sites of the ACE [Cotton et al. 2002] and provide different levels of Bk-potentiating activity [Hayashi et al. 2003; Ianzer et al. 2004]. However, studies showed that similarities in the amino acid sequences are not related to Bk-potentiating activity [Hayashi et al. 2003; Ianzer et al. 2007].
Many studies suggested that the mechanisms of Bk potentiation may not be entirely due to ACE inhibition [Camargo and Ferreira, 1971; Greene et al. 1972; Hayashi et al. 2003; Mueller et al. 2005]. Mueller and colleagues using BPP-9a and analogs demonstrated that ACE inhibition and the Bk-potentiating activity might be evoked by different mechanisms [Mueller et al. 2005].
Concerning the Bk potentiation and ACE inhibition activities, we demonstrated the opposite effects for similar BPPs [Ianzer et al. 2007]. Despite these differences, BPP-7a and BPP-10c, which contain the motif Ile–Pro–Pro, produced a sustained antihypertensive effect in spontaneously hypertensive rats (SHRs) unrelated to their ability to inhibit somatic ACE (sACE) or to potentiate Bk in vivo [Ianzer et al. 2007]. Recently, biodistribution studies found high concentrations and permanence of BPP-10c in mice kidneys, even when co-administered with a commercial ACE inhibitor [Silva et al. 2008]. Guerreiro and colleagues [Guerreiro et al. 2009] demonstrated that argininosuccinate synthetase (AS) from mice kidneys is the major binding protein for the BPP-10c. This enzyme represents a potential limiting step in nitric oxide (NO)–citrulline cycle [Solomonson et al. 2003]. In vitro, BPP-10c was able to activate the AS and its antihypertensive effect was partially reverted by an AS inhibitor. These results showed that AS plays an important role in the antihypertensive effect evoked by this decapeptide [Guerreiro et al. 2009].
The short pentapeptide BPP-5a shows a high susceptibility to the cleavage by proteolytic enzymes and does not present the Ile–Pro–Pro motif at the C-terminal [Cheung and Cushman, 1973; Ferreira et al. 1970a]. This study was undertaken in order to ascertain whether BPP-5a has antihypertensive activity and to evaluate the mechanism of its cardiovascular effects.
Methods
Reagents
BPP-5a (<Glu–Lys–Trp–Ala–Pro), Bk, angiotensin I (Ang I), angiotensin II (Ang II), acetylcholine, L-NAME and HOE-140 were purchased from Bachem Chemical Co (USA). The peptides were dissolved in isotonic sterile saline (0.9% NaCl; Asterflex, Brazil) just before use.
Experimental animals
Experiments were carried out in male SHRs (280–350 g), the strain control Wistar rats (WTs; 250–320 g) and C57BL/6 mice (20–25 g) bred at the animal facility of the Biological Science Institute (CEBIO, Universidade Federal de Minas Gerais, Minas Gerais, Brazil). The animals had free access to food and water and were submitted to a light–dark cycle (12 hours each) before the preparation for the experiments. All experimental protocols were performed in accordance with the Guide for the Care and Use of Laboratory Animals as adopted and promulgated by the United States National Institutes of Health. In vivo experimental procedures were performed in accordance with the guidelines established by our local institutional animal welfare committee (CEUAIB/Butantan Institute, protocol 520/08).
AS activity assay
The effect of BPP-5a upon AS activity was determined according to Guerreiro and colleagues [Guerreiro et al. 2009]. The reaction is based on the accumulation of the product pyrophosphate as inorganic phosphate, following cleavage with pyrophosphatase. AS (1 µg) was added to the reaction buffer (20 mmol/l Tris-HCl, pH 7.8, 2 mmol/l ATP, 2 mmol/l citrulline, 2 mmol/l aspartate, 6 mmol/l MgCl2, 20 mmol/l KCl and 0.2 units of pyrophosphatase) to a final volume of 0.2 ml. We incubated 0.5 µmol/l of BPP-5a at 37°C in 96-well microtiter plates, and the reactions were stopped after 30 minutes by the addition of an equal volume of molybdate buffer (10 mmol/l ascorbic acid, 2.5 mmol/l ammonium molybdate, and 2% sulfuric acid). Accumulation of phosphate was determined by spectrophotometry at 650 nm and concentration was extrapolated from a standard curve of inorganic phosphate.
In vitro receptor autoradiography of 125I-angiotensin II binding to mice kidneys
Mice (C57BL/6) were decapitated and their kidneys were rapidly removed. Kidneys of mice were snap-frozen in cooled isopentane (−32°C). Sections (16 µm) were serially cut, starting from the central area of the kidney (HM 505N; Microm), mounted on gelatin-coated slides, and dried at 4°C, before the assay.
The binding assay was performed as described previously [Santos et al. 2003]. Sections were incubated with high-performance liquid chromatography (HPLC)–purified radioligand. Assay buffer with 5 × 10−10 mol/l 125I-Ang II and 10−5 mol/l of PD123319 or 10−8 and 10−6 mol/l of BPP-5a. After incubation, sections were rinsed in 0.05 mol/l Tris-HCl, pH 7.4 (3 × 30 seconds), and in distilled water (1 × 20 seconds), dried at room temperature, and exposed to autoradiographic film (Fuji Image Plate) for 24 hours. After exposure, films were scanned and quantified by densitometry (Bio-Imaging Analyzer; Fujifilm). Illustrations were made with Osiris Imaging software (version 3.12).
Cell culture
Commercially available cell lines of Chinese hamster ovary (CHO), obtained from the American Type Culture Collection, Manassas, VA, were used for cell culture experiments. Cells were cultured for 3 days or more as a monolayer (94/16-mm Petri dish) in the culture medium recommended for each cell type.
Binding studies in cell culture
Competition experiments were performed with AT1 receptor-transfected cells incubated with 125I-Ang II (0.4 nmol/l) in 24-well plates for 60 min at 4°C in 300 µl of serum-free medium (Dulbecco’s modified eagle medium [DMEM]) supplemented with 0.2% BSA, 0.005% bacitracin, 100 µmol/l phenylmethylsulfonyl fluoride, and 500 µmol/l o-phenanthroline. Competition experiments were performed by pre-incubating the cells with Ang II (10−11 to 10−6 mol/l) or BPP-5a (10−12 to 10−5 mol/l). After two washes with ice-cold serum-free DMEM, cells were disrupted with 0.1% Triton X-100 in water at 22–24°C. Bound radioactivity in the cell lysate was measured in a γ-counter (1275 MINIGAMMA LKB Wallac). Experiments were made in duplicate (n = 3–6 for each peptide). Ang II was labeled with 125I by the chloramine T method and purified by HPLC, as described previously [Pinheiro et al. 2004].
Imaging of intracellular NO production in CHO and Mas, B1 and B2 transfected CHO cells
Experiments were conducted as described previously [Carvalho et al. 2007; Sampaio et al. 2007]. For fluorescence experiments, confluent cells between the second and fourth passages were plated in six-well plates. CHO-transfected and CHO-nontransfected cells were preincubated in freshly prepared Hanks’ balanced salt solution (HBSS) containing 10−6 mol/l 4,5-diaminofluorescein-diacetate (DAF-2DA; Molecular Probes) for 20 minutes. After washing with HBSS, cells were incubated with 10−8 to 10−6 mol/l of BPP-5a or 10−8 and 10−6 mol/l of agonist for 30 minutes at 37°C in a humidified incubator under an atmosphere with 5% CO2. Control cells were incubated in the same media without any drug. As a negative control, cells were incubated in media lacking DAF-2DA. After the incubation time, the medium was discarded, and the cells were then washed in PBS and cover slipped using 10% glycerol (Tris, pH 7.4). Fluorescent images were obtained using a Zeiss 510 meta laser scanning confocal microscope equipped with an oil-immersion objective lens (×63).
Blood pressure recording in rats
Arterial pressure measurements
The cardiovascular parameters, pulsatile arterial pressure (PAP), mean arterial pressure (MAP), and heart rate (HR) were monitored using a solid-state strain gauge transducer connected to a computer through a data acquisition system (MP 100; BIOPAC Systems, Inc, Santa Barbara, CA, USA). The PAP, MAP, and HR were monitored simultaneously during experiments in different monitor channels and recorded for late analysis.
Effect of BPP-5a on blood pressure of conscious rats
This assay was performed according to Ianzer and colleagues [Ianzer et al. 2007]. Under anesthesia with tribromoethanol (2.5 g/kg) i.p., 20 hours before the experiment polyethylene catheters were placed into the abdominal aorta through the femoral artery and vein to measure cardiovascular parameters and to inject drugs, respectively. After recovery from anesthesia, the rats were kept in individual cages with free access to water and chow until the end of the experiments.
Cardiovascular parameters were initially followed for 1 hour (baseline period). Subsequently, intravenous bolus injection of the BPP-5a or vehicle (NaCl 0.9%) in a total volume of 0.5 ml was erformed. BPP-5a doses of 710, 71, 14.2, 2.37 and 0.47 nmol/kg were used in SHRs (n = 5–6 for each dose) and doses of 71 and 0.47 nmol/kg were used in WTs (n = 5 for each dose), as a control for SHRs. The cardiovascular parameters were monitored continuously for 6 hours after injection. MAP and HR values were sampled every 2 minutes during the whole experiment.
Potentiation of Bk hypotensive effects and inhibition of Ang I pressor effect during cardiovascular effects of the BPP-5a in conscious SHRs
Surgical procedures were the same as described above. Cardiovascular parameters were monitored for 40 minutes (baseline period). Following, intravenous bolus injection of Bk (0.5 and 1.0 µg) and Ang I (20 and 40 ng) in a total volume of 0.1 ml were made to obtain the control responses. BPP-5a (2.37 nmol/kg) or saline (0.9%) in a total volume of 0.5 ml were then administrated in SHRs (n = 4–5). Bk (0.5 µg) was given at 10 and 210 minutes; and Ang I (40 ng) was administered at 20 and 220 minutes after BPP-5a or vehicle injection.
Indirect systolic blood pressure measurements
For 4 days, SHRs were acclimated to restraint and tail-cuff inflation. After the adaptation period, rats were submitted for the catheterization of the femoral vein. The day after, the cardiovascular parameters were sampled by tail-cuff plethysmography (IITC, Life Scientific Instruments, CA, USA) before and 24 hours after intravenous injection of BPP-5a (71 nmol/kg) or vehicle (n = 5). During measurements, the warming chamber was maintained at 36–37°C. The data acquisition software showed systolic blood pressure, diastolic blood pressure, MAP, and HR.
Rat aortic rings assays
The vasorelaxant evaluation was performed as described previously [Verano-Braga et al. 2008]. The BPP-5a was tested in aortic rings (3–4 mm) from SHRs and WTs with or without functional endothelium precontracted to the same tension level (about 1.0 g) induced by submaximal concentrations of phenylephrine (0.003 or 0.01 µmol/l). BPP-5a was added in increasing cumulative concentrations (0.0001 to 1 µmol/l) once the response to phenylephrine had stabilized. Experiments were performed in the presence of L-NAME (100 µmol/l), HOE 140 (1 µmol/l), or indomethacin (10 µmol/l), added to the bath 20 minutes before the addition of phenylephrine. As a control for all of the above-mentioned protocols, another vessel segment from each rat was simultaneously monitored only in the presence of pentapeptide. Indeed, the effects of BPP-5a and acetylcholine (0.01 µmol/l) were compared for 6 minutes to reveal the time course of these responses. Mechanical activity, recorded isometrically by a force transducer (World Precision Instruments, Inc., Sarasota, FL, USA), was fed to an amplifier-recorder (Model TMB-4; World Precision Instruments, Inc.) and to a personal computer equipped with an analog-to-digital converter board (AD16JR; World Precision Instruments, Inc.), using CVMS data acquisition/recording software (World Precision Instruments, Inc.). The vasodilator effect of BPP-5a was expressed as a percentage decrease in maximal contraction induced by phenylephrine.
Statistical analysis
Results are presented as mean ± SEM. The comparisons were made using Student’s unpaired t-test or one-way analysis of variance (ANOVA) followed by Dunnett’s post hoc test when appropriate. Two-way ANOVA with a Bonferroni multiple comparison post hoc test was used to compare concentration–response curves obtained in aortic rings. The GraphPad Prism 5.0 (GraphPad Software, Inc.) was software program used in all statistical analysis. Significance was set at p < 0.05.
Results
Antihypertensive effect of BPP-5a in SHRs
Tables 1 and 2 shows baseline, maximal values and changes in blood pressure and HR, respectively, caused by intravenous injection of BPP-5a in WTs and SHRs. There were no differences in the pre-injection values (baseline) of MAP and HR within the same strain groups.
Baseline, maximal values and changes of MAP in SHRs and WTs produced by different doses of BPP-5a.

p < 0.05; **p < 0.01 and ***p < 0.001 when compared with the control group.
MAP, mean arterial pressure; SHR, spontaneously hypertensive rat; WT, Wistar rat; ND, not determined
Baseline, maximal values and changes of HR in SHRs and WT produced by different doses of BPP-5a.

p < 0.05 when compared with the control group.
HR, heart rate; SHR, spontaneously hypertensive rat; WT, Wistar rat; ND, not determined
The cardiovascular effects of BPP-5a were evaluated in conscious SHRs and WTs. Figures 1 and 2 show the time course of MAP and HR before and after bolus injection of BPP-5a at different doses in SHRs. BPP-5a showed potent and sustained antihypertensive activity. The effect of a single i.v. bolus injection of the pentapeptide lasted for at least 6 hours for all administered doses. The reduction of blood pressure was not associated with reflex tachycardia. Indeed, BPP-5a decreased HR. However, there was no clear relationship between the changes in MAP and HR (Tables 1 and 2). BPP-5a at the dose of 2.37 nmol/kg produced the largest decrease in blood pressure (−38 ± 4 mmHg, p < 0.01) and in HR (−71 ± 17 bpm, p < 0.05). No further changes in the MAP fall were obtained when the dose given was increased up to 300-fold (710 nmol/kg) (Table 2). The maximal changes in MAP and HR usually occurred 60 minutes after BPP-5a administration. In WTs, BPP-5a caused no significant changes in MAP or HR when compared with the control group (Tables 1 and 2). In order to verify whether the cardiovascular effects evoked by the pentapeptide were still evident 24 hours later, tail-cuff lethysmography was conducted in SHRs. We observed that 24 hours after acute i.v. injection of BPP-5a (710 nmol/kg), the blood pressure and HR were not statistically different from basal values (Table 3).
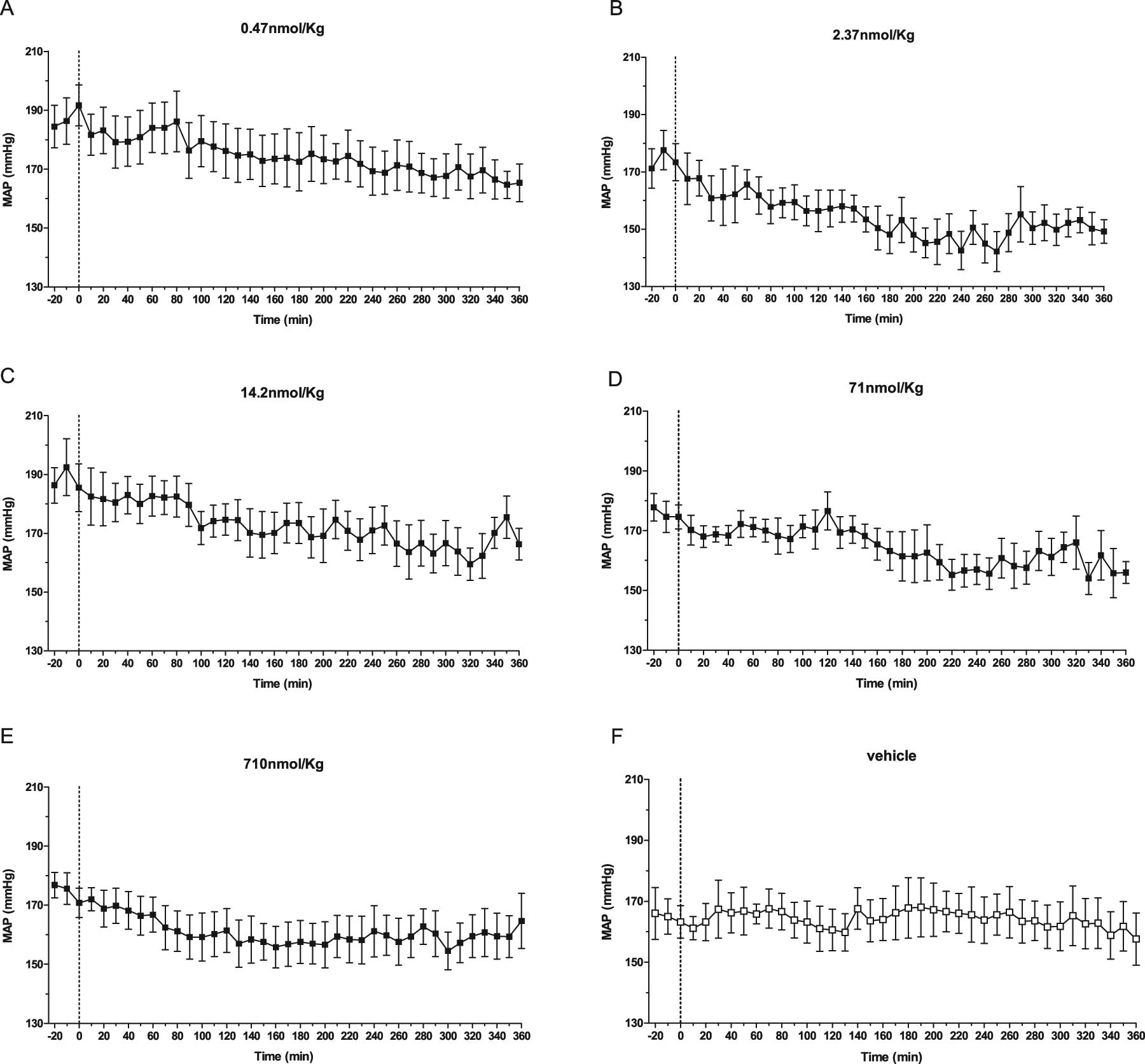
Time-course changes on MAP produced by BPP-5a in SHR. Pentapeptide was given in doses ranging from 0.47 to 710 nmol/kg. The data show the mean SEM changes observed with each dose.

Time-course changes on HR produced by BPP-5a in SHR. Pentapeptide was given in doses ranging from 0.47 to 710 nmol/kg. The data show the mean SEM changes observed with each dose.
Cardiovascular parameters sampled before and 24 hours after i.v. injection of BPP-5a (710 nmol/kg) in SHR. Tail-cuff measurements were performed in conscious rats (n = 5).

MAP, mean arterial pressure; HR, heart rate
Evaluation of BPP-5a activity upon Bk hypotensive effect and ACE
In order to verify whether the antihypertensive effect of BPP-5a was due Bk potentiation and/or ACE inhibition, we evaluated the hypotensive effect of Bk and the hypertensive effect of Ang I injections after BPP-5a or vehicle administration in SHR

Effect of BPP-5a (2.37 nmol/kg) on the MAP change produced by i.v. injection of Bk and Ang I in SHRs. Panels A and B show influence of BPP-5a and vehicle, respectively on the depressor effect of Bk and the pressor effect of Ang I. Gray bars, hypotensive effect of bradykinin (0.5 g); black bars, pressor response of angiotensin I (40 ng). The data are presented as mean SEM (n = 4–6).
AS catalytic activity measurement
Aiming to verify whether BPP-5a was similar to BPP-10c in provoking cardiovascular effects through AS activation [Guerreiro et al. 2009], we evaluated the functional interaction of the pentapeptide with AS in vitro. BPP-5a caused only a small increase (17%) of the catalytic activity of the AS. For comparisons, Table 4 presents results from in vitro and in vivo assays with BPP-5a and BPP-10c.
Effects of BPPs on somatic ACE inhibition, AS activation and Bk potentiation. The in vitro inhibition of ACE corresponds to the average value of three independent Ki(app) values determined according to Cotton et al. [2002]. The Bk potentiation unit (PU) is defined as the amount of potentiator (nanomoles) necessary to transform the effects of the single dose of Bk into that produced by the double dose.
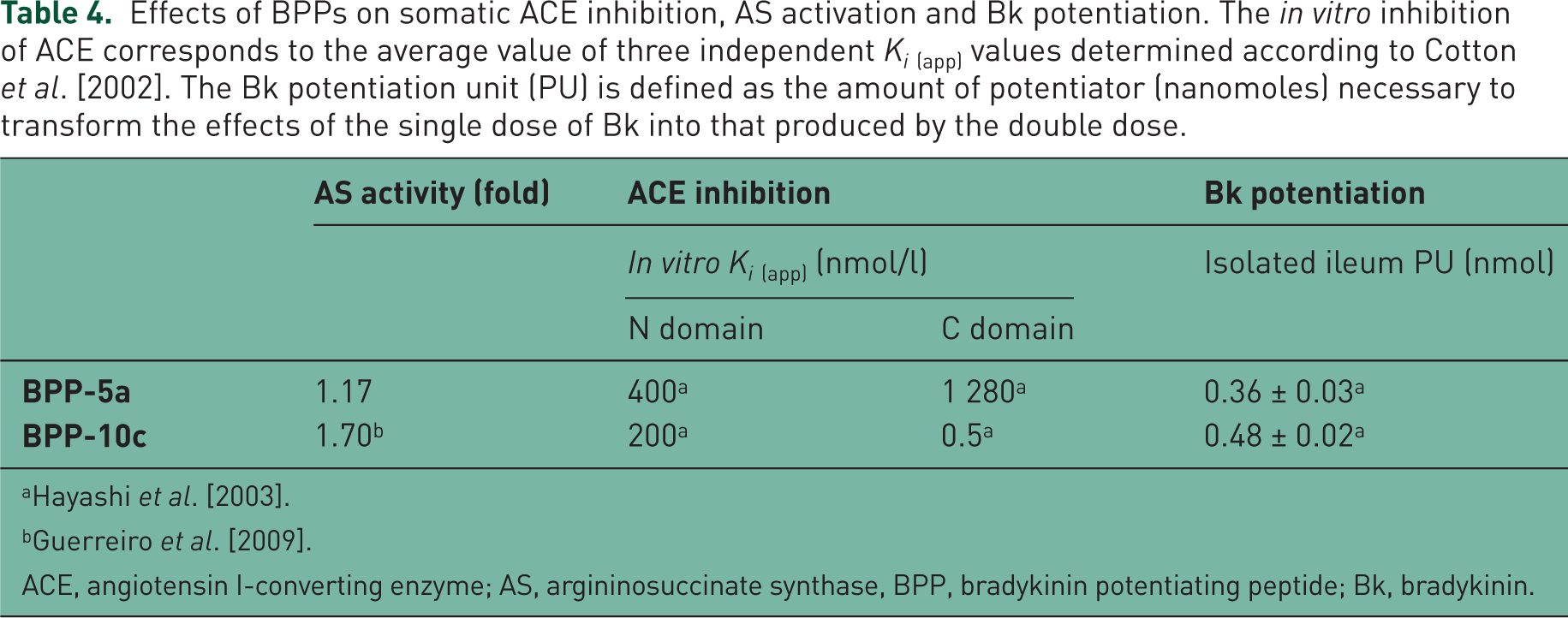
ACE, angiotensin I-converting enzyme; AS, argininosuccinate synthase, BPP, bradykinin potentiating peptide; Bk, bradykinin.
Binding studies
Herein, our objective was to investigate whether BPP-5a might exert an antihypertensive effect by blocking Ang II receptors. BPP-5a was not able to bind in AT1 or AT2 receptor. No significant displacement of 125I-Ang II from the two receptors was observed with the pentapeptide in concentrations ranging from 10−12 to 10−5 mol/l (Figure 4). Moreover, to evaluate a possible interaction of BPP-5a with Bk receptors and angiotensin-(1-7) receptor, we used CHO cells transfected with B1, B2, or Mas. BPP-5a did not provoke significant increase of NO releasing in these cells (Figure 5).

A. Competition for 125I-Ang II binding to AT1-transfected CHO cells by Ang II and BPP-5a. Competition curves were generated by adding increasing concentrations of Ang II (10-11 to 10-6 mol/L) or BPP-5a (10-12 to 10-5 mol/L) to the incubation buffer containing 0.4 nmol/L of 125I-Ang II. Data are presented as mean ± SEM of three to six independent experiments. B. Competition for AT2 binding of Ang-II to slices of Wistar kidney. Displacement of 125I-Ang II was evaluated with PD123319 (10-5 mol/L) or BPP-5a (10-8 and 10-6 mol/L). The displacement by peptides was compared to kidney slices incubated with buffer containing 0.4 nmol/L of 125I-Ang II (total binding). Data are presented as mean ± SEM; **p < 0.01.

Measurements of nitric oxide production by transfected-CHO cells. Confocal microphotographs of B1-transfected CHO cells (A), B2-transfected CHO cells (C) and Mas-transfected CHO cells (E) treated with BPP-5a (10–8 to 10-6 mol/L) and its respective agonists (10−8 and 10−6 mol/L). Production of NO as per cent over the control (baseline) of B1-transfected CHO cells (B), B2-transfected CHO cells (D) and Mas-transfected CHO cells (F) treated with BPP-5a (10−8 to 10-6 mol/L) and respective agonists (10−8 and 10−6 mol/L). NO production was visualized and estimated using the DAF-2 DA probe as described in methods.
Vasorelaxation in rat aorta rings
In order to evaluate whether BPP-5a cause cardiovascular effects by a direct vasorelaxing action, we used endothelium-containing and endothelium-denuded aortic rings of SHRs and WTs. BPP-5a produced concentration-dependent vasorelaxation in rat aorta rings of SHR. This effect was abolished in the absence of functional endothelium (Figure 6A). Maximal values (%) for the relaxant effect produced by BPP-5a (Emax) were 46.4 ± 5.4 and 2.2 ± 2.1, p < 0.001 for vessel with and without endothelium, respectively. To further address the possibility of involvement of Bk B2 receptors in the effect of BPP-5a in aortic rings of SHRs, we performed assays in the presence of the antagonist, HOE 140. As show in Figure 6B, HOE 140 (1 µmol/l) did not change the vasorelaxation induced by BPP-5a. To evaluate the possible participation of NO in the response induced by the pentapeptide, additional experiments in aortic rings of SHRs were performed in the presence of L-NAME (100 µmol/l). The nitric oxide synthase (NOS) inhibition completely abolished the vasodilator effect of BPP-5a (Figure 6C). BPP-5a caused slight endothelium-dependent vasodilator effect in aortic rings of WT rats, which was abolished in absence of functional endothelium (Figure 6D). It was also evaluated whether COX is involved in the vasorelaxing effects evoked by BPP-5a. Vasorelaxing effect provoked by BPP-5a was not inhibited by indomethacin (10 µmol/l) (Figure 6E).
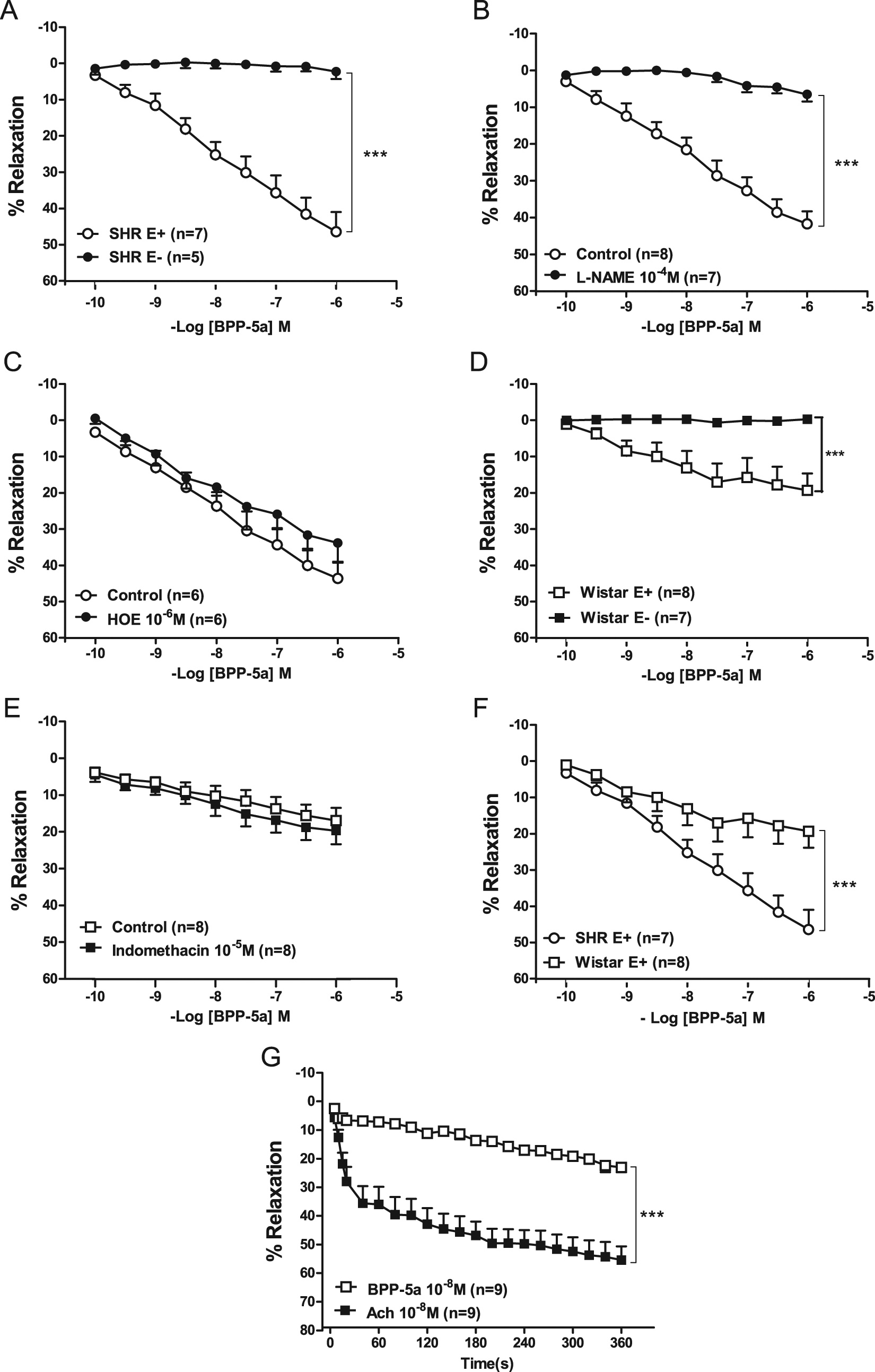
Vasodilator effects of BPP-5a in aortic rings from SHR and Wistar rats. A. Effects of BPP-5a on aortic rings of SHR containing endothelium (E+) or lacking functional endothelium (E-); B. Effects of BPP-5a on aortic rings of SHR in the absence and presence of HOE 140 (1 mol/L); C. Effects of BPP-5a on aortic rings of SHR in the absence and presence of L-NAME (100 mol/L); D. Effects of BPP-5a on aortic rings of Wistar rats containing endothelium (E+) or lacking functional endothelium (E-); E. Effects of BPP-5a on aortic rings in the absence and presence of indomethacin (10 mol/L); F. Comparison of effects evoked by BPP-5a on aortic rings of SHR and Wistar rats containing endothelium (E+); G. Comparison of the time-course effects of BPP-5a and acetylcholine (0.01µmol/L) on aortic rings of Wistar rats; Each point represents mean ± SEM generated from five separated experiments (n=6-9); ***p < 0.001 (two-way ANOVA followed by Bonferroni test).
A comparison of the vasorelaxing effect of BPP-5a in aortic rings of both strains revealed greater responses in SHRs than those from WTs (Emax: 46.4 ± 5.4 versus 19.3 ± 4.6, p < 0.001; respectively) (Figure 6F).
Figure 6G shows the time course of the vasorelaxing effects evoked from both BPP-5a and acetylcholine in aortic rings. The vasorelaxation induced by BPP-5a was slower and the Emax was fourfold lower than acetylcholine.
Discussion
The main finding of this study was that the BPP-5a caused a potent and long-lasting antihypertensive effect, which was not due to Bk potentiation or ACE inhibition.
Several studies attributed the BPPs effects on blood pressure to their capacity to inhibit ACE catalytic activity, which has two major consequences: decrease of the Ang II formation and reduction of Bk degradation [Keim et al. 1972; Loyke, 1977; Marcic et al. 1999]. However, in recent years, ossible mechanisms that go beyond interaction with ACE have been raised to explain the cardiovascular effects of ACE inhibitors, including BPPs. For instance, it has been proposed that ACE inhibitors can act as direct and indirect allosteric B1 and B2 receptors agonists, respectively [Erdos et al. 2010]. In addition, we have recently described ACE-independent antihypertensive effects of Bk-potentiating peptides [Ianzer et al. 2007].
In the current study we observed that BPP-5a did not interact with B1 and B2 Bk receptors, which clearly suggest that the proposed allosteric effects upon Bk receptors [Lameu et al. 2010] are not involved in its antihypertensive effect.
It had been shown that the capacity of several peptides from snake venom to potentiate Bk ex vivo and in vivo seems to be not directly proportional to their efficacy to inhibit ACE in vitro [Cotton et al. 2002; Hayashi et al. 2003; Mueller et al. 2005]. It was also shown that a synthetic compound similar to BPP-9a presented high Bk-potentiating activity and weak ACE inhibition [Mueller et al. 2005]. Accordingly, we demonstrated that BPP-7a markedly reduces blood pressure of hypertensive rats, without changing ACE activity in vitro or in vivo [Ianzer et al. 2007]. Taken together, these observations clearly show that the Bk-potentiating activity of BPPs can be dissociated from their ACE inhibitor activity and, more importantly, that both activities are not directly related to their potency as antihypertensive peptides or to their cardiovascular effects in normotensive animals.
As observed before for BPP-7a and 10c, administration of BPP-5a produced mild bradycardia, which may have contributed to its antihypertensive effect. This may be related to a central effect, as recently suggested for BPP-10c [Lameu et al. 2010; Silva et al. 2008].
Krieger and colleagues observed that BPP-5a infusion (0.82 µmol/min) reduced the blood pressure of hypertensive rats. Despite that, BPP-5a was considered a nonappropriate antihypertensive compound due to its short time of action [Krieger et al. 1971]. However, this conclusion was obtained from a small period of observation (< 60 minutes). We observed that in keeping with this early observation the antihypertensive effect in SHRs tends to diminish approximately 60 minutes after BPP-5a bolus injection (Figure 1). Subsequently, the antihypertensive effect increased, lasting for at least 6 hours. In addition, the tail-cuff measurements in SHR treated with the highest dose of BPP-5a (710 nmol/kg) showed that the cardiovascular parameters did not differ from basal levels after 24 hours (Table 3). The antihypertensive effect of BPP-5a was already evident with the smallest dose used (0.47 nmol/kg) which is far below the dose used previously in the seminal studies [Krieger et al. 1971; Stewart et al. 1971]. It is worth noting that increases in the doses up to 1500-fold (710 nmol/Kg) did not produce further decreases in blood pressure.
In order to test the involvement of ACE in the antihypertensive effect of BPP-5a we probed the pressor effect of Ang I and the vasodepressor effect of Bk in rats treated with a dose of 2.37 nmol/kg of BPP-5a, which is fivefold higher than the lowest dose used in this study. Neither the pressor effect of Ang I nor the hypotensive effect of Bk were altered in this condition, indicating that interference with the catalytic activity of ACE is not contributing to the antihypertensive effect of the pentapeptide.
Recently, AS was proposed as a new target for the antihypertensive effect of BPP-10c [Guerreiro et al. 2009]. AS is known as an important player of the citrulline–NO cycle [Solomonson et al. 2003]. Unlike BPP-10c, we showed that BPP-5a produced slightly but not significant activation of AS (Table 4). Thus, the antihypertensive effect of BPP-5a does not appear to be due to the activation of AS. In addition, our results in CHO cells transfected with different receptors indicate that the antihypertensive activity of BPP-5a is also not related to any direct effect on AT1, AT2, and Mas receptors or Bk B1 and B2 receptors.
Considering the lack of evidence for the involvement of classical humoral systems in the antihypertensive effect of BPP-5a, we tested its direct effect in blood vessels. Interestingly, BPP-5a produced NO endothelium-dependent vasorelaxation in aortic rings of SHRs unraveling a pivotal role for endothelial cells and NO releasing activity in the BPP-5a cardiovascular effects. Furthermore, our data shows that the vasorelaxing effect of BPP-5a was greater in SHRs than in WTs, which explain the antihypertensive but not hypotensive effect observed in our in vivo assays.
The vasorelaxation evoked by BPP-5a did not involve B2 receptor or prostaglandins as evaluated by their inhibitors, HOE 140 and indomethacin, respectively. Conversely, the NOS inhibitor, L-NAME, completely blocked the vasorelaxing effect caused by BPP-5a indicating that the cardiovascular effect of the pentapeptide is due to increases in endothelial NO releasing. Whether these effects of BPP-5a on the endothelium are mediated by an unknown receptor, remains to be further elucidated.
The comparison of the amplitude and time course effects of BPP-5a and acetylcholine on aortic rings showed differential features. The vasorelaxation evoked by the BPP-5a was less potent and slower than that produced by acetylcholine. This suggests that the BPP-5a cardiovascular effects may not involve cholinergic-like mechanisms. This is further strengthened, as we could not abolish vasorelaxation induced by BPP-5a with atropin in Wistar rats (data not shown). A recent report showed that this pentapeptide presents a quite unspecific action, upon both M1 muscarinic and Bk B2 receptors [Morais et al. 2010]. However, present findings are in disagreement with it because the antihypertensive and vasodilator effects could not be explained by agonism on M1 peripheral receptors, once it is well known that these receptors are prominently expressed in lung vessels and brain, playing a significant role in these tissues [Beny et al. 2008; Lamping et al. 2004; Langmead et al. 2008; Norel et al. 1996; Sheffler et al. 2009; Shirey et al. 2009; Walch et al. 2000]. Also, agonism of the M1muscarinic receptor has been related to increases in blood pressure and HR [Martin, 1996]. Indeed, current data showed that BPP-5a did not act on B2-transfected CHO cells and the B2 antagonist, HOE-140, did not abolish pentapeptide vasorelaxation effect. Thus, our results (ex vivo and in vitro) consistently demonstrate that B2 receptors are not involved in BPP-5a effects. Nevertheless, it has also been suggested that BPP-5a promotes crosstalk between Bk B2 and M1, resulting in a synergistic regulation of NO signaling [Morais et al. 2010]. This meets the NO-dependent vasorelaxing mechanism described in the present article.
As discussed above, strong evidence for an ACE-independent antihypertensive effect of BPPs has been described [Guerreiro et al. 2009; Ianzer et al. 2007; Verano-Braga et al. 2010]. Considering that some of these peptides were used as model for the development of ACE inhibitors and that even captopril appears to have ACE-independent effects [Attoub et al. 2008; Pandey and Anand-Srivastava, 1996; Ujhelyi et al. 1989; Verano-Braga et al. 2010], a systematic re-evaluation of the antihypertensive mechanisms elicited by these compounds can lead to the development of novel cardiovascular drugs (Camargo et al. 2011).
The evidence obtained in this study indicates that the antihypertensive and vasorelaxation effects of BPP-5a are due to an endothelium- and NO-dependent mechanism unrelated to the inhibition of the hydrolytic activity of ACE. The results presented here further support the need for reformulating the concept of sACE inhibition as the single mechanism of action for BPPs and related peptides/compounds. The novel identified targets for the powerful effects of different oligopeptides from venoms [Guerreiro et al. 2009; Verano-Braga et al. 2010] may lead to the development of new antihypertensive drugs, which could have an important impact on the therapeutic control of cardiovascular diseases and comorbidities.
Footnotes
Acknowledgements
We would like to acknowledge José Roberto Silva for technical assistance.
This work was supported funds provided by the Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP) through the Center for Applied Toxinology (CAT-Cepid) Program (grant number 1998/14307-9) and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq).
The authors declare that there is no conflict of interest.
