Abstract

The diagnosis of right ventricular hypertrophy (RVH) in adulthood remains a challenge for the clinical cardiologist.
We present an ECG tracing obtained in a 70-year-old woman with idiopathic pulmonary fibrosis and respiratory failure in whom RVH was confirmed by echocardiography.
The patient had been admitted to the Emergency Department because of a moderate respiratory distress. Blood pressure was 160/100 mmHg, heart rate was 85/min, respiratory rate was 30/min and oxygen saturation was 87% despite a 6 l/min oxygen supplementation. Blood gas analysis revealed severe hypoxemia and normocapnia, with normal pH value. The patient was evaluated in our outpatient clinic and later admitted to the pneumologic ward.
The ECG (Figure 1) showed sinus rhythm at 88 beats per minute with right bundle branch block (RBBB). In lead V1, the R’ wave measured 19 mm and QRS duration was 120 ms. Criteria for right atrial enlargement were not met, and the QRS axis was apparently indeterminable on the frontal plane. Nonetheless, if the prominent late electrical forces directed extremely rightwards (between +150° and +180°) and due to the RBBB were excluded, the QRS axis related to left ventricular activation was patently normal. On the precordial leads, RS morphology were present from lead V2 to lead V6, and the delayed RV activation, indicated by RBBB, was appreciable in all leads. Of note, in leads V2–V3 the intrinsecoid deflection was delayed (more than 40 ms) and the prominent R waves likely represented the summation of left and right ventricular activation, both directed frontwards. Finally, right precordial leads V3R–V5R demonstrated a rsR’ pattern with prominent R’ waves similar to those in lead V1 and representing RVH. ST-T changes in the inferior and precordial leads up to V4 were consistent with ‘right ventricular strain’.
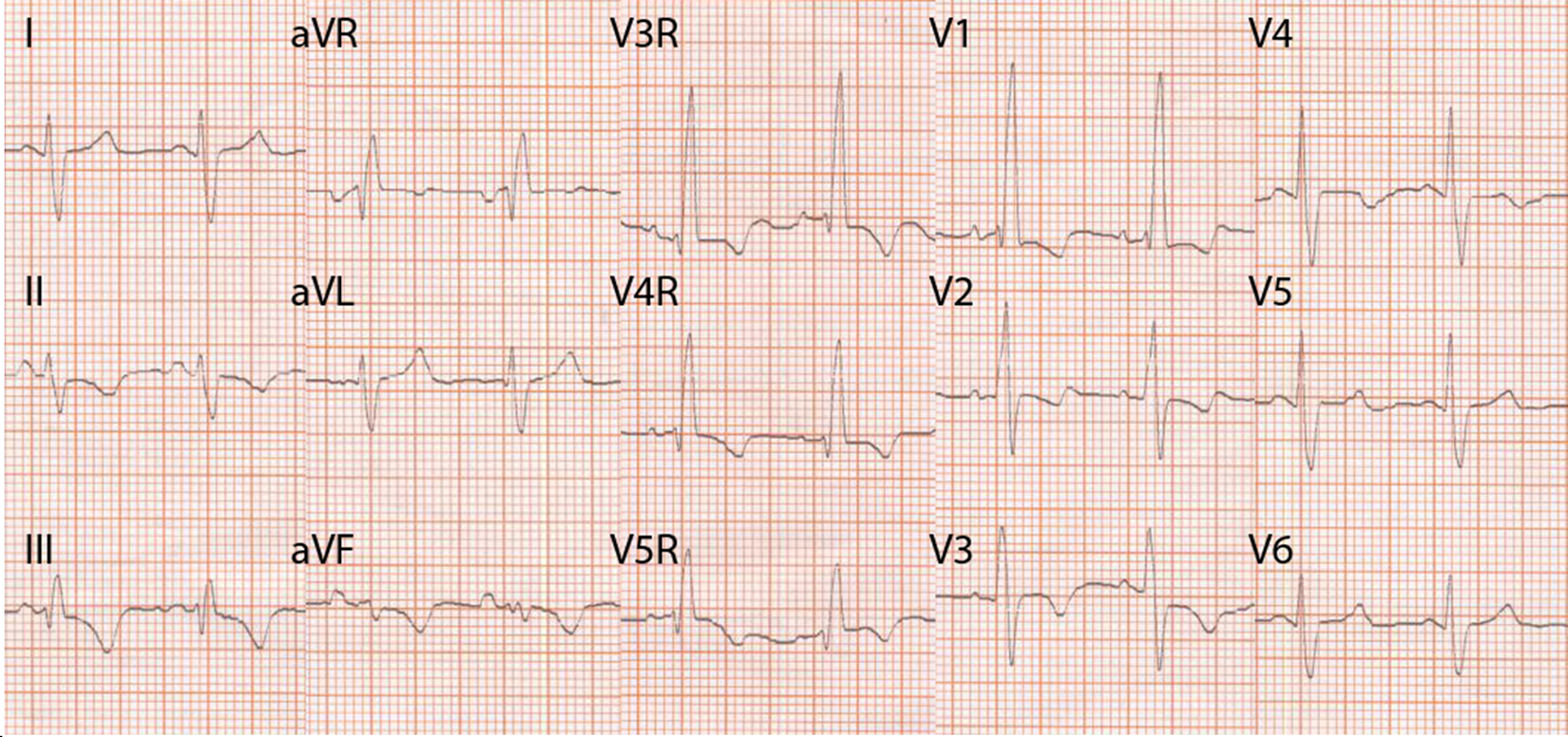
ECG at admission consistent with right ventricle hypertrophy (see the text for a detailed description).
At echocardiography left ventricle was hyper- contractile (ejection fraction was higher than 0.75) with normal wall thickness and chamber size; no significant mitral or aortic flow abnormalities were detected. Right ventricle (Figure 2, left) was moderately dilated (end-diastolic diameter was 38 mm) and hypertrophic (thickness of the free wall was 9 mm in the subcostal view). The longitudinal systolic function, assessed with tricuspid annular plane systolic excursion (TAPSE), was not severely impaired (17 mm). Of note, the right atrium was not enlarged (area was 10 cm2). A grade 1–2/4 tricuspidal regurgitation was present, and right systolic ventricular pressure was estimated to be 85 mmHg. The inferior vena cava was not dilated (15 mm) but respiratory changes in size were not maintained. Pulsed wave Doppler analysis of the hepatic veins flow pattern was indicative of very high right ventricular filling pressures (Figure 2, right): the D wave was higher than the S wave and a prominent reversal A wave was present. Moreover, a prominent inward wave, independent from cardiac cycle and synchronous to inspiration, was detected. This inward flow wave had to be considered the main determinant of right ventricular filling and the echocardiographic equivalent of Kussmaul’s sign.

Left, subcostal four-chamber view showing right ventricle dilatation. RV, right ventricle; LV, left ventricle. Right, pulsed wave Doppler interrogation of the hepatic vein. Ar, reverse atrial flow; S, systolic flow; D, diastolic flow; Inward, inward flow synchronous with inspiration.
The most common ECG pattern associated with pulmonary disease in adults is ‘cor pulmonale’ [Scott et al. 1955]. It is classically observed in chronic obstructive pulmonary disease (COPD) and it reflects the electric consequences of the downward displacement of the diaphragm due to pulmonary emphysema. This pattern includes low voltages in the limb leads, a QRS axis that may be indeterminate in the frontal plane, a rightward P wave axis and persistent S waves in all precordial leads with low R wave voltage in leads V5–V6. RVH is suggested, in the presence of the COPD pattern, only if R wave voltage in lead V1 is ‘relatively’ increased [Hancock et al. 2009].
The actual uncertainty in identifying RVH in the picture of pulmonary disease mainly comes from three factors.
First, the sensitivity of the ECG criteria for RVH is generally low, and there is a sort of gradient of accuracy from congenital heart disease to primary pulmonary hypertension to chronic lung disease.
Second, RBBB is often present when right ventricular dilatation and dysfunction is associated with pulmonary disease: provided that ECG criteria are essentially based on voltage measurements, the ‘disturbance’ generated on the QRS complex in the right precordial leads by terminal right ventricular forces directed rightwards and frontwards has never been investigated. In other words, it is not formally established whether RVH voltage criteria can be applied whenever RBBB is present. Of note, in a study carried out in the 1950s, the height of R’ waves exceeding 15 mm had been taken as evidence of RVH in addition to complete RBBB [Scott et al. 1955].
Third, the pattern of RVH in patients with pulmonary disease is not as common as RBBB, at least in cases of mild to moderate disease severity [Holtzman et al. 2011].
In contrast, RVH is a common finding in young patients with congenital heart disease. Two different patterns of RVH have been actually described: the former is similar to incomplete RBBB and suggests volume overload, the latter consists of predominantly tall R waves in the right precordial leads as part of an Rs, R, or Qr complex and suggests pressure overload. In other words, QRS complex prolongation due to various degrees of RBBB could be considered a manifestation of right ventricle dilation, whereas R wave voltage would be more directly related to right ventricle wall thickness [Hancock et al. 2009]. Moreover, although in congenital heart disease right ventricle involvement may consist either of ‘pure’ hypertrophy (isolated pulmonary stenosis) or ‘pure’ dilatation (nonrestrictive ventricular septal defect), in adulthood diseases of the right ventricle, secondary to pulmonary or left-sided cardiac disorders, a combination of right ventricle dilatation with a less evident degree of hypertrophy is invariably present.
As a matter of fact, the description and validation of the long-standing ECG criteria of RVH is mainly based on autoptic series [Lehtonen et al. 1988] and has never been reassessed more recently with noninvasive imaging techniques, mainly because of the inherent difficulties in evaluating the complex three-dimensional geometry of the right ventricle. Actually, echocardiography has important limitations in the study of right ventricle wall thickness, dimensions and remodeling, especially with coexistent lung disease.
Nonetheless, in this particular case RVH was identified with ECG and echocardiography without the need of confirmatory finding by more complex investigations. A possible explanation is that pulmonary fibrosis itself did not displace the diaphragm downwards, as it is not typically associated with emphysema, despite pulmonary arterial hypertension caused the pressure overload of the right ventricle that, in turn, adapted its thickness to counterbalance an abnormal wall stress. Therefore, right axis deviation and prominent anterior forces were maintained and could be easily detected with ECG, especially in the right precordial leads [Camerini et al. 1956]. Moreover QRS duration was not particularly prolonged and right atrial enlargement criteria were not present. Actually, the mild regurgitation of the tricuspid valve despite pressure overload confirmed that the right ventricle had somehow adapted to the unfavorable hemodynamics by a combination of hypertrophy and dilation that, in turn, were not compromising valve competence and were not causing right atrial dilatation.
In conclusion, although the presence of RBBB is known to be a limitation factor in the ECG diagnosis of RVH, the occurrence in all leads others than I, aVL and V5–V6 of both prominent electrical vectors directed frontwards and rightwards and widespread secondary ST-T changes may be considered as strong indicators of RVH.
Footnotes
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
The authors declare no conflicts of interest in preparing this article.
