Abstract
The early recognition and appropriate management of diseases affecting the retina and choroid is important to optimise visual outcomes. However, treating these conditions in pregnant women may pose unique challenges and requires multidisciplinary collaboration between obstetricians and ophthalmologists. This review provides an overview of chorioretinal conditions that may arise or worsen during pregnancy or labour, their clinical presentation, current evidence on management in pregnant women, as well as their relevance to maternal–fetal health.
Introduction
The physiological, hormonal and immunological changes of pregnancy affect multiple organ systems including the eyes. 1 Pregnancy may therefore exacerbate pre-existing chorioretinal diseases, induce new chorioretinal pathology, or contribute to the development of conditions also seen in non-pregnant patients, all of which may impact vision. Early recognition and appropriate treatment may help optimise visual outcomes for these women.
This review aims to cover the presentation and management of common chorioretinal conditions (apart from diabetic retinopathy, which will be covered in a separate review) to increase the awareness of management needs and complexities for non-ophthalmic clinicians involved in the care of this cohort of patients.
Pathophysiology of chorioretinal changes in pregnancy
Pregnancy induces a broad spectrum of progressive physiological changes, including cardiovascular, metabolic, hormonal, immunological and haematological adaptations.2,3 The increased metabolic demands induce a cascade of haemodynamic changes which affect the systemic and ocular circulation. Despite the activation of compensatory mechanisms to increase the circulating plasma volume and cardiac output, there is arterial underfilling, with 85% of blood volume residing within the venous circulation. In addition, both haemodynamic and hormonal changes lead to upregulation of nitric oxide, contributing to a decrease in systemic vascular resistance and peripheral vasodilation. Retinal blood flow is maintained through the release of vasoactive mediators from the vascular endothelium, allowing the capillary vascular tone to adapt to changes in perfusion pressure or metabolic needs (autoregulation). However, endothelial cell function may be compromised in pregnancy, resulting in changes to both the systemic vasculature and the ocular microcirculation, including retinal capillary plexuses and choroidal vessels. In addition, the production of angiopoietic factors including angiopoietin-1, angiopoietin-2 and vascular endothelial growth factor (VEGF) is increased, contributing to retinal capillary dilation and perfusion changes. These factors may also be modulated by growth hormone and insulin-like growth factors, which promote fetal development.4,5
Ophthalmic evaluation of the choroid and retina
Chorioretinal diseases may be asymptomatic, or may produce symptoms such as blurred vision, metamorphopsia (distorted vision), or scotomata (blind spots in the field of vision). If chorioretinal pathology is suspected, a comprehensive eye examination should be conducted. This comprises visual acuity and intraocular pressure measurements, followed by fundoscopic examination of the posterior segment of the eye. Instillation of topical agents that induce pupil dilation (such as a combination of tropicamide 1% and phenylephrine 2.5% eye drops) is often done to facilitate visualisation of the retinal fundus. However, as a selective α1-adrenoceptor agonist, phenylephrine is a vasoconstrictor, meaning that systemic absorption could theoretically affect placental blood flow in pregnancy. Historically, some ophthalmologists may have opted to avoid phenylephrine and instil a single agent (tropicamide) for pupil dilation because of these theoretical concerns. There is no evidence to support the avoidance of phenylephrine at the doses used in eyedrops, so these can be used as normal if required. 6
In recent years, the development of high-resolution and non-invasive retinal imaging has greatly improved our ability to diagnose and monitor chorioretinal disease. Imaging modalities include the following (Figure 1):
Optical coherence tomography (OCT) scans provide cross-sectional images of the retina, allowing for a detailed and non-invasive assessment of macular pathology. Enhanced depth imaging (EDI) OCT techniques can enable better evaluation of the underlying choroid. OCT angiography (OCTA) is a newer non-invasive imaging modality which utilises motion contrast to detect blood flow, which enables efficient 3D imaging of the retinal vasculature in vivo without the need for contrast dye.
7
Fundus fluorescein angiography (FFA) has traditionally been the gold standard investigation for diagnosing retinal vascular pathology. This is an invasive procedure that involves serial retinal photography after intravenous injection of fluorescein dye, which can theoretically enter the fetal circulation via the transplacental route. While older studies have not found any direct associations between FFA and obstetric complications or birth abnormalities,
8
with the advent of newer technologies such as OCT and OCTA, many ophthalmologists may prefer these over FFA in pregnant patients in whom a diagnosis can be made using either modality. Ultimately, if there is no alternative, FFA can be used in pregnancy and breastfeeding. Indocyanine green angiography (ICG) is a similarly invasive test involving intravenous injection of indocyanine green dye to evaluate the choroidal circulation. The risk of transplacental transfer is low, and this can be used in pregnancy if no alternative imaging modality is available to support investigation.9,10
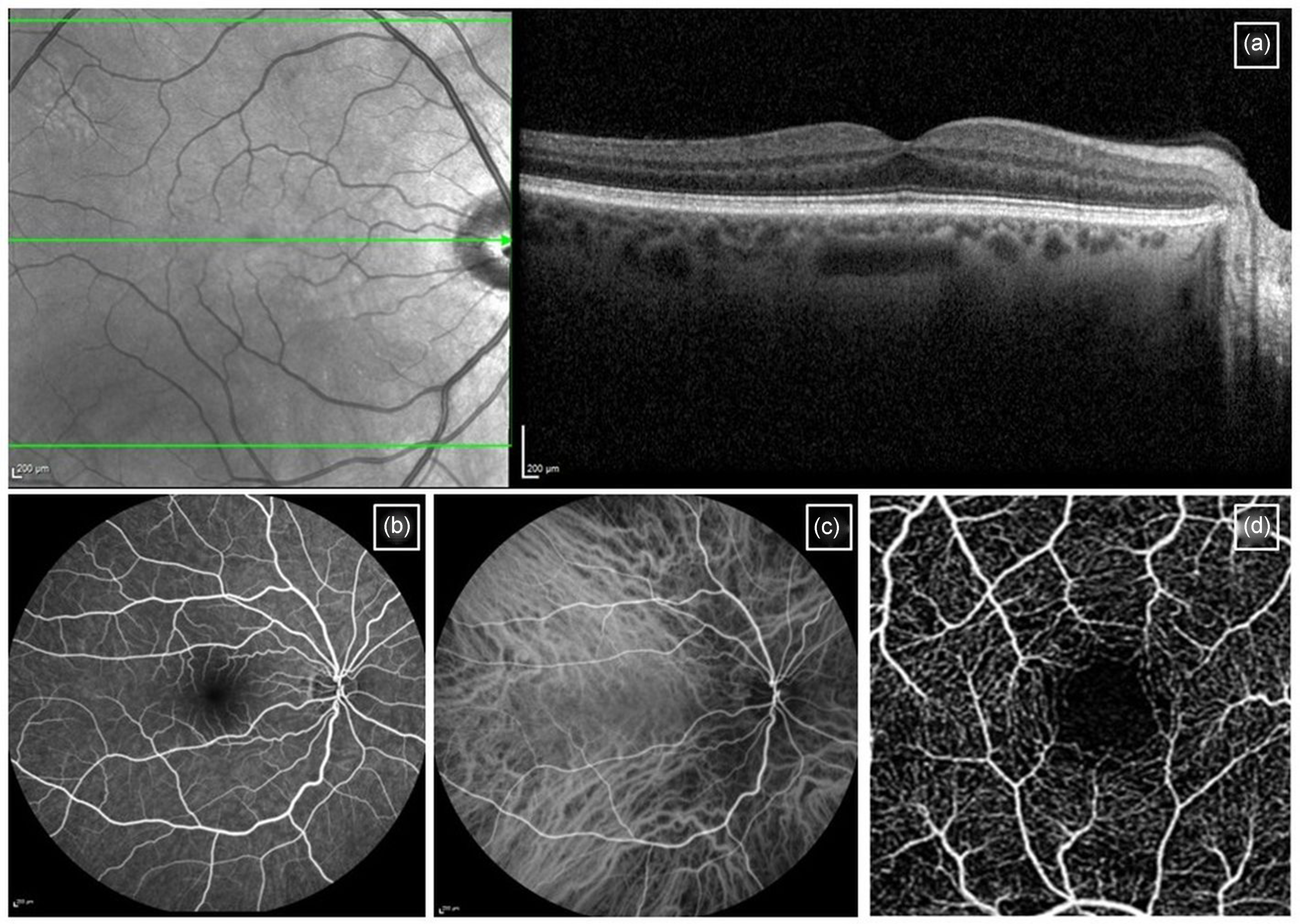
Retinal imaging modalities: (a) normal OCT scan, (b) normal fluorescein angiography, (c) normal indocyanine green angiography and (d) normal OCT angiography scan.
Pre-existing chorioretinal diseases
Macular neovascularisation
Macular neovascularisation (MNV; previously known as choroidal neovascularisation or CNV) typically occurs as a result of pathologic new blood vessels that arise from the choroidal vasculature. The neovascularisation breaches Bruch's membrane (which separates the choroid from the retinal pigment epithelium, RPE) passing into the avascular sub-retinal or sub-RPE space. Rarely, new blood vessels may arise within the retina itself. The resultant leakage of fluid or blood from these new vessels can compromise retinal function and result in visual loss, which can occur rapidly over a course of days or weeks. Patients may report blurred vision, metamorphopsia, scotomata, or be asymptomatic if the central part of the macula (fovea) is unaffected.
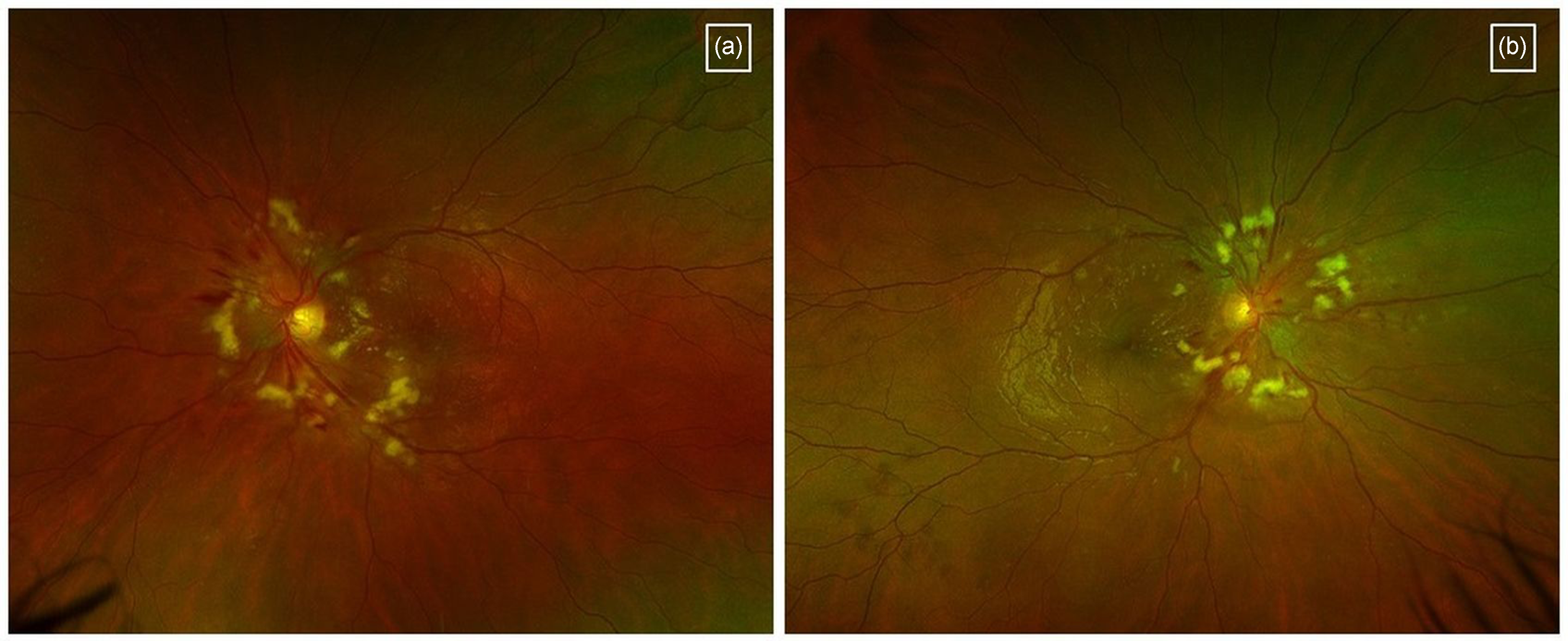
In pregnancy, the increase in angiogenic factors such as VEGF or placental growth factor may theoretically promote the development or recurrence of MNV in susceptible eyes. However, there are insufficient data to establish causality between pregnancy and the types of MNV seen in women of childbearing age. MNV is most commonly related to age-related macular degeneration, which is seen in older adults.11,12 In women of childbearing age, MNV may be related to degenerative conditions such as pathologic myopia or angioid streaks (Figure 2(a) and (b)) or inflammatory chorioretinopathies such as punctate inner choroidopathy (PIC) or multifocal choroiditis (MFC) (Figure 2(c) and (d)). It may also be related to central serous chorioretinopathy (CSCR). Less commonly, MNV may develop in association with inherited retinal diseases (macular dystrophies), choroidal tumours or choroidal rupture secondary to trauma. 11 Sometimes, no apparent antecedent cause may be evident (idiopathic).
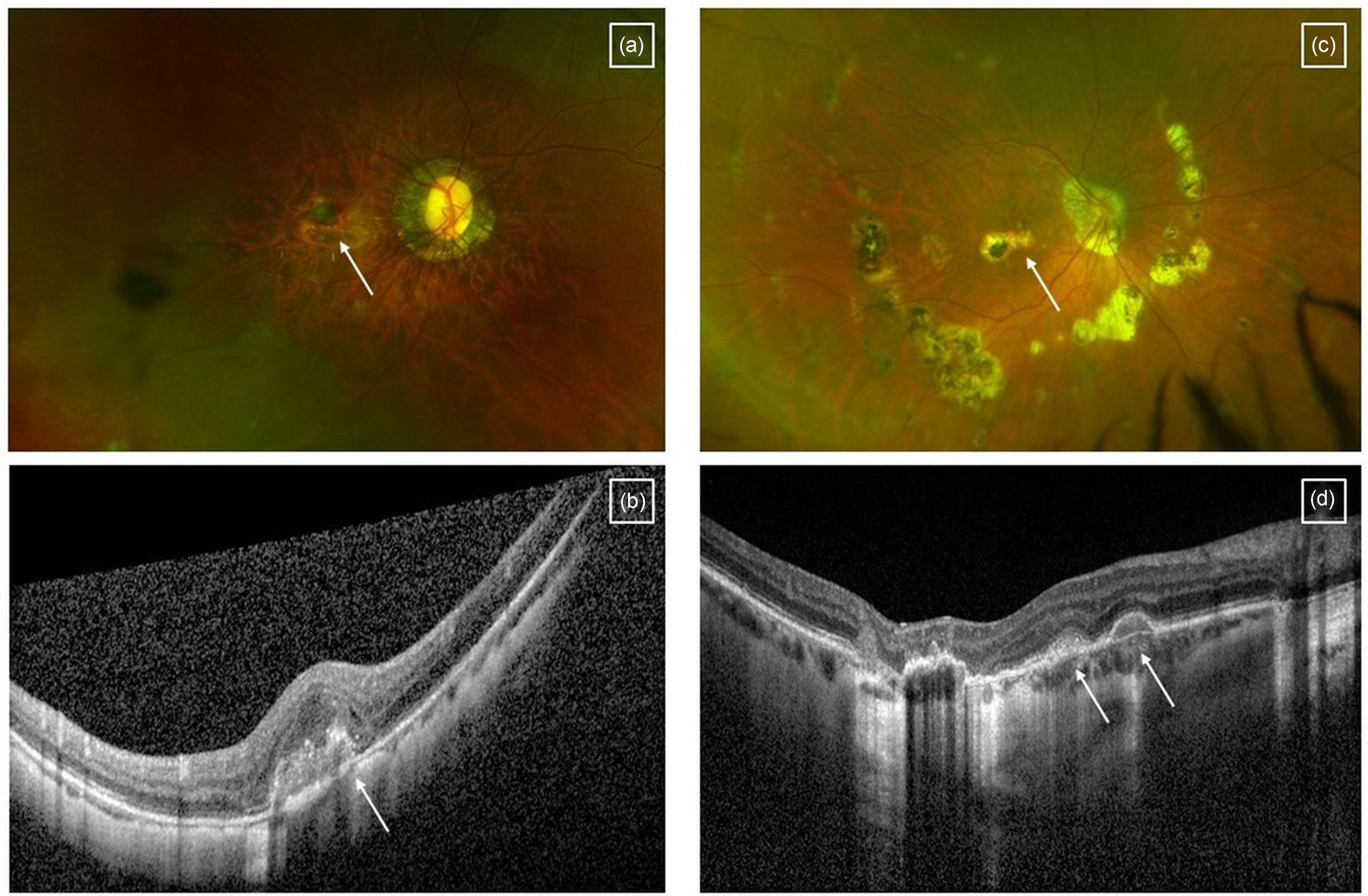
Macular neovascularisation secondary to myopia featured on (a) ultra-widefield imaging and (b) OCT scan. Macular neovascularisation secondary to punctate inner choroidopathy featured on (c) ultra-widefield imaging and (d) OCT scan. Arrows indicate areas of macular neovascularisation.
Following initial OCT scans, some ophthalmologists may prefer OCTA to invasive tests such as FFA and/or ICG for diagnosing MNV in pregnancy. 13 In the non-pregnant population, management options for MNV include observation (if not visually significant) or intravitreal anti-VEGF injections to preserve vision and minimise the risk of permanent structural damage. Anti-VEGF treatment should be administered promptly, ideally as soon as possible (clinical guidelines suggest within 2 weeks) to reduce the risk of vision loss. The risks of intravitreal anti-VEGF use in pregnancy are less certain. Although the anti-VEGF agent is injected directly into the vitreous at the back of the eye, a degree of systemic absorption occurs up to 1 month or more after treatment, and the effect of systemic VEGF suppression can increase with cumulative doses. 14 There may therefore be concerns about the effects of this medication on the fetus, particularly as VEGF plays an important role in placentation and embryogenesis. A recent case series and systematic review of the literature showed that intravitreal anti-VEGF injections were not associated with adverse pregnancy outcomes in women with no other risk factors for poor pregnancy outcomes, although this was limited by the small numbers of cases included. 15 Whilst systemic anti-VEGF therapy would be avoided in pregnancy, it is reasonable to consider the use of intravitreal anti-VEGF if clinically indicated and if no suitable alternative exists. Given the limited safety data, multidisciplinary team (MDT) discussion followed by careful patient counselling is warranted to support pregnant women with vision-threatening MNV in making a considered decision about appropriate treatment that is right for their individual circumstances. 16
Non-infectious posterior uveitis
Uveitis comprises a group of inflammatory or infective conditions affecting the uveal tissue within the eye: the iris and/or ciliary body (leading to anterior uveitis), the vitreous and peripheral retina (intermediate uveitis), the retina and choroid alone (posterior uveitis, or all of the uveal tissue (panuveitis). During pregnancy, maternal immunity undergoes modulation to promote tolerance of the allogeneic fetus, with the maternal immune response varying by stage of pregnancy. 17 While flares of non-infectious anterior uveitis occur more frequently in the first trimester of pregnancy and decline over the second and third trimester, data specific to posterior uveitis are more limited.18,19 The latter comprises a diverse range of conditions including punctate inner choroidopathy (PIC), MFC, serpiginous choroidopathy, Birdshot chorioretinopathy, Behcet's disease, Vogt–Koyanagi–Harada disease and many others. It is outside the scope of this review to discuss all these conditions in detail, but we will focus on PIC and MFC (see below) since they are examples of posterior uveitis most relevant to women of childbearing age. In general, management of non-infectious posterior uveitis requires a combination of local and systemic therapy, which may be in the form of oral steroids in the short term and systemic immunosuppression in the medium to long term. In general, certain types of systemic immunosuppressants may need to be adjusted or avoided in pregnancy depending on their specific safety profile. In some cases, local treatment with intravitreal corticosteroids (whilst not always being the optimal treatment over the long-term) may offer a pragmatic alternative to maintain temporary disease control while limiting systemic exposure.
PIC and MFC are idiopathic chronic inflammatory disorders of the choriocapillaris believed by some to be part of the same spectrum of disease, 20 and typically affect young myopic women in their childbearing years.21,22 Patients may report blurred vision, metamorphopsia, photopsias (flashing lights) or be asymptomatic. 23 Clinical features include multiple small discrete yellow-white lesions affecting the choroid, RPE and outer retina in the macula and/or peripheral fundus, usually in the absence of significant intraocular inflammation.21,22
PIC/MFC lesions may resolve spontaneously within a few weeks, leaving atrophic scars which may fade with time. However, they may have an impact on vision if the lesions affect the central macula. These lesions can often recur and/or be complicated by MNV, which is thought to result from the underlying inflammation and/or choroidal hypoperfusion. 24 The resultant structural damage may compromise visual outcomes.25,26 Clinical management should therefore take into account both the inflammatory and neovascular components, and be tailored according to the extent and course (including recurrence) of the disease, risk of progression, complications and the patient's unique circumstances. 27 MNV can be treated with intravitreal anti-VEGF injections, while treatment of the inflammatory component may involve close monitoring, short courses of local or systemic steroids in the presence of acute disease activity, or systemic immunosuppression with steroid-sparing disease-modifying anti-rheumatic drugs (DMARDs); rarely, biologic agents may also be considered to minimise the long-term risk of recurrence.28,29
Data on the optimal treatment regime for PIC/MFC in pregnancy is limited. Systemic corticosteroid use in pregnancy is considered to be relatively safe, 30 with little evidence of an increased risk of adverse obstetric outcomes such as pre-term birth, pre-eclampsia or low birth weight, although it may contribute to hyperglycaemia in pregnancy and the development of gestational diabetes mellitus. In a retrospective study of 16 women (26 affected eyes) with PIC/MFC, 44% experienced a relapse in disease activity during pregnancy, most frequently in the third trimester, in contrast to the conventional understanding of a lower risk of uveitis reactivation in pregnancy. 28 There was a trend towards fewer relapses in women who received continuous systemic immunosuppression before and during pregnancy as compared to short courses of periocular/systemic corticosteroids, although numbers were too small to be conclusive. Six patients continued treatment with azathioprine during pregnancy, having discontinued other teratogenic DMARDs such as methotrexate and mycophenolate mofetil prior to conception; there was one stillbirth in a patient with a complex obstetric history, but no birth defects were reported. In general, the rates of maternal, obstetric, and fetal complications were comparable to the general population. 28
Infectious uveitis: Toxoplasma chorioretinitis
The Toxoplasma gondii parasite is endemic in many countries worldwide, thriving particularly in hot humid climates in Latin America and tropical African countries, where seroprevalence often exceeds 50%. 31 Toxoplasma infection may be congenital (via vertical transmission in utero) or acquired later in life, in association with the ingestion of water or undercooked meat contaminated with T. gondii oocysts.
Acute toxoplasma infection in pregnancy confers low rates of vertical transmission (6%) in the first trimester, but tends to result in a more severe form of congenital toxoplasmosis which involves neurological and ocular complications. Despite the higher rates of transmission if infected in the third trimester (72%), newborns born to these women are more likely to be asymptomatic. 32 Reactivation of systemic toxoplasmosis during pregnancy is uncommon in immunocompetent patients. 33 The vast majority of congenital toxoplasmosis occurs in infants of women who acquire a primary infection in pregnancy. 34
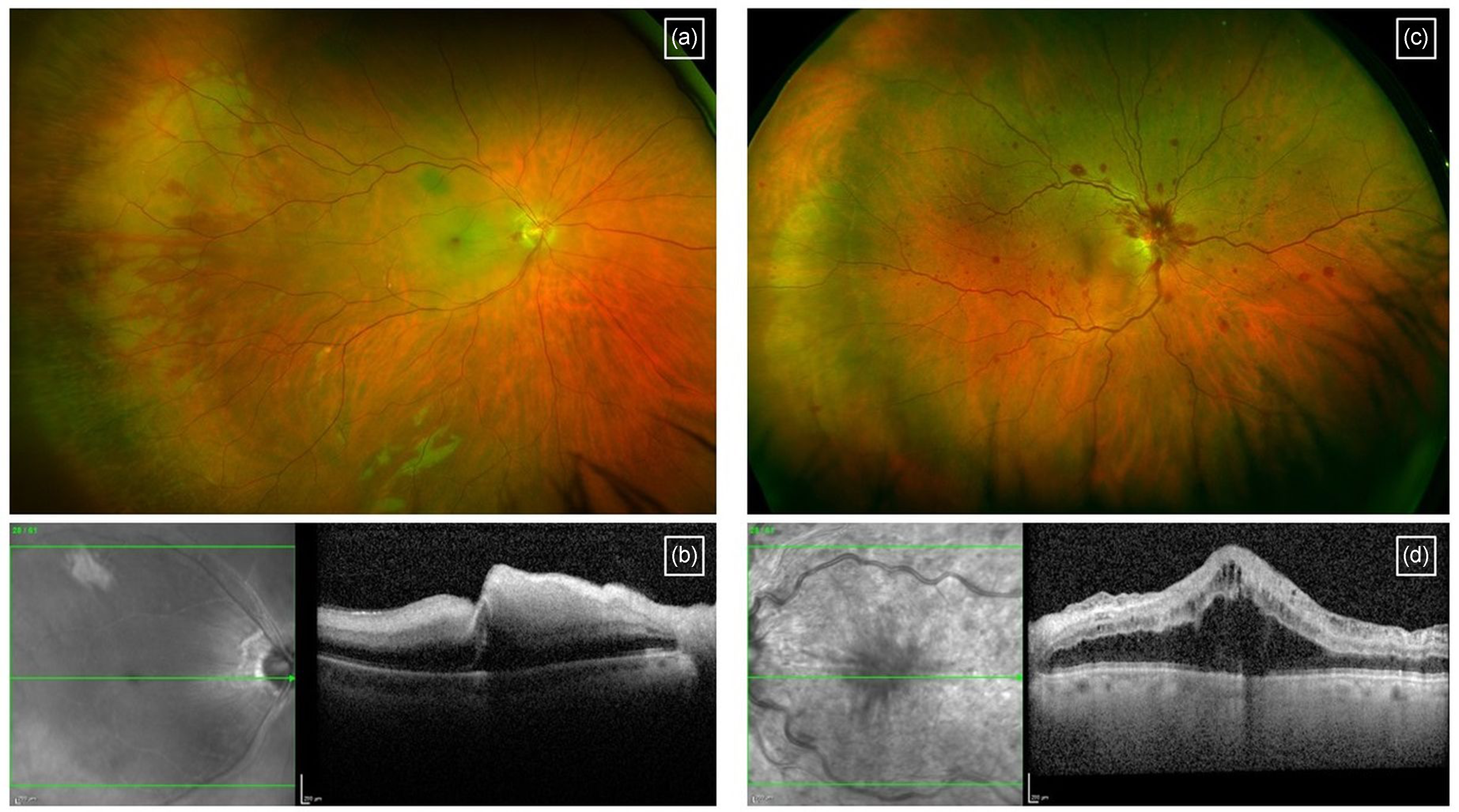
Toxoplasma chorioretinitis may be acquired de novo, or present as a reactivation. There is conflicting evidence as to whether pregnancy is associated with an increased incidence of reactivation. 35 Active toxoplasma chorioretinitis presents as a full-thickness area of retinitis with overlying vitritis, which may be adjacent to an old chorioretinal scar (in cases of reactivation) (Figure 3(a)–(d)). Affected patients may report blurred vision, floaters or even be asymptomatic.
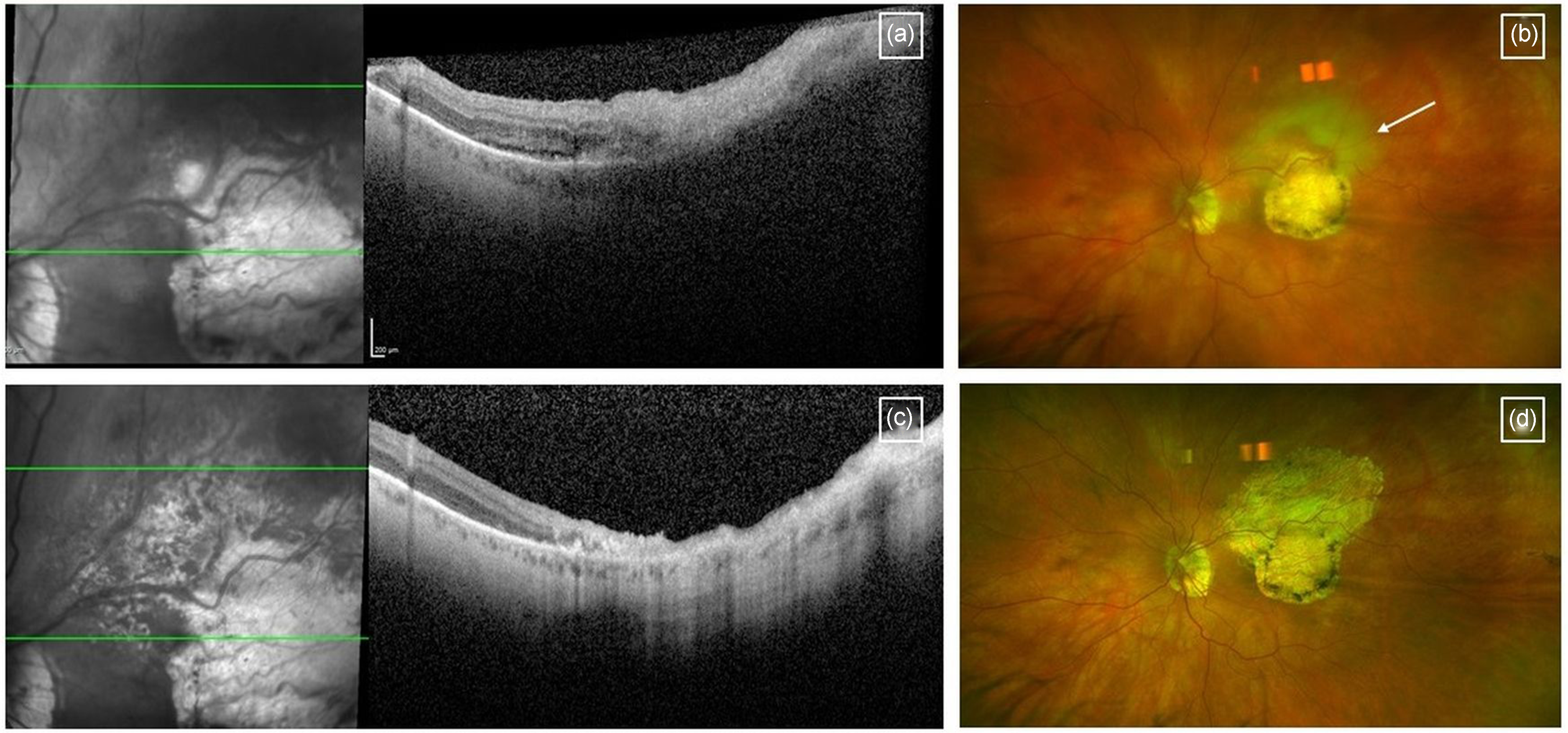
Toxoplasma chorioretinitis: acute presentation demonstrating (a) full-thickness retinitis on cross-sectional OCT scan and (b) an indistinct white patch adjacent (new, active) adjacent to a discrete white patch (old toxoplasma scar) on ultra-widefield imaging; note that the view is hazy due to vitreous inflammation. (c) and (d) Feature the same patient 6 months later – the active disease has resolved and developed into atrophy.
Toxoplasma chorioretinitis tends to be self-limiting in immunocompetent patients, and often does not require treatment if the lesion is peripheral or if vision is preserved. 36 Indications for treatment include reducing the risk of vertical transmission and hence congenital toxoplasmosis in pregnancy, as well as sight-threatening cases with significant vitritis or chorioretinal lesions involving the macula, peripapillary region or major retinal vessels. 37 However, a recent international survey of ocular inflammation specialists found that not all would recommend treatment in the pregnant population. 38
Spiramycin, a macrolide antibiotic, is the first-line treatment for toxoplasmosis in pregnancy due to the lower risk of teratogenicity and fetotoxicity. Azithromycin and Clindamycin are suitable alternatives. Clindamycin is known to be safe in pregnancy – it is used to treat group A streptococcal infections (as second line treatment) and antenatal sepsis for penicillin-allergic women who are pregnant, and is not known to cause adverse events in the second and third trimester of pregnancy. 39 Other standard treatment regimes in non-pregnant populations are either teratogenic or should be avoided in the first trimester for other reasons (pyrimethamine/sulfadiazine combination and trimethoprim-sulfamethoxazole (cotrimoxazole), respectively), or their efficacy has not been adequately studied in pregnant women with toxoplasmosis (atovaquone). 35 Adjunctive treatment with systemic corticosteroids is often given to treat the ocular inflammation and reduce visual morbidity depending on local practice patterns, and if indicated, pregnancy should not alter this treatment plan. 40 Prednisolone is the corticosteroid of choice in pregnancy, and pregnant women can be reassured that prednisolone is largely metabolised by the placenta, with only 10% of the dose reaching the fetus.
Chorioretinal diseases potentially exacerbated by pregnancy
Diabetic retinopathy
This complex topic will be covered in a separate review.
Central serous chorioretinopathy
CSCR is an idiopathic condition characterised by serous retinal detachments and RPE detachments in the context of choroidal thickening, with detection of these features on OCT scans often confirming the diagnosis (Figure 4). Patients may report blurred central vision, metamorphopsia, micropsia (images appearing smaller than normal), or may occasionally be asymptomatic.
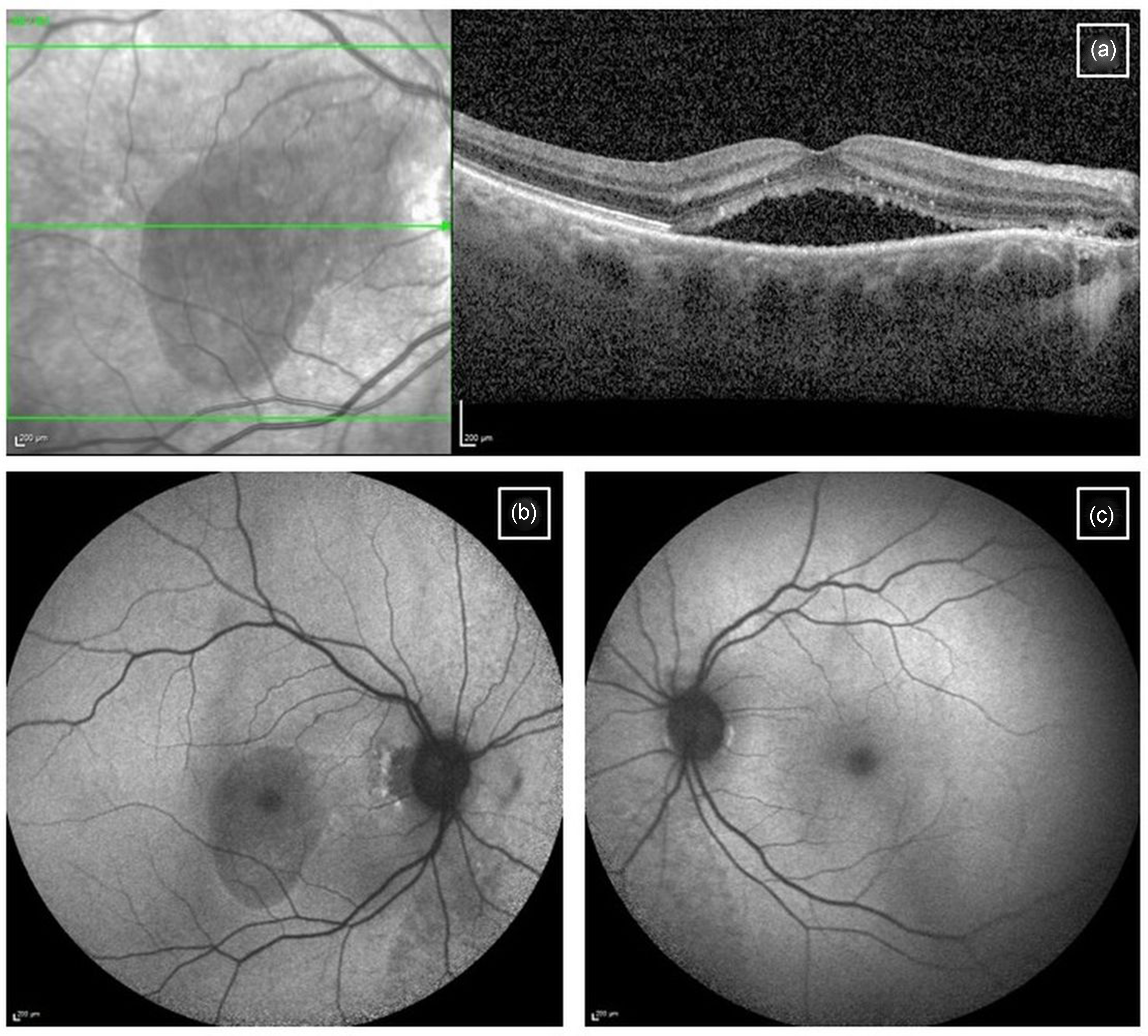
Central serous chorioretinopathy: (a) Acute presentation featured on OCT scan with central sub-retinal fluid; (b) Blue auto-fluorescence imaging of the affected eye and (c) unaffected fellow eye for comparison.
CSCR occurs more commonly in males, and in those treated with exogenous steroid medications (orally, topically and via steroid inhalers or nasal sprays). However, pregnancy is also known to be a risk factor. 41 While the pathogenesis of CSCR in pregnancy remains to be established, this association has been attributed to an increase in endogenous cortisol levels, vascular hyper-permeability or haemodynamic changes present in pregnancy. 42 CSCR in pregnancy typically occurs in the third trimester, and is more commonly associated with atypical features of subretinal fibrin deposition and RPE changes that may mimic MNV.43–45 This may complicate management, as uncomplicated CSCR can often be monitored, while the presence of MNV may prompt a discussion about anti-VEGF treatment. OCTA may therefore be very useful in confirming or excluding the presence of concurrent MNV.
In the majority of non-pregnant patients, CSCR resolves spontaneously and can be observed. However, acute CSCR can persist and become chronic. There are limited treatment options for chronic CSCR, but the most effective is photodynamic therapy (PDT) with verteporfin dye, which leads to resolution in approximately two-thirds of patients. 46 In pregnancy, uncomplicated CSCR also tends to resolve spontaneously soon after delivery and would not be an indication for pre-term delivery, 47 although there is a broad spectrum of severity, and cases of structural damage resulting in permanent visual loss have been reported. 48 Evidence on verteporfin use in pregnancy is limited, and PDT with verteporfin dye in pregnancy would not be advisable unless the benefits clearly outweigh the potential risks to both the woman and fetus, particularly since spontaneous resolution often occurs. Verteporfin exposure has been demonstrated to be teratogenic in rats but not rabbits, which may be related to the increased drug exposure from the former's smaller body surface area. 49 A recent review of the published literature yielded only two case reports of inadvertent exposure to verteporfin PDT in pregnant women, neither of which resulted in teratogenicity nor adverse obstetric outcomes.50,51
Pre-eclampsia may present with similar symptoms to CSCR and serous retinal detachments in the third trimester. 52 It should therefore be considered in the differential diagnosis for CSCR. In view of the obstetric and systemic implications of pre-eclampsia, pregnant patients presenting with suspected CSCR should be also assessed by an obstetrician or midwife at the time of presentation or assessment.
Chorioretinal diseases that may be induced by pregnancy or labour
Hypertensive disorders in pregnancy
Systemic hypertension can lead to abnormalities of the retinal and choroidal circulation, including vasoconstriction, ischaemia, hypoperfusion and increased microvascular permeability. This condition may be exacerbated in pregnancy, with vascular changes resulting in variable ocular manifestations mainly affecting the posterior segment circulation of the eye. Pregnancy-induced hypertension occurs in 5–11% of pregnancies, while mild and severe preeclampsia is reported to affect 0.5% and 3% of women, respectively. 3 Most of these women present with variable stages of retinopathy (mild, moderate or severe), and less than half are symptomatic.3,53,54 A comprehensive ophthalmic examination, including a dilated retinal fundus examination, should be performed to assess the severity of vascular changes and to evaluate the retinal microcirculation.53–56
Hypertensive retinopathy/choroidopathy
Hypertensive retinopathy is the most common ocular manifestation of systemic hypertension. Patients may be asymptomatic, or may present with acute or subacute onset blurring of vision. In the early stages, retinal changes may include focal or diffuse narrowing of the retinal arteriolar vessels indicated by a decreased ratio of the retinal artery-to-vein diameter. This may be associated with retinal haemorrhages, diffuse retinal oedema or lipid exudation that occurs due to the breakdown of the blood-retinal barrier. In addition, hypertension may induce focal areas of ischaemia of the retinal nerve fiber layer manifesting as cotton-wool spots (Figure 5(a) and (b)). In severe cases, hypertension may lead to increased intracranial pressure, causing optic disc swelling, ischemic optic neuropathy, and later optic atrophy.3,53,54
Hypertensive retinopathy. Images are shown of the left (a) and right (b) eyes showing cotton wool spots and retinal haemorrhages around the optic disc.
Hypertensive choroidopathy may be associated with hypertensive retinopathy, and mainly affects younger pregnant women with acute elevation of blood pressure. 3 The choroid is a highly vascularised tissue, and choroidal manifestations may have important implications for vision if prompt treatment is delayed. Clinical manifestations of hypertensive choroidopathy include serous retinal detachment, Elschnig spots (RPE lesions overlying occluded choriocapillaris vessels with significant leakage on fluorescein angiography) and Siegrist streaks (linear hyperpigmented streaks over choroidal arteries). The presence of choroidopathy may indicate a more aggressive form of hypertension that requires prompt intervention to preserve vision.3,44,53,57
The primary goal is medical control of hypertension. The majority of retinal changes will resolve following normalisation of blood pressure. However, severe changes at baseline may result in atrophic changes within the retina or optic nerve and limit the visual outcome. Hypertensive retinopathy would not normally be regarded as an indication for emergency delivery. Instead, this would be determined by the trajectory and severity of co-existent pre-eclampsia. In exceptional circumstances, severe progressive hypertensive retinopathy or concurrent retinal disease (e.g. retinopathy related to diabetes mellitus) may be an indication for expediting delivery, mainly in the context of preeclampsia. This decision should be supported by a multidisciplinary team discussion with the patient which weighs up the fetal risks from premature delivery and the maternal risks of visual deterioration or permanent visual loss.3,54
Retinopathy associated with pre-eclampsia and eclampsia
Up to 25% of patients with severe pre-eclampsia and 50% with eclampsia report experiencing visual symptoms. These include deterioration of vision (and occasionally severe vision loss), photopsia, scotomata, diplopia and visual field defects.3,53,54
Ophthalmic examination in symptomatic pregnant women with pre-eclampsia may reveal hypertensive retinopathy and choroidopathy, or exudative (also known as serous) retinal detachments. 3 In particular, retinal vascular changes have been reported in 30–100% of patients with pre-eclampsia . The presence of hypertensive choroidopathy, even in the absence of papilloedema, suggests the need for more aggressive blood pressure control to prevent permanent visual impairment.3,49
Exudative retinal detachment related to uncontrolled hypertension is uncommon and can occur in the third trimester of pregnancy or soon after delivery.3,48 Affected women may present with sudden loss of vision or blind spots in their visual field. It has been postulated that choroidal ischaemia secondary to uncontrolled hypertension results in thickening of the choroid and subsequent damage to the RPE. The resultant impairment of the RPE pump function causes the accumulation of fluid beneath the retina, leading to detachment of the retina from the RPE. Management is mainly conservative and involves treatment of the underlying hypertension. Spontaneous resolution of the exudative retinal detachment usually occurs within 2–12 weeks with good visual prognosis. 58
Causes of acute blindness in women with pre-eclampsia and eclampsia may include cortical blindness, which presents with normal fundoscopic findings and pupillary function. The underlying pathophysiology is attributed to vasoconstriction of vessels of the posterior cerebral circulation. Usually, most women will have a full visual recovery by 48–72 h postpartum, but those with severe eclampsia may develop retinal and optic nerve atrophy, which may permanently affect visual function.53,54,57
HELLP syndrome
Women with haemolysis, elevated liver enzymes and low platelets (HELLP) syndrome, a thrombotic microangiopathic vasculopathy complicating up to 15% of cases of pre-eclampsia, are more likely to develop severe ocular manifestations. 59 The commonest ocular findings include severe arteriolar spasm and hypertensive retinal vascular changes. In severe cases, women may develop exudative retinal detachment (0.9% of patients) and a uveal effusion-like syndrome. HELLP syndrome can also cause neurological manifestations in the form of occipital lobe infarction leading to cortical blindness.
Overall, uncontrolled hypertension is considered the main factor involved in the pathogenesis of vision loss in all hypertensive disorders of pregnancy including HELLP syndrome, therefore prompt blood pressure control is essential.60,61 In addition, increased inflammatory biomarkers have been reported in these women, and it has therefore been proposed that systemic administration of corticosteroids might be considered in severe cases.62,63 Corticosteroids might help to optimise RPE pump function, thereby promoting resolution of exudative retinal detachment. 60 In view of the above, a comprehensive evaluation and considered approach with regards to urgent delivery may help optimise systemic and visual outcomes in HELLP syndrome.
Retinal vascular occlusion
Pregnancy is a hypercoagulable state which is thought to be a physiological adaptation to prevent postpartum haemorrhage. Pregnant women are therefore at a higher risk of retinal vascular occlusive disorders, and this risk may increase in association with pre-existing hypercoagulable states such as primary antiphospholipid antibody syndrome and protein S deficiency.3,64,65
Pregnancies complicated by retinal artery occlusion are usually already affected by preeclampsia; however, the incidence is not well documented. 3 The risk of retinal arterial occlusion depends on underlying conditions such as systemic hypertension or hypercoagulability, as well as anatomical variations. Visual symptoms depend on the anatomic distribution – central retinal artery occlusion results in a global reduction in vision, hemi-retinal artery occlusion causes an altitudinal visual field defect, and branch retinal artery occlusions result in sectoral visual field loss. Fundoscopically, the affected area of the retina is pale and swollen, and the retinal arteries may appear attenuated with a ‘boxcar’ or ‘cattle-track’ appearance due to segmented blood flow. (Figure 6(a)) Visual prognosis is typically poor. Pregnant women with retinal artery occlusions should receive multidisciplinary management as per ischaemic stroke and be investigated thoroughly for underlying risk factors including hypertension and cardiovascular pathology. 65
Central retinal artery occlusion featured on (a) ultra-widefield imaging demonstrating cattle-trucking of the retinal arteries and whitening of the affected retina with (b) inner retinal thickening on the OCT scan. Central retinal vein occlusion featured on (c) ultra-widefield imaging demonstrating dilated tortuous retinal veins and widespread retinal haemorrhages, with (d) an OCT scan showing associated cystoid macular oedema.
Retinal vein occlusions (RVO) are another key type of retinal vascular occlusion. RVOs occur more commonly in the third trimester, and the risk of RVO during pregnancy is reported to be 0.5–1.8%. 3 Fundoscopically, central retinal vein occlusion (CRVO) is characterised by extensive retinal haemorrhages in all quadrants, dilated and tortuous veins, cotton-wool spots and possibly optic disc swelling (Figure 6(b)). In branch retinal vein occlusion (BRVO), there are localised retinal haemorrhages along the distribution of the affected vein, with the involved vein being dilated and tortuous. Macular oedema may be present if the macula is involved.
Management of uncomplicated RVO is typically conservative, but may involve sectoral or panretinal photocoagulation (a form of retinal laser treatment) if significant retinal ischaemia or neovascularisation is present. Intravitreal steroids or anti-VEGF injections could be considered if there is visually significant macular oedema, although the limited evidence base for safety in pregnancy belies the need for careful consideration of the potential associated treatment risks.3,15,66 RVO is associated with atherosclerotic risk factors in the general population, and so investigations for underlying systemic causes including hypertension, diabetes, hypercholesterolaemia and pro-thrombotic conditions should be considered. 3 These may need to be deferred until after pregnancy due to pregnancy-related changes in lipid levels, clotting factors and anticoagulants. It has been suggested that RVOs may be associated with pre-eclampsia/eclampsia rather than the state of pregnancy itself. 67 Importantly, RVOs are not associated with venous thromboembolism (VTE), so a history of this does not require VTE prophylaxis and should not count as a risk factor for VTE in pregnancy. 68
Purtscher-like retinopathy
Purtscher retinopathy is a rare condition usually associated with severe head trauma, although similar findings have been described in patients with acute pancreatitis or renal failure. Purtscher-like retinopathy has been occasionally associated with labour in the context of pre-eclampsia or HELLP syndrome. The exact pathophysiological mechanism is unknown, but appears to involve the occlusion of retinal arterioles by aggregated granulocytes or platelets following complement activation, micro-emboli or fibrin clots.69,70
Patients usually present with reduced visual acuity which is typically accompanied by visual field loss in the form of central, paracentral or arcuate scotomata, while the peripheral visual field is usually preserved. Fundoscopic examination shows cotton wool spots, retinal haemorrhages and Purtscher flecken (pathognomonic retinal lesions). Visual prognosis is variable – most cases tend to resolve spontaneously within 1 to 3 months with full recovery of vision, but the prognosis is guarded in cases with optic disc swelling, or outer retinal or choroidal involvement.71,72
There is no definitive treatment for Purtscher-like retinopathy in pregnancy. Treatment with high-dose intravenous steroids has been reported to confer better visual outcomes, although there is no conclusive evidence to support this, meaning that observation is the most frequently recommended option.69,70
Conclusion
Ophthalmic conditions affecting the choroid and retina in pregnancy present distinct challenges for both ophthalmologists and obstetricians. This is because of the dynamic physiological changes in pregnancy that may exacerbate pre-existing chorioretinal conditions or lead to new pathologies, and the need for cautious management balancing competing maternal and fetal risks. Early recognition and treatment is crucial for preserving vision. It is important not to omit key treatments in pregnancy because of theoretical concerns about fetal exposure. By carefully considering the role of the available imaging techniques as well as the risks and benefits of various treatment options, obstetricians and ophthalmologists can work together to optimise visual and systemic outcomes for pregnant women as well as fetal outcomes in these complex situations.
Footnotes
Declaration of conflicting interest
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: WT has a consultancy agreement with Roche unrelated to the submitted work, which has not influenced the content or conclusions of the review. CF is the Editor-in-Chief of Obstetric Medicine: The Medicine of Pregnancy.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Contributorship
AYO, CF, CK and SdS conceptualised the review. AYO and EEC curated and analysed data and wrote the original draft which was revised and approved by all authors.
