Abstract
Objectives
To determine the necessary screening interval for retinopathy in diabetic patients with no retinopathy based on time to laser therapy and to assess long-term visual outcome following screening.
Methods
In a population-based community screening programme in North Wales, 2917 patients were followed until death or for approximately 12 years. At screening, 2493 had no retinopathy; 424 had mostly minor degrees of non-proliferative retinopathy. Data on timing of first laser therapy and visual outcome following screening were obtained from local hospitals and ophthalmology units.
Results
Survival analysis showed that very few of the no retinopathy at screening group required laser therapy in the early years compared with the non-proliferative retinopathy group (p < 0.001). After two years, <0.1% of the no retinopathy at screening group required laser therapy, and at three years 0.2% (cumulative), lower rates of treatment than have been suggested by analyses of sight-threatening retinopathy determined photographically. At follow-up (mean 7.8 ± 4.6 years), mild to moderate visual impairment in one or both eyes due to diabetic retinopathy was more common in those with retinopathy at screening (26% vs. 5%, p < 0.001), but blindness due to diabetes occurred in only 1 in 1000.
Conclusions
Optimum screening intervals should be determined from time to active treatment. Based on requirement for laser therapy, the screening interval for diabetic patients with no retinopathy can be extended to two to three years. Patients who attend for retinal screening and treatment who have no or non-proliferative retinopathy now have a very low risk of eventual blindness from diabetes.
Introduction
Historically, diabetic retinopathy (DR) was the leading cause of loss of vision in people of working age in the UK. 1 Recent UK data indicate that this is no longer the case,2,3 probably as a result of modern management of risk factors, active treatment by ophthalmologists and, recently, population-based screening for DR. UK programmes have aimed to screen patients annually, although there is little clear evidence to support this screening interval. There remain substantial uncertainties over whether there should be a universal screening interval and, if so, how long it should be or whether certain categories of patient should be screened more or less frequently. The advent of population-based screening programmes has generated the opportunity to examine these questions.
Previous studies have looked at time to sight-threatening retinopathy (STR) or “referable retinopathy” determined photographically.4–12 However, many such patients do not need immediate laser therapy when assessed by an ophthalmologist, usually because there is no macular oedema. This cannot be easily assessed by standard retinal cameras. Use of the STR criteria determined photographically exaggerates the number of potentially serious cases needing treatment. Thus, analysis of the timing of laser treatment, the clinical endpoint of screening, is required to determine how many patients need treatment at given time periods, and how often screening should take place.
From 2001, we ran a population-based DR screening programme (previously described) in North Wales. 13 The programme covered a total population of 278,000 with a population prevalence of diabetes then of 2.76%. Subsequently, an all-Wales screening programme was established. Here, we study the need for eventual laser therapy in patients who had no DR when screened in 2001–2002. By examining length of time from screening to first laser treatment we aimed to determine whether these patients need annual screening or if a longer time interval could be instituted. Other large screening programmes have found that 65% to 70% of their diabetic population have no retinopathy at screening.10,14 Less frequent screening for this patient group would have an impact on the economics of DR screening.
Methods
The no retinopathy and non-proliferative retinopathy groups at initial screening for diabetic retinopathy.
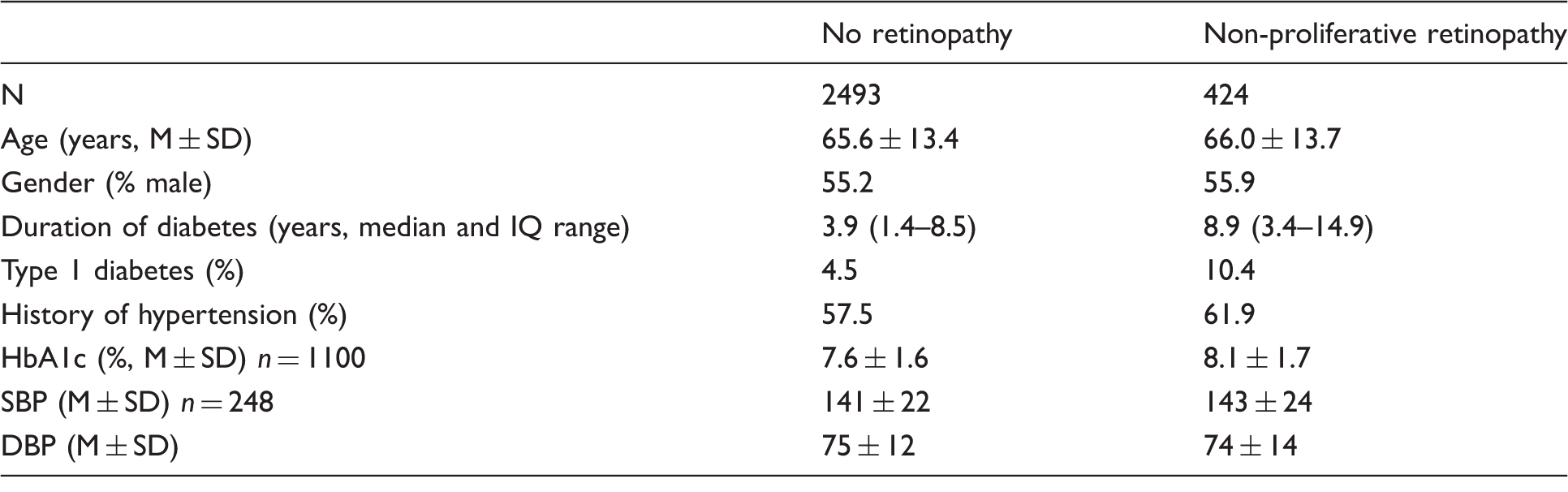
SD: standard deviation; M: mean; IQ: intelligence quotient; SBP: systolic blood pressure; DBP: diastolic blood pressure.
The two groups were significantly different only in terms of duration of known diabetes and percentage with type 1 diabetes (both p < 0.001).
HbA1c and blood pressure data recorded within three years of screening were available for a sample. Patients were followed until death, until they left the area (information from Welsh Demographic Services NHS database), or for approximately 12 years. The primary end-point was time from screening to first retinal laser therapy undertaken specifically for DR. Baseline data, date of any laser treatment, and visual outcome were determined from individual hospital records, computerized clinical letter systems, and appointment systems at all local hospitals and eye clinics, diabetes registers, and pathology laboratory systems. Follow-up screening occurred approximately annually. Where there was no other information, and we could show that a patient had not attended an ophthalmology clinic, no significant decline in visual acuity (VA) from screening was assumed. Patients with previous retinal laser treatment for DR or who had proliferative retinopathy were excluded. Corrected VA was deemed normal if it was 6/9 (logMar 0.2) or better in both eyes tested separately. As a broad equivalent to the UK definition of legal blindness, we deemed patients who had VA 6/60 (logMar 1.0) or worse in their better eye to be blind. Visual field assessment was mostly not performed.
Baseline characteristics and visual outcome in the two groups were compared by t test or chi square as appropriate. The survival probability without laser treatment in patients with no retinopathy at baseline was compared with those who had non-proliferative retinopathy by Kaplan–Meier analysis and log rank test. From this analysis, we compared the survival probabilities of patients having laser treatment at one to six years of follow-up in each group. Factors predicting laser treatment were assessed using Cox proportional hazards regression analysis (backwards stepwise). Retinopathy at screening, age at screening, gender, duration of known diabetes, diabetes type, and history of hypertension were the predictor variables modelled. A similar analysis was conducted including HbA1c in patients where those data were available. Statistical analysis was undertaken using SPSSv22. Data are reported as mean ± SD unless otherwise stated.
Results
We were able to obtain information on 2917 of the 2955 patients identified. At screening, 2493 had no retinopathy and 424 had non-proliferative retinopathy. High street optometrists undertook screening by slit lamp biomicroscopy in 86% of visits. Some patients were screened at care homes, where slit lamp was not used. The patient population was > 99% white Caucasian. Table 1 shows the baseline parameters at screening for each group. Of the total, 155 (5.3%) had type 1 diabetes, 1828 (63%) were treated with oral agents, 644 (22%) were treated by diet alone, and a further 260 of those with type 2 diabetes (9%) were treated with insulin. Treatment was unknown in 30 (1%). Follow-up to death was carried out in 42% of patients, 41% and 50% in the no retinopathy and retinopathy groups, respectively. In the retinopathy group, the grade of retinopathy at screening was 20 or 3 (microaneurysms, retinal haemorrhages, exudates well displaced from the fovea) in 80%, preproliferative (grades 30–50) in 8%, and STR by photographic criteria (grade 4–5: exudates within one disc diameter of the fovea or ischaemic maculopathy) in 11%.
Duration of follow-up (for all patients) was 9.1 ± 3.6 years (n = 2917); follow-up was until death in 1233, and 11.4 ± 1.5 years in the remainder. The proportion treated with laser was 76/2493 (3%) in the no retinopathy group, and 99/424 (23%) in the retinopathy group. In the latter group, 65 were graded at screening as having STR (ETDRS grades 40, 50, and 4), but only 30 required eventual laser treatment at any future time. Figure 1 shows Kaplan–Meier analysis of laser treatment over time. In the no retinopathy group, no patients required laser therapy one year after screening. Two patients, with duration of known diabetes of 8 and 10 years, had had laser therapy by two years after screening (<0.1% of the cohort). A further three patients had had laser therapy by three years (duration of known diabetes 2–15 years, Table 2). Of those with no retinopathy at baseline, 1% of patients had required laser therapy by 5.8 years, but in those with retinopathy at baseline, 1% required laser therapy by 0.06 years. For 2% requiring laser therapy, the times were 7.9 and 0.23 years, respectively.
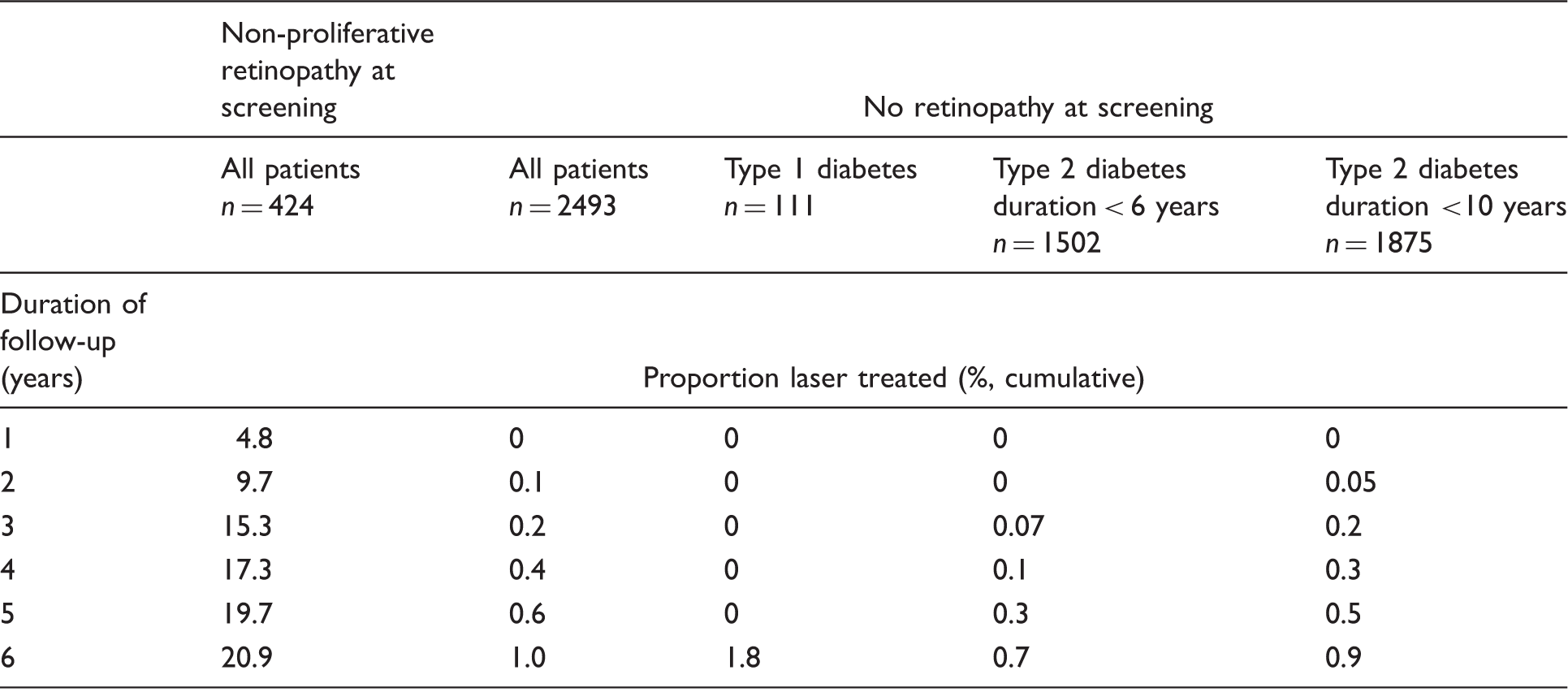
The proportions of patients in the two groups with and without any retinopathy at screening undergoing laser therapy over time (Kaplan–Meier analysis). Survival without laser treatment was significantly different between the two groups (p < 0.001, log rank test). The proportions of patients undergoing laser treatment in the six years following screening, from Kaplan–Meier analysis, according to presence of retinopathy at screening. The three right hand columns show the rates of laser treatment in patients with no retinopathy at screening and type 1 diabetes or type 2 diabetes with duration of diabetes <6 years or <10 years.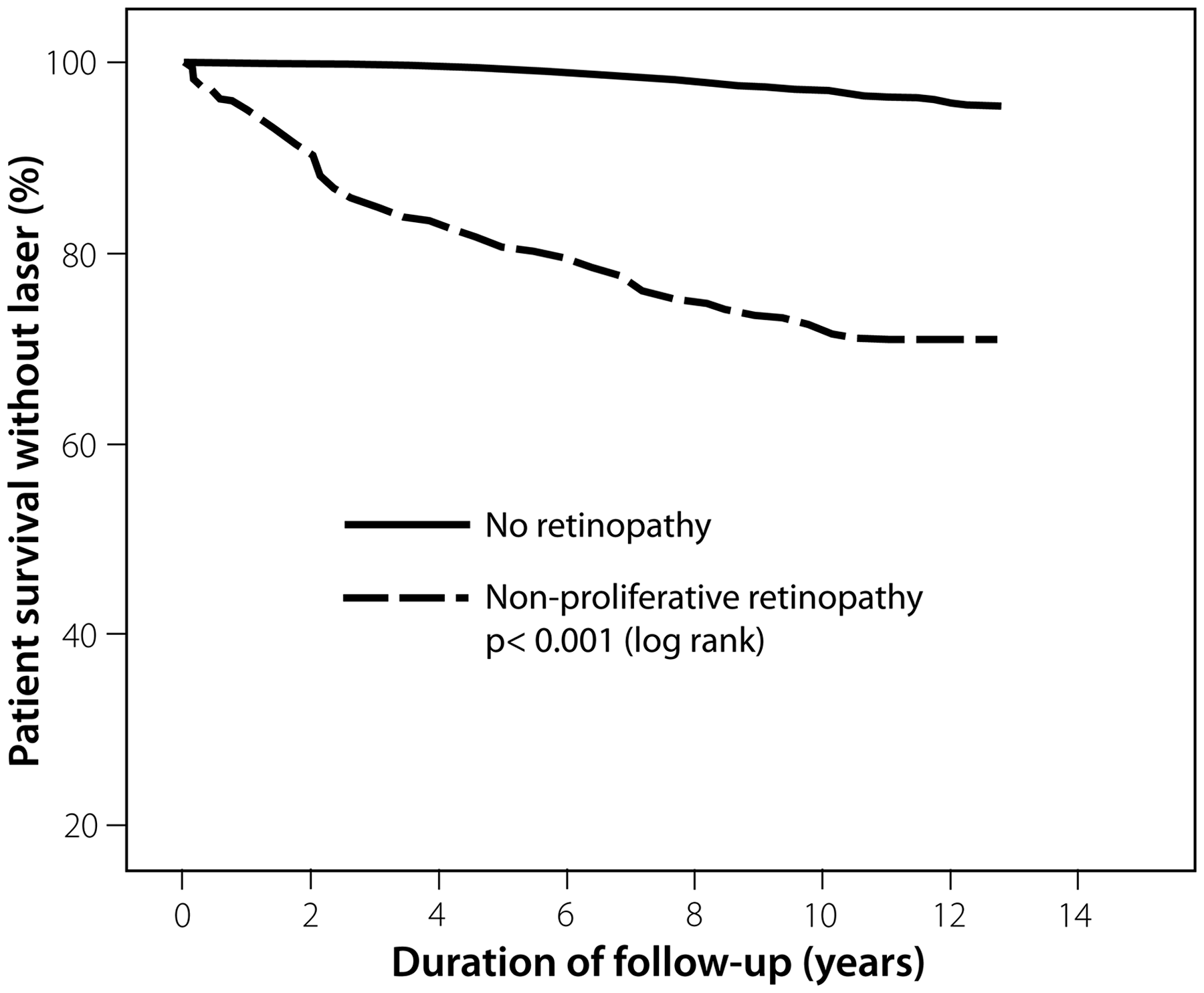
Cox regression indicated that predictors at screening of future laser treatment were presence of retinopathy at screening (odds ratio (OR) = 9.6, p < 0.001), age at screening (per year, OR = 0.96, p < 0.001), duration of diabetes (per year, OR = 1.035, p < 0.001), and type of diabetes (type 1 vs. others, OR = 1.9, p = 0.04). If insulin therapy was used as a predictor instead of type 1 diabetes, the OR was 1.3 (not significant). Among type 1 patients, 16.1% required laser therapy compared with 6.4% of the remainder (χ2 = 21.0, p < 0.001). If the analysis was restricted to those with an HbA1c value (n = 1100), then presence of retinopathy (OR = 9.5, p < 0.001), age at screening (OR = 0.96, p < 0.001), and duration of diabetes (per year, OR = 1.04, p = 0.008) were predictors of future laser therapy as before, but HbA1c (per 1%, OR = 1.38, p < 0.001) and history of hypertension (OR = 2.40, p = 0.006) were stronger predictors than type of diabetes (not significant).
We determined the effect of the type and duration of diabetes on numbers needing laser therapy. In patients with type 1 diabetes and no retinopathy at screening, none needed laser therapy for at least five years from screening. No patient with type 1 diabetes required laser therapy until they had had diabetes for at least nine years, irrespective of whether retinopathy was identified at screening or not. The proportion of type 2 diabetic patients with no retinopathy at screening requiring laser therapy when duration of known diabetes is < 6 years or < 10 years is shown in Table 2.
Long-term visual acuity (VA) outcome according to presence of retinopathy at screening.
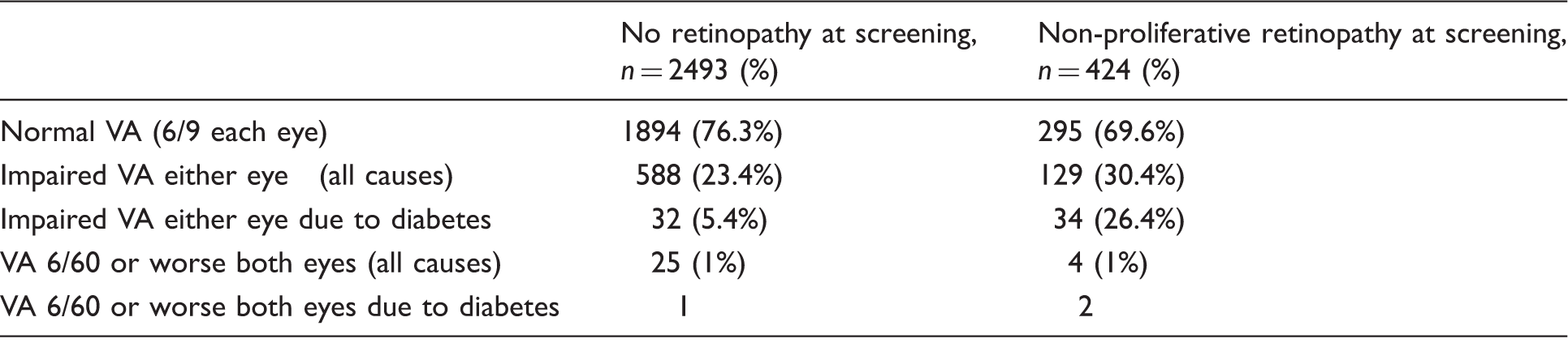
Impaired VA is defined as VA worse than 6/9 (logMar 0.2) in one or both eyes. Impaired VA due to diabetes is expressed as a percentage of all cause impaired vision. In the no retinopathy at screening group VA outcome was unknown in 11. At follow-up the proportion of patients with impaired VA (all cause) was less in the patients with no retinopathy at screening (p = 0.003). The proportion with impaired VA due to diabetes was also less (p < 0.001).
Discussion
Although this is a retrospective analysis, outcomes were successfully ascertained in a high proportion of patients. The number undergoing laser therapy in the no retinopathy at screening group was low but there was a high proportion among patients with retinopathy at screening indicating that we were not failing to identify laser treatments. A small number of patients went to specialist units out of our area but they were always referred by local ophthalmologists and so we were able to track their progress. The assumption that deteriorating VA would prompt eye clinic referral seems likely to be true. Deteriorating acuity due to DR would always result in referral so we will not have missed blindness due to diabetes even if there were incomplete referral of reduced vision from other causes.
This is a population-based study of all classes of diabetic patients presenting a reasonably typical cross section as found in many communities. Primary care physicians were encouraged to refer all diabetic patients not under specialist ophthalmology care. Hospital diabetic clinic patients were also included. We do not believe that there is any selection bias.
The difference in need for laser therapy between the two groups was very clear, with the no retinopathy at screening group having few patients needing laser in the early years. There is debate over whether patients without retinopathy require annual screening. Sweden, Iceland, and New Zealand have already taken the decision to screen such patients every two years.7,17 The current evidence in support of this comes primarily from large screening programmes assessing numbers who progress to STR by photographic criteria. Progression from no retinopathy to STR occurred in 0.8% at two years in the Liverpool programme.4,5 Norwich identified an annual incidence of <0.5% per year, with cumulative incidence of STR of 5% at five years in patients with short diabetes duration. 8 In Wales, 0.485% of people with type 2 diabetes had STR by two years. 14 The Turin group found STR after two years in 0.47% of young onset insulin-treated patients with duration <10 years, but 1.59%–3.59% in other categories of diabetic patient. 11 Data from Scotland showed lower proportions of patients progressing if they had two preceding negative examinations: <0.3% for T1DM and <0.2% in T2DM to STR at two years.10,12 We found <0.1% needing laser treatment at two years. Our figures for treatment at two years and subsequent time points were substantially lower than those suggested by analyses based on STR. These data illustrate the difference between analyses based on STR and analysis based on laser treatment. Laser therapy was the clinical end-point of the screening pathway. STR and referable retinopathy are surrogates derived from the screening process. They may be easier to measure and have better reproducibility (due to standardized grading), but they are ultimately less definitive. Many patients are referred because of maculopathy, retinopathy within one disc diameter of the macula on retinal photograph, but are subsequently found to have no macula thickening on optical coherence tomography with laser treatment not indicated. Our own data showed that less than half of those graded at screening as having STR required laser therapy at any point in the future. Thus, these surrogate measures (photographic STR) substantially overestimate the numbers of patients needing specialist treatment and cannot be used to accurately estimate screening intervals. For direct comparison, the UK Prospective Diabetes Study seems to be the only other analysis of time to laser therapy, but the subjects in that study were newly diagnosed type 2 diabetic patients. 18 The rates of laser therapy (0.2% of patients at three years, 1.1% at six years and 2.6% at nine years) are virtually identical to ours.
An alternative approach has been to institute the longer screening interval and assess results. Kristinsson et al. 19 followed 205 type 1 and 2 patients in Iceland. No STR or need for laser therapy was identified after two years. Olafsdottir and Stefansson 17 reported 10-year follow-up in Iceland on 296 patients screened biennially as long as they had no retinopathy. No patient had undue delay in the treatment of STR. Agardh and Tababat-Khani suggested a three-year screening interval but based on data from a population of patients with mean HbA1c 6.4%. 7 A recent systematic review suggested more data were needed before making a general recommendation to increase screening intervals. 20
In multivariate analysis, the presence of any retinopathy at screening was the strongest predictor of future laser therapy. This finding was independent of other predictors: duration of diabetes, younger age, diabetes type, glycaemic control, and history of hypertension. Laser therapy was more common in type 1 than other types of diabetes. Older patients were less likely to undergo laser therapy, consistent with other Welsh data. 14 The population studied here was typical of many diabetic populations, so the results have general applicability.
Our results support calls to change screening policy. Overall, patients with no retinopathy can safely be screened every two to three years irrespective of other factors. Laser treatment is very unlikely to be required before this. After presence of any retinopathy, multiple regression showed type and duration of diabetes to be significant predictors of future need for intervention. Therefore, we determined the numbers of patients in these subgroups requiring laser therapy. In type 1 diabetes, the data suggest that screening is not necessary before nine years duration of diabetes or for five years following screening negative for DR. The numbers with type 1 diabetes were not large (total 155), so these findings should be treated with caution. Duration of diabetes is much less clear in type 2 diabetes, but even here, patients with a known duration of diabetes <6 years have very low rates of laser treatment in the early years following a negative screen, and could reasonably be rescreened after three years.
Determining which screening interval to adopt involves an arbitrary decision on the acceptable level of risk without screening. The data indicate that the proportion of patients likely to have active treatment delayed as a result of longer screening intervals would be very small. The advantage of such a change would be better targeting of screening and reduction in costs. The Welsh Screening Service found that 67% of patients with type 2 diabetes screened between 2005 and 2009 had no retinopathy. 14 Data from Scotland also indicate that such a change in policy would lead to a substantial reduction in the numbers to be screened each year. 10
Visual outcome for patients in this programme was good. At follow-up, there was mild to moderate visual impairment in either eye due to diabetes in 1.3% of the no retinopathy at screening group but 8.0% of the retinopathy group. Blindness by our measure (6/60 vision in the best eye) due to diabetes occurred only in 3/2917 patients (Table 2). Thus, screening and treatment in those with no or mild to moderate non-proliferative DR was largely successful in preventing blindness. These results are consistent with a documented fall in blind registrations due to diabetes in the UK.2,3 Large screening programmes have also shown a reduction in the numbers of cases of STR by photographic criteria. 21
Conclusions
Analysis of screening intervals should be based on time to laser therapy in the screened population. This indicates lower rates of laser treatment than inferred from analysis of STR determined photographically, which overestimates the incidence of disease needing treatment. For all patients with no retinopathy, a change to follow-up screening at two to three years results in almost zero delay in treatment. Patients with no or non-proliferative retinopathy undergoing regular screening and treatment when indicated now have a very low risk of blindness from diabetes when assessed in long-term follow-up.
Footnotes
Acknowledgments
We are grateful to Mrs Petula Whittle for help with identifying patient records.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
