Abstract
Background:
Interstitial lung diseases (ILDs) are a heterogeneous group of disorders with complex etiologies and nonspecific clinical manifestations, often requiring histopathological confirmation for accurate diagnosis. Transbronchial lung cryobiopsy (TBLC) has emerged as a less invasive alternative to surgical lung biopsy (SLB) for obtaining lung tissue, yet its role in multidisciplinary discussion (MDD) remains underevaluated.
Objectives:
This study aimed to assess the diagnostic yield and safety of TBLC when integrated into MDD for diagnosing ILDs.
Design:
A retrospective dual-center study was conducted on 208 patients with undiagnosed ILDs who underwent TBLC at Shanghai Chest Hospital and Fuzhou Pulmonary Hospital from May 2021 to December 2024.
Methods:
TBLC procedures were performed under general anesthesia, with samples evaluated by pathologists and integrated into MDD involving pulmonologists, radiologists, and rheumatologists. Diagnostic yield, complication rates, and concordance were analyzed.
Results:
TBLC achieved a histological diagnostic yield of 88.9% (185/208 cases), which improved to 97.6% (203/208 cases) after MDD. The most common diagnosis was hypersensitivity pneumonitis (HP, 18.3%) followed by idiopathic nonspecific interstitial pneumonia (iNSIP, 15.4%). Complications were rare, with pneumothorax occurring in 5.3% of cases and moderate bleeding in 3.4%. No procedure-related mortality was observed.
Conclusion:
TBLC is a safe and effective diagnostic tool for ILDs, with high diagnostic yield when integrated into MDD. TBLC reduces the need for SLB while maintaining low complication rates. These findings support TBLC as a valuable component of the MDD approach for ILDs.
Plain language summary
Background Interstitial lung disease (ILD) is often difficult to diagnose and may require a lung tissue sample. Surgical biopsy is effective but invasive. A less invasive procedure, called transbronchial lung cryobiopsy (TBLC), is now available, but its value in a team-based diagnostic approach needs further confirmation. Objective This study evaluated how effective and safe TBLC is when its results are reviewed by a multidisciplinary team (MDD) for diagnosing ILD. Methods We reviewed data from 208 patients with undiagnosed ILD at two hospitals in China. All patients underwent TBLC. Their biopsy results were then discussed by a team of lung specialists, radiologists, and pathologists to reach a final diagnosis. Key Results TBLC alone provided a clear histological diagnosis in 88.9% of cases. After the multidisciplinary team discussion (MDD), a final diagnosis was reached in 97.6% of cases. The most common final diagnoses were Hypersensitivity Pneumonitis and Idiopathic Nonspecific Interstitial Pneumonia. The procedure was safe: pneumothorax occurred in 5.3% and moderate bleeding in 3.4% of patients. No deaths were related to the procedure. Conclusion Transbronchial lung cryobiopsy (TBLC) is a safe and effective diagnostic tool for ILD. When its results are integrated into a multidisciplinary team discussion, the diagnostic success rate is very high. This supports TBLC as a valuable alternative to more invasive surgical biopsy.
Background
Interstitial lung diseases (ILDs) represent a heterogeneous group of disorders characterized by inflammation and fibrosis of the pulmonary interstitium, leading to impaired gas exchange and progressive respiratory failure. 1 Clinical presentation typically includes progressive dyspnea and dry cough, with systemic manifestations such as fever, arthralgia, or skin changes often indicating underlying connective tissue disease (CTD).2,3
Given their complexity and nonspecific presentations, ILDs require multidisciplinary collaboration for accurate diagnosis. In 2002, the American Thoracic Society (ATS) and the European Respiratory Society (ERS) established the multidisciplinary discussion (MDD) model for the diagnosis of ILDs. 4 Subsequent studies have shown that MDD improves interobserver agreement, diagnostic confidence, and accuracy in idiopathic interstitial pneumonias (IIPs) and idiopathic pulmonary fibrosis (IPF).5,6 MDD plays a crucial role in the classification and diagnosis of ILDs by integrating clinical, radiological, and histopathological data through collaboration among specialists from different disciplines, thereby enhancing diagnostic accuracy and therapeutic decisions.7–9 Current guidelines recognize MDD as the diagnostic gold standard for IPF and other ILDs.10–12
Histopathological confirmation is essential for many ILDs, with specimen acquisition methods including surgical lung biopsy (SLB), transbronchial lung cryobiopsy (TBLC), and conventional transbronchial lung biopsy (TBLB).10,13–16 SLB, usually performed through video-assisted thoracic surgery, is the gold standard for identifying histopathological patterns of ILDs, with SLB data significantly enhancing MDD diagnostic accuracy.6,10,17,18 However, SLB requires hospitalization and is associated with complications such as bleeding, infection, and acute exacerbation,19,20 thus limiting its use in clinical practice. TBLC has emerged as a less invasive alternative to SLB, achieving a diagnostic concordance of 70.8% with SLB, which increases to 76.9% after MDD.21–25 TBLC can retrieve larger tissue samples with a higher percentage of alveolar tissue, fewer crush artifacts, and less atelectasis compared to traditional methods.26,27 The diagnostic yield of TBLC in patients with unclassified ILDs is approximately 79%.25,28,29 While TBLC offers a high diagnostic yield, it has some risks, including bleeding and pneumothorax, which may necessitate hospitalization and chest tube drainage.22,26,27,30
This study aims to describe our experience with TBLC in undiagnosed ILD patients from May 2021 to December 2024. We evaluated diagnostic consistency among initial clinical, histological, and MDD diagnoses, as well as procedure-related complications. By examining real-world data, this study seeks to support the integration of TBLC into the diagnostic algorithm for ILDs, ultimately enhancing the precision and efficacy of MDD.
Patients and methods
A descriptive retrospective study was conducted on all patients with inconclusive diagnoses who underwent TBLC at Shanghai Chest Hospital, Shanghai Jiao Tong University, and Fuzhou Pulmonary Hospital. This study has been approved by the ethics committees in both institutes, and informed consent was waived. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 31 The completed STROBE checklist is provided as Supplemental File.
Inclusion criteria (indication of TBLC) were as follows: (a) patients with suspected ILDs based on clinical presentation, radiological examination, pulmonary function test (PFT), and laboratory examination; (b) histologic confirmation required (for diagnosis or treatment decisions); (c) no absolute contraindications, such as severe cardiopulmonary dysfunction, uncorrectable coagulation dysfunction, etc.; and (d) signed informed consent for TBLC.
Exclusion criteria included (a) age >80 years; (b) the presence of severe comorbidities; (c) the diagnosis of connective tissue disease-associated ILD (CTD-ILD) or interstitial pneumonia with autoimmune features (IPAF); and (d) patients who are unable to engage in MDD due to various reasons such as patient refusal, incomplete information, etc.
The patients underwent three sequential diagnostic evaluations: (1) Initial clinical diagnosis: patients were evaluated by a multidisciplinary team (MDT) comprising pulmonologists, thoracic surgeons, rheumatologists, and radiologists, based on imaging and laboratory findings. (2) Histological diagnosis: tissue samples obtained via TBLC were analyzed by lung pathologists to establish a definitive pathological diagnosis (typical histopathological images are shown in Figure 1). (3) MDD diagnosis: Patients were re-evaluated by the same MDT, now augmented with lung pathologists, to integrate clinical and pathological data into a consensus diagnosis.

Histopathological features of various interstitial lung diseases. (a) UIP—normal lung tissue alternating with fibrotic areas, showing honeycombing and fibroblast foci. (b) HP—inflammatory cell infiltration and fibrosis with focal organization and atypical granulomas. (c) OP—inflammatory cell infiltration with prominent intra-alveolar organization. (d) NSIP—alveolar septal widening due to fibrosis and mild lymphocytic infiltration. (e) Sarcoidosis—non-necrotizing granulomas predominantly located along the lobular periphery. (f) SR-ILD—lymphocytic infiltration with mild anthracosis and fibrosis, accompanied by pigmented macrophage accumulation in alveoli. (g) Lipoid pneumonia—mild lymphocytic infiltration with abundant lipid-laden macrophages in alveolar spaces. (h) Occupational exposure-related ILDs—fibrosis with mild lymphocytic infiltration and focal deposition of black particulate matter. (i) PAP—focal alveolar spaces are filled with eosinophilic granular material which is positive in both PAS staining and digested PAS staining.
The MDD process was standardized across both centers as follows:
Pre-MDD Materials: Prior to the meeting, the following de-identified materials were systematically compiled for each case. (1) Complete clinical history and physical examination findings, including drugs and occupational exposure. (2) PFT, including forced vital capacity (FVC), forced expiratory volume in 1 second (FEV1), and diffusing capacity of the lung for carbon monoxide (DLCO). (3) Laboratory results (including autoantibody serology). (4) High-resolution computed tomography (HRCT) scans. (5) Histopathological slides and report from the TBLC specimens (this item is not required at initial clinical diagnosis).
Discussion and Decision-Making Protocol: Each de-identified case underwent a standardized two-step diagnostic process: Step 1 involved presenting clinical data, HRCT findings, laboratory results, and PFTs, after which MDD participants independently recorded preliminary diagnosis and convened to establish a consensus-based initial clinical diagnosis. Step 2 comprised a blinded histopathological interpretation of TBLC specimens, with pathologists concealing specimen dimensions to maintain blinding; participants again recorded individual diagnosis before reaching a final consensus MDD diagnosis. In rare cases of persistent disagreement, the case was reviewed by a senior ILD pulmonologist (F.L) who made the final diagnostic determination after deliberating all viewpoints. A sample was considered non-diagnostic when insufficient histopathologic features were present to identify a disease. A sample was considered invalid if lung parenchyma was absent from the sample.
All TBLC procedures were performed by experienced bronchoscopists in the operating room under general anesthesia, following a standardized protocol across both institutions to ensure consistency and safety. Anesthesia & Airway: Patients underwent endotracheal intubation (tube caliber 8–8.5 mm). Equipment: A standard flexible bronchoscope (BF-160, Olympus, Japan) and a 1.9-mm cryoprobe (Erbokryo CA, ERBE, Germany) were used in all cases. Target Selection: The biopsy site was selected based on pre-procedural HRCT findings, prioritizing the lobe with the most active or representative disease, while avoiding areas of honeycombing or severe fibrosis. Biopsy Technique: A standard bronchoscope was used to conduct an airway examination, a balloon catheter was inserted into the planned location, and a cryoprobe was inserted through the working channel and into the target bronchial segment. Biopsies were performed with the cryoprobe tip guided to a position about 2 cm away from the pleura. The cryoprobe was cooled down for 3–4 s using liquid nitrous oxide, and pulled back with the frozen tissue. Specimen Acquisition & Hemostasis: A minimum of one biopsy was mandated, with the goal of obtaining 3–5 samples from the same target area to maximize diagnostic material while balancing procedural risk. The final number was at the operator’s discretion, considering factors such as specimen size and early bleeding. Post-biopsy Management: The catheter balloon was inflated to prevent hemorrhage. The presence and severity of bleeding were assessed after deflating the balloon, and cold saline or adrenaline could be used to treat active bleeding. Bleeding severity was classified as mild (single bleeding volume less than 20 ml), moderate (bleeding 20–150 ml in 24 h), or massive (bleeding more than 150 ml in 24 h). Post-procedure Chest X-ray was used to identify pneumothorax.
Complications, such as pneumothorax, bleeding, subcutaneous emphysema, mediastinal emphysema, pneumonia, acute exacerbation of ILDs, intraoperative hypoxemia, and 30-day post-interventional mortality, were reported.
The medical records were analyzed, and demographic data, occupational or personal exposure, CTD serology, PFT, HRCT scans, procedure details, size and number of biopsies, initial clinical diagnosis, histological diagnosis, and final MDD diagnosis results were recorded. Diagnostic concordance among these diagnoses was assessed and quantified using Cohen’s kappa (κ) statistic. Cases were considered concordant if the specific disease entity was identical between the two diagnoses.
Data are presented as the mean ± standard deviation (SD) or median (interquartile range, IQR). Analysis was performed using IBM SPSS Statistics 22 (IBM Corp., Armonk, NY, USA) or GraphPad Prism 5 (GraphPad Software, San Diego, CA, USA). Student t-test, Mann–Whitney U test, or the chi-square (χ2) test was used to compare differences between different groups according to their distribution. To identify factors independently associated with the histological diagnosis, the MDD diagnosis, and diagnostic concordance, variables with p < 0.1 in univariate analyses or of clinical relevance were entered into a multivariate binary logistic regression model. Results are presented as odds ratios (OR) with 95% confidence intervals (CI). A two-sided p-value <0.05 was considered statistically significant.
Results
Demographic and clinical data
A total of 208 patients with suspected ILDs who underwent TBLC at both institutes were included in this study. The demographic and clinical characteristics of the study cohort are summarized in Table 1. The median age of patients was 61 years (IQR: 54–67), with a predominance of males (64.9%). Regarding smoking status, 60.1% were nonsmokers, 27.9% were current smokers, and 12.0% were former smokers. The median BMI was 24.1 kg/m2 (IQR: 22.2–26.6). A history of potential antigenic exposure (e.g., birds, molds, occupational dusts) was identified in 41 patients (19.7%). Most patients presented with respiratory symptoms at baseline, with 60.1% experiencing multiple symptoms concurrently, 15.4% reporting cough, 13.5% presenting with dyspnea, 1.4% with fever, and 0.5% with expectoration. Notably, 9.1% of patients were asymptomatic, with their ILDs detected incidentally during routine examinations. CTD serological testing was performed in all patients, and the results were negative. PFT revealed a mean FVC of 2.8 ± 0.9 L, representing a median of 83.8% of predicted values (IQR: 67.0–95.0%). The median FEV1/FVC ratio was 83.9% (IQR: 79.2–88.1), indicating normal airflow. Median total lung capacity (TLC) was 93.2% of predicted values (IQR: 76.3–110.7%). DLCO was mildly to moderately reduced, with a mean of 68.5 ± 23.3% of predicted values. (Due to some patients being unable to perform PFTs, FVC and FEV1 were recorded in 193 patients, and DLCO was recorded in 183 patients.)
Patients’ demographics and clinical data at baseline.

DLCO, diffusing capacity of the lungs for carbon monoxide; FEV1, forced expiratory volume in 1 s; FVC, forced vital capacity; PFT, pulmonary function test; TLC, total lung capacity.
Procedural characteristics and complications
The procedural details of TBLC are outlined in Table 2. The right lower lobe was the most frequent biopsy site (53.8%), followed by the left lower lobe (26.4%), right upper lobe (6.3%), and left upper lobe (3.8%). No biopsies were obtained from the right middle lobe, while multiple lung lobes were sampled in 9.6% of cases. The median number of biopsy samples per patient was 3 (IQR: 2–5). Regarding specimen size, 76.4% of biopsies yielded samples ⩾5 mm in diameter, while 23.6% were <5 mm.
Procedure details of patients undergoing TBLC.

ILDs, interstitial lung diseases; TBLC, transbronchial lung cryobiopsy.
Complications were infrequent, with pneumothorax occurring in 5.3% of cases (11 patients) and moderate bleeding in 3.4% of cases (7 patients). No cases of acute exacerbation of ILDs, subcutaneous emphysema, mediastinal emphysema, or pneumonia were observed. Importantly, there were no procedure-related deaths, with a 30-day mortality rate of 0%.
Distribution of initial clinical diagnoses, histological diagnoses from TBLC, and MDD diagnoses
The initial clinical diagnoses, based on clinical and radiological data prior to biopsy, are presented in Table 3. Unclassified ILD (UILD) was the most frequent pre-TBLC categorization, observed in 33.7% of cases, followed by idiopathic nonspecific interstitial pneumonia (iNSIP) in 22.6% and pulmonary infection in 9.6%. Clinically suspected usual interstitial pneumonia (UIP) was recorded in 6.3% of cases, while suspected IPF and hypersensitivity pneumonitis (HP) accounted for 4.8% and 3.8%, respectively. Other clinical impressions included unclassifiable IIP (2.9%), sarcoidosis (2.4%), alveolar proteinosis (2.4%), smoking-related ILD (SR-ILD) (2.4%), and occupational exposure-related ILD (2.4%).
Distribution of initial clinical, histological, and MDD diagnoses.

ACIP, airway-centered interstitial fibrosis; AFOP, acute fibrinous organizing pneumonia; AIP, acute interstitial pneumonia; COP, cryptogenic organizing pneumonia; DR-ILD, drug-related ILD; EGPA, eosinophilic granulomatous polyangiitis; FIPF, familial idiopathic pulmonary fibrosis; HP, hypersensitivity pneumonitis; IIP, idiopathic interstitial pneumonias; ILA, interstitial lung abnormalities; ILDs, interstitial lung diseases; iNSIP, idiopathic nonspecific interstitial pneumonia; IPF, pulmonary fibrosis; LAM, lymphangioleiomyomatosis; LCH, langerhans cell histiocytosis; MDD, multidisciplinary discussion; OP, organizing pneumonia; PAM, pulmonary alveolar microlithiasis; SR-ILD, smoking-related interstitial lung disease; UIP, usual interstitial pneumonia; Unclassifiable IIP, unclassifiable idiopathic interstitial pneumonia.
The histological diagnoses based on TBLC specimens are presented in Table 3. NSIP was the most frequent histological pattern, observed in 20.2% of cases, followed by HP in 16.8%, and UIP in 11.1%. SR-ILD was diagnosed in 10.1% of cases, while organizing pneumonia (OP) accounted for 8.2%. Occupational exposure-related ILD was identified in 5.8% of cases, alveolar proteinosis in 3.8%, and sarcoidosis in 3.4%. Less common diagnoses included bronchiolitis, pneumonia, acute interstitial pneumonia (AIP), and various other entities. In 12.0% of cases, histological examination yielded nonspecific findings insufficient for a definitive diagnosis.
The MDD diagnoses, formulated by integrating clinical, radiological, laboratory, and histological data, are presented in Table 3. HP became the most common diagnosis (18.3%), followed by iNSIP (15.4%) and SR-ILD (11.1%). IPF and occupational exposure-related ILD were diagnosed in 8.7% and 6.7% of cases, respectively. Other diagnoses included OP (4.8%), sarcoidosis (4.3%), alveolar proteinosis (4.3%), unclassifiable IIP (3.8%), DR-ILD (3.4%), and pneumonia (2.9%). Following MDD, only 2.4% of cases remained unidentified, representing a significant reduction from the 12.0% of cases that were histologically unidentified based on TBLC alone.
In our study, 77 cases showed discrepancies between the initial clinical diagnosis and the final MDD diagnosis. Among the 17 cases initially classified as Unclassified ILD, MDD reclassified them as iNSIP (4 cases), HP (3 cases), SR-ILD (2 cases), occupational exposure-related ILD (2 cases), sarcoidosis (1 case), alveolar proteinosis (1 case), FIPF (1 case), ACIF (1 case), with 2 cases remaining unidentified. For the 26 cases with an initial diagnosis of iNSIP, MDD revised them to SR-ILD (8 cases), IPF (6 cases), HP (5 cases), occupational exposure-related ILD (4 cases), sarcoidosis (2 cases), and DR-ILD (1 case). Among the 6 UIP cases, MDD reclassified them, including OP (3 cases), non-tuberculous mycobacterial infection (1 case), alveolar proteinosis (1 case), HP (1 case). Of the 12 pulmonary infection cases, MDD established specific ILD diagnoses in 11, most commonly OP (2 cases), with single cases of AFOP, HP, DR-ILD, occupational exposure-related ILD, pulmonary ossification, radiation pneumonia, bronchiolitis, UILD, and sarcoidosis, while 1 case remained unidentified. In addition, 6 other IIP cases were revised to SR-ILD (3 cases), iNSIP (2 cases), and lipoid pneumonia (1 case); 4 IPF cases were reclassified as iNSIP (2 cases), SR-ILD (1 case), and occupational exposure-related ILD (1 case); and other individual changes included pulmonary granuloma to sarcoidosis, sarcoidosis to HP, HP to EGPA, EGPA to HP, bronchiolitis to COP, and occupational exposure-related ILD to IPF.
Discrepancies between histological and final MDD diagnoses were also observed in our cohort, 19 cases with inconclusive histological diagnoses were finally diagnosed in MDD as iNSIP (3 cases), pulmonary alveolar proteinosis (1 case), HP (2 cases), and sarcoidosis (2 cases), IPF (1 case), DR-ILD (1 case), FIPF (1 case), unclassifiable IIP (1 case), pulmonary ossification (1 case), LAM (2 cases), pneumonia (3 cases), and UILD (1 case). In addition, 8 cases pathologically diagnosed as NSIP were reclassified after MDD as SR-ILD (3 cases), IPF (2 cases), HP (2 cases), and occupational exposure-related interstitial lung disease (1 case), reflecting the limitations of pathological diagnosis in distinguishing NSIP from other types of ILDs, and highlighting the diagnostic advantages of MDD when incorporating patient exposure history and clinical manifestations. Furthermore, 2 cases of pathologically diagnosed as HP were revised to iNSIP (1 case) and pneumonia (1 case), 3 cases of OP were revised to unclassifiable IIP, 1 case of UIP was also revised to unclassifiable IIP, and 1 case of alveolitis was revised to non-tuberculous mycobacterial infection after MDD, suggesting potential misjudgments in pathological diagnosis, while MDD was able to more accurately identify the nature of the disease.
Diagnostic yield among initial clinical, histological, and MDD diagnoses
The diagnostic yields of initial clinical, histological, and MDD diagnoses are presented in Table 4. The initial clinical diagnoses were established in all 208 patients as the prerequisite for TBLC. The histological diagnoses were made in 88.9% (185/208 cases), with 11.1% (23 cases) remaining undetermined. Following MDD, the diagnoses were improved to 97.6% (203/208 cases), leaving only five cases (2.4%) unidentified.
Diagnostic yield between different diagnostic pathways.

MDD, multidisciplinary discussion.
Analysis of diagnostic yield and complications based on procedural variables
Table 5 presents the diagnostic yield and complications, stratified by biopsy location, sample number, and maximum sample diameter.
Diagnostic yield, complications, and 30-day mortality stratified by biopsy location, sample number, the maximum sample diameter.
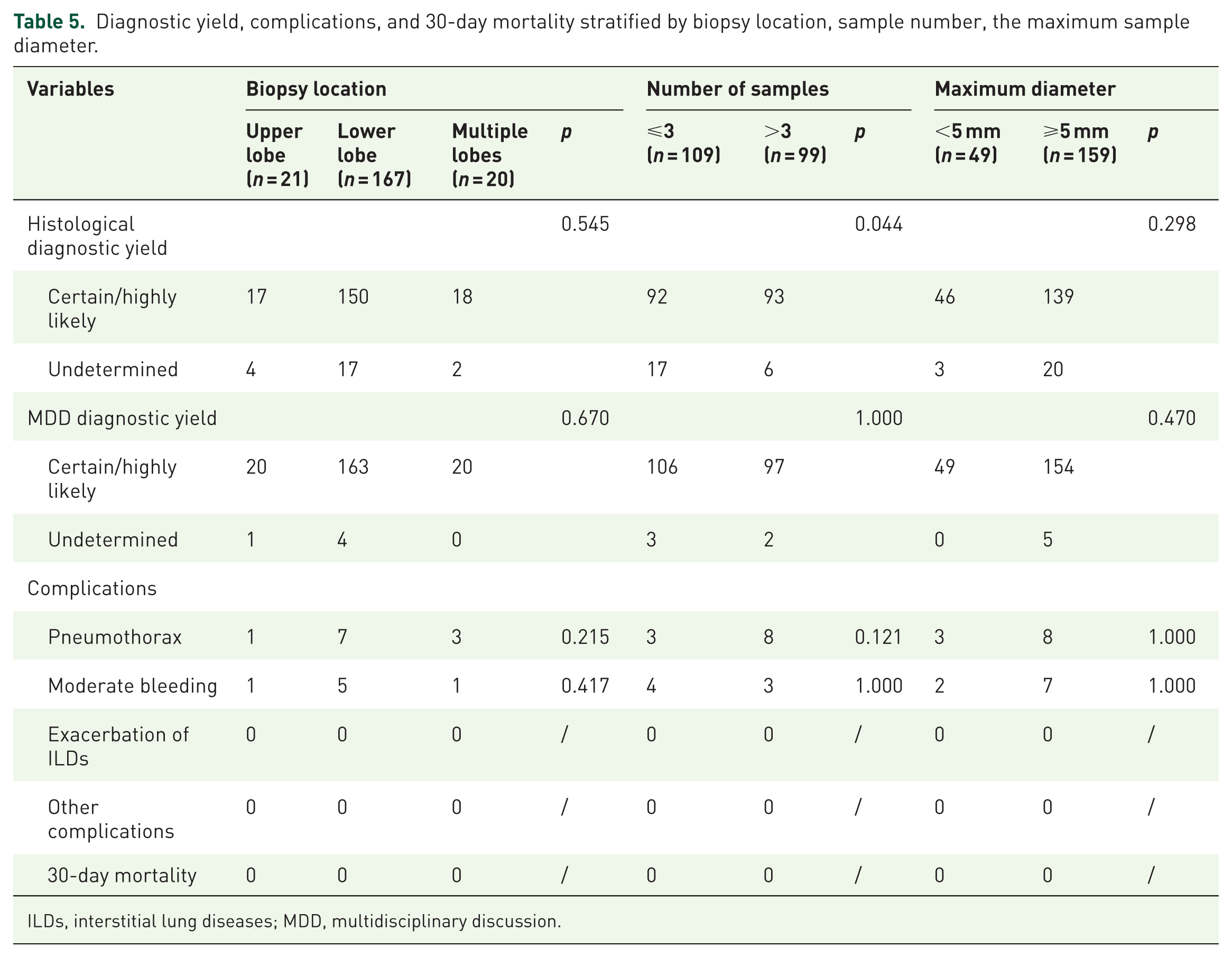
ILDs, interstitial lung diseases; MDD, multidisciplinary discussion.
For biopsy location, no significant differences were observed in histological diagnostic yield between upper lobe (17/21, 81.0%), lower lobe (150/167, 89.8%), and multiple lobe (18/20, 90.0%) biopsies. MDD diagnostic certainty was achieved in 95.2% of upper lobe cases (20/21), 97.6% of lower lobe cases (163/167), and 100% of multiple lobe cases (20/20), with no significant difference.
Regarding sample quantity, obtaining more than three samples significantly improved histological diagnostic yield (93/99, 93.9% for >3 samples vs 92/109, 84.4% for ⩽3 samples; p = 0.044) but did not affect MDD diagnostic yield (97/99, 98.0% for >3 samples vs 106/109, 97.2% for ⩽3 samples; p = 1.000).
Sample size analysis revealed that specimens ⩾5 mm in diameter did not demonstrate significantly higher histological diagnostic yield (139/159, 87.4%) compared to specimens <5 mm (46/49, 93.9%; p = 0.298). MDD diagnostic yield was similarly high regardless of specimen size (154/159, 96.9% for ⩾5 mm vs 49/49, 100% for <5 mm; p = 0.470).
Complication rates were low across all procedural variables. Pneumothorax occurred in 4.8% of upper lobe biopsies (1/21), 4.2% of lower lobe biopsies (7/167), and 15.0% of multiple lobe biopsies (3/20) (p = 0.215). Moderate bleeding was observed in 4.8% of upper lobe biopsies (1/21), 3.0% of lower lobe biopsies (5/167), and 5.0% of multiple lobe biopsies (1/20) (p = 0.417).
Association of diagnostic yield with patient characteristics
Table 6 presents the diagnostic yield of the histological diagnosis and MDD diagnosis, across different demographic characteristics, symptoms, and occupational exposure. Male patients demonstrated significantly higher histological diagnostic yield (125/135, 92.6%) compared to females (60/73, 82.2%; p = 0.022).
Diagnostic yield across demographic characteristics, symptoms, and occupational exposure.

MDD, multidisciplinary discussion.
Neither histological nor MDD diagnostic yield demonstrated any significant association with patient age, smoking status, or body mass index (all p > 0.05). Symptoms, including cough, dyspnea, fever, multiple symptoms, and asymptomatic status, had no significant impact on histological or MDD diagnostic yield. Occupational exposure was reported in 41 patients (19.7%). No significant differences in histological yield or MDD yield were observed between the exposed and non-exposed groups.
Association of complications, demographic characteristics, and symptoms
Table 7 presents the occurrence of pneumothorax and moderate bleeding across demographic characteristics and symptoms. Patients who experienced pneumothorax had lower BMI (p = 0.037) compared to those without pneumothorax. Contrary to our expectations, age, male gender, and smoking history do not increase the incidence of pneumothorax and moderate bleeding. For moderate bleeding, patient age, BMI, sex, and smoking history were not significantly associated with bleeding risk. No significant relationships were identified between specific symptoms (cough, expectoration, dyspnea, fever, multi-symptoms, and asymptomatic) and the occurrence of either pneumothorax or moderate bleeding.
Analysis of pneumothorax and moderate bleeding across demographic characteristics and symptoms.

Discussion
Our study assessed the role, performance, and safety of TBLC in the MDD diagnosis of ILDs at two specialized pulmonary hospitals in China. In this series of 208 patients with undiagnosed ILDs who required lung biopsy, a histological diagnosis was obtained in 185 cases (88.9%). This diagnostic yield appears higher than the previously reported rates, which range from 79% to 80%.30,32,33 When integrated into MDD, 203 cases (97.6%) were sufficient to establish a diagnosis, which is also higher than the diagnostic yield of 80%–85.2% described in the literature for TBLC-MDD.34–36 Our findings confirm that MDD is essential to enhance the diagnostic yield of TBLC in patients with unclassified ILDs, consistent with previous studies emphasizing the critical role of MDD.36–38 In our study, the incidences of pneumothorax and moderate bleeding were 5.3% and 3.4%, respectively, with no severe complications requiring ICU admission or surgery.
As a less invasive alternative to SLB, TBLC demonstrates comparable diagnostic impact within the MDD framework. Compared with SLB, TBLC demonstrated comparable diagnostic impact when integrated into the MDD process. 39 The COLDICE study showed that histopathological agreement between TBLC and SLB was 70.8%, and diagnostic agreement at MDD was 76.9%. 25 Both TBLC and SLB similarly improve diagnostic confidence and inter-observer agreement when integrated into MDD, with TBLC increasing diagnostic confidence in 78% of suspected IPF cases—comparable to SLB. 40 Pathologists’ independent analysis showed that TBLC was considered informative in the vast majority of cases. Although SLB-MDD was not included in this study, previous research has demonstrated that TBLC-MDD diagnoses made with high confidence are highly reliable, demonstrating excellent concordance with SLB-MDD diagnoses.24,25
Building on the well-established role of MDD as the diagnostic gold standard for ILDs,41–44 our study further highlights the contribution of TBLC in bridging the gap between preliminary clinical assessment and final MDD consensus (Supplemental Table 4). The diagnostic concordance between the initial clinical and MDD diagnoses was 63.0% (131/208 cases), indicating low inter-method agreement (κ = 0.386, 95% CI: 0.373–0.399). After excluding cases that were undiagnosed at the clinical stage, this concordance was 64.5% (131/203, κ = 0.389, 95% CI: 0.376–0.402). The overall diagnostic concordance between the histological and MDD diagnoses was 81.3% (169/208 cases), with moderate inter-method agreement (κ = 0.448, 95% CI 0.440–0.456). After excluding histologically undetermined cases, the diagnostic concordance increased to 91.3% (169/185, κ = 0.477, 95% CI 0.471–0.483).
We further examined whether procedural or demographic factors influenced this concordance between histology and MDD (Supplemental Tables 5 and 6). The diagnostic concordance was numerically lower for upper lobe biopsies (16/21, 76.2%) compared to lower lobe biopsies (137/167, 82.0%) and multiple lobe biopsies (16/20, 80.0%), though this difference did not reach statistical significance. The diagnostic concordance was significantly higher in the > 3 samples group (87/99, 87.9% vs 82/109, 75.2%; p = 0.021). Diagnostic concordance was comparable between larger (128/159, 80.5%) and smaller specimens (41/49, 83.7%). Diagnostic concordance between histology and MDD was higher in males (116/135, 86.0%) than in females (53/73, 72.6%; p = 0.019). The diagnostic concordance was also unaffected by age, smoking, BMI, or specific symptoms (all p > 0.05). No significant differences in diagnostic concordance were observed between the exposed and non-exposed groups.
The increased size and tissue integrity from TBLC improve the detection of key pathological features.30,45 In our series, IPF accounted for a significant proportion (8.7%) of cases diagnosed after MDD, consistent with previous studies demonstrating increased IPF diagnoses through MDD,7,46 and the diagnosis was most frequently revised from iNSIP and HP to IPF.40,47,48 While SLB is traditionally recommended when HRCT is nondiagnostic for IPF,22,49 TBLC achieves a 78% diagnostic yield and significantly enhances diagnostic confidence for IPF within MDD.40,50
Regarding procedural factors, obtaining > 3 samples significantly improved histological diagnostic yield and diagnostic concordance, but did not affect MDD yield. While a greater number of samples (⩾3) significantly improved the histological diagnostic yield and diagnostic concordance, it had no effect on the MDD diagnostic yield. After adjusting for variables including sex, BMI, biopsy characteristics, and PFT parameters, more than three biopsy samples remained an independent predictor for both higher histological diagnostic yield (OR 4.84, 95% CI 1.44–16.30, p = 0.011) and greater diagnostic concordance (OR 2.80, 95% CI 1.11–7.07, p = 0.029) (Supplemental Table 3). Notably, specimen diameter ⩾5 mm and biopsy location were not associated with diagnostic outcomes, supporting current recommendations for multi-sample acquisition. 51 Notably, the association between male sex and higher diagnostic yield observed in univariate analysis was attenuated after multivariable adjustment. These analyses provide a more robust, confounder-adjusted perspective on factors influencing TBLC diagnostic performance within the MDD framework.
Approximately 11.1% of cryobiopsies were non-diagnostic due to insufficient alveolar tissue, normal lung, or nonspecific findings. While the optimal specimen size for pattern recognition remains undefined, some experts recommend ⩾5 mm in diameter for adequate assessment. 52 However, in our study, samples with a diameter ⩾5 mm did not show a significant increase in diagnostic yield, whether in histopathology or MDD. No significant association was found between biopsy site and either diagnostic efficacy or complications in our cases, consistent with the observations of Jose et al. 49
In our study, patients with certain MDD diagnoses exhibited significantly lower FVC (% predicted) values than those with undetermined diagnoses, while higher DLCO (% predicted) was observed in cases with discordant histopathology-MDD diagnoses compared to concordant ones (Supplemental Table 1). Our findings suggest that while most conventional PFT parameters do not influence diagnostic performance, DLCO may warrant further investigation regarding its relationship with diagnostic consistency in ILD evaluation. However, impaired lung function is associated with an elevated risk of under-diagnostic yield (both pathological and multidisciplinary) in TBLC procedures, which may be attributed to more complex and difficult clinical, pathological, and/or radiological presentations and more sampling restrictions. 51
In our study, 11 patients (5.3%) developed pneumothorax, 7 of which were treated with chest tube drainage (median air leak time: 1 day). The reported incidence of pneumothorax following TBLC varies across studies, with some reporting no cases while others document rates as high as 33%.30,32,40,50,53–55 The overall incidence of pneumothorax after TBLC is approximately 9%. 10 Studies reporting high pneumothorax rates share notable methodological characteristics: the procedures were performed under spontaneous breathing, and the cryoprobe was typically cooled for approximately 5–6 s.40,56 Our sampling procedures were performed under general anesthesia, which may explain the lower pneumothorax rate compared to studies utilizing spontaneous breathing. In addition, our cryoprobe freezing time was shorter. Our technique aligns with Bango-Álvarez et al., who reported a 4.7% pneumothorax rate by advancing the cryoprobe until resistance, then retracting 1–2 cm. 54 Based on our institutional experience, we recommend general anesthesia, shorter cryoprobe freezing time, and maintaining a safe distance from the pleura to minimize pneumothorax risk. We identified lower BMI and FEV1/FVC ratio as potential risk factors for pneumothorax (Supplemental Table 2), which have not been previously described, and require further confirmation in future studies.
The incidence of moderate bleeding with TBLC was 3.4% (7/208 cases), which is much lower than the reported rates in other series.26,27,50,55,57 Among these, two cases involved approximately 100 mL blood loss, one case about 50 mL, and the remaining cases about 20 mL, all successfully resolved with local endoscopic measures without requiring additional interventions. This favorable safety profile was achieved through multiple precautionary measures: (1) strict discontinuation of antiplatelet/anticoagulant medications for at least 7 days prior to procedure; (2) preferential biopsy sampling from dependent lung areas to facilitate natural tamponade; and (3) use of endobronchial balloon after sampling completion, which has been associated with a lower rate of significant bleeding. 58 Similarly, we analyzed potential factors associated with bleeding and found that neither sampling strategies nor clinical parameters had any impact on bleeding outcomes. Other studies also suggest that bleeding risk is independent of baseline functional status, probe size, number of samples, or multiple site sampling. 51
Our study has several limitations. First, the predominance of lower-lobe biopsies may introduce sampling bias, potentially underrepresenting upper-lobe-predominant diseases. Second, patients with preserved lung function were preferentially selected, limiting generalizability to critically ill populations. Third, the absence of direct SLB comparison is mitigated by high-quality evidence demonstrating comparable diagnostic utility of TBLC within MDD.25,40 Fourth, the dual-center design at specialized institutions may limit generalizability to general hospitals. Fifth, diagnostic confidence was not systematically assessed. Future prospective studies with standardized confidence scales would further characterize TBLC-guided MDD precision.
Conclusion
In conclusion, this dual-center study supports that TBLC is a safe and effective procedure that provides high-quality histological samples for MDD in undiagnosed ILDs. When integrated into a structured MDD, TBLC demonstrates a high diagnostic yield and can significantly reduce the need for more invasive SLB.
Supplemental Material
sj-doc-1-tar-10.1177_17534666261438213 – Supplemental material for The role of transbronchial lung cryobiopsy in multidisciplinary discussion for interstitial lung diseases: a dual-center retrospective study
Supplemental material, sj-doc-1-tar-10.1177_17534666261438213 for The role of transbronchial lung cryobiopsy in multidisciplinary discussion for interstitial lung diseases: a dual-center retrospective study by Qi Liu, Ting Wang, Qing Chang, Ning Xu, Ruolin Mao, Mengnan Li, Ding Sun, Yuqing Chen, Hai Zhang, Hongying Zhang and Feng Li in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-docx-2-tar-10.1177_17534666261438213 – Supplemental material for The role of transbronchial lung cryobiopsy in multidisciplinary discussion for interstitial lung diseases: a dual-center retrospective study
Supplemental material, sj-docx-2-tar-10.1177_17534666261438213 for The role of transbronchial lung cryobiopsy in multidisciplinary discussion for interstitial lung diseases: a dual-center retrospective study by Qi Liu, Ting Wang, Qing Chang, Ning Xu, Ruolin Mao, Mengnan Li, Ding Sun, Yuqing Chen, Hai Zhang, Hongying Zhang and Feng Li in Therapeutic Advances in Respiratory Disease
Footnotes
Acknowledgements
The authors thank Shanghai Municipal Hospital Respiratory and Critical Care Medicine Specialist Alliance for support.
Declarations
Supplemental material
Supplemental material for this article is available online.
Patient and public involvement
Patients or the public were not involved in the design, conduct, reporting, or dissemination plans of our research.
