Abstract
Background:
Central bronchopleural fistula (cBPF) is one of the most severe complications after pulmonary resection, with high morbidity and mortality. Bronchoscopic occlusion has emerged as a minimally invasive treatment for cBPF, but systematic evidence to guide optimal material selection and to predict treatment efficacy is lacking.
Objectives:
To evaluate how anatomical characteristics of cBPF influence the choice of bronchoscopic occlusion materials, and to develop a risk model for individualized prediction of long-term occlusion outcomes.
Design:
Single-center retrospective observational study.
Methods:
We analyzed clinical data of 56 patients with postoperative cBPF. Fistula anatomical features and their matching with various occlusion materials were evaluated. Occlusion strategies were categorized as pure pharmacologic therapy, stent-only, or combined approaches, according to the actual combinations of materials used during bronchoscopic occlusion. An outcome prediction model was constructed using the least absolute shrinkage and selection operator (LASSO) for feature selection and a Bayesian cumulative logit mixed-effects model for longitudinal outcomes at multiple time points, and key risk factors for long-term outcomes were identified during follow-up.
Results:
Fistula size and type largely determined the choice of occlusion strategy. Occlusion outcomes evolved dynamically over time, with the 3-month postocclusion mark representing a key inflection point in outcome stability. The model showed that the 1–3-month period after bronchoscopic occlusion was a high-risk window for unfavorable occlusion grades. Patients with occult fistulas and those treated with pure pharmacologic therapy were more likely to maintain or regain better occlusion status. A visual risk-scoring nomogram was constructed based on this model to support risk stratification and individualized outcome prediction.
Conclusion:
This study provides new evidence to guide material selection for bronchoscopic cBPF occlusion and to predict therapeutic outcomes and long-term prognosis. The findings support an individualized treatment strategy and dynamic management approach for patients with bronchial stump fistula.
Plain language summary
After lung surgery, a small number of patients develop an unwanted opening between the main airway and the space around the lung. This problem—called an airway leak or bronchopleural fistula—lets air and fluid move where they should not, causing cough, breathlessness and infection. Traditionally, surgeons had to reopen the chest to fix the leak. Today, doctors can often seal it from inside the airway with a flexible camera (a bronchoscope) and special sealing materials such as tissue glue or tiny stents. We reviewed the records of 56 patients who received this bronchoscopic treatment in one hospital. By comparing the size of each leak, the material used to seal it and the patient’s infection status, we discovered three practical lessons: Size matters. Small leaks (under 5 mm) usually healed when only a chemical “scar-forming” agent was injected. Larger leaks needed a stent or a combination of materials to stay closed. The first three months are critical. Most treatment failures happened within 1–3 months after the procedure, especially if infection in the chest cavity was still present. A simple risk score can guide follow-up. We created an easy bedside tool that combines leak size, infection markers and treatment type to predict each patient’s chance of long-term healing. These findings can help doctors choose the right sealing method, schedule closer check-ups during the high-risk early period and act sooner if healing is at risk.
Keywords
Introduction
Bronchopleural fistula (BPF) is one of the most serious complications following lung resection. It is defined as an abnormal passage between the bronchial stump and the pleural cavity after pulmonary resection, which allows air and purulent fluid to shuttle between the pleural space and the remaining lung. BPF is commonly classified into central and peripheral types. 1 In this study, central bronchopleural fistula (cBPF) was defined as a postoperative fistula caused by dehiscence or incomplete healing of the bronchial stump after anatomic lung resection, forming a direct communication between the central bronchus and the pleural cavity that can be reached and directly visualized with flexible bronchoscopy. Peripheral BPF referred to fistulas arising after nonanatomic sublobar procedures or treatment of pleural-based lesions, in which the abnormal communication is located between distal bronchioles or alveoli and the pleural space, essentially within the spectrum of alveolar–pleural fistula and usually not directly visible bronchoscopically. This study primarily focused on cBPF, the subtype most amenable to bronchoscopic occlusion. 2 Clinically, cBPF leads to severe cough and dyspnea and can rapidly progress to life-threatening conditions such as pulmonary infection, empyema, or even tension pneumothorax. 2 Although BPF develops in only a small proportion of patients after pulmonary resection, large contemporary series and registry data suggest an overall incidence of roughly 0.4%–2% after anatomical lung resection and up to about 7%–8% after pneumonectomy, with reported mortality rates ranging from approximately 15% to over 50% despite modern perioperative care. For example, Fuso et al. reported a 2.2% overall incidence of BPF in 835 anatomic resections, and more recent large cohorts from Japan have found BPF incidences of 0.4%–0.7% but still 90-day mortality around 15% and 5-year survival below 30% among patients who develop BPF.3–5 Advances in surgical techniques and perioperative management have somewhat improved overall outcomes, but BPF remains a formidable clinical challenge. 7
Traditionally, BPF has been managed primarily with surgical re-intervention, such as re-thoracotomy to repair or reconstruct the bronchial stump. However, surgical repair is highly invasive and risky; even if the fistula is successfully closed, recurrence is possible, and patients are often in poor condition with ongoing infection or respiratory failure, contributing to high postoperative mortality. 6 Therefore, there is urgent clinical interest in effective bronchoscopic occlusion techniques that achieve closure of the fistula with less trauma. In recent years, bronchoscopic interventional techniques have transformed the management of BPF. A variety of bronchoscopic occlusion methods have been applied, including endobronchial injection of sclerosing agents (e.g., polidocanol) or other chemical irritants to promote local scar formation, placement of various plug devices (such as absorbable collagen sponges), insertion of endobronchial one-way valves to allow unidirectional drainage and reduce air leak, deployment of covered airway stents to span the fistula, and even placement of cardiac atrial septal defect occluders in the bronchus to seal the fistula. 7 Multiple recent case series and reviews have shown that customized covered airway stents, Amplatzer-type occluder devices and other bronchoscopic materials can achieve high technical closure rates (often > 70%) and improve short-term outcomes compared with conservative therapy, although most data come from single-center retrospective studies.8–11 Despite this expanding arsenal of bronchoscopic occlusion techniques, there is still no unified evidence-based guidance on selecting the most appropriate occlusion material or device based on fistula characteristics. Moreover, tools for outcome evaluation and prognosis prediction are lacking—it is challenging for clinicians to identify, before treatment, which patients are at high risk of occlusion failure or recurrence. Long-term management is especially problematic, as some patients who initially achieve successful closure later experience re-opening of the fistula due to underlying infection or chronic pleural disease.
Materials and methods
Study design and patients
This study was a single-center retrospective observational study. We included patients diagnosed with cBPF after lung resection who underwent bronchoscopic occlusion therapy at the Respiratory Disease Center of Dongzhimen Hospital, Beijing, between September 2020 and December 2024. All cases were transferred from the thoracic surgery departments of other hospitals. Data sources included the inpatient electronic medical record system and a dedicated follow-up database. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement (Supplemental Material). 12
Inclusion criteria:
Patients were eligible if they met all of the following conditions: (1) diagnosis of cBPF confirmed by imaging and bronchoscopy; (2) had undergone an anatomic lung resection (segmentectomy, lobectomy, combined lobar/segmental resection, or pneumonectomy); (3) postoperatively confirmed as having a secondary cBPF by chest computed tomography (CT) combined with bronchoscopy; (4) underwent at least one bronchoscopic occlusion treatment for the fistula; (5) completed at least one follow-up bronchoscopy after occlusion, with complete initial data records.
Exclusion criteria:
Patients were excluded if any of the following applied: (1) pleural fistula due to nonsurgical causes; (2) postoperative follow-up lacked bronchoscopic examination (diagnosis based only on imaging without bronchoscopic confirmation); (3) malignancy was present at the fistula site; (4) active immunosuppression (e.g., ongoing chemotherapy, organ transplant, long-term corticosteroid therapy).
We defined the main follow-up time points as 1, 3, 6, and 12 months after the initial bronchoscopic occlusion. All patients who had at least one postocclusion bronchoscopic evaluation were eligible for the longitudinal analysis. For each patient, all available visits corresponding to the predefined time windows were included; visits that were not performed were treated as missing and were not imputed. Incomplete follow-up was mainly due to patients not returning to the hospital for scheduled bronchoscopic examinations, rather than death. The process of patient screening, exclusion, and follow-up completion is summarized in Figure 1.
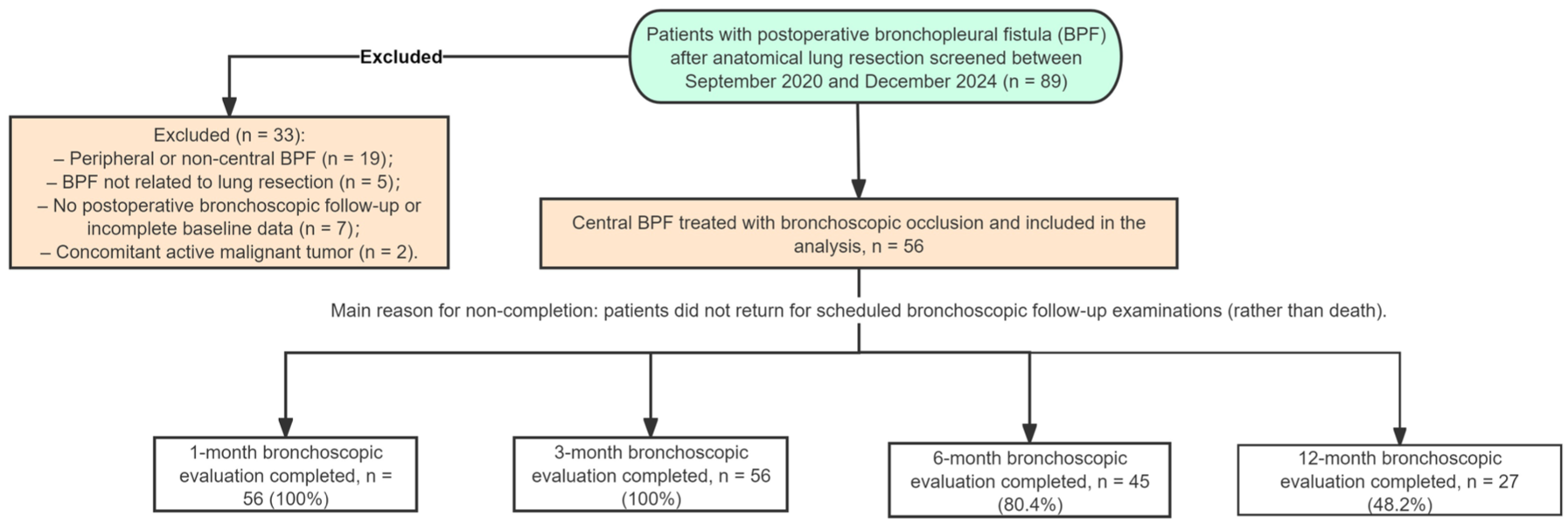
Flowchart.
Variables and data collection
We collected the following categories of variables for each patient: Demographic characteristics (age and sex). Fistula characteristics: assessed bronchoscopically during the occlusion procedure, including fistula classification, shape, and size. Fistulas were classified according to bronchoscopic visibility and size into three types, all representing central postoperative cBPF with openings located within the reach of routine flexible bronchoscopy at the bronchial stump or within bronchial segments accessible to the bronchoscope:
(1) Large visible fistula: the fistulous opening was clearly identified at the initial bronchoscopic examination, with a maximum diameter ⩾5 mm;
(2) Small visible fistula: the opening was directly identified at the initial examination, with a maximum diameter <5 mm;
(3) Occult fistula: the suspected fistulous region was bronchoscopically reachable, but no discrete opening contour was seen on initial inspection. The presence of a fistula was confirmed only after adjunctive maneuvers, such as instilling diluted methylene blue or saline via the chest tube or airway and observing intermittently emerging dyed fluid or bubbles in the same region, so that the exact morphology and size of the opening could not be measured. Fistula shape was subjectively categorized as round, ovoid, or elongated based on bronchoscopic appearance, and fistula size was recorded as the largest diameter (in millimeters).
Infection and inflammation indicators: results of bronchoalveolar lavage fluid (BALF) microbiology (categorized as bacterial infection, fungal infection, or mixed infection—with mixed infection defined as simultaneous detection of ⩾2 different classes of pathogens such as bacteria with fungi or viruses), C-reactive protein (CRP), white blood cell count (WBC), and serum albumin. BALF was obtained during the bronchoscopic treatment procedure, while the other laboratory values were collected before each bronchoscopic occlusion session.
Functional status indicators: Karnofsky Performance Status (KPS) score, body mass index (BMI), and subjective dyspnea grade (on a 0–4 scale, with 0 = no dyspnea and 4 = very severe dyspnea).
Occlusion treatment variables included details of materials used during bronchoscopic occlusion. We recorded the type and combination of occlusion materials applied, including polidocanol-type sclerosing agents, biologic sealants (e.g., fibrin glue), stent devices, silicone prostheses, and atrial septal occluders (multiple types could be combined). According to the actual combination of materials, the treatment approach was categorized into three occlusion strategies: (a) pure pharmacologic therapy—use of sclerosing agents and/or biologic sealants without any mechanical device; (b) stent only—use of stent-based devices alone without other materials; and (c) combined—a composite strategy using two or more types of materials (e.g., silicone prosthesis occlusion combined with fibrin glue). Representative bronchoscopic findings and occlusion techniques are illustrated in Figure 2. Other clinical indicators: presence of fever, nature of sputum (purulent sputum or serous sputum resembling pleural fluid), daily chest tube drainage volume (ml/24 h), and pleural space status (categorized as none, hydropneumothorax, empyema, or both hydropneumothorax and empyema). All other variables were recorded at each follow-up visit except for demographic and fixed fistula characteristics. Two researchers verified all data independently and entered it into a unified database.

Representative bronchoscopic findings and occlusion techniques in central bronchopleural fistula (cBPF). Representative bronchoscopic views of central bronchopleural fistula (cBPF) and occlusion techniques. (a) Fistulous opening. (b) Postoperative residual cavity. (c) Right upper lobe fistula after occlusion. (d) Fistulous opening. (e) Submucosal pharmacologic injection. (f) After placement of a silicone prosthesis. (g) Fistulous opening. (h) Bronchial stump fistulous opening. (i) Fistula after stent occlusion.
Outcome definition
The primary outcome was the postocclusion efficacy of fistula occlusion, evaluated at follow-up based on clinical symptoms, imaging findings, and bronchoscopic examination (including a methylene blue test, if performed). Outcomes were classified into four grades: complete response (CR): the fistula is fully healed (closed), the intrathoracic residual cavity is partially absorbed or has disappeared, and clinical symptoms are completely resolved, with the improvement sustained for ⩾1 month; clinical complete response (CCR): The fistula is not fully healed but is entirely covered by the occlusion material (no residual air leak visible bronchoscopically), the residual cavity is partially absorbed or disappeared, and clinical symptoms are completely resolved; partial response (PR): The fistula is only partially occluded (incomplete closure), an intrathoracic cavity persists, and symptoms have improved but not fully resolved; no response (NR): Occlusion is ineffective—the fistula remains partially open or the intrathoracic cavity persists or has enlarged, and clinical symptoms show no significant improvement or have worsened. 13
Treatment outcome at each follow-up was independently assessed by two experienced pulmonologists (Jiaping Liu and Lixin Cui) using the above criteria and reviewing imaging, bronchoscopic findings, and methylene blue leak test results. In disagreement, a third expert was consulted to adjudicate the outcome classification.
Statistical analysis
Continuous variables were compared between groups using the t-test or Mann–Whitney U test, depending on distribution normality. Categorical variables were compared using the chi-square or Fisher’s exact test, as appropriate. Univariate analyses explored the association of each variable with the outcome category. For multivariable analysis, LASSO regression selected candidate predictors, with cross-validation identifying the optimal penalty parameter λ. Variables with nonzero coefficients at the optimal λ were retained as candidate predictors and included as fixed effects in the model. A Bayesian cumulative logit mixed-effects model was constructed with occlusion outcome grade (from best to worst: CR → CCR → PR → NR) as the dependent variable. Fixed effects included follow-up time (1, 3, 6, 12 months) and predictors selected by LASSO (e.g., lab values, pleural complications, occlusion strategy), with a random intercept to account for patient variability. Bayesian cumulative logit mixed-effects models were fitted in Statistical analyses were performed using R software (version 4.3.1; R Foundation for Statistical Computing, Vienna, Austria) using the brms package, which implements a No-U-Turn Sampler (4 chains, 4000 iterations, including 2000 warm-up, adapt_delta = 0.90). Convergence was assessed using the potential scale reduction factor (Rˆ ≈ 1.00) and effective sample sizes (>1000). Model adequacy was assessed using posterior predictive checks and graphical diagnostics (posterior distributions and trace plots), and Bayesian estimates are reported as posterior means with 95% highest-density intervals. All data management, descriptive analyses, LASSO regression, and visualizations were performed in R (including glmnet and ggplot2).
Results
Baseline characteristics
A total of 56 patients with cBPF were included in the study. Baseline characteristics are summarized in Table 1. The mean age was 55, and 64.3% were male (36/56). Regarding fistula classification, the large visible fistula type was most common (46.5% of cases), followed by small visible fistula (41.4%) and occult fistula (12.1%). In terms of shape, a round fistula opening was most frequent (71.4% of cases), with ovoid and elongated shapes in 10.7% and 5.4%, respectively; in 12.5% of cases, shape was not determined. Fistula size distribution: 62.5% of patients (35/56) had a fistula diameter <5 mm, while 37.5% (21/56) had a diameter ⩾5 mm. Follow-up completion was 56/56 (100%) at 1 month, 56/56 (100%) at 3 months, 45/56 (80.4%) at 6 months, and 27/56 (48.2%) at 12 months.
Baseline characteristics of the study population.

Percentages for occlusion materials exceed 100% because multiple material types could be used in combination for a single patient.
Concerning the use of individual occlusion materials (counting each material separately regardless of combinations), pharmacologic agents—including polidocanol-type sclerosing agents and biologic sealants such as fibrin glue—were most frequently employed and were used in 35 treatment episodes, followed by stent devices (23 episodes), silicone prostheses (7 episodes), and atrial septal occluders (1 episode). Because some patients received more than one type of occlusion material, the sum of these counts exceeds the total number of patients. According to the occlusion strategy grouping at the patient level, 25 patients (44.6%) received pure pharmacologic therapy (sclerosing agents and/or biologic sealants without mechanical devices), 19 (33.9%) were treated with a stent-only approach, and 12 (21.4%) with a combined-material strategy involving two or more types of occlusion materials.
Fistula features and choice of occlusion material
Table 2 summarizes the use of occlusion materials by fistula type, shape, and size. Pharmacologic agents were most frequently used across all fistula types (p = 0.219). Stents were more common in large visible fistulas, while silicone prostheses were mainly applied in small visible and occult types (p = 0.006 and p = 0.020). Atrial septal occluder use was rare and not type-dependent (p = 0.816). Only silicone prostheses showed a significant difference by shape, being more used in elongated fistulas (p = 0.012); other materials showed no association. Use of pharmacologic agents was more frequent in fistulas <5 mm, whereas stents were more often used in those ⩾5 mm (p < 0.001 and p = 0.005); other materials were not size-dependent. To further assess the impact of fistula characteristics on treatment choice, occlusion strategies were ordered from simple to complex (pure pharmacologic therapy, stent-only, and combined strategies) and analyzed using logistic regression (Figure 3). A fistula diameter ⩾5 mm independently predicted selection of a more complex (stent-based or combined) strategy over a simple one (p = 0.002), whereas smaller fistulas were more likely to receive a simple occlusion approach (p = 0.009). No significant interaction between fistula shape and size was observed, indicating that strategy selection was driven primarily by fistula size rather than shape.
Chi-square analysis of fistula characteristics versus material usage.


Associations of fistula size, type, and shape with occlusion strategy selection.
Temporal changes in occlusion outcomes
Occlusion efficacy showed clear dynamic changes over time (Figure 4). The average times to achieve each outcome were: 1.41 months for PR, 1.74 months for CCR, and 5.59 months for CR. Early after occlusion, most patients showed good efficacy. At 1 month, CCR remained dominant, but the proportion of PR increased, indicating early efficacy fluctuations. By 3 months, CCR slightly decreased, and NR outcomes appeared in a few patients, indicating that initial improvements were not sustained in some cases. The PR proportion decreased, with CR increasing, suggesting overall stabilization and improvement. At 6 months, the combined CR/CCR rate increased, while NR slightly rose, reflecting late failure in some patients. By 12 months, most patients were in CR or CCR, with PR and NR nearly disappearing, indicating long-term stability in survivors.
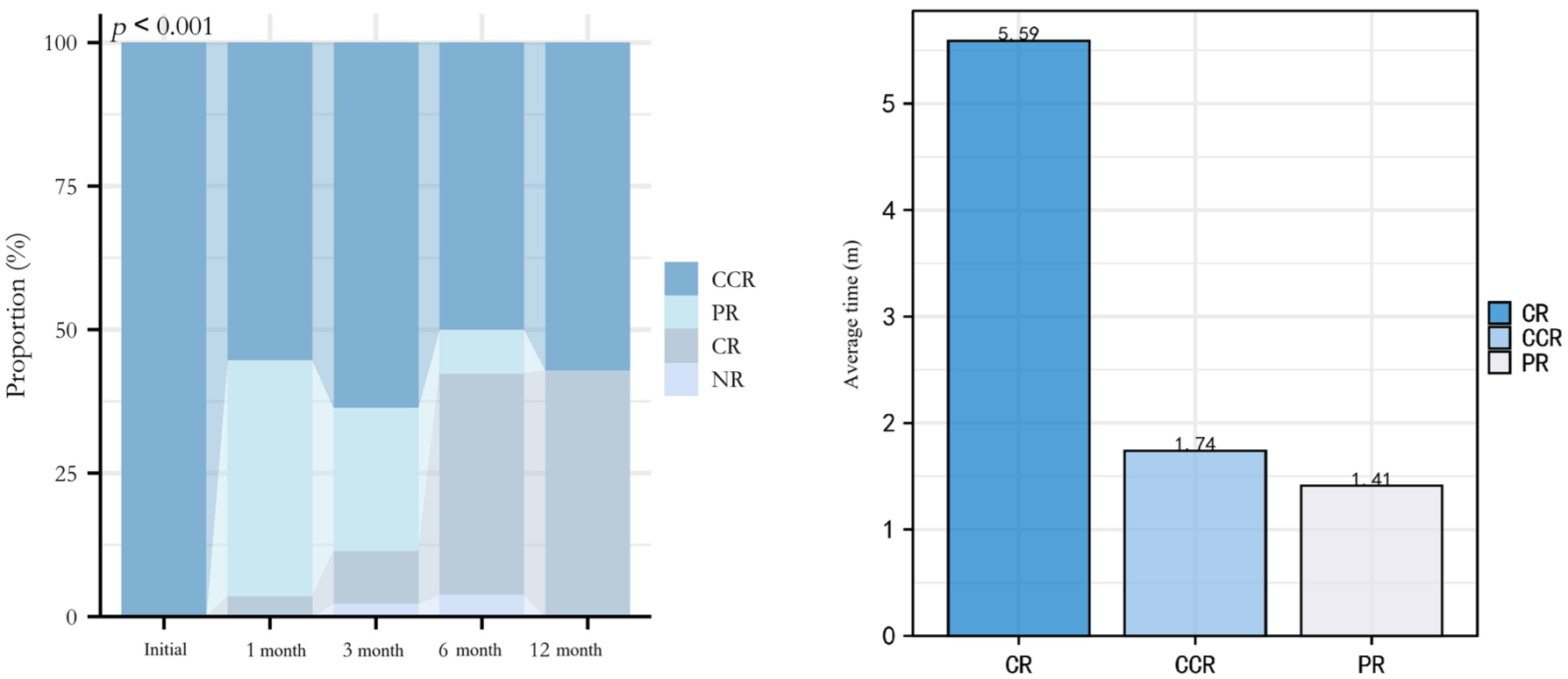
Longitudinal changes in bronchoscopic occlusion outcomes.
Factors influencing occlusion outcome
Next, we analyzed potential factors associated with the graded occlusion outcomes (CR/CCR/PR/NR) across all follow-up observations. Table 3 presents the results of univariate association tests for both categorical and continuous variables. Among categorical factors, pleural space complications were strongly associated with poorer occlusion outcomes. Patients with hydropneumothorax or empyema (or both) had worse outcomes compared to those without pleural complications (p < 0.001). Specifically, patients without pleural complications had higher rates of CR/CCR, while those with pleural complications were more likely to have CCR/PR outcomes. The occlusion strategy was borderline associated with clinical outcomes (p = 0.058), without a consistent directional trend across treatment groups. Factors such as purulent sputum, fever, albumin, and infection etiology did not significantly correlate with outcomes (p > 0.1). Dyspnea score had no significant association with outcomes (p = 0.997). For continuous variables, CRP and drainage volume increased with worse outcomes, with CRP significantly higher in NR patients (p < 0.001). KPS declined with worse outcomes (p = 0.152), but this was not statistically significant. WBC and BMI showed no significant differences (p > 0.2). Univariate analysis suggested that pleural space complications and high inflammatory burden (CRP) were key to poor outcomes. Patients with NR often had elevated CRP, WBC, and higher pleural drainage, while those with CR had controlled inflammation and minimal drainage.
Univariate analysis of different variables in relation to clinical evaluation of occlusion outcome.

Normal distribution: BMI, KPS (CR group) expressed as mean ± standard deviation; nonnormal distribution; WBC, CRP, and priming volume expressed as median (interquartile spacing).
CCR, clinical complete response; CR, complete response; CRP, C-reactive protein; KPS, Karnofsky performance status; NR, no response; PR, partial response; WBC white blood cell count.
A Bayesian cumulative logit mixed-effects model with a patient-level random intercept was fitted, including follow-up time (1, 3, 6, and 12 months) and LASSO-selected covariates (pleural complications, occlusion strategy, fistula type, BMI, drainage volume, albumin, KPS, WBC, and CRP). The 4-level occlusion grade was treated as an ordered outcome, with higher levels indicating worse status. The LASSO coefficient profiles and variable selection process are shown in Figure 5(a) and (b), and the posterior mean estimates with 95% credible intervals from the Bayesian model are summarized in Figure 5(c). All parameters had Rhat values close to 1.00 and adequate effective sample sizes, indicating satisfactory convergence (diagnostics shown in Supplemental Figure S1 and Table S1).

LASSO-based variable selection and Bayesian ordinal mixed-effects modeling of longitudinal occlusion outcomes. (a) LASSO coefficient profiles. (b) LASSO-based variable selection. (c) Posterior mean estimates with 95% credible intervals from the Bayesian ordinal mixed-effects model.
After multivariable adjustment, the early postoperative period emerged as a high-risk window. Compared with baseline, the coefficients for time at 1 and 3 months were 2.53 (95% CrI 1.64–3.42) and 1.95 (95% CrI 0.96–2.93), respectively, both clearly positive, indicating a higher probability of remaining in worse occlusion grades during the first 1–3 months. At 6 and 12 months, the coefficients were −0.33 (95% CrI −1.42 to 0.76) and −0.58 (95% CrI −1.97 to 0.82), with credible intervals crossing zero, suggesting only a possible tendency toward recovery in the mid- to longer term. Using combined materials as the reference, the coefficient for pure pharmacologic therapy was −1.86 (95% CrI −2.70 to −1.02), indicating that patients treated with pure pharmacologic therapy were, overall, more likely to achieve and maintain better occlusion grades. This apparent advantage may at least partly reflect treatment selection, as pure pharmacologic therapy was preferentially used in patients with smaller fistula orifices and more favorable local conditions for healing. The coefficient for stent-only occlusion was −0.68 (95% CrI −1.75 to 0.40), pointing in the same favorable direction but with insufficient evidence to conclude superiority over combined materials. For fistula type, taking small visible fistulas as the reference, occult fistulas had a coefficient of −1.24 (95% CrI −2.28 to −0.20), indicating a higher likelihood of maintaining or regaining better occlusion status, which is consistent with their typically smaller orifice and lower local inflammatory burden. In contrast, the coefficients for pleural complications, BMI, drainage volume, albumin categories, KPS, WBC, and CRP were close to zero with 95% credible intervals generally spanning zero, suggesting no stable independent effects in this cohort.
Based on the final Bayesian cumulative logit mixed-effects model, we used the posterior mean estimates of the regression coefficients and cut-points to construct a prognostic risk scoring flowchart (Figure 6). All continuous variables marked with an asterisk (*) (e.g., BMI, WBC) are standardized using Z-score normalization: X_std = (X − μ)/σ. This flowchart provides a user-friendly tool for clinicians to input a patient’s key clinical variables and the follow-up time point to obtain a risk score and estimate the probability of each outcome category. It enables rapid risk stratification and outcome prediction for individual patients, facilitating personalized decision-making and timely intervention for those at high risk of occlusion failure or recurrence.
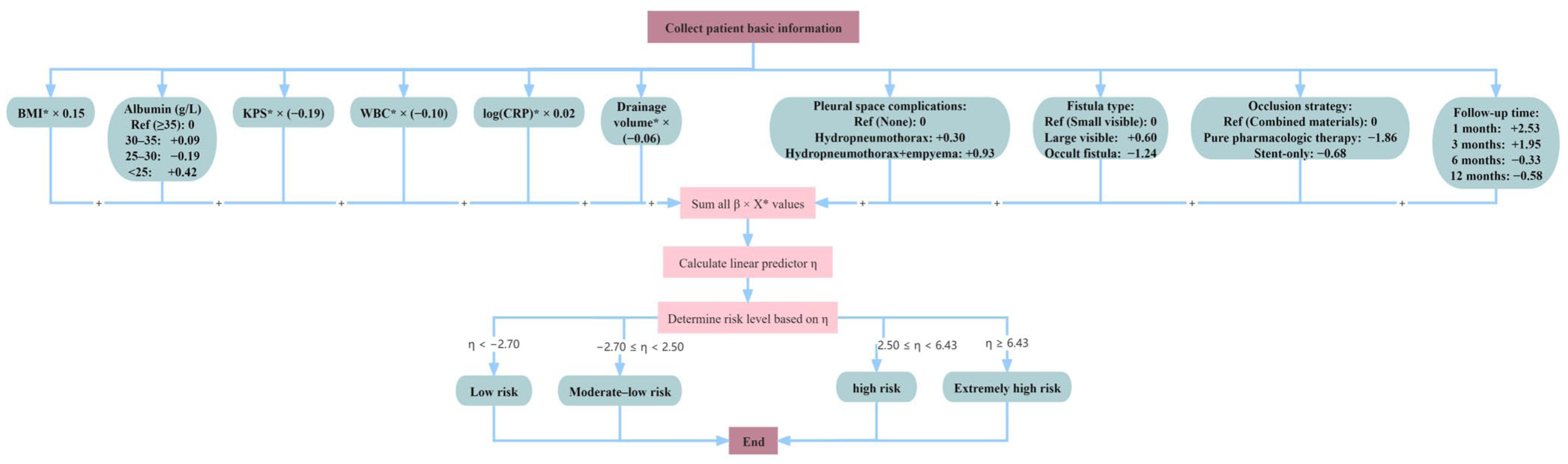
Clinical risk scoring algorithm derived from the final Bayesian cumulative logit model.
Discussion
In this study, we systematically reviewed the bronchoscopic occlusion treatment of patients with cBPF and analyzed longitudinal data at multiple time points to identify factors influencing occlusion strategy and efficacy. Several significant findings emerged. First, fistula anatomical characteristics—especially classification and size—were decisive factors in selecting occlusion materials. Fistulas with a diameter ⩾5 mm (large fistula type) tended to require a more complex occlusion approach, often involving stents or combinations of materials. In contrast, smaller fistulas were more often successfully managed with simpler measures such as sclerosing agents alone. This underscores the importance of careful assessment of fistula size and morphology in planning the occlusion strategy; choosing appropriate materials that provide sufficient mechanical support for larger fistulas is fundamental to improving success rates.
Another key conclusion of this research is the demonstration that the efficacy of BPF occlusion therapy evolves dynamically over time. We observed that approximately 3 months postprocedure is a critical window during which the treatment outcome can fluctuate. Some patients experienced delayed failure or recurrence of the fistula after an initial period of apparent success. The primary reasons for such late failure likely include persistent infection and inherent limitations of the occlusion materials. Residual pleural or pulmonary infection can impede scar formation or weaken existing scar tissue, leading to re-opening of the fistula. 14 Chronic inflammatory responses triggered by infection may cause pathological remodeling of local tissue. Neutrophil-rich inflammation and increased MMP-9 activity may promote extracellular matrix degradation around the fistula and impair durable healing.15,16 There are also limitations to the materials themselves. A long-term indwelling bronchial stent can lead to granulation tissue proliferation or migration of the stent; a metallic stent, being a foreign body, may continuously irritate the airway mucosa and provoke chronic inflammation.17,18 Likewise, biological glues may gradually degrade, allowing the air leak to recur. In an infected environment, pathogens such as Pseudomonas aeruginosa produce elastases that can specifically cleave fibrinogen α chains, 19 dramatically reducing the tensile strength of fibrin glue. 20 On the other hand, the fact that most patients who achieved initial successful occlusion maintained long-term closure suggests that with effective infection control and regular follow-up, durable healing is attainable. These observations highlight that clinicians should not be complacent after an initial successful bronchoscopic occlusion (“one-time success”); instead, long-term monitoring and proactive management are required, with readiness to intervene if signs of recurrence or delayed failure appear. These temporal patterns highlight the need for prognostic tools that go beyond static, single time-point outcomes and can support treatment decisions throughout the disease course. Several prognostic tools have been proposed for thoracic surgery, including a simple score by Hu et al. that predicts early BPF after pneumonectomy using only age ⩾70 years, diabetes and neoadjuvant therapy. 21 Such preoperative models are useful to identify high-risk patients and guide preventive strategies, but they do not inform management once BPF has already occurred. Our model, by contrast, focuses on patients with established central postoperative BPF undergoing bronchoscopic occlusion and uses longitudinal, ordinal outcomes (CR/CCR/PR/NR at 1, 3, 6, and 12 months) together with fistula anatomy, pleural complications, inflammatory burden and occlusion strategy to stratify prognosis. It is therefore designed as a complementary tool that shifts the emphasis from predicting “who will develop BPF” to guiding “how to individualize bronchoscopic treatment and follow-up when central BPF is present.”
In clinical practice, prevention and early control of cBPF begin with meticulous perioperative management. Preoperatively, optimization of nutritional status, serum albumin, glycemic control, and active pulmonary infection is essential to promote bronchial stump healing. Intraoperatively, careful bronchial stump closure, preservation of blood supply, and selective reinforcement with vascularized tissue flaps in high-risk resections help reduce the risk of cBPF. Postoperatively, strict management of the pleural space is fundamental: adequate and continuous chest drainage, timely relief of tube obstruction, and appropriate negative pressure are crucial to prevent hydropneumothorax, empyema, and secondary stump breakdown. Once cBPF has developed, management should be stepwise and individualized. Adequate drainage and targeted antimicrobial therapy are the first priorities, followed by bronchoscopic occlusion in hemodynamically stable patients with controllable infection, particularly those with small or occult fistulas who are poor candidates for reoperation. By contrast, thoracic surgical intervention is generally reserved for patients in whom pleural and systemic inflammation cannot be adequately controlled—for example, those with persistent or progressive hydropneumothorax or empyema, extensive stump dehiscence or destroyed residual lung, or failure of well-conducted conservative and bronchoscopic therapy. Close collaboration between thoracic surgeons and interventional pulmonologists, especially during the high-risk 1–3-month period after bronchoscopic occlusion, is essential to optimize timing and selection of each treatment modality.
Conclusion
In cBPF, fistula size and type can be used to guide occlusion strategy—pure pharmacologic therapy for small or occult fistulas and stent-based or combined approaches for larger defects—while the 1–3-month period after bronchoscopic occlusion should be regarded as a critical window for close surveillance. The proposed Bayesian risk-scoring flowchart offers a practical tool to stratify this risk and individualize bronchoscopic management.
Limitations
However, several issues should be considered when extrapolating our findings. This was a single-center retrospective study, and the case-mix, surgical techniques, occlusion materials, and infection-control practices of our tertiary center may differ from those in other institutions, so the absolute risks and calibration of the model may change when applied elsewhere. At the same time, the main predictors in our model—fistula size, pleural space complications, inflammatory burden, and occlusion strategy—are pathophysiologically plausible and routinely available, suggesting that the direction of their effects and the model’s ability to discriminate higher- from lower-risk patients may be at least partly preserved in other settings. Prospective multicenter studies are therefore needed to externally validate the model in different institutions and patient populations, and to determine whether recalibration or updating is required before wider clinical use.
Supplemental Material
sj-docx-1-tar-10.1177_17534666261431091 – Supplemental material for Bronchoscopic occlusion for central bronchopleural fistula after pulmonary resection: material selection and risk model for long-term prognosis
Supplemental material, sj-docx-1-tar-10.1177_17534666261431091 for Bronchoscopic occlusion for central bronchopleural fistula after pulmonary resection: material selection and risk model for long-term prognosis by Jiaping Liu, Lixin Cui, Heng Zou and Hongwu Wang in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-docx-2-tar-10.1177_17534666261431091 – Supplemental material for Bronchoscopic occlusion for central bronchopleural fistula after pulmonary resection: material selection and risk model for long-term prognosis
Supplemental material, sj-docx-2-tar-10.1177_17534666261431091 for Bronchoscopic occlusion for central bronchopleural fistula after pulmonary resection: material selection and risk model for long-term prognosis by Jiaping Liu, Lixin Cui, Heng Zou and Hongwu Wang in Therapeutic Advances in Respiratory Disease
Footnotes
Acknowledgements
We sincerely thank all staff from the Department of Pulmonary and Critical Care Medicine, Dongzhimen Hospital, Beijing University of Chinese Medicine, for their dedicated work in bronchoscopic procedures and patient care throughout this study.
Declarations
Supplemental material
Supplemental material for this article is available online.
Artificial intelligence involvement
In preparing this manuscript, we have utilized artificial intelligence tools (ChatGPT) for language polishing and editing to enhance the text’s clarity, fluency, and academic expression. The use of AI tools was strictly limited to improving grammar, wording, and overall expression and did not involve any alteration of research data or the generation of academic content. We ensure that all research data and conclusions are independently generated by the authors in compliance with academic integrity and ethical standards. The use of AI tools was aimed solely at improving language quality and did not affect the academic honesty of the paper.
