Abstract
Bronchopleural fistula (BPF) leading to persistent air leak (PAL), be it a complication of pulmonary resection, radiation, or direct tumor mass effect, is associated with high morbidity, impaired quality of life, and an increased risk of death. Incidence of BPF following pneumonectomy ranges between 4.4% and 20% with mortality ranging from 27.2% to 71%. Following lobectomy, incidence ranges from 0.5% to 1.5% in reported series. BPFs are more likely to occur following right-sided pneumonectomy, while patients undergoing bi-lobectomy were more likely to suffer BPF than those undergoing single lobectomy. In addition to supportive care, including appropriate antibiotics and nutrition, management of BPF includes pleural decontamination, BPF closure, and ultimately obliteration of the pleural space. There are surgical and bronchoscopic approaches for the management of BPF. Surgical interventions are best suited for large BPFs, and those occurring in the early postoperative period. Bronchoscopic techniques may be used for smaller BPFs, or when an individual patient is no longer a surgical candidate. Published reports have described the use of polyethylene glycol, fibrin glues, autologous blood products, gel foam, silver nitrate, and stenting among other techniques. The Amplatzer device, used to close atrial septal defects has shown promise as a bronchoscopic therapy. Following their approval under the humanitarian device exemption program for treatment of prolonged air leaks, endobronchial valves have been used for BPF. No bronchoscopic technique is universally applicable, and treatment should be individualized. In this report, we describe two separate cases where we use an Olympus© 21-gauge EBUS-TBNA (endobronchial ultrasound–transbronchial needle aspiration) needle for directed submucosal injection of ethanol leading to closure of the BPF and subsequent successful resolution of PAL.
Background
Bronchopleural fistula (BPF) leading to persistent air leak (PAL), be it a complication of pulmonary resection, radiation, or direct tumor mass effect, is associated with high morbidity, impaired quality of life, and an increased risk of death. Incidence of BPF following pneumonectomy ranges between 4.4% and 20%,1,2 with mortality ranging from 27.2% to 71%.3,4 Following lobectomy, incidence ranges from 0.5% to 1.5% in reported series. BPFs are more likely to occur following right-sided pneumonectomy, while patients undergoing bi-lobectomy were more likely to suffer BPF than those undergoing single lobectomy. 3 In addition to supportive care, including appropriate antibiotics and nutrition, management of BPF includes pleural decontamination, BPF closure, and ultimately obliteration of the pleural space. 4
There are surgical and bronchoscopic approaches for the management of BPF. Surgical interventions are best suited for large BPFs, and those occurring in the early postoperative period.2,5 Bronchoscopic techniques may be used for smaller BPFs, or when an individual patient is no longer a surgical candidate. Published reports have described use of polyethylene glycol, fibrin glues, autologous blood products, gel foam, silver nitrate, and stenting among other techniques. 6 The Amplatzer device, used to close atrial septal defects has shown promise as a bronchoscopic therapy. 7 Following their approval under the Humanitarian device exemption program for treatment of prolonged air leaks, endobronchial valves have been used for BPF.8,9 No bronchoscopic technique is universally applicable and treatment should be individualized.
In this report, we describe two separate cases where we use an Olympus 21-gauge EBUS-TBNA (endobronchial ultrasound–transbronchial needle aspiration) needle for directed submucosal injection of ethanol leading to closure of the BPF and subsequent successful resolution of PAL.
Case 1
A 65-year-old man, active smoker with right-lower-lobe squamous cell carcinoma presented for elective lobectomy. A bi-lobectomy was performed, given the mass location at the confluence of the right middle and lower lobes. On postoperative day (POD) #7, bronchoscopy was performed for mucus plugging at the level of the right mainstem. The bronchus intermedius (BI) stump appeared healthy, as expected postoperatively. Two days later, the patient developed systemic evidence of infection, and broad spectrum antibiotics were started. On POD #12, a brisk air leak and purulent pleural fluid drainage were noted. On POD #13, bronchoscopy revealed BI dehiscence with copious air and purulence emanating from the fistulous space.
The patient was taken to the operating room for pleural decontamination, Eloesser flap placement, and sutured repair of the dehisced BI. After suffering a cardiac arrest postrepair, a significant air leak was noted on the mechanical ventilator, with delivered and expired tidal volumes differing by 200 mL. Owing to this, the patient was unable to successfully wean from the ventilator. Flexible bronchoscopy was performed and showed a dehisced BI stump, estimated at 5 mm in diameter, with visible nylon sutures (Figure 1).

Fistulous tract with surgical suture visible.
Procedure
A flexible therapeutic bronchoscope was introduced via the endotracheal tube (ETT) and navigated to the dehisced BI. The mucosa surrounding the dehiscence was irritated using a cytologic specimen brush. A 21-g EBUS needle (Olympus NA-201SX-4021) was inserted via the bronchoscope working channel. Fifteen 0.1-mL aliquots of 98% ethanol were injected in a clockwise fashion into the submucosal layer. Mucosal edema developed immediately. By procedure completion the fistula orifice had fully approximated (Figure 2) and ventilator volumes equilibrated. A left mainstem intubation was performed to protect the edematous BI stump. On postprocedure day 2, the ETT migrated into the right mainstem without volume loss. The ETT was then retracted to the main carina. Although the patient recovered from a respiratory mechanics standpoint, they underwent tracheostomy insertion due to new onset encephalopathy. An inspection bronchoscopy performed 1 week following ethanol injection demonstrated and intact BI, absent fistulous lesions. Ultimately, the patient was discharged to a long-term acute care facility.

Resolution of bronchopleural fistula immediately following submucosal ethanol injection.
Case 2
A 69-year-old man with prolonged nicotine dependence and a history of stage-III squamous cell carcinoma on immunotherapy and status postradiation was transferred to our institution after thoracic bi-lobectomy. He developed PAL and received subsequent talc pleurodesis without any improvement in the PAL. Initial bronchoscopic evaluation included serial balloon occlusion of the right main stem and subsequent subsegments to identify the location of the fistulous tract. Without improvement in the air leak distal to the right main bronchus, inspection bronchoscopy identified a right BI fistula (Figure 3) endoscopically measured to be approximately 3.0 mm in diameter. Due to the fistula exiting out the posterior membrane of the right BI and its proximity to the right-upper-lobe takeoff, stenting and endobronchial valves were deemed not appropriate. He underwent two-step bronchoscopic ethanol injection procedures (Figure 4). Due to the large size of the fistula, the second set of injections was coupled with bronchoscopic injection of a foam sealant (Floseal, Baxter) directly into the fistulous orifice. Serial computed tomography (CT) scans 1 day before (Figure 5, black arrow) the first and 1 day after (Figure 6, black arrow) the second bronchoscopic procedures demonstrates apposition of bronchial mucosa and subsequent closure of the fistula. In addition, PAL had resolved.

Right bronchus intermedius fistula (arrow).
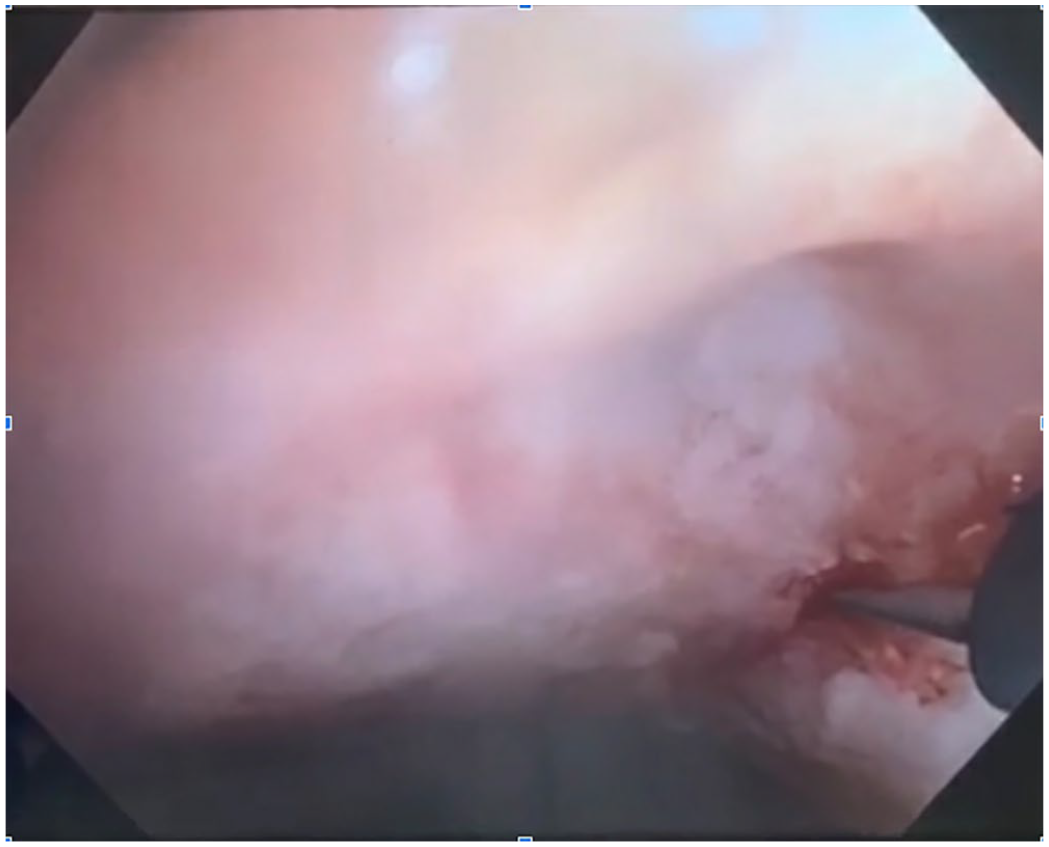
Injection of ethanol with 21-g EBUS needle. Injections are made circumferentially around the fistula.

CT scan after therapeutic injections demonstrating fistula (black arrow).

Axial CT scan showing closure of fistula (black arrow).
Procedure
Similar to the procedural steps outlined above in Case 1, using a flexible ultrathin bronchoscope through an ETT, a 21-g EBUS needle was inserted via the working channel. Approximately 10 0.3-mL aliquots of 98% ethanol were submucosally injected in a clockwise fashion around the peripheral rim of the fistula. Subsequently, using a guide sheath (Olympus) in the working channel, a foam sealant was injected directly through the sheath into the fistula. Using the lever of the bronchoscope, the distal end was flexed then gently extended over the foam to pack and evenly spread the sealant into the fistula.
Discussion
Consequences of a large BPF may include prolonged air leak, inefficient ventilation, ventilator dependence, pleural infections, sepsis, and death. Management of BPF centers upon prompt recognition and initiation of appropriate therapy, which is dictated by patient and clinical factors.
BPF may occur early in the postoperative period or in a delayed fashion. BPF can be associated with new fevers, dyspnea, or new onset sputum purulence. Physical examination may reveal subcutaneous emphysema, new air leak or change in chest tube drainage output. Thoracic imaging may demonstrate new or increasing pneumothorax, change in pleural fluid level, and mediastinal shift. Imaging must be interpreted in the context of expected postsurgical pneumothorax and effusion. CT scan findings may demonstrate disruption of the bronchial tree. Flexible bronchoscopy is an expedient form of BPF diagnosis in proximal fistulae and can simultaneously be used for therapeutic intent. 6
Takaoka and colleagues described the successful resolution of five small (<3 mm) central BPFs following submucosal injection of absolute ethanol. These patients developed central BPF postresection of malignant lesions, 1 day to 9 months following operation. The procedure included de novo endotracheal intubation, therapeutic flexible bronchoscopy, peri-fistula mucosal irritation with an endobronchial brush, and subsequent submucosal injection of absolute ethanol. Sessions involved 12 to 41 injections of 0.1 mL of ethanol, until demonstration of fistula closure. Three patients required multiple sessions. Initial survival was excellent, with death occurring in one patient 9 months postprocedure. 10
In this abstract, we outline two cases: the first, of a 65-year-old patient with BPF following bi-lobectomy for squamous cell carcinoma of the lung. This patient had recurrent BI dehiscence despite surgical intervention. Ethanol injection of the BI stump was able to rapidly resolve this patient’s air leak. To our knowledge, this is the first case of the use of an EBUS needle in bronchoscopic intervention for a BPF in a mechanically ventilated patient.
The second case, of a 69-year-old man who also underwent bi-lobectomy for primary lung squamous cell carcinoma. He had a PAL that was refractory to talc pleurodesis. Due to progressive deconditioning and tumor burden, he was deemed nonoperative. Two separate bronchoscopy procedures for ethanol injection were performed with bronchoscopic, radiographic, and clinical improvement. To our knowledge, this is the first endoscopic case using combination therapy with ethanol and foam sealant leading to successful closure of a large BPF.
Administration of ethanol for BPF has previously been reported. However, the use of an EBUS needle for injection is less apparent. The EBUS needle has a stylet that keeps the needle lumen clear of bronchial cells and any debris and thus allows for precise submucosal injections at exactly desired quantities. The adjustable needle facilitates an exact depth of penetration. An adjustable safety mechanism on the handle allows the bronchoscopist to limit the depth of needle penetration. This is in contrast to needles typically used in diagnostic bronchoscopy such as the Wang biopsy needle. Needle deployment in traditional devices is cumbersome and less controlled than the EBUS needle. Traditional needles lack an anchoring mechanism for attachment to the bronchoscope’s working channel and lack safety features meant to limit excess needle penetration depth. The EBUS needle is superior for interventions requiring precision and controlled injection.
Ethanol’s therapeutic effect for BPF is that it creates a localized inflammatory reaction, tissue edema, and eventual scar formation. Thus creating an overlying mechanical shelf at the site of the fistula. Ethanol causes rapid tissue dehydration and protein denaturation with subsequent fibrotic scar formation. 8 Permanent fibrosing effects of ethanol injection may take days to weeks to occur. Despite the relatively large size of the fistula orifice, ethanol injection was successful in the management of BPF. Benefits of ethanol injection include its low cost and rapid therapeutic effect. Moreover, whereas placing endobronchial valves and stents may in certain cases be anatomically impossible and poses no side effects associated with foreign body placement (migration, fracture, and mucosal shearing). This technique requires no additional resources than those readily available for therapeutic flexible bronchoscopy. Ethanol injection can be performed in a critically ill mechanically ventilated patient at the bedside, obviating the need for rigid bronchoscopy as in the Amplatzer device technique. These favorable characteristics led us to choose ethanol submucosal injection therapy to treat BPF in our two patients. Drawbacks include the need for significant bronchoscopic expertise, risk of tissue injury including ischemia and necrosis, and catastrophic injury to neighboring major vessels due to improper needle deployment and ethanol injection. Owing to this, we suggest preprocedure contrast-enhanced CT scanning. In addition, foam sealant sets quickly and runs the risk of potential damage to the working channel of the bronchoscope. Therefore, the use of a disposable guide sheath is advised. Inadvertent injection of ethanol can cause unintended tissue necrosis, and spillage of ethanol can cause scarring of normal endobronchial mucosa.
There are no definitive guidelines for management of BPF, though a myriad of approaches exist. We advocate for the submucosal injection of ethanol, precisely delivered via EBUS needle as an effective therapeutic option. We consider this technique in appropriate patients, including those for whom surgery has failed, or is not an option.
Footnotes
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
