Abstract
Background:
Postoperative recovery following lung cancer surgery can be challenging owing to various cardiorespiratory complications. Pulmonary rehabilitation is crucial during both the preoperative and postoperative periods. However, because of limited resources and accessibility, innovative approaches are necessary to improve rehabilitation outcomes. This study aimed to evaluate the effectiveness of the RehabLung App, a novel AI-based mobile application integrating vision recognition, gamified feedback, and web-based patient management to support personalized lung rehabilitation through telerehabilitation and real-time monitoring.
Objectives:
This study outlines the protocol for a randomized controlled trial involving patients with lung cancer undergoing thoracic surgery.
Design:
Assessor-blinded (single-blind), two-arm, parallel-group randomized controlled trial (1:1 allocation) conducted at a single medical center.
Methods and analysis:
The intervention will include individualized exercise prescriptions delivered five times per week for 8 weeks via the App, which adapts based on weekly assessments of clinical parameters and real-time performance data. Primary outcomes include forced expiratory volume in 1 second (FEV1) and forced vital capacity (FVC), as well as cardiopulmonary fitness assessed at baseline and at postoperative weeks 5 and 8. Secondary outcomes include respiratory muscle strength, diaphragmatic function, and user satisfaction, which are evaluated using a custom questionnaire informed by technology acceptance models. In addition, clinically meaningful endpoints will be collected, including morbidity, mortality, and length of stay. Frailty will be assessed preoperatively using the Clinical Frailty Scale, and validated patient-reported outcomes (EORTC QLQ-C30) will be administered to evaluate quality of life and symptom burden.
Ethics:
The study has been approved by the Institutional Review Board at National Cheng Kung University Hospital (A-ER-111-055).
Discussion:
We hypothesize that patients using the RehabLung App may demonstrate improvements in pulmonary function, exercise tolerance, diaphragmatic function, and rehabilitation adherence compared with those receiving standard care. The system’s real-time feedback and risk alerts may facilitate timely intervention and could potentially improve engagement.
Conclusion:
The implementation of the RehabLung App in lung cancer surgery care may enhance the effectiveness and accessibility of pulmonary rehabilitation. This protocol describes an AI-enhanced telerehabilitation system that may support individualized recovery strategies and could potentially improve both clinical outcomes and patient engagement.
Trial Registration:
Registration Date;2024,09,19; ClinicalTrials.gov (Identifier: NCT06600503).
Keywords
Introduction
Lung cancer is one of the leading causes of morbidity and mortality worldwide. Thus, the current clinical survey requires effective treatment strategies to improve patient outcomes. 1 For early-stage lung cancer, resection is a realistic therapeutic option that provides the best chance of survival. 2 However, patients’ cardiorespiratory performance is severely compromised during the postoperative phase, which makes it crucial. These difficulties include reduced physical activity, deterioration in overall quality of life, and diminished lung capacity. Thus, postoperative treatment must be effective in promoting healing and providing the best possible long-term health outcomes.
Respiratory function and lung capacity are frequently reduced after lung cancer surgical operations such as pneumectomies and lobectomies.3,4 Removing lung tissue may reduce lung volume, making it more difficult for the patient to oxygenate blood effectively. Furthermore, atelectasis can be induced by surgical stress and anesthesia, further impairing lung function. 5 In addition, postoperative discomfort and fatigue may limit a patient’s physical activity and lead to deconditioning. When these factors come together, the overall respiratory function declines significantly, making it necessary to implement appropriate rehabilitation measures to speed up recovery.
Patients with pulmonary diseases, especially those who recuperate from surgery, have been treated mainly through traditional lung rehabilitation. Physical therapy, breathing exercises, and education are commonly incorporated into rehabilitation programs to enhance lung function and overall physical well-being. Extensive research has demonstrated that these regimens can significantly improve lung function, exercise capacity, and the overall quality of life. Patients receiving classical lung rehabilitation frequently report improved management of concurrent illnesses, enhanced endurance, and decreased dyspnea symptoms. 6 Despite its well-established advantages, traditional rehabilitation requires substantial resources, such as specialized facilities and medical staff, which may prevent certain patients from receiving this program. The process of improving a patient’s physical and mental health before surgery to improve postoperative outcomes is known as prehabilitation or preconditioning exercise. Prehabilitation has significantly increased cancer patients’ muscle strength, cardiorespiratory fitness, and general resilience. Prehabilitation activities have been shown to reduce postoperative problems, shorten hospital stays, and expedite patient recovery times. The advantages of prehabilitation emphasize the importance of proactive management of cancer treatment.7,8
The limitations of traditional pulmonary rehabilitation are potentially resolved through telerehabilitation. Using digital AI technology, telerehabilitation makes rehabilitation programs more accessible to clinical patients by enabling remote monitoring and supervision. 9 Wearable technology, virtual coaching platforms, and smartphone applications offer real-time feedback and prescribe personal rehabilitation programs. These developments enable continuous monitoring of patient progress, act quickly when necessary, and increase patient involvement. 10 Thus, telerehabilitation has the potential to be as effective as traditional in-person treatment in rehabilitating patients, while also providing affordability and convenience. It is anticipated that its significance in the postoperative care of patients with lung cancer will increase as technology continues to advance. 11 In addition, telerehabilitation programs for lung cancer patients are safe and practical, with evidence showing improvements in quality of life, muscle mass, depression, and anxiety. However, they show limited or no impact on physical capacity (except in the preoperative period), symptom severity, survival, lung function, or dyspnea. After surgery, telerehabilitation enhances quality of life, depression, and anxiety; during systemic treatment, it also benefits symptom severity and muscle mass. Nonetheless, recruitment and attendance rates often fall below 70%. 12 AI-based tools have been used to create personalized exercise plans and track real-time heart rate in telerehabilitation for lung cancer survivors, leading to better cardiorespiratory fitness, quality of life, and symptom management compared to standard care. 13
The primary purpose of this research is to evaluate the effectiveness of a novel AI-based application utilizing vision recognition techniques, the RehabLung App, designed to assist in lung rehabilitation for patients following lung cancer surgery through real-time monitoring. Considering the established barriers to postoperative cardiorespiratory recovery, it is imperative to investigate novel approaches to enhance patient outcomes. This study aimed to evaluate the potential benefits and practical applications of the RehabLung App in medical practice by comparing patients’ cardiorespiratory function before and after lung cancer surgery. By highlighting the App’s performance, we seek to contribute to the growing body of evidence supporting the potential effectiveness of tele-rehabilitation for improving the quality of life and postoperative recovery of patients with lung cancer.
Method and materials
Study design
This study outlines the protocol for a randomized controlled trial designed to evaluate the effectiveness of the RehabLung App on lung function in patients undergoing thoracic surgery. The trial will be conducted in accordance with the SPIRIT 2013 Statement, a standard checklist for defining key protocol items in clinical trials. 14 The study is conducted in accordance with the Declaration of Helsinki, and all participants will provide written informed consent before they participate in the study. The study will consist of two arms: one group will receive the intervention through the RehabLung App, and another group will receive usual care. The measurement time points will include baseline, postoperative assessment at week 5, and follow-up assessment at week 8 after enrollment.
Study sample and setting
Participants will be referred to this study after being evaluated for anatomical lung resection surgery at least 1 month prior to the scheduled procedure by the attending physician in the Outpatient Department of the Thoracic Medicine Department at National Cheng Kung University Hospital, Tainan, Taiwan. Eligibility screening will be conducted upon referral, and once confirmed, a physical therapist on the study team will provide detailed information about the study protocol. Upon obtaining informed consent, participants will be randomly allocated to either the intervention group (RehabLung App with a web-based patient-care system plus video-based healthcare education) or the standard care group (video-based healthcare education only; Figures 1 and 2).
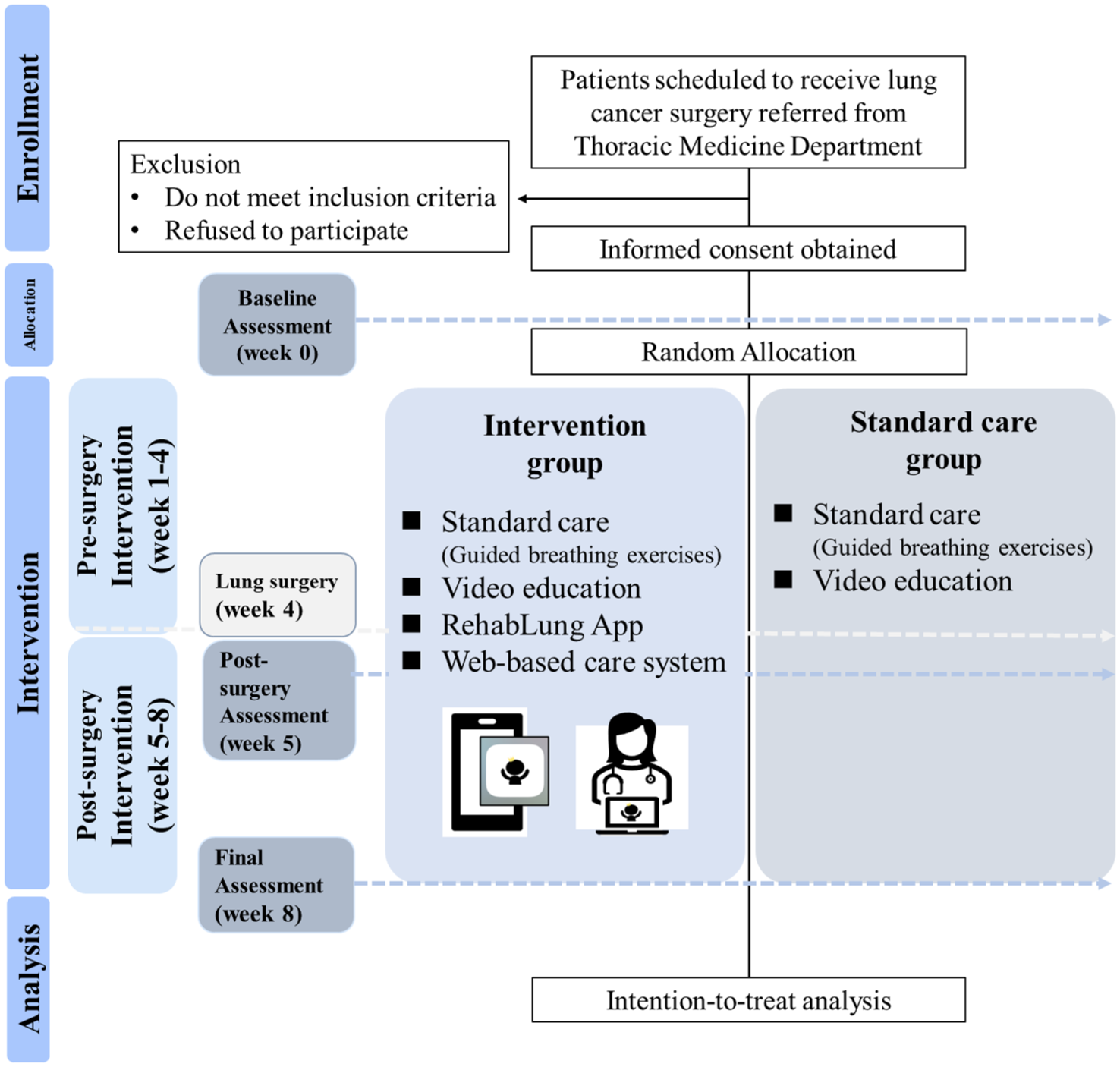
Flow chart of the study.
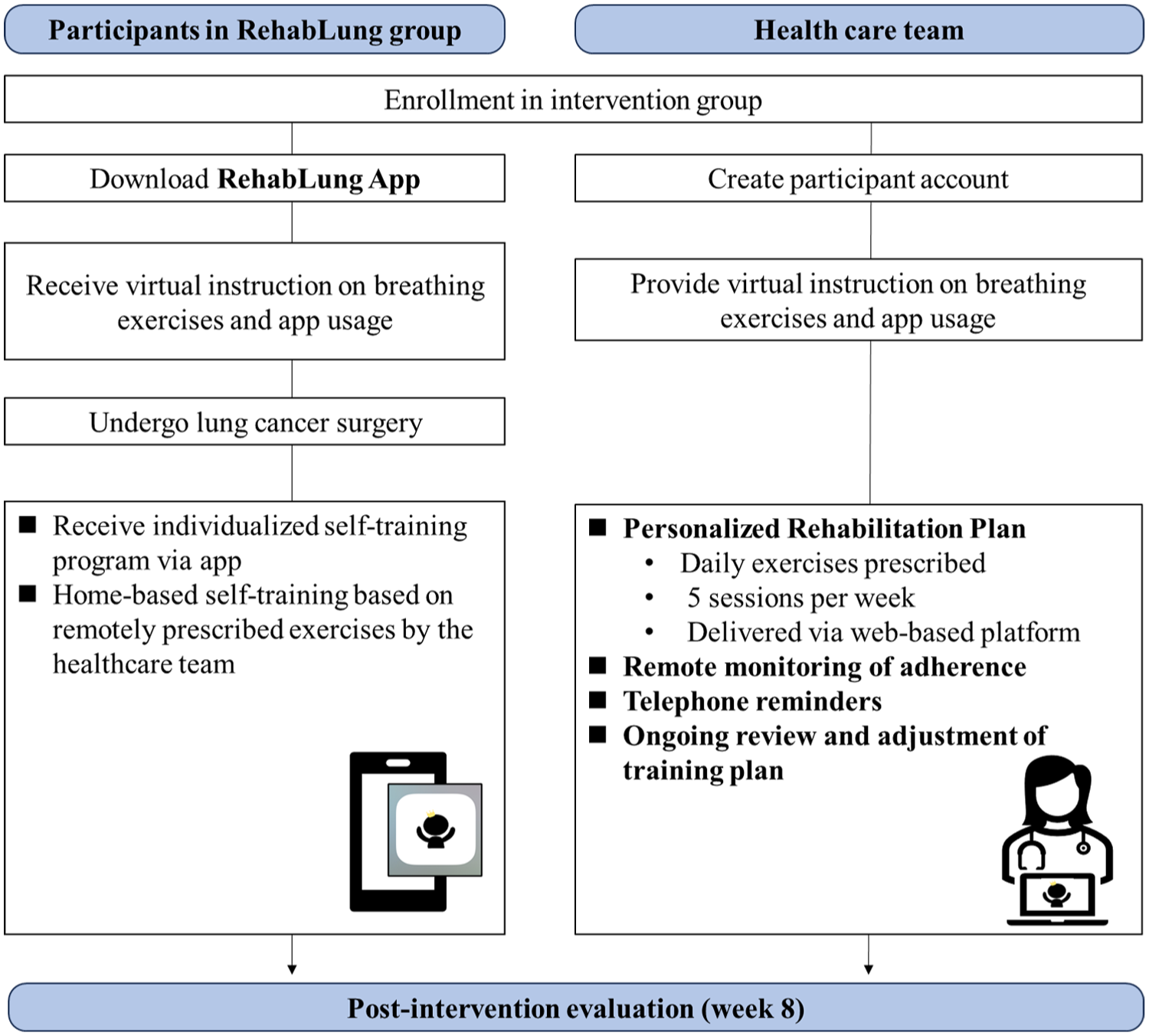
Intervention process for the RehabLung group.
The control group will receive only video-based healthcare education. This education included surgery-related Q&A, pre- and postoperative instructions, and guided breathing exercises via video. These exercises covered breathing control, inspiratory muscle training, chest expansion, segmental chest breathing with pursed-lip breathing, and stepping exercises.
Throughout the study period, participants in both groups were allowed to continue receiving standard postoperative care, including routine follow-up visits, medication management, and general health education as determined by their healthcare providers. However, any structured pulmonary rehabilitation programs or use of other digital exercise platforms outside of the study protocol were not permitted.
Intervention components
The intervention group will receive care through a combination of video-based healthcare education and a mobile application-guided postoperative pulmonary rehabilitation program. The RehabLung App (version 1.0; developed by the J. M. Su Lab in 2022) delivers a tailored exercise prescription that considers each participant’s surgical type and clinical condition. All participants begin with foundational exercises, including pursed-lip breathing, diaphragmatic breathing, thoracic mobility training, and general stretching routines. Building on this base, the program is further customized according to factors such as the type and location of lung resection (e.g., lobectomy or wedge resection), physical examination findings such as diminished breath sounds or asymmetrical thoracic movement, pulmonary function metrics such as FEV1%, patient-reported symptoms including dyspnea and fatigue, and evidence of thoracic tightness or local restrictions. This individualized approach integrates targeted thoracic expansion breathing exercises (unilateral/lateral costal expansion) and position-facilitated deep-breathing strategies to enhance regional ventilation and re-expand the lungs in surgically affected areas.
The exercise prescription framework was developed in accordance with clinical practice guidelines and informed by international standards from the American Thoracic Society (ATS) and European Respiratory Society (ERS) on pulmonary rehabilitation, which emphasize individualized treatment planning and progressive adaptation based on patient response.
Exercise prescription details (FITT-VT)
The exercise program follows the FITT-VT framework to ensure standardization and reproducibility. Participants in the intervention group are prescribed five training sessions per week, each lasting approximately 20–30 min. Exercise intensity is monitored using the Rate of Perceived Exertion (RPE) scale; if a participant reports an RPE score below 4 out of 10 after a session, the subsequent week’s prescription is adjusted by increasing the number of repetitions or introducing additional exercises. This self-regulated feedback mechanism facilitates gradual progression while maintaining safety.
Throughout the 8-week intervention period, the prescription is reviewed and modified weekly based on the patient’s clinical status, AI-generated performance data, and therapist evaluations. All exercises are delivered via the RehabLung App and monitored through a web-based platform for real-time supervision of adherence, frequency, and movement accuracy. This integrated digital system ensures that each participant receives personalized rehabilitation guidance and facilitates timely intervention if deviations from the expected recovery trajectory occur.
Monitoring and personalization
All exercise instructions and feedback are delivered through the RehabLung App and monitored in real-time via an integrated web-based patient-care system (Figure 3). This system enables healthcare professionals to track training frequency, adherence, and movement accuracy remotely.

Body, hand, and mouth position analysis in RehabLung application. The RehabLung application analyzes body and hand positions and captures mouth shape using a mobile camera to ensure correct breathing patterns during rehabilitation exercises. (a) Exhalation through pursed-lip breathing during stretching exercises. (b) Exhalation of segmental breathing through pursed-lip breathing.
If a participant is flagged as high risk for complications, alerts are sent to the clinical team. Based on scoring and progress evaluation, modifications are made to exercise intensity or functional goals. These updates ensure that the program remains aligned with the patient’s evolving clinical needs.
Performance evaluation and feedback
The RehabLung App uses AI-based vision recognition via the smartphone camera to analyze trunk and upper limb position, mouth shape and aperture, inhalation/exhalation duration, and repetition counts. Before each session, the system calibrates according to individual anatomical differences. These data are used to calculate accuracy and completion scores. A scoring algorithm assigns weights to exercises based on their clinical relevance, guiding both patient feedback and clinician decisions.
Engagement and motivation
To enhance engagement, the App integrates gamified elements, including personalized daily tasks, real-time feedback, verbal cues, synchronized music, and a point-based reward system. Participants can unlock features and share progress on social media, making rehabilitation more interactive and motivating.
Technical summary of the RehabLung system
The RehabLung App (clinically deployed version), developed through iterative refinement by a multidisciplinary team comprising clinical experts and software engineers from National Cheng Kung University and the National University of Tainan, was designed with internationally recognized pulmonary rehabilitation guidelines (e.g., ATS/ERS). 15 Its development emphasizes core components for patients with pulmonary impairment. The App consists of four main modules: (a) personalized exercise prescription tailored to preoperative status and postoperative progression; (b) AI-based motion detection using computer vision to analyze posture, breathing patterns, and movement accuracy; (c) gamified elements including real-time feedback, point accumulation, and motivational tools; and (d) a web-based clinician dashboard enabling real-time performance tracking and risk alerts. This integrated design ensures that the system is both clinically relevant and technically robust, supporting remote pulmonary rehabilitation.
Eligibility criteria
Eligible participants will be adults aged over 20 years scheduled to undergo lung resection surgery, including both minimally invasive procedures (e.g., video-assisted thoracoscopic surgery, thoracoscopic lobectomy, segmentectomy, wedge resection) and traditional open thoracotomy approaches. All participants must be conscious and cognitively intact. Patients with severe visual or hearing impairments, neuromuscular disorders, acute respiratory illnesses, or major comorbidities that could interfere with participation will be excluded.
Although feasibility studies often focus solely on minimally invasive cases, we included both surgical types to capture a broader spectrum of functional changes, especially as more invasive surgeries often result in more pronounced pulmonary and cardiopulmonary function declines that are clinically meaningful for evaluating intervention effects.
Sample size calculation
This study adopts two co-primary outcomes: lung function (spirometry) and cardiorespiratory fitness (CPET parameters). Sample size estimation was based on one of the co-primary outcomes—forced expiratory volume in one second (FEV1)—due to the availability of reliable published data with clinically relevant effect sizes. We referenced a randomized controlled trial by Tao et al. 16 involving patients with postoperative non-small cell lung cancer. Their study reported a mean FEV1 of 2.27 ± 0.48 L in the intervention group, which received pulmonary rehabilitation, and 1.75 ± 0.53 L in the control group at 3 months post-surgery. Using this difference (Δ = 0.52 L) and a pooled standard deviation of approximately 0.505 L, we calculated the required sample size with a two-sided α of 0.05 and 80% power using G*Power (version 3.1.9.6). In addition to FEV1, this study also reported clinically relevant functional and symptom outcomes (such as 6MWT performance and dyspnea-related measures), supporting the clinical significance of the pulmonary function difference.
The analysis indicated a need for 16 participants per group. To account for a potential dropout rate of 10%, we increased the target to 17 participants per group, resulting in a total of 34 participants for the trial.
Randomization
This randomized controlled trial will employ stratified randomization based on the Eastern Cooperative Oncology Group (ECOG) performance status scale to ensure balanced group assignment. Participants will be categorized into three strata: ECOG 0-1, representing asymptomatic or mildly symptomatic individuals who are fully ambulatory; ECOG 2, including symptomatic individuals capable of self-care but unable to work; and ECOG 3-4, covering patients with more severe disabilities who have limited self-care or are completely disabled. A computer-generated randomization list will allocate participants to either the intervention or the control group in a 1:1 ratio within each stratum. An independent researcher, not involved in participant enrollment, intervention delivery, or outcome assessment, will generate the randomization sequence using block randomization. Participants will be enrolled by a physical therapist on the study team, who will also assign participants to the allocated group using sealed opaque envelopes. Allocation will remain concealed until the completion of the baseline assessment, at which point the envelopes will be opened to reveal the group assignment.
Although ECOG status was the primary stratification factor, additional data on age, surgery type, and cancer stage were collected for potential post-hoc analyses. In the event of participant dropout, the study will apply an intention-to-treat analysis, with appropriate statistical techniques used to address any significant imbalances arising from missing data. This trial design ensures that groups are balanced in terms of preoperative functional status, which is a crucial factor influencing post-surgery rehabilitation outcomes in patients with lung cancer, thus enhancing the study’s overall reliability.
Blinding
Because of the nature of a behavioral, app-based rehabilitation intervention, participants and the treating physical therapist cannot be blinded to group allocation. Therefore, this trial employs a single-blind (assessor-blinded) design. During the informed-consent process, participants will be told that they will be randomly assigned to one of two postoperative rehabilitation programs and will be informed of their assignment after randomization and baseline assessment. Outcome assessments at baseline, postoperative, and follow-up stages will be performed by an outcome assessor who is not involved in participant enrollment, intervention delivery, or routine participant support, and who will remain unaware of group assignment. Participants will be reminded at each assessment not to share any information about their intervention (e.g., app features, training content, or monitoring feedback) with the assessor. Group labels will be anonymized using neutral identifiers (e.g., Group A/Group B) for data management, and any accidental unblinding incidents will be recorded.
Mobile application intervention
The daily training program was tailored and prescribed by the physical therapist, including specific chest breathing exercises and repetitions, with recommended rest periods between each exercise to prevent discomfort that may arise from hyperventilation. After completing each exercise, the participants will be guided to self-evaluate their perceived effort using a RPE scale ranging from 1 to 10. The therapist can then adjust the program based on the participant’s feedback from the system (Table 1).
The components of chest rehabilitation in the RehabLung application include a visual and audio training guide, along with key evaluation focus areas to assess the accuracy of exercises.

Outcome measures
All study measurements were conducted by the same outcome assessor at baseline within one week after enrollment, postoperative assessment, and follow-up assessment at week 8.
Primary outcome
This study defines two co-primary outcomes to comprehensively evaluate patients’ recovery following thoracic surgery: lung function and cardiorespiratory fitness (CRF). These outcomes were selected to reflect both the pulmonary and systemic cardiovascular effects of the mobile rehabilitation intervention, in alignment with its multidimensional goals.
Cardiorespiratory fitness will be assessed through a cardiopulmonary exercise test using a cycle ergometer aimed at obtaining a comprehensive evaluation of cardiopulmonary function during physical exertion. Participants will be encouraged to undergo a cycle ergometer stress test to evaluate cardiopulmonary function. During the test, the workload gradually increased by 10 W/min until the participants reached their peak exercise capacity. Throughout the exercise test, the participants will be carefully monitored for echocardiography and heart rate while wearing a mask that analyzes ventilatory outcomes using a computed gas analysis system. Key measures of cardiorespiratory fitness will include peak oxygen consumption (VO2 peak) and workload achieved during the test, recorded as peak work rate (in watts) and peak work rate (% predicted). Gas analysis will provide insights into oxygen exchange efficiency, including maximal ventilation (L/min), maximal ventilation (% predicted), ventilatory equivalents, VE/VCO2 slope, and anaerobic threshold (AT), which is defined as a nonlinear increase in CO2 production during the incremental exercise test. Predicted values for peak oxygen uptake, peak work rate, and other measures will be referenced based on the participants’ race, age, height, biological sex, and weight.
Pulmonary function will be evaluated using spirometry to assess the lung volume and ventilatory capacity over time. Experienced therapists will guide participants to perform maximal inspiration, followed by maximal effort exhalation for as long and as forcefully as possible. The measurements obtained included forced vital capacity (FVC [L, % predicted]), forced expiratory volume in one second (FEV1 [L, % predicted]), and the ratio of these two volumes (FEV1/FVC [% predicted]). The predicted values for these measurements will be derived from large population studies of healthy adults matched by race, age, height, biological sex, and weight. These tests will assess differences in pulmonary function between the groups before and after surgery, with assessments conducted at baseline, postoperatively, and at the follow-up endpoint. To account for variability in recovery trajectories, follow-up assessments will be scheduled within predefined windows (e.g., week 5 ± 7 days and week 8 ± 10–14 days postoperatively). If postoperative complications or clinical instability occur, spirometry and CPET will be delayed until the treating physician determines testing is safe. Complications (e.g., pulmonary infections or other issues) and length of stay will be extracted from medical records and incorporated into secondary outcomes and sensitivity analyses.
Secondary outcome
The secondary outcomes included global respiratory muscle strength and diaphragm function. Global respiratory muscle strength was assessed using a pressure gauge and a mouthpiece. Participants were instructed to perform five maneuvers, starting with maximal exhalation, followed by full inhalation, followed by maximal inhalation, followed by full exhalation through the mouth. Measurements included maximal inspiratory pressure (PImax [cmH2O, % predicted]) and maximal expiratory pressure (PEmax [cmH2O, % predicted]).
Diaphragmatic function was assessed by diaphragm excursion, and diaphragm thickness was assessed by ultrasonography (NextGen LOGIQ e Ultra-sound, GE Healthcare, USA) at the right hemidiaphragm. A curve 5–12 MHz transducer set to two-dimensional mode was first used to depict the right diaphragm, a motion-mode beam was performed perpendicular to the posterior part of the diaphragm, and a motion-mode image was used to measure the distance of diaphragm excursion between end-expiration and end-inspiration. A linear 6–13 MHz transducer was set to the B mode to assess changes in diaphragmatic thickness (Δtdi%)
Quality of life
The European Organization for the Research and Treatment of Cancer Quality of Life Questionnaire C30 (EORTC QLQ-C30) is an instrument that includes 30-item measuring health-related quality of life among cancer patients in five main domains incorporated five functional scales (physical, role, cognitive, emotional, and social), three symptom scales (fatigue, pain, and nausea and vomiting), five single symptom items and financial difficulty. THE EORTC QLQ-C30 questionnaire is validated in a wide range of languages, including traditional Chinese. All items are transformed into scores ranging between 0 and 100, with a higher score indicating better functioning in five functional scales and a lower score indicating fewer symptoms and better quality of life. 19 Participants will perform the EORTC QLQ-C30 survey through Google Form. The median duration to finish the EORTC questionnaire by internet is 6.0 min.
Frailty
Frailty status will be assessed using the Clinical Frailty Scale (CFS). 20 The evaluation will include baseline and postoperative stages to assess changes in patient functional resilience and vulnerability from preoperative to postoperative stages.
Clinical outcome measures
The study will obtain clinical outcomes from patients’ medical records to determine how the intervention affects real-world results. The study will track the morbidity of postoperative complications, including cardiopulmonary complications and infections, mortality, and the length of hospital stay.
Additional variables
We developed the Mobile Rehabilitation System Satisfaction Questionnaire, which consists of 25 items rated on a 5-point Likert scale. This questionnaire evaluated several key aspects of the rehabilitation system, focusing on user satisfaction and its effectiveness in the self-rehabilitation process. The items assess whether the system meets rehabilitation needs, provides practical guidance, offers immediate feedback on user movements, and analyzes the rehabilitation status. Additionally, the questionnaire explored user perceptions of the system’s convenience, enjoyment, and ease of use, as well as user engagement and confidence in the self-rehabilitation process. The design of these questions aimed to comprehensively assess the system’s role in facilitating self-rehabilitation and to understand users’ overall perspectives on the system.21–25
The questionnaire was developed based on key theoretical constructs from the Technology Acceptance Model (TAM), NASA-TLX, and usability evaluation literature, focusing on domains such as perceived usefulness, ease of use, cognitive load, and user engagement. Although the questionnaire has not yet undergone psychometric testing, it was reviewed by a multidisciplinary panel for content relevance and face validity. Future work will focus on formal validation and refinement. In this study, feasibility is evaluated by adherence rates, dropout rates, and user satisfaction. Adherence is defined as the proportion of completed sessions out of the total prescribed sessions during the 8-week program, tracked automatically via the RehabLung App. Dropout is defined as participants who withdraw before completing the final follow-up. Acceptability is assessed using the Mobile Rehabilitation System Satisfaction Questionnaire, which captures user experience and perceptions of the App’s usability, convenience, and effectiveness in supporting rehabilitation.
Although our primary outcomes focus on clinical parameters, these additional process indicators allow us to evaluate the real-world applicability and patient engagement of the intervention.
Compliance of intervention
Compliance and adherence to the study protocol will be encouraged through reminder phone calls from the study team. The application system provides the healthcare team with individual training results for each participant and generates alerts if a participant fails to engage in the prescribed sessions. In such cases, the study team will discuss the reasons for their lack of engagement, aiming to understand any barriers they may be facing, whether related to health, scheduling, or personal circumstances. This proactive approach fosters open communication and support, which may help enhance adherence to the program.
Data collection, management, and analysis
All participant data will be stored confidentially in accordance with the Taiwan Personal Data (Information) Protection Act (PDPA) and the General Data Protection Regulation. Data collection was followed by a series of statistical analyses. Normality of the data was assessed using the Shapiro–Wilk test. Data are presented as mean ± SD or median [interquartile range], as appropriate. Intention-to-treat analysis will be employed to mitigate biased comparisons between groups and to address potential dropout effects.
Between-group comparisons will use unpaired Student’s t-tests or Mann–Whitney U tests, while categorical data will be analyzed using Pearson’s chi-square or Fisher’s exact test. Within-group changes over time will be assessed using the Wilcoxon signed-rank test. Bonferroni correction will be applied for multiple comparisons. A two-sided p-value of < 0.05 will be considered statistically significant.
To control variability introduced by different surgical approaches, subgroup analyses will be conducted based on surgical type (minimally invasive vs open thoracotomy). In addition, potential confounding factors such as baseline lung function and tumor stage will be considered in future multivariable analyses. All analyses will be performed using SPSS Statistics for Mac OS X, Version 26.0 (IBM, USA).
Participant and data safety
Throughout the study period, the physical therapist and attending physician closely monitored the participants for any adverse events and assessed their impact on both physical and mental health. In the event of adverse effects, appropriate medical care is provided.
To ensure the safety of digital data, our mobile application complies with the Institutional Review Board guidelines of National Cheng Kung University Hospital, specifically the “Basic Information Security Checklist for Mobile Applications in Human Research Projects,” which protects user privacy and data security in human research. We adhere to stringent data protection regulations to ensure that the application does not access any other information on the user’s device. During installation, users are prompted to confirm the permissions required for various functionalities and have the option to revoke any granted permissions afterward to safeguard their privacy. The application actively alerts participants to obtain their consent, and all data collected by the system are encrypted, requiring decryption for access. Thus, in the event of a data breach, the information remains unreadable. Furthermore, all personnel involved in the system will sign a confidentiality agreement to ensure that no data are disclosed externally.
Clinical trial registration
This study was approved by the Institutional Review Board of National Cheng Kung University Hospital (NCKUH) (Registration Number: A-ER-111-055) and was registered at ClinicalTrials.gov (Identifier: NCT06600503). The record was first verified on October 1, 2024, and the anticipated study start date is October 10, 2025, with full study completion by December 31, 2025.
Discussion
It is well established that lung rehabilitation has many benefits in patients undergoing lung surgery. The digital AI technology of telerehabilitation provides remote monitoring and supervision, making rehabilitation programs more accessible to patients. This study was designed to assess the potential efficacy of a new AI-based mobile application for lung recovery in patients after lung cancer surgery (RehabLung App). While no definitive data currently support specific improvements in cardiorespiratory function, the design of the RehabLung App suggests that it may be beneficial for enhancing recovery outcomes. The anticipated improvements in exercise tolerance, lung capacity, and overall quality of life highlight how telerehabilitation could address the limitations of conventional lung rehabilitation methods. The RehabLung App aims to facilitate both preoperative and postoperative interventions through a comprehensive survey approach. Patients can engage in planned prehabilitation exercises before surgery, track their progress, and facilitate adherence to prescribed regimens. By optimizing lung function and overall physical fitness, patients may be better prepared for the demands of surgery, potentially reducing postoperative complications. Improved patient outcomes may be achieved by increasing patient involvement and motivation through personalized workout routines and real-time feedback.
With real-time data on lung capacity, respiratory function, and physical activity, healthcare professionals were able to monitor patients’ recovery progress and tailor rehabilitation plans as needed. 26 By promoting adherence to prescribed exercises, the App’s personalized feedback may reduce the risk of complications such as atelectasis and deconditioning. This study developed the RehabLung App to bridge the gap between preoperative planning and surgical recovery through continuous remote monitoring and individualized management, which may improve overall patient outcomes.
Although the RE-AIM framework was not formally adopted, our study design and evaluation procedures addressed several of its core domains. The reach was reflected in the clearly defined inclusion criteria and the successful recruitment of eligible participants. Adoption and implementation were captured through objective adherence data, participant satisfaction, and App usage metrics. Effectiveness was evaluated through changes in lung function parameters and self-reported outcomes. These elements together suggest that the intervention was designed with practical translation and implementation in mind.
Future studies may consider formally operationalizing the full RE-AIM framework to strengthen implementation of science perspectives and guide scalability assessments. Building upon previous research indicating that telerehabilitation can be as effective as traditional in-person programs—while offering advantages in accessibility and continuity of care—the RehabLung App’s real-time monitoring and tailored feedback functions are anticipated to enhance adherence and optimize rehabilitation outcomes.
Moreover, remote monitoring allows for timely interventions, particularly in the vulnerable postoperative phase when complications may arise. However, the single-group design of the current study limits the generalizability of our findings. Larger cohort studies are needed to validate these results across diverse patient populations. Future research should also explore the long-term benefits and cost-effectiveness of AI-supported mobile rehabilitation applications compared to conventional strategies. Future investigations should also explore the long-term benefits of AI-based mobile applications and evaluate their cost-effectiveness relative to conventional rehabilitation techniques.
In conclusion, the RehabLung App is designed to support postoperative recovery and may enhance respiratory function following lung cancer surgery. Although preliminary observations suggest that telerehabilitation may serve as a viable alternative to conventional lung rehabilitation and prehabilitation programs, further investigation is required to substantiate these claims. The App aims to facilitate real-time monitoring and provide tailored feedback that could potentially improve patient engagement and recovery. As the study progresses, it is essential to assess the effectiveness of apps in diverse patient populations and their long-term benefits compared to traditional rehabilitation methods. Ultimately, this AI-based mobile application has the potential to advance postoperative care and enhance the quality of life of patients with lung cancer, warranting further research to explore its efficacy.
Interpretation
This study aimed to develop an AI-based mobile application for patients undergoing lung cancer surgery to enhance accessibility and potentially improve the efficiency of pulmonary rehabilitation. By leveraging AI, the application was designed to provide a comprehensive platform for preoperative and postoperative care, enabling patients to engage in tailored rehabilitation exercises while facilitating remote monitoring by healthcare professionals. The anticipated outcomes of this study may contribute to a better understanding of how digital AI interventions can support recovery in this patient population and improve overall rehabilitation experiences.
Supplemental Material
sj-docx-1-tar-10.1177_17534666261427319 – Supplemental material for The effectiveness of the AI-based RehabLung mobile rehabilitation system on cardiopulmonary function and user satisfaction in lung cancer patients undergoing thoracic surgery: a protocol for a two-arm randomized clinical trial
Supplemental material, sj-docx-1-tar-10.1177_17534666261427319 for The effectiveness of the AI-based RehabLung mobile rehabilitation system on cardiopulmonary function and user satisfaction in lung cancer patients undergoing thoracic surgery: a protocol for a two-arm randomized clinical trial by Hsin-Lun Yang, Ching-Hsia Hung, Yu-Ting Huang, Hui-Ching Cheng, Yau-Lin Tseng, Jun-Ming Su and Kun-Ling Tsai in Therapeutic Advances in Respiratory Disease
Footnotes
Acknowledgements
None.
Declarations
Supplemental material
Supplemental material for this article is available online.
Declaration of generative AI and AI-assisted technologies in the writing process
During the preparation of this work, the author(s) used OpenAI’s ChatGPT to assist with language refinement, including grammatical corrections, and to improve the clarity and conciseness of the content. After using this tool, the author(s) reviewed and edited the content as required and took (s) full responsibility for the content of the publication.
Guarantor statement
Dr. Kun-Ling Tsai assumes responsibility as the guarantor and acknowledges the integrity of this study.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
