Abstract
Background:
Chronic obstructive pulmonary disease (COPD) with an eosinophilic phenotype is linked to frequent exacerbations and increased oral corticosteroid (OCS) use. Mepolizumab, an anti–IL-5 monoclonal antibody, has shown efficacy in eosinophilic airway disease. This study assessed its real-life effectiveness in COPD patients with frequent OCS-requiring exacerbations.
Objectives:
To evaluate the effectiveness of mepolizumab in reducing exacerbations and corticosteroid use in eosinophilic COPD patients in a real-world setting.
Design:
Retrospective, single-centre cohort study with two-year follow-up.
Methods:
The study included 13 consecutive COPD patients treated with mepolizumab (100 mg SC every 4 weeks) from 2020 to 2022 and followed for two years. Inclusion required ⩾2 OCS-treated exacerbations or maintenance OCS use in the prior year and blood eosinophils > 300 cells/µL. Response was defined as a ⩾50% reduction in annual OCS-requiring exacerbations.
Results:
At one year, 9 of 13 patients (69%) were responders. Median annual moderate-to-severe exacerbations decreased from 4.0 (IQR: 1.5–10.0) to 1.0 (IQR: 0.0–2.0; p = 0.007). COPD-related hospitalizations declined from 2.0 (IQR: 0.0–4.0) to 0.0 (IQR: 0.0–0.0; p = 0.007). Among the eight patients who continued therapy into the second year, moderate-to-severe exacerbations declined from a median of 5.0 (IQR: 3.0–12.0) at baseline to 2.0 (IQR: 1.0–2.8) in year one and 0.5 (IQR: 0.0–2.8) in year two (p = 0.018), representing 60% and 90% reductions, respectively. Hospitalisations dropped from 2.0 (IQR: 0.3–4.0) to 0.0 (IQR: 0.0–0.0) in both years (p = 0.008). Maintenance OCS was discontinued in 2 of 4 patients (50%) after one year. Responders did not differ from non-responders in type 2 markers, but tended to have a higher baseline frequency of exacerbations.
Conclusion:
Mepolizumab significantly reduced exacerbations and hospitalizations in eosinophilic COPD patients with frequent OCS use. A 69% response rate at one year and sustained benefit in the second year support its role in this difficult-to-treat population.
Background
Chronic obstructive pulmonary disease (COPD) is a progressive lung condition, primarily caused by long-term cigarette smoking, characterized by persistent airflow limitation, chronic inflammation and recurrent exacerbations. 1 Acute exacerbations contribute to disease progression, increased healthcare utilization and higher morbidity and mortality rates. While infections are a major trigger, some exacerbations are intrinsic and associated with elevated blood and sputum eosinophils.2,3 Current guidelines recommend triple inhaled therapy combining inhaled corticosteroids (ICS), long-acting β2-agonists (LABAs) and long-acting muscarinic antagonists (LAMAs)—for COPD patients with frequent exacerbations and elevated blood eosinophils. 4 However, despite this regimen, 30%–40% of patients continue to experience moderate-to-severe exacerbations.4,5
COPD is a heterogeneous disease with multiple clinical phenotypes and inflammatory endotypes. 6 Eosinophilic COPD, defined by blood eosinophil counts ⩾150–200 cells/μL, is associated with a higher exacerbation risk but shows a better response to corticosteroid therapy. 3 Given its pathophysiological similarities to eosinophilic asthma, targeted anti–IL-5 therapy has been investigated for eosinophilic COPD. Mepolizumab, a humanized monoclonal antibody against interleukin-5 (IL-5), reduces blood and tissue eosinophils by blocking IL-5 signalling. In severe eosinophilic asthma, it has significantly lowered exacerbation rates and improved quality of life. 7 However, its role in COPD remains less clear. Two randomized controlled trials (METREX and METREO) assessed mepolizumab in COPD patients with moderate-to-severe exacerbations. A significant reduction in annual exacerbation rates was observed in METREX (p = 0.04), particularly in patients with higher baseline eosinophil counts. 8 In the subsequent MATINEE trial, which included only patients with consistently high eosinophil counts (⩾300/μl at baseline), mepolizumab was confirmed to be moderately efficacious, showing a significant 21% reduction in exacerbation rates (p = 0.01). 9 Similar findings were reported for benralizumab, another anti–IL-5 biologic, in the GALATHEA and TERRANOVA trials, although statistical significance was not reached for overall exacerbation reduction. 10 Based on post hoc analyses of the METREX and METREO studies, mepolizumab treatment appears to be more effective specifically in highly eosinophilic COPD patients with frequent exacerbations requiring oral corticosteroids (OCS), providing a potential targeted therapeutic option for this difficult-to-treat phenotype. 11
Recent trials have explored dupilumab, a monoclonal antibody targeting interleukin-4 (IL-4) and interleukin-13 (IL-13), key drivers of Type 2 inflammation. The BOREAS trial demonstrated that dupilumab significantly reduced moderate-to-severe COPD exacerbations (rate ratio 0.70; p < 0.001) in patients with eosinophilic COPD (⩾300 eosinophils/μL), alongside improvements in lung function and quality of life. 12 The NOTUS trial further confirmed these findings, showing a 34% reduction in exacerbation risk (p < 0.001), with sustained FEV1 improvements at weeks 12 and 52. 13 These findings reinforce the role of Type 2 inflammation in COPD pathophysiology. However, optimal patient selection criteria remain a challenge.
This study aims to evaluate the efficacy of mepolizumab in a real-life setting in a highly selected cohort of COPD patients with eosinophilic inflammation and a high exacerbation and OCS use burden, providing further insight into its role in exacerbation reduction and overall clinical benefit.
Methods
Study design and participants
This study included consecutive COPD patients initiated on mepolizumab therapy at the University Clinic for Respiratory and Allergic Disease, Golnik, between 2020 and the end of 2022. For inclusion in this specific analysis, to ensure enrichment for smoking-related disease, patients were required to have a confirmed diagnosis of COPD, defined by post-bronchodilator FEV1/FVC < 0.70 and compatible clinical symptoms, significant smoking history (⩾20 pack-years) and radiological evidence of emphysema on high-resolution computed tomography HRCT. Eligibility for mepolizumab treatment was determined based on two key criteria:
(1) clinical instability, defined as either ⩾2 exacerbations in the previous year requiring a short course of OCS (defined as ⩾3 consecutive daily doses of 40 mg/day prednisone-equivalent, for example, 32 mg/day methylprednisolone), or dependence on maintenance oral corticosteroids (mOCS) (defined as ⩾5 mg/day prednisone-equivalent for ⩾3 months), despite receiving ⩾6 months of triple inhaled therapy that included a high-dose inhaled corticosteroid; and 2) eosinophilic phenotype (blood eosinophil count > 300 cells/μL documented within the last year). Exacerbation severity was defined according to GOLD criteria; however, only events requiring OCS were recorded in this study. Moderate exacerbations were defined as those treated with OCS, with or without antibiotics, while severe exacerbations were defined as OCS-treated events requiring hospitalization, potentially associated with acute respiratory failure. Mild exacerbations managed with SABA alone were not captured. 14
Patients were excluded if they had other significant respiratory diseases (e.g., interstitial lung disease, lung cancer or known bronchiectasis not attributable to COPD), a primary diagnosis of asthma rather than COPD, were current smokers at the time of treatment initiation, or declined to participate in the study.
Markers of type 2 (T2) inflammation included blood eosinophil count (BEC, cells/μL), fractional exhaled nitric oxide (FeNO, ppb) and total serum IgE (kU/L). BEC was obtained at baseline (within 1 month prior to treatment initiation), and historical BEC values from the preceding 12 months were also recorded. FeNO and IgE levels were recorded.
HRCT was performed in all patients prior to treatment initiation. Scans were interpreted by a single experienced thoracic radiologist (IP) who was blinded to treatment response. Excessive dynamic airway collapse (EDAC) was defined as a > 70% reduction in the cross-sectional area of the trachea or main bronchi during forced expiration on dynamic expiratory CT imaging.
Bronchial wall thickening (BWT) and mucus plugging were evaluated on baseline HRCT. BWT was assessed in the lobar bronchi using a semi-quantitative 4-point visual scale: absent (normal wall thickness for age and airway generation), mild (subtle thickening without luminal narrowing), moderate (definite thickening with mild luminal narrowing) or severe (marked thickening with significant luminal narrowing or airway distortion), similar as described before. 15 For analysis, BWT was considered present if moderate or severe thickening was identified in any lobar bronchus.
Mucus plugging was defined as complete occlusion of the airway lumen on HRCT, involving medium- to large-calibre bronchi (estimated lumen diameter 2–10 mm). 16 The extent of plugging was categorized by the number of involved lung segments: none, 1–2 segments (mild) and ⩾3 segments (extensive).
Treatment was approved by a multidisciplinary team specializing in obstructive lung diseases. During the study period, fourteen consecutive patients meeting these criteria were initiated on mepolizumab and followed to assess clinical response.
Intervention
Patients received mepolizumab (100 mg sc every 4 weeks) while continuing their standard COPD treatment, including inhaled bronchodilators and corticosteroids, as indicated.
Patients were regularly followed at each application visit and underwent additional comprehensive assessments every 6 months. Clinical evaluations at 1 year and 2 years post-treatment initiation included exacerbation frequency (number of moderate/severe exacerbations per year, focused on those requiring OCS) and OCS use (requirement for maintenance therapy). Treatment was discontinued at 1 year if exacerbation frequency did not decrease by at least 50%, if the severity of exacerbations did not decrease or if the dose of mOCS was not significantly reduced.
Patients were managed according to a standardized protocol throughout the duration of mepolizumab treatment, with clinical data collected retrospectively. Pharmacological treatments were assessed by a clinical pharmacist (TM). One patient was excluded from the final analysis due to a short treatment duration and insufficient data.
Outcomes and data analysis
The primary outcome assessed was the response to mepolizumab. Patients were classified as responders based on their clinical outcomes at 12 months of follow-up if exacerbation frequency or OCS maintenance dose decreased by at least 50% or who were able to successfully discontinue mOCS without relapse. Partial responders showed a 25%–49% reduction in annualized OCS-treated exacerbations. Non-responders experienced < 25% reduction or discontinued mepolizumab due to lack of efficacy.
This study was reported in accordance with the STROBE guidelines (see Supplemental File 1). 17
Continuous variables were expressed as medians and interquartile ranges (IQRs) due to the small sample, while categorical variables were summarized as percentages. Exploratory comparisons between groups were performed using nonparametric tests. Exacerbation rates before and after treatment were compared using the Wilcoxon signed-rank test or the Friedman test as appropriate. Due to the limited sample size, we did not perform multivariable analyses. A two-tailed p-value < 0.05 was considered statistically significant.
Results
Demographic and clinical characteristics of patients
Thirteen patients were included (9 responders, 4 non-responders). Overall, median age was 67.7 years (IQR, 63.8–73.1), with a median age at COPD diagnosis of 55.0 years. Females represented 66.7% of the cohort, more frequent among non-responders (75%) than responders (55.6%). The entire cohort had a substantial smoking history, with a median of 40 pack-years (IQR, 30–60). All patients were on high-dose ICS containing triple therapy and had ceased smoking by the time mepolizumab treatment was being considered.
Baseline and peak historical BEC were higher in non-responders compared with responders (180 (IQR, 132–460) vs 130 (90–315) cells/μL and 890 (585–1015) vs 700 (510–1175) cells/μL, respectively), although neither difference was statistically significant. Overall, FeNO and total serum IgE levels were elevated, with median values of 93 ppb (IQR, 51–100) and 123 kU/L (IQR, 115–266), respectively. Lung function was lower in non-responders. While FEV1 was reduced (855 mL (34%) vs 1120 mL (50%) in responders), the difference was not statistically significant. In contrast, FVC was significantly lower in non-responders (1875 mL (58.5%) vs 3110 mL (89.0%), p = 0.003). DLCO was also numerically lower in non-responders (46.5% vs. 67.0%). At 6 months, responders demonstrated numerically greater median improvement in lung function (ΔFEV1: 119.1 mL vs 55.0 mL). Detailed baseline characteristics are presented in Table 1.
Baseline patient characteristics.

Data are number of participants (%), median (IQR) or mean ± SD; p < 0.05 are in bold.
AE COPD H, Acute exacerbation of chronic obstructive pulmonary disease requiring hospitalization; AE COPD OCS, Acute exacerbation of chronic obstructive pulmonary disease requiring oral corticosteroids; BEC, blood eosinophil count; BMI, body mass index; DLCO, Diffusing capacity of the lungs for carbon monoxide; FeNO, fractional exhaled nitric oxide; FEV1, forced expiratory volume in 1 second; FVC, forced vital capacity; ICS, inhaled corticosteroid; at 6 M, values measured at 6 months; mOCS, maintanence oral corticosteroids.
At baseline, the mean annual exacerbation rate was higher in responders compared to non-responders (7.22 ± 4.68 vs 1.75 ± 2.06; p = 0.05) and the median number of OCS-treated exacerbations was significantly greater in responders (3 (IQR 1–4.5) vs 0 (IQR 0–1.5); p = 0.035). In addition, severe exacerbations requiring hospitalization were numerically more common among responders (median 3 (IQR 1–4.5)) vs (0.5 (IQR 0–3.25), p = 0.26). While mOCS use was present in both groups, it was more frequent among non-responders (2 of 4, 50%) than responders (2 of 9, 22%). These findings suggest that baseline OCS burden—particularly exacerbation frequency and severity—may be more predictive of treatment response than BEC alone in eosinophilic COPD.
All patients underwent HRCT within 1 year preceding treatment initiation. Imaging revealed emphysema and varying degrees of BWT and mucus plugging in all patients, with detailed quantitative assessments presented in Table 2. Bronchiectasis was present in at least a minimal form in 7 of 12 patients (58.3%). Concurrent EDAC was identified in 5 cases (41.7%).
Airway features according to response.

Data are presented as frequencies. p-Values for comparisons between responders and non-responders were > 0.05.
Mepolizumab treatment significantly reduced exacerbation frequency in the majority of patients
Using predefined response criteria (⩾50% reduction in exacerbation requiring OCS rate or decreased mOCS dose within the first year), 9 patients (69%) were classified as responders, 3 (23%) as non-responders and 1 (8%) as a partial responder. The partial responder showed clinical improvement and a reduction in exacerbation severity but did not meet the predefined criteria (Figure 1). Two non-responders died by the end of the first year. Treatment discontinuation occurred in three patients: one non-responder due to lack of efficacy, one responder for reasons unrelated to treatment and one responder who voluntarily discontinued despite clear clinical benefit. Notably, the latter patient experienced recurrent exacerbations following discontinuation of mepolizumab.

Patient flow and treatment outcomes over 2 years of treatment.
Mepolizumab treatment was associated with a statistically significant reduction in exacerbation requiring OCS frequency. Median annual moderate-to-severe exacerbations decreased from 4.0 (IQR: 1.5–10.0) to 1.0 (IQR: 0.0–2.0; p = 0.007), corresponding to an overall 75% reduction from baseline. COPD-related hospitalizations declined from 2.0 (IQR: 0.0–4.0) to 0.0 (IQR: 0.0–0.0; p = 0.007) - Figure 2. Among the eight patients who continued mepolizumab therapy into the second year, median moderate-to-severe exacerbations decreased from 5.0 (IQR: 3.0–12.0) at baseline to 2.0 (IQR: 1.0–2.8) in the first year (60% reduction) and further to 0.5 (IQR: 0.0–2.8) in the second year (p = 0.018), a 90% reduction from baseline. Hospitalizations similarly decreased from 2.0 (IQR: 0.3–4.0) at baseline to 0.0 (IQR: 0.0–0.0) in both years (p = 0.008)—Table 1, Figures 2 and 3. Maintenance OCS was discontinued in 50% (2 of 4) of patients after one year.
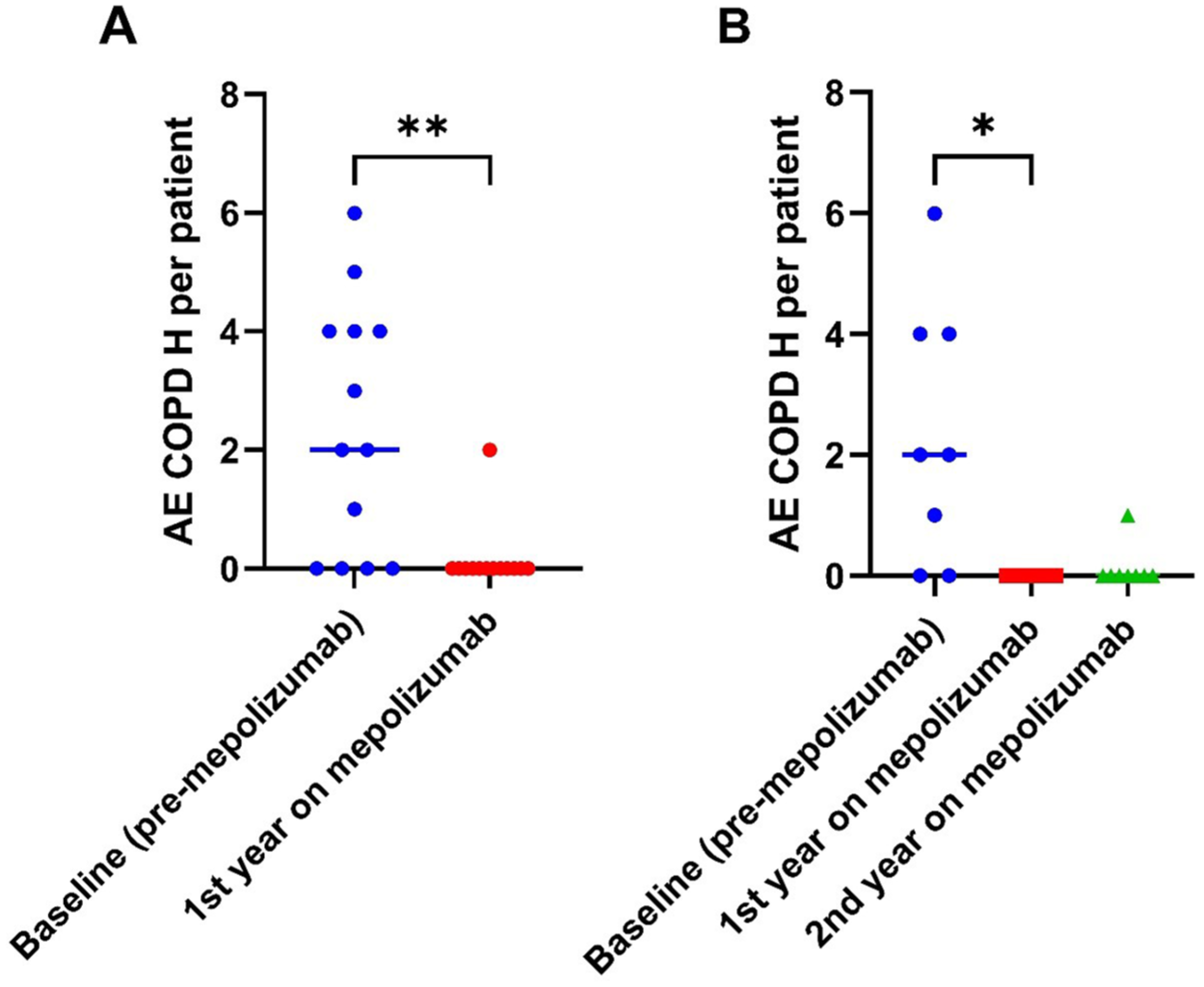
Number of AE COPD requiring hospitalization per patient. (a) In all included patients (n = 13); (b) in patients who continued with mepolizumab treatment into the second year (n = 8).

Total AE COPD per patient during continued mepolizumab therapy (n = 8). (a) Scatter plot with medians. (b) Individual trajectories.
Discussion
Our study demonstrated a significant reduction in moderate and severe exacerbations requiring OCS in a small cohort of patients with severe eosinophilic COPD who experienced frequent systemic steroid-requiring exacerbations and were treated with mepolizumab in a real-world clinical setting. Significantly, 69% of patients responded to treatment within the first year, and these effects persisted into the second year in patients who continued with mepolizumab treatment. These findings provide valuable insights into the effectiveness of mepolizumab in this high-risk population.
Our study adds to the growing evidence supporting the role of anti–IL-5 therapies in eosinophilic COPD. In prior randomized trials, mepolizumab significantly reduced exacerbations in METREX and MATINEE, though with modest absolute effects, while the METREO trial showed a similar but non-significant trend.8,9 In contrast, benralizumab did not reduce exacerbation rates in GALATHEA or TERRANOVA (10). Compared with these trials, our cohort had substantially higher baseline exacerbation rates (mean 5.5 per year vs ~1.2–2.7 per year in RCTs), reflecting a more severe and selectively enriched population. We observed an approximately 75% overall reduction in OCS-treated exacerbations after 1 year of mepolizumab. In patients who continued therapy, the reduction deepened further—by 60% in the first year and 90% from baseline by the second year. This magnitude of benefit exceeds that reported in prior trials and underscores the potential value of careful patient selection, particularly among individuals with high BEC and frequent OCS-treated exacerbations despite maximal inhaled therapy.
Our cohort differed from those enrolled in randomized trials in several important respects. Although spirometric disease severity, as measured by FEV1, was comparable to that reported in RCTs (mean FEV1 approximately 40%–50% predicted), responders in our study exhibited significantly higher FVC values, suggesting that better preserved lung volumes may be associated with greater responsiveness to treatment. Responders also exhibited a significantly higher baseline frequency of exacerbations than non-responders, indicating that individuals with more unstable disease may derive greater clinical benefit. Furthermore, our analysis focused specifically on OCS-treated exacerbations, whereas randomized trials also included events managed with antibiotics. This narrower definition may have enriched the cohort for individuals with a more eosinophilic, corticosteroid-responsive phenotype. Notably, a post hoc analysis of the METREX and METREO trials similarly reported greater benefit among patients with elevated BEC and OCS-requiring exacerbations, consistent with our findings. 11 Similarly, a post hoc analysis of benralizumab clinical trials found that patients receiving triple therapy, with elevated baseline blood eosinophil counts and a history of three or more exacerbations in the prior year, were most likely to benefit from treatment. 18 These findings further underscore the need for a more complex characterization of the patient subgroup most likely to respond, incorporating both clinical and biomarker data.
In our study, both responders and non-responders had high historic BEC preceding initiation of mepolizumab treatment, with no significant differences observed between groups. This raises questions about the reliability of BEC as a standalone predictive biomarker of anti–IL-5 response in COPD. Recently, Leung et al. 19 demonstrated that BEC correlate only weakly with airway T2 inflammation markers such as IL-13 and mast cell gene expression, and that one-third of patients exhibited discordant eosinophilia status between blood and airways in COPD. Furthermore, airway eosinophilia was a strong predictor of ICS-responsive T2 inflammation. Taken together, this highlights the need for a better biomarker to predict response to ICS and biological therapy in COPD. Post hoc RCT analyses, along with our data, suggest that a history of frequent OCS-requiring exacerbations serves as a better clinical indicator of future response to mepolizumab and may reflect significant airway eosinophilia.
Meanwhile, therapies targeting a broader spectrum of T2 inflammation, such as interleukin (IL)-4 and IL-13 inhibition with dupilumab, have demonstrated promising results in COPD patients with elevated BEC and/or FeNO. The BOREAS and NOTUS trials reported both reductions in exacerbation rates and improvements in lung function within this broader T2 inflammatory phenotype.11,12 Distinct clinical COPD subphenotypes may differentially benefit from these targeted biological therapies. In particular, eosinophilic COPD patients experiencing a very high frequency of exacerbations and OCS burden potentially gain significant benefit from anti–IL-5 treatment, as observed in our study. In a recent large cohort of 43,753 COPD patients on maintenance treatment, 5.2% needed four or more OCS courses annually; in the same study, among 1287 patients with ⩾3 exacerbations and BEC ⩾250, 37% required ⩾4 OCS courses, representing a considerable number of patients with unmet clinical needs despite maximal inhaled therapy. 5 This highlights a significant population with frequent exacerbations and eosinophilic inflammation who may benefit from targeted biologic therapies.
Although limited by sample size, we analyzed HRCT airway features across the entire cohort. While differences did not reach statistical significance, the data suggest that specific airway characteristics may be associated with treatment response. Responders to mepolizumab treatment more frequently demonstrated moderate-to-severe BWT, whereas non-responders more commonly exhibited extensive mucus plugging. This may reflect differences in biologic pathway mechanisms. Anti–IL‑5 therapy primarily targets eosinophil-mediated airway wall inflammation, which remains pharmacologically modifiable. In contrast, mucus plugging is more closely associated with epithelial alarmins (IL‑33/TSLP) and downstream IL‑13–driven goblet cell hyperplasia and mucus hypersecretion, leading to fixed luminal obstruction that may be less responsive to eosinophil depletion alone. Consistent with this, trials targeting IL‑4/IL‑13 (e.g., dupilumab) and IL‑33 have demonstrated effects on mucus-related physiology and outcomes, whereas the benefits of anti–IL‑5 appear greatest in eosinophilic, wall-predominant disease.11,12,18 These findings support a multimodal approach to selecting biological therapy that incorporates clinical history, imaging features, exacerbation patterns and biomarkers. HRCT features may aid in identifying patients more likely to respond to mepolizumab; however, confirmation in larger, prospective studies is needed. Data on the predictive value of HRCT airway features are scarce, but our findings align with a recent study showing greater response to inhaled therapy in asthma patients with increased airway wall thickness. 20
This study has several limitations. First, the small sample size, single-centre retrospective design and lack of a control group limit the generalizability of the findings, and between-group comparisons should be interpreted with caution. Second, although clinical follow-up extended into the second year, data collection relied on medical records, which may underreport milder or self-managed exacerbations. Third, imaging findings were evaluated semi-quantitatively and based on retrospective HRCT review, which may introduce subjectivity. Fourth, given that severe asthma can present with fixed airflow obstruction, 21 we cannot exclude the possibility that the observed T2 inflammation is driven by an underlying asthmatic pathology rather than COPD, especially in light of recent negative results of the RESOLUTE trial involving benralizumab in COPD. 22 Finally, the study did not assess airway-specific biomarkers such as sputum eosinophils or IL-13 expression, which might have better predicted response to anti–IL-5 therapy than BEC alone. 23 These limitations underscore the need for prospective, controlled studies with larger sample sizes and biomarker-integrated designs.
However, a key strength of our study is the extended follow-up beyond 1 year, allowing for the assessment of longer-term treatment effects. The observed trend toward a further reduction in exacerbations requiring OCS rates in the second year among patients who continued with mepolizumab is an important finding. Although the small sample size limits the statistical power of this observation, it suggests that the benefits of treatment may not only persist but potentially increase over time in a carefully selected population of unstable patients with severe eosinophilic COPD.
Conclusion
In conclusion, despite limitations including a small sample size and the lack of a control group, our study provides valuable real-world evidence of mepolizumab treatment benefits in a distinct subgroup of eosinophilic COPD patients. In this cohort, higher baseline rates of OCS-treated exacerbations and greater overall corticosteroid burden were more strongly associated with treatment response than blood eosinophil counts alone. This suggests that clinical exacerbation history—particularly events requiring systemic corticosteroids—may better capture disease activity and treatment benefit than biomarkers in isolation. These findings complement observations from larger clinical trials and contribute to the evolving understanding of patient selection for anti–IL5 therapy. Future larger prospective studies are warranted to confirm our observations, evaluate the long-term effects of mepolizumab in this population and compare its efficacy with other emerging biologic therapies targeting T2 inflammation.
Supplemental Material
sj-docx-1-tar-10.1177_17534666251414352 – Supplemental material for Mepolizumab for eosinophilic COPD with frequent exacerbations: single-centre real-world experience in a phenotype-directed approach
Supplemental material, sj-docx-1-tar-10.1177_17534666251414352 for Mepolizumab for eosinophilic COPD with frequent exacerbations: single-centre real-world experience in a phenotype-directed approach by Irena Šarc, Tina Morgan, Igor Požek and Peter Kopač in Therapeutic Advances in Respiratory Disease
Footnotes
Acknowledgements
None.
Declarations
Supplemental material
Supplemental material for this article is available online.
Use of artificial intelligence
During the preparation of this work, the authors used ChatGPT version GPT-4o to enhance the readability and language of the manuscript. After using this tool/service, the authors reviewed and edited the content as needed and take full responsibility for the content of the publication.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
