Abstract
Inhaled bronchodilator therapy with long-acting muscarinic antagonists (LAMAs) and long-acting β2-agonists (LABAs) in combination is currently the mainstay of treatment for chronic obstructive pulmonary disease (COPD). Treatment guidelines recommend the addition of inhaled corticosteroids (ICS) to LABA/LAMA only in patients with a history of frequent/severe exacerbations and high blood eosinophil counts, or in those with concomitant asthma. Despite this, real-world data suggest that clinicians are not adhering to this guidance and that ICS are frequently overused. This is possibly due to the incorrect assumption that when LABA/LAMA therapy is not sufficient, adding an ICS to the treatment regimen is the logical next step. In this narrative review, we describe global and country-specific guideline recommendations from Germany, Spain, and Japan and compare these with real-world data on LABA/LAMA and ICS use in clinical practice. We also provide a clinical guide to the use of add-on therapies with LABA/LAMA for different patient phenotypes, including (1) patients still symptomatic (but not exacerbating) despite LABA/LAMA treatment; (2) patients still exacerbating despite LABA/LAMA treatment who have high blood eosinophil counts; and (3) patients still exacerbating despite LABA/LAMA treatment who do not have high blood eosinophils or concomitant asthma.
Plain language summary
Treatment guidelines for chronic obstructive pulmonary disease (COPD) recommend dual bronchodilator therapy for the majority of patients, consisting of an inhaled combination of long-acting β2-agonist (LABA) and long-acting muscarinic antagonist (LAMA). Patients whose COPD is not well controlled on LABA/LAMA require further clinical intervention, which may or may not involve treatment with additional drugs.
Data from observational studies reflecting routine clinical practice suggest that inhaled corticosteroids (ICS) are often added to LABA/LAMA, even though treatment guidelines recommend only adding ICS in a specific group of patients with a history of exacerbations and high levels of eosinophils (a type of inflammatory cell) in the blood, or in those with current asthma. As long-term ICS use may be associated with an increased risk of side effects such as pneumonia, it is important to avoid overuse of ICS. When a patient’s COPD is not well controlled on LABA/LAMA, other treatable conditions should first be ruled out, and factors such as medication adherence, inhaler technique, and co-existing health conditions should also be considered.
This review gives advice on what follow-up options physicians should consider when LABA/LAMA is not providing adequate control of a patient’s COPD. Specifically, recommendations are given for three different patient profiles:
1. Patients who still have symptoms of COPD (but no acute/sudden worsenings of symptoms, known as exacerbations).
2. Patients who have exacerbations, as well as high levels of eosinophils in the blood.
3. Patients who have exacerbations, but without high levels of eosinophils in the blood or current asthma.
Background
Chronic obstructive pulmonary disease (COPD) is characterized by progressive deterioration of lung function and airflow limitation. Long-term maintenance treatment is required for relief of symptoms,1,2 the most characteristic of which is chronic and progressive dyspnoea.1,3 COPD may also be punctuated by periods of acute worsening of respiratory symptoms, called exacerbations.1,4
Inhaled bronchodilator therapy with long-acting muscarinic antagonists (LAMAs) and long-acting β2-agonists (LABAs) in combination is currently the mainstay of COPD treatment.1,5,6 The Global Initiative for Chronic Obstructive Lung Disease (GOLD) 2023 report recommends first-line treatment with LABA/LAMA for patients with high symptom burden (modified Medical Research Council [mMRC] score ⩾2 and/or COPD Assessment Test score ⩾10), or frequent or severe exacerbations (⩾2 moderate exacerbations/year or ⩾1 exacerbation leading to hospitalization) and a blood eosinophil count< 300 cells/µL. 1 According to GOLD 2023, triple therapy (ICS in combination with LABA/LAMA) should only be considered as initial pharmacological treatment in patients with both frequent or severe exacerbations and blood eosinophils ⩾300 cells/µL, and the use of LABA/ICS is no longer encouraged. 1 However, if patients with COPD have concomitant asthma, ICS use is mandatory. 1
Global and specific national guidelines are largely harmonized in their recommendations that most patients with COPD should be on bronchodilator monotherapy or dual therapy.1,7–9 However, despite guidelines showing that LABA/LAMA is central to the management of COPD, this is often not the case.
In this review, we compare guidelines for the pharmacological treatment of COPD with real-life prescribing patterns reported in registries and other published studies. We also include a brief clinical description of when and why add-on therapies to LABA/LAMA should be considered as a second step in treatment for the following patient phenotypes: (1) patients still symptomatic (but not exacerbating) despite LABA/LAMA treatment; (2) patients still exacerbating despite LABA/LAMA treatment who have high blood eosinophil counts; and (3) patients still exacerbating despite LABA/LAMA treatment but who do not have high blood eosinophil counts or concomitant asthma.
Recommendations in guidelines: How should patients be treated?
According to the GOLD 2023 report, LABA/LAMA is recommended as first-line therapy for the majority of patients with COPD. 1 In line with these recommendations, the American Thoracic Society (ATS) and Japanese Respiratory Society (JRS) also recommend first-line therapy with LABA/LAMA for patients with COPD and dyspnoea or exercise intolerance.10,11 In terms of follow-up treatment, GOLD recommends the addition of ICS (LABA/LAMA/ICS) if patients continue to have exacerbations despite LAMA or LABA monotherapy (if blood eosinophils ⩾300 cells/µL), or LABA/LAMA dual therapy (if blood eosinophils ⩾100 cells/µL). However, in patients with persistent dyspnoea despite LABA/LAMA, ICS is not recommended; instead, switching inhaler type or molecules should be considered, as well as non-pharmacological approaches (e.g. pulmonary rehabilitation). Other potential causes of dyspnoea should also be investigated and treated. 1
Similar to GOLD, the German National Guideline for COPD recommends that the majority of patients are initially treated with a long-acting bronchodilator or LABA/LAMA dual therapy. 9 Patients classified as GOLD A are recommended either short-acting muscarinic antagonists or short-acting β2-agonists, LAMA or LABA as initial therapy. 9 For patients who continue to experience exacerbations despite adequate treatment with long-acting bronchodilators, ICS should be considered. 9
The Spanish COPD guidelines stratify patients into two levels of risk – low and high – according to the degree of airflow obstruction, degree of dyspnoea, and frequency or severity of exacerbations. 7 For the initial treatment of low-risk patients (i.e. forced expiratory volume in 1 s ⩾50%, mMRC 0–1, and ⩽1 moderate exacerbation in the previous year), a LAMA monotherapy is recommended, with LABA/LAMA as follow-up treatment; for high-risk patients, three phenotypes are included in the pharmacological treatment scheme, namely non-exacerbators, non-eosinophilic exacerbators, and eosinophilic exacerbators. 7 For non-exacerbators (⩽1 exacerbation per year) and non-eosinophilic exacerbators, LABA/LAMA is recommended as both initial and follow-up inhaled treatment, with triple therapy an option for non-eosinophilic exacerbators with blood eosinophils >100 cells/µL, taking into account the frequency, severity, and aetiology of the exacerbations and the risk of pneumonia. 7 For eosinophilic exacerbators, step-up from LABA/ICS to LABA/LAMA/ICS is recommended. 7
According to the JRS guidelines, LAMA (or LABA) is recommended for patients with mild COPD; patients with moderate-to-severe COPD should be treated with LABA/LAMA, with the addition of macrolides, theophylline, or a mucolytic agent if further control of symptoms is required. 11 ICS is recommended in patients with concomitant asthma-like features and/or in patients with frequent exacerbations and blood eosinophil counts of more than 300 cells/µL.11,12
Largely, the recommendations are aligned between GOLD, ATS, Germany, Spain, and Japan, as well as between guidelines from many other countries or regions.1,7,9,11,13–17 For example, the ATS practical guidelines strongly recommend that any patient with dyspnoea and exercise intolerance should be given LABA/LAMA as first-line therapy. 10
It is important to acknowledge that guideline recommendations always refer to classes of drugs rather than specific LAMA, LABA, or ICS molecules (and their combinations) within each class. Although some studies have suggested that different LABA/LAMA fixed-dose combinations have specific efficacy profiles in COPD that might allow more personalized therapy, 18 systematic reviews evaluating intra-class efficacy for LABA/LAMA and LABA/LAMA/ICS have not shown any clear evidence of greater efficacy associated with specific combinations.19–23 A summary of international and national treatment guidelines for COPD is presented in Table 1.
Summary of treatment recommendations from international and national COPD guidelines.

FEV1 ⩾50%, mMRC 0–1, and ⩽1 moderate exacerbation in the previous year.
FEV1 <50%, mMRC 2–4, and ⩾2 moderate exacerbations (or ⩾1 exacerbation leading to hospitalization) in the previous year.
Taking into account the frequency, severity, and aetiology of the exacerbations, as well as the risk of pneumonia.
BEC, blood eosinophil count; COPD, chronic obstructive pulmonary disease; FEV1, forced expiratory volume in 1 s; GOLD, Global Initiative for Chronic Obstructive Lung Disease; ICS, inhaled corticosteroids; JRS, Japanese Respiratory Society; LABA, long-acting β2-agonist; LAMA, long-acting muscarinic antagonist, mMRC, modified Medical Research Council.
Data from registries: How are patients being treated in real-life practice?
When looking at country-specific real-world data regarding the use of LAMA, LABA, and ICS, it appears that clinicians are not always adhering to the current recommendations, as illustrated by the examples below.1,7,9–11,24–40
In Germany, results from two large registry studies indicate discrepancies between real-world findings and current treatment guidelines.27–30 For example, in the COSYCONET (COPD and SYstemic consequences-COmorbidities NETwork) COPD cohort, 67.6% of patients were treated with LABA/LAMA; 28 however, when these patients were classified according to 2017 GOLD criteria (ABCD classification scheme), 66% of patients in groups A and B (low exacerbation rates) were treated with ICS, despite ICS treatment being considered inappropriate in these patients in most circumstances. 28 Conversely, there was evidence of undertreatment in groups C and D (high exacerbation rates), with many patients not treated with LAMA or LABA/LAMA as recommended. 28 Similar results have been found in another German registry, the DACCORD registry.29,30,37–40 For patients with data available at the end of the 1-year follow-up period, LAMA monotherapy or LABA/LAMA/ICS was the most common therapy despite most patients being classified as GOLD B, 29 for whom treatment with LAMA, LABA, or LABA/LAMA is recommended. 9 In a further analysis carried out after 2 years of follow-up, the majority of patients were on LAMA monotherapy (36%) or LABA/LAMA/ICS (30%). 30 Only a minority of patients (16%) were on LABA/LAMA at the end of 2 years, despite 75% of patients in DACCORD being non-exacerbators. 30
Overuse of ICS has also been reported in Spanish registries.24–26 Among patients with COPD initiating LABA/LAMA/ICS in primary care, 70% were classified as GOLD A or B. 24 These patients were on triple therapy despite 54% being non-exacerbators at baseline, and they continued on the same treatment over time regardless of disease severity. 24 In another primary care study from Spain, almost half (48%) were treated with ICS, although only 27% experienced moderate or severe exacerbations in the previous year (34% during the previous 2 years). 25
A number of observational/registry studies have also been conducted in Japan.31–34 Analysis of data from patients with COPD in the COPD Assessment in Practice (CAP) study, 65% of whom had no previous exacerbations in the last year, 31 showed that 28% of patients were treated with LABA/LAMA/ICS and 21% with LABA/LAMA. 34 When the patients in the study were classified according to GOLD 2017 criteria, 36% of patients in group A were receiving an ICS-containing treatment regimen. 32
Data from other regions, such as Latin America and Central and Eastern Europe, suggest that overuse of ICS is not unique to Germany, Spain, and Japan. In the LASSYC (Latin American Study of 24-h Symptoms in Chronic Obstructive Pulmonary Disease) study, 32.7% of GOLD A patients and 19.8% of GOLD B patients were treated with LABA/ICS, and 17.3% of GOLD A patients and 30.2% of GOLD B patients were treated with LABA/LAMA/ICS. 35 In the POPE (Phenotypes of COPD in Central and Eastern Europe) study in Central and Eastern Europe, 34% of GOLD A patients and 41.6% of GOLD B patients were prescribed ICS-containing therapies. 36
Use of LABA/LAMA as maintenance therapy: What are the treatment pathways?
There are three main pathways leading to LABA/LAMA therapy: (1) stepping up treatment from short-acting bronchodilators, LAMA monotherapy, or LABA monotherapy; (2) withdrawing ICS from LABA/LAMA/ICS or switching from LABA/ICS; and (3) LABA/LAMA as initial therapy (Figure 1).

The role of LABA/LAMA and add-on therapies in the management of COPD.
In the first pathway, guidelines recommend escalation to dual therapy if patients remain symptomatic despite short- or long-acting bronchodilator monotherapy.7–9 In the second pathway, withdrawal of ICS, either from LABA/ICS or from triple therapy (i.e. switching to LABA/LAMA), is recommended for patients without a history of frequent exacerbations and with a blood eosinophil count <300 cells/µL 41 (ICS withdrawal in patients with eosinophils ⩾300 cells/µL is more likely to be associated with the development of exacerbations). 1 In the third pathway, LABA/LAMA should be initiated if patients have a high symptom burden,1,7,9 impaired physical activity,10,42–44 or frequent/severe exacerbations (⩾2 moderate exacerbations or ⩾1 exacerbation leading to hospitalization in the past year) and blood eosinophils <300 cells/µL. 1
In patients for whom LABA/LAMA treatment is not enough, the first steps to take before changing therapy are to check patient adherence, inhaler compatibility/technique, alternative combinations of LABA/LAMA, and inhalation ability.1,45 It is important to ensure that patients are adherent and using their inhalers correctly before considering changing their medication or device. Poor adherence may be unintentional or intentional: unintentional non-adherence can often be remedied using patient education, simplification of treatment regimens, or the use of a reminder system, whereas intentional non-adherence may be more complex and challenging to address. 46 Some patients actively engage with disease management but may not receive full benefit from their medication due to incorrect inhalation technique, or other reasons beyond their control. 45 Some studies have indicated that around 50–70% of patients are not using their inhalers correctly.47,48 It is also necessary to check that patients can achieve the required peak inspiratory flow for their device.49–51
Once these factors have been checked, it is important to take into account the phenotype of the patient before considering the next steps; for example, whether the key factor is symptoms (e.g. dyspnoea) or exacerbations, and what the exacerbation phenotype is (eosinophilic or non-eosinophilic; Figure 1).
For patients with dyspnoea, it is important to look for cardiovascular disease, which is a common comorbidity in COPD. For example, in the DACCORD study, >50% of patients with COPD had cardiovascular comorbidities, with a rise in prevalence corresponding to increasing age groups. 40 For patients with exacerbations, it is imperative to recognize that not all exacerbations are the same and that different add-on therapies are required for different types of exacerbation. 52
While the majority of patients who receive LABA/LAMA plus add-on therapy are on triple therapy (LABA/LAMA/ICS), this may not be suitable for all, and there are other add-on options available for certain patient phenotypes.1,53 Tools that can help phenotype patients in order to select the most appropriate therapy to add to LABA/LAMA are described in Table 2 and considered further in relation to three distinct patient phenotypes.
What diagnostic tools can be used to phenotype patients?
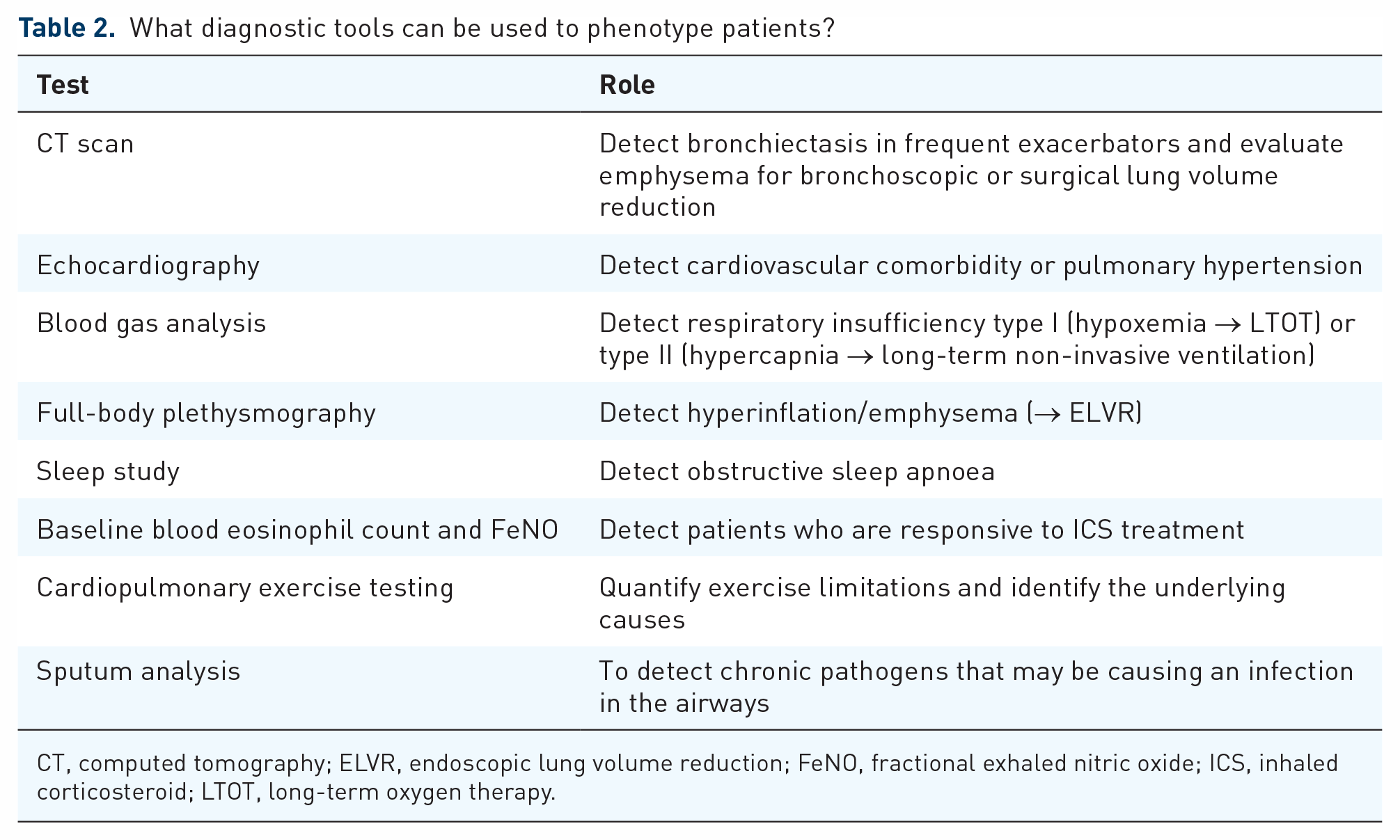
CT, computed tomography; ELVR, endoscopic lung volume reduction; FeNO, fractional exhaled nitric oxide; ICS, inhaled corticosteroid; LTOT, long-term oxygen therapy.
Phenotype 1: patients still symptomatic (but not exacerbating) despite LABA/LAMA
For patients with this phenotype, management is difficult and options are limited. In patients without a history of exacerbations, ICS has only a modest additional impact on lung function and symptoms.54,55
Key strategies for phenotype 1
It is important to evaluate and provide suitable interventions for pulmonary complications and systemic comorbidities as these can have an impact on mortality.56,57
For example, prefrailty and frailty have been reported in up to 56% and 20% of patients with COPD, respectively; 58 in addition, frailty and respiratory impairment together substantially increase the risk of death. 59 Patient-reported outcome measures such as the PROMs-D (patient-reported outcome measures for dyspnoea-related behaviour and activity limitation scale) are effective in stratifying patients for frailty, 60 which has led to their inclusion in the JRS guidelines. 11
Check inhaler technique and adherence, lung function (e.g. post-bronchodilator reversibility test to rule out the possibility of asthma phenotype).
Late/end-stage COPD: consider opiates/morphine, lung volume reduction surgery, and lung transplantation.
Assessment/management of comorbid conditions.
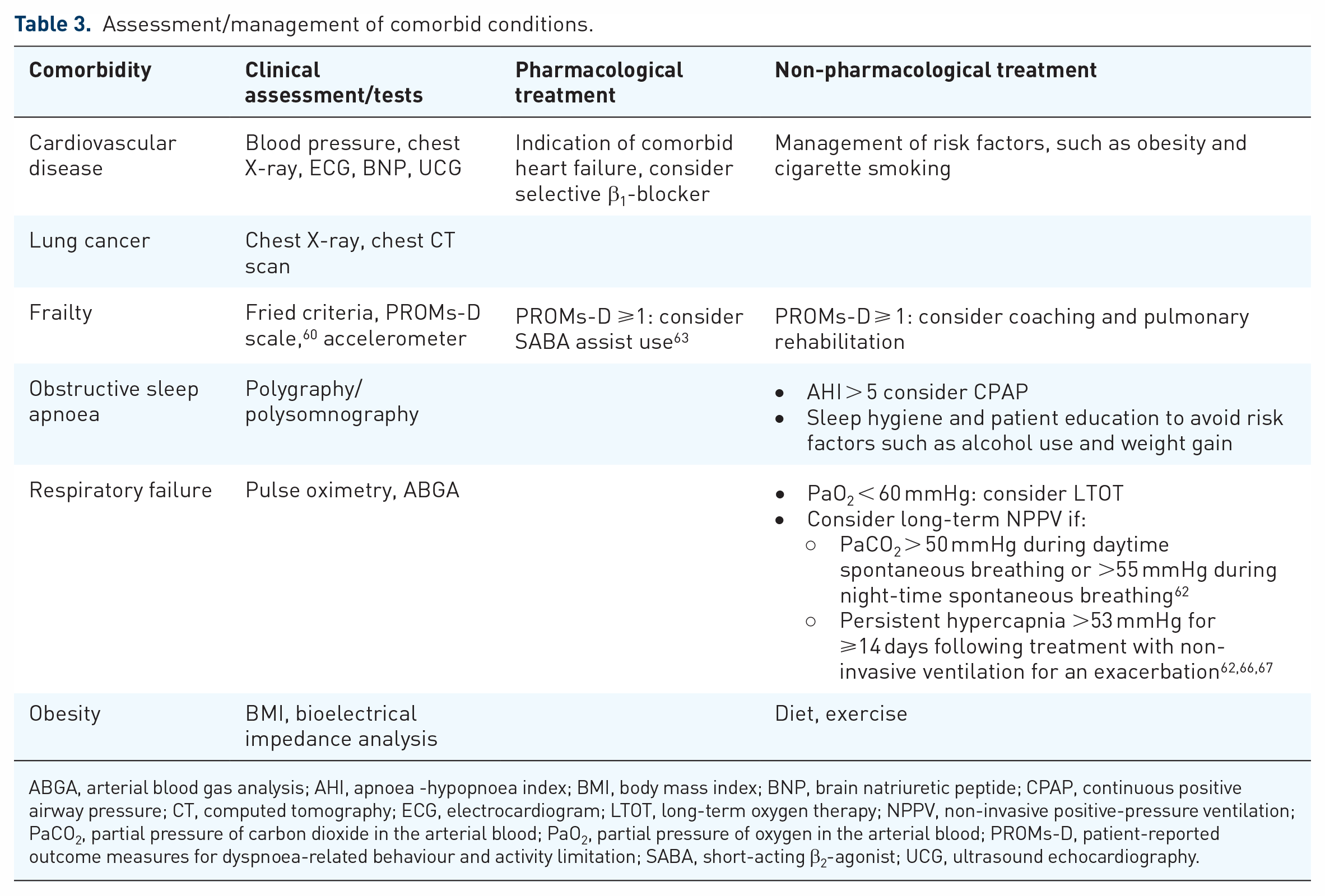
ABGA, arterial blood gas analysis; AHI, apnoea -hypopnoea index; BMI, body mass index; BNP, brain natriuretic peptide; CPAP, continuous positive airway pressure; CT, computed tomography; ECG, electrocardiogram; LTOT, long-term oxygen therapy; NPPV, non-invasive positive-pressure ventilation; PaCO2, partial pressure of carbon dioxide in the arterial blood; PaO2, partial pressure of oxygen in the arterial blood; PROMs-D, patient-reported outcome measures for dyspnoea-related behaviour and activity limitation; SABA, short-acting β2-agonist; UCG, ultrasound echocardiography.
Phenotype 2: patients still exacerbating despite LABA/LAMA and with high blood eosinophil counts
In patients with COPD who continue to exacerbate whilst prescribed LABA/LAMA, biomarkers can be used to guide therapy.61,68–70 Assessment of blood eosinophil count in combination with exacerbation history can be used to predict responsiveness to ICS, and further markers such as fractional exhaled nitric oxide (FeNO) can indicate that ICS might be beneficial. 68 Physicians should also consider allergic (atopic) asthma as an alternative diagnosis by testing for immunoglobulin E. 71 The JRS guidelines recommend measurement of this type 2 inflammation biomarker to confirm asthma-like features in patients with COPD. 11
In combination with an exacerbation history, a high blood eosinophil count is a predictive biomarker that helps to identify patients who would benefit from ICS treatment.72,73 However, the definition of high blood eosinophils has been debated,74–79 and greater variability has been reported at higher eosinophil thresholds.80,81 The GOLD 2023 report recommends that patients with a blood eosinophil count ⩾300 cells/μL and frequent or severe exacerbations (⩾2 moderate exacerbations or ⩾1 requiring hospitalization per year) should be treated with ICS in addition to LABA/LAMA. 1 The addition of ICS can also be considered at a lower eosinophil threshold (⩾100 cells/µL) if patients continue to have exacerbations despite LABA/LAMA. 1 However, these values should not be used as precise cut-offs, but rather to identify individuals with the greatest likelihood of ICS benefit. 82
Key strategies for phenotype 2
An ICS is indicated in addition to LABA/LAMA.
It is useful to interpret different biomarkers using a reference range to predict the effect of ICS (Table 4).1,68,69,83
High-dose ICS is not always necessary, and medium doses should be considered during follow-up.
Current smoking decreases FeNO levels by approximately 30% 84 and may impair ICS efficacy.85–87
Tests and treatment pathways for initiating/continuing ICS treatment.

FeNO, fractional exhaled nitric oxide; ICS, inhaled corticosteroid; LABA, long-acting β2-agonist; LAMA, long-acting muscarinic antagonist; ppb, parts per billion.
Phenotype 3: patients still exacerbating despite LABA/LAMA but without high eosinophils or concomitant asthma
Not all exacerbations are alike:88–93 around 50–70% of exacerbations are due to respiratory infections, whereas 10% are due to environmental pollution and up to 30% are of unknown aetiology. 94
ICS are not beneficial for patients with low blood eosinophil counts (i.e. a neutrophilic phenotype) who experience exacerbations. As exacerbations in these patients tend to be associated with a greater presence of bacterial pathogens, 95 detection and management of potential airway infection is key for this patient phenotype,61,96,97 as well as assessment of potential bronchiectasis and comorbidities.
Patients with purulent sputum have been found to have greater neutrophilic inflammation and a higher presence of bacterial pathogens. 95 Purulent sputum is a key sign to differentiate between bacterial causes as opposed to inflammatory, viral, or environmental causes,94,98 and serial sputum cultures help identify patients with chronic bacterial infection, especially when caused by unusual bacteria.99,100 For the detection of possible bronchiectasis, high-resolution chest computed tomography (CT) is useful. 99 The mean prevalence of bronchiectasis in patients with COPD has been reported to be 54.3% (ranging from 4% to 72% in some studies).97,101
Vaccination is also important in patients with COPD given the link between viral infections and exacerbations. The influenza vaccine is recommended for all patients with COPD, whereas pneumococcal vaccines are recommended for patients aged >65 years and for younger patients with significant comorbidities. 1
Key strategies for phenotype 3
A chest CT scan is recommended for the identification of bronchiectasis as well as other pathologies, for example, bronchiolitis.
Sputum culture should be implemented in exacerbating patients with purulent sputum or bronchiectasis present on a chest CT scan.
Roflumilast and/or macrolides can be considered for patients with frequent exacerbations and a low eosinophil count. 1
For patients with chronic bronchitis, roflumilast is recommended to reduce exacerbations, particularly in patients with a prior hospitalization for an exacerbation, greater exacerbation frequency, and higher baseline blood eosinophil counts. 102
If pathogenic microorganisms are identified in the airways, targeted antibiotic therapy should be initiated before consideration of long-term macrolide use (both roflumilast and macrolides often cause side effects and/or bacterial resistance). 103
A subgroup of particularly challenging patients with frequent bacterial exacerbations and showing colonization (e.g. with Pseudomonas aeruginosa) need eradication therapy, which can include inhaled antibiotics. 104
Long-term macrolides can be considered in patients with frequent exacerbations; 64 however, patients’ sputum should be regularly tested for bacterial resistance and should be re-evaluated if non-tuberculous mycobacterium is identified.64,65 Currently, there is no consensus regarding how long this treatment should be applied, and therefore this treatment should be initiated and monitored by a respiratory specialist. 65 Mucoactive drugs are other options to consider for this patient profile. Considering the increase in non-tuberculous mycobacterial infections, the use of erythromycin, which does not develop cross-resistance with clarithromycin, is recommended. 11
The potential limitations to this review are that whilst we did look at global and country-specific guidelines, we predominately focused on registry data from three specific countries, which may not necessarily be extrapolated to all populations. Furthermore, there are differences between the three countries, both environmentally and clinically, that may have an impact on the patient phenotypes.
Conclusions
Despite global and national recommendations that most patients with COPD should be on LABA/LAMA, real-world data suggest that countries are not adhering to this guidance and that ICS are frequently overused.
In patients currently on LABA/LAMA who continue to experience symptoms or exacerbations, evaluation of current treatment and intervention for comorbidities should be considered as early as possible. In accordance with treatment guidelines, the addition of ICS to LABA/LAMA must be considered if patients have frequent or severe exacerbations with high eosinophil counts (⩾300 cells/µL), or concomitant asthma, and if other treatable pathologies such as bronchiectasis or chronic infections have been ruled out.
Footnotes
Acknowledgements
Paul Todd, PhD, of Nucleus Holdings provided medical writing and editorial support. Nucleus also submitted the manuscript under the authorization of the authors. All statements and disclosures were reviewed and approved by the authors prior to submission.
