Abstract
Background:
Hermansky-Pudlak syndrome (HPS) is a rare disease characterized by excessive bleeding, oculocutaneous albinism, and pulmonary fibrosis (PF). However, few studies have systematically summarized the clinical characteristics of HPS.
Objectives:
To summarize the clinical characteristics, risk factors of PF, radiological and pathological presentations, and prognostic factors in patients with HPS.
Design:
A systematic review.
Data sources and methods:
We searched PubMed, Embase, Web of Science, and Scopus for eligible studies and extracted patient-level data of clinical characteristics, diagnosis of PF, radiological and pathological features, outcomes, and survival time. Categorial variables were presented as numbers (proportions) and compared using the chi-square test. Univariate and multivariate logistic regression analyses were applied to identify potential risk factors of PF. Kaplan–Meier curve, log-rank test, and Cox regression models were performed for survival analysis and prognostic factors.
Results:
A total of 186 patients from 112 eligible studies were included. Ocular albinism was associated with increased risk of PF (OR 9.08, 95% CI 2.26, 36.41, p = 0.002), while nystagmus was associated with reduced risk of PF (OR 0.11, 95% CI 0.03, 0.42, p = 0.001). Ground glass opacity (77.9%) was the most common radiological pattern, and ceroid deposition (66.7%) was the most common pathological pattern in HPS-associated PF (HPS-PF). Significant improvements in survival time were observed in patients who received an antifibrotic drug or lung transplantation (p = 0.042). However, no significant prognostic factor was identified in multivariate Cox regression analyses.
Conclusion:
Ocular albinism may serve as a risk factor, while nystagmus may serve as a protective factor of PF in HPS patients. Applying antifibrotic drugs or lung transplantation may improve the outcome and survival time of patients with HPS-PF. Future prospective studies with a large sample size were needed to verify these results and identify potential prognostic factors.
Trial registration:
This systematic review was registered in PROSPERO (CRD42024623580)
Plain language summary
This study systematically reviewed 186 patients with Hermansky-Pudlak syndrome (HPS), a rare genetic disorder that can cause pulmonary fibrosis (PF). Results showed that patients with pale eyes had a higher risk of developing PF while those with rhythmic oscillation of the eyes appeared to be at lower risk. The most common lung imaging finding was ground glass opacities, and ceroid deposition was the most frequent tissue abnormality. Treatment with anti-fibrotic drugs or lung transplantation was associated with significantly improved survival, suggesting these interventions may help HPS patients with PF live longer. However, more researches are still needed to confirm these findings and better understand factors affecting outcomes.
Introduction
Hermansky-Pudlak syndrome (HPS), which was first reported in 1959, is a rare, monogenic autosomal recessive disorder characterized by excessive bleeding, oculocutaneous albinism and potential pulmonary involvement.1,2 Prevalence of HPS was estimated to be 1–2 per million worldwide and 1 per 1800 individuals in Puerto Rico. 3 Currently, 11 genetic subtypes of HPS (HPS-1 to HPS-11) have been identified in humans, which may be associated with different clinical manifestations and long-term outcomes.2,4 However, few studies have summarized the proportion and clinical characteristics of HPS patients with different genetic subtypes.
Pulmonary fibrosis (PF) is an abnormal lung condition characterized as alveolar epithelial cell damage and excessive extracellular matrix accumulation.5,6 For patients with HPS, PF was regarded as an important source of mortality, and patients with HPS-1, HPS-2, and HPS-4 were reported to be more susceptible to PF than other subtypes. 7 Pathological examinations of lung tissue have been reported to observe foamy type II alveolar epithelial cells and ceroid-like deposition in some Hermansky-Pudlak syndrome–associated Pulmonary Fibrosis. 4 However, the radiological and pathological characteristics of HPS-associated PF (HPS-PF) were still inadequately understood. Moreover, although certain studies reported the potential effect of antifibrotic drugs and lung transplantation in treating HPS-PF, the survival gains attributable to these methodologies were still under investigation.8,9
To address the above issues, we conducted a systematic review to investigate the clinical characteristics, radiological and pathological features, and prognostic factors of HPS patients with or without PF.
Methods
This systematic review was registered in PROSPERO (CRD42024623580) and conducted following the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) guidelines. 10
Literature search
Two investigators independently searched Web of Science, Embase, Scopus, and PubMed for eligible articles from the inception of the database to December 2024. The search results were then updated in June 2025. The search strategies were listed as follows: ((((Hermanski-Pudlak Syndrome) OR (Hermanski Pudlak Syndrome)) OR (Hermansky-Pudlak Syndrome)) OR (Hermansky Pudlak Syndrome)). References of related reviews were also carefully screened to prevent omissions.
Inclusion and exclusion criteria
The inclusion criteria can be listed as follows: (1) Case reports, case series, or original articles with a clear diagnosis of PF or without PF for HPS patients. (2) Studies with available patient-level data of HPS patients. The following exclusion criteria were also applied: (1) Original articles without patient-level data. (2) Studies not published in English. (3) Review, comments, or meta-analysis. (4) Studies with unavailable full text.
Quality assessments
Quality assessments were applied via a revised scale for case studies proposed by Murad et al., with four domains and eight questions. 11 As three out of eight questions were originally designed for cases of adverse drug events, these three questions were not used in this study. The remaining five questions regarding selection, ascertainment, causality, and reporting of cases were finally applied for quality assessments.11,12
Data extraction
Two investigators independently extracted the following data for further analyses: (1) Baseline characteristics of eligible studies, including the first author, publication year, and number of cases with or without PF, respectively. (2) Baseline characteristics of cases: including age, sex, geographical location, HPS mutation, and related symptoms. (3) Radiological and pathological characteristics, including findings of X-ray, computed tomography (CT), and lung biopsy. (4) Outcome (Live or death) and survival time. We also tried to contact the corresponding authors of eligible articles for more detailed patient-level data.
Statistical analyses
We first compared the clinical characteristics of HPS patients with or without PF, with categorial variables presented as numbers (proportions) and analyzed via chi-square test. Continuous variables with normal distribution were presented as mean ± standard error, and those with skewed distribution were presented as median (quartile 1, quartile 3). Univariate and multivariate logistic regression were applied to investigate factors that may increase the risk of PF, the results of which were presented as odds ratio (OR) with 95% confidence interval (CI). Radiological and pathological characteristics of HPS patients were also summarized and presented via numbers (proportions). The survival time was measured via the Kaplan–Meier method and virtualized via a survival curve. Log-rank test was applied to evaluate the difference in survival time among different groups. Univariate and multivariate Cox regression analyses were then performed to identify potential prognostic factors, the results of which were presented as hazard ratio (HR) with 95%CI. All statistical analyses were performed via R and EmpowerStats software (EmpowerStats | Data Analysis for Biostatistics & Epidemiology), and a p < 0.05 was considered statistically significant.
Results
Study screening and baseline characteristics
A total of 5023 records were identified after a literature search, with 569 records entered into full-text screening. Eventually, 112 eligible studies including 186 patients (111 with PF and 75 without) were included in this systematic review (Figure 1). Baseline characteristics of all HPS patients are shown in Table 1. Significant differences were observed in age (p = 0.012), location of patients (p = 0.015), type of mutation (p < 0.001), cough (p < 0.001), dyspnea (p < 0.001), nystagmus (p < 0.001), excessive bleeding (p = 0.010), crackles (p < 0.001), pulmonary hypertension (p < 0.001) and clubbing fingers (p < 0.001) between HPS patients with or without PF. Baseline characteristics of included studies are shown in Table S1. Quality assessments suggested that most studies were of moderate to high quality, with a mean score of 3.72 (range, 2–5).

Selection of eligible records in this systematic review.
Baseline characteristics of eligible HPS patients with or without pulmonary fibrosis.
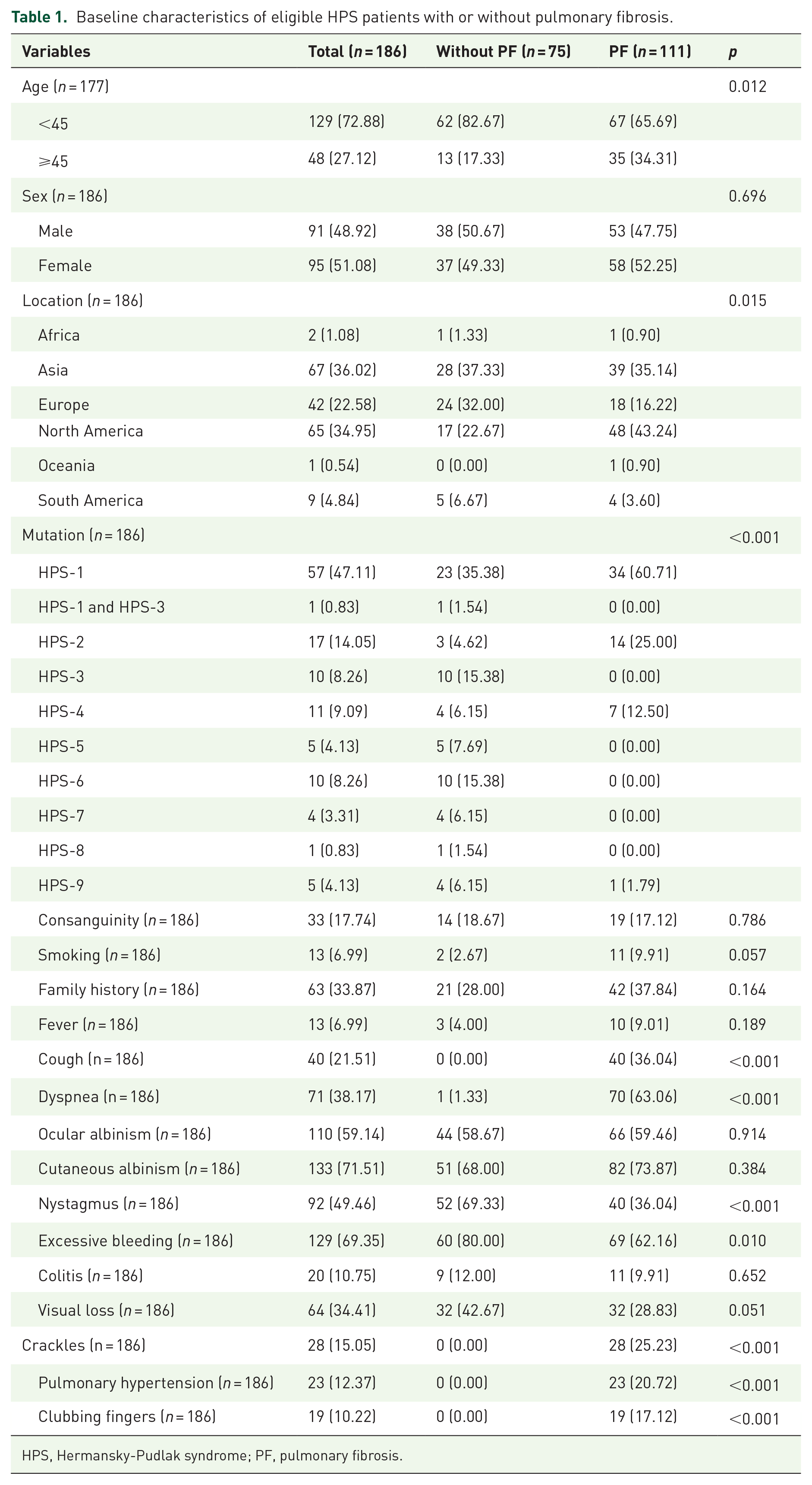
HPS, Hermansky-Pudlak syndrome; PF, pulmonary fibrosis.
Risk factors of PF in HPS patients with susceptible mutation
Most of the HPS-PF patients in this review carried a mutation of HPS-1, HPS-2, or HPS-4, which was consistent with previous evidence. However, there were also HPS patients who did not develop PF despite carrying PF-related mutations (Table 1). Therefore, univariate and multivariate logistic regression were conducted in HPS patients carrying HPS-1, HPS-2, or HPS-4 to identify potential risk factors of PF, with baseline characteristics of whom are shown in Table S2. Both univariate and multivariate analyses suggested that ocular albinism was associated with increased risk of PF (OR 9.08, 95% CI 2.26, 36.41, p = 0.002), while nystagmus was associated with reduced risk of PF (OR 0.11, 95% CI 0.03, 0.42, p = 0.001) (Table 2). Age ≥ 45 was also associated with increased risk of PF (OR 3.53, 95%CI 1.07, 11.62, p = 0.044). Subgroup analyses of HPS-1, HPS-2, and HPS-4 were not performed due to insufficient sample size.
Univariate and multivariate logistic regression analysis of fibrosis risk in patients with HPS-1, HPS-2 and HPS-4.

Significant values were shown in BOLD.
CI, confidence interval; HPS, Hermansky-Pudlak syndrome; OR, odds ratio.
Radiological and pathological characteristics of HPS-PF
Clinical presentations of chest X-ray, chest CT, and lung histopathology were summarized in Table 3. Fifty patients with PF underwent X-ray, and 80.0% reported reticulation. Seventy-two patients completed chest CT. Ground glass opacity (GGO) (77.9%) was the most commonly reported pattern, followed by reticulation (73.6%) and traction bronchiectasis (50.0%). Thirty patients reported available information on lung histopathology. Ceroid deposition was the most common presentation (66.7%), followed by infiltration of inflammatory cells (53.3%). Only a small proportion of studies reported pathological subtypes of PF. The pattern of usual interstitial pneumonia and nonspecific interstitial pneumonia occupied 16.7% and 6.7%, respectively. Radiological and pathological information of HPS patients without PF was not available.
Radiological and pathological characteristics of HPS patients with pulmonary fibrosis.

CT, Computed Tomography; GGO, Ground Glass Opacity; NSIP, Nonspecific Interstitial Pneumonia; UIP, Usual Interstitial Pneumonia.
Outcomes and survival predictors of HPS-PF
Sixty-two patients with PF provided data on clinical outcomes, with 28 alive and 34 deaths. Among these 62 patients, 36 did not receive antifibrotic treatments, 12 received antifibrotic drugs, and 14 underwent lung transplantation, with their baseline characteristics shown in Table S3. The median survival time was 48 months (Figure 2(a)), and lung transplantation and antifibrotic drugs were shown to improve the total survival time of HPS patients (p = 0.042, Figure 2(b)). Thirty-six patients with available data on all prognostic factors were included in further Cox regression analyses. Although univariate analyses suggested that cough (OR 2.77, 95%CI 1.04, 7.41, p = 0.042) and excessive bleeding (OR 3.13, 95%CI 1.17, 8.40, p = 0.023) may serve as potential survival predictors, these results were not supported in multivariate Cox regression analyses (all p > 0.05) (Table 4). Few studies reported the outcomes of HPS patients without PF. These results warranted further investigations due to the limited number of patients.

Kaplan–Meier curve of patients with HPS-PF. (a) Total survival time. (b) Survival time of patients treated without antifibrotic drug, with antifibrotic drug, and lung transplantation.
Univariate and multivariate Cox regression analyses in HPS patients with pulmonary fibrosis.
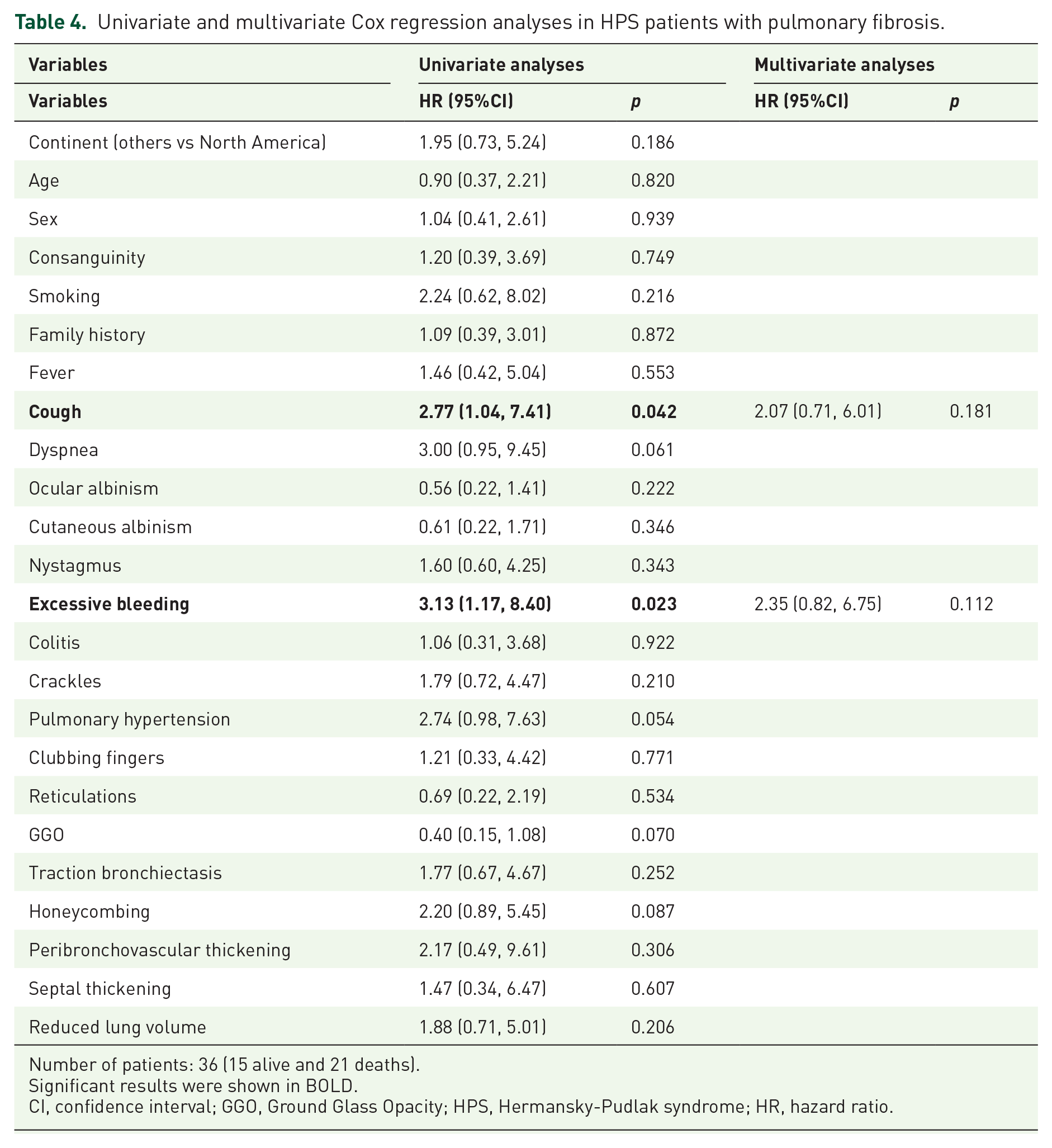
Number of patients: 36 (15 alive and 21 deaths).
Significant results were shown in BOLD.
CI, confidence interval; GGO, Ground Glass Opacity; HPS, Hermansky-Pudlak syndrome; HR, hazard ratio.
Discussion
Although accumulating cases of HPS have been reported in recent decades, the low incidence and prevalence of this disease have made it hard to evaluate its characteristics in a relatively large sample. With a total of 112 eligible studies and 186 patients, this systematic review suggested that ocular albinism was associated with increased risk of PF, while nystagmus was associated with reduced risk of PF. GGO was the most common radiological pattern, and antifibrotic treatment was beneficial for the overall survival of patients with HPS-PF. These findings may improve our understandings and help to identify more effective treatment approaches for this disease.
Previous studies showed that the molecular function of HPS genes was associated with different HPS protein-associated complexes, which may participate in endolysosomal trafficking via regulating lysosomal activities. 2 HPS-1 and HPS-4 may affect biogenesis of lysosome-related organelles complex-3 (BLOC-3), while HPS-2 may affect adaptor-related protein complex 3 (AP-3). Defects of BLOC-3 and AP-3 may disrupt the formation of lamellar bodies, thereby promoting the process of PF. The potential mechanisms linking HPS mutations (especially HPS-1, HPS-2, and HPS-4) and PF together were summarized in Figure 3. HPS mutations may inhibit the maturation of lamellar bodies, leading to abnormal accumulation of surfactants and apoptosis of alveolar type II epithelial cells, thereby increasing the risk of PF.6,13 Lysosomal and endoplasmic reticulum stress was observed in mice with HPS mutation, which was associated with apoptosis of alveolar epithelial cells. 14 Mitochondrial dysfunction was also observed in samples with HPS-1 or HPS-2 mutations.15,16 As the association between mitochondrial dysfunction and PF has been widely reported,17,18 mitochondrial dysfunction may serve as another essential driver of HPS-related PF. Excessive reactive oxygen species caused by mitochondrial dysfunction may also promote PF via fibroblast activation and pulmonary inflammation. 6 Additionally, immune dysregulation may also serve as a potential promoter of PF in patients with HPS. Elevated levels of lymphocytes and pro-inflammatory cytokines have already been reported in HPS-PF.6,19 Mechanically, type 2 innate lymphoid cells have been reported to promote the differentiation and proliferation of lung fibroblasts via amphiregulin-epidermal growth factor receptor-dependent pathways. 20 Another study reported that patients with HPS-1 aged 40 or older were associated with elevated levels of neutrophil extracellular traps and neutrophil granule proteins, showing that neutrophil-mediated inflammation may serve as a potential age-related driver of fibrotic progression in HPS-1 patients. 21 Lastly, metabolic reprogramming was also reported in patients with PF. Although an abnormal metabolic pattern was observed in macrophages of HPS patients, further studies were needed to investigate the underlying mechanisms. 6
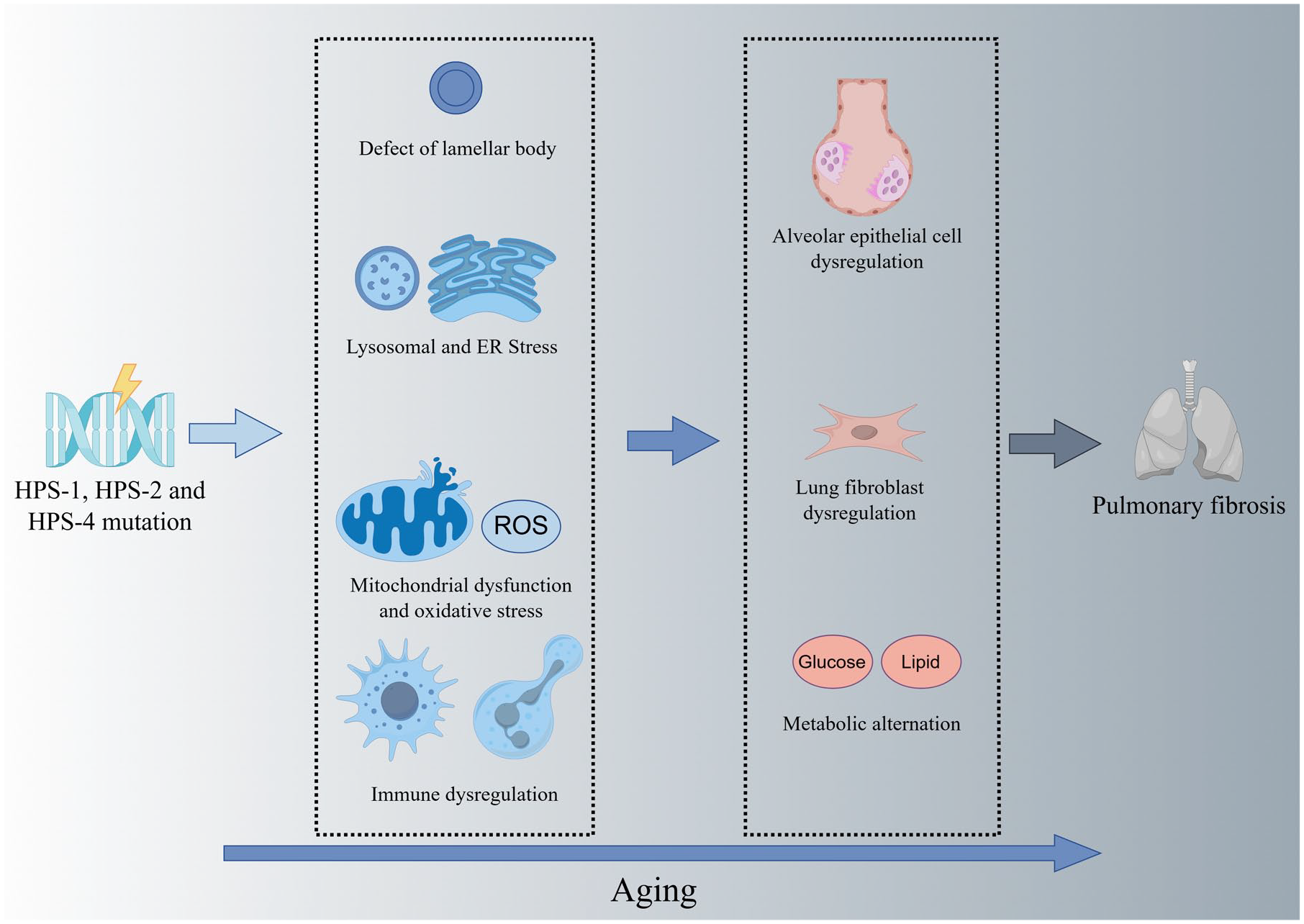
Potential mechanisms linking HPS and pulmonary fibrosis together.
Moreover, this review found that ocular albinism may be associated with increased risk of PF, while nystagmus may be associated with reduced risk of PF. Although the potential mechanisms were still unclear, these findings suggested that chest CT and pulmonary function tests may be performed for patients with ocular albinism to identify potential pulmonary complications in the early stage. A systematic physical examination was also needed for suspicious HPS patients to identify other coexisting symptoms. In addition, significant improvements in survival time were observed in patients receiving antifibrotic drugs and lung transplantations compared with other patients. This suggests that antifibrotic approaches should be performed along with symptomatic treatments when managing HPS-PF. Preparation for lung transplantation may also be considered for patients with rapidly progressive PF. Specifically, the relative scarcity of donor lungs makes patient selection crucial for prioritizing HPS cases requiring transplantation. Considering the bleeding tendency of patients with HPS-PF, procoagulant drugs may be prescribed for hemorrhage prevention. Blood or platelet transfusion should also be taken into consideration to prevent worsening anemia or bleeding. 4 Lastly, as the Cox regression analyses were conducted in a small sample size due to a lack of available data, further studies or systematic reviews with a larger sample size were still needed to explore potential prognostic factors.
This systematic review also has some limitations. First of all, the data of this study were based on reports of previous studies, which were associated with heterogeneity and potential risk of bias. 22 Second, not all studies provided sufficient pathological and follow-up details of HPS patients. Therefore, the sample size was still small when evaluating pathological features and prognostic factors, which may make our results less reliable. For example, as the Cox regression analyses only included a small number of patients, further studies were still needed to verify our results. Thirdly, studies with unavailable full tests or not in English were also excluded. These missed article sources may also be relevant to our topic.
Conclusion
In conclusion, ocular albinism may serve as a risk factor of PF, while nystagmus may serve as a protective factor of PF in HPS patients. Ground glass opacities and reticulations may serve as two common radiological patterns in patients with HPS-PF. Antifibrotic drugs and lung transplantation were two effective approaches to improve the survival outcomes of HPS-PF. Future prospective studies with a large sample size are still needed for verification of our results and identification of potential prognostic factors.
Supplemental Material
sj-docx-1-tar-10.1177_17534666251374241 – Supplemental material for Clinical characteristics and prognostic factors of Hermansky-Pudlak syndrome with or without pulmonary fibrosis: a systematic review
Supplemental material, sj-docx-1-tar-10.1177_17534666251374241 for Clinical characteristics and prognostic factors of Hermansky-Pudlak syndrome with or without pulmonary fibrosis: a systematic review by Dongru Du, Ting Yang, Huajing Wan and Fengming Luo in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-docx-2-tar-10.1177_17534666251374241 – Supplemental material for Clinical characteristics and prognostic factors of Hermansky-Pudlak syndrome with or without pulmonary fibrosis: a systematic review
Supplemental material, sj-docx-2-tar-10.1177_17534666251374241 for Clinical characteristics and prognostic factors of Hermansky-Pudlak syndrome with or without pulmonary fibrosis: a systematic review by Dongru Du, Ting Yang, Huajing Wan and Fengming Luo in Therapeutic Advances in Respiratory Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
