Abstract
Background:
Pulmonary hypertension (PH) is a complex disorder associated with various underlying conditions, including cardiac and respiratory diseases. PH is classified into five groups based on etiology, disease mechanisms, hemodynamic data, and treatment options. Preliminary data suggest that sodium-glucose cotransport-2 inhibitors (SGLT2-I), known for their benefits in chronic kidney disease, heart failure, and type-2 diabetes mellitus, may have therapeutic implications in PH through their metabolic effects, which include reducing aerobic glycolysis, improving mitochondrial function, and enhancing fatty acid oxidation.
Objective:
This study aimed to evaluate the clinical effects of SGLT2-I in PH by analyzing a large multicenter database of medical records from the TriNetX Network.
Design:
The cohort included adult patients with PH diagnosed between January 1, 2012, and January 1, 2023, classified by PH group and treatment with SGLT2-I. Propensity score matching (PSM) was used to balance baseline characteristics between the SGLT2-I and non-SGLT2-I groups.
Methods:
The primary endpoint was a composite of all-cause mortality, RHF, and hospital admissions over 365 days. Secondary endpoints included the individual components of the primary endpoint, intubations, RHF incidence, IV diuretic use, and NT-Pro-BNP levels. PSM was used to adjust for baseline differences between cohorts.
Results:
A total of 771,490 patients with PH were identified, with 58,303 treated with SGLT2-I. After PSM, each cohort of treated and untreated patients included 58,302 patients. Patients treated with SGLT inhibitors had a significant reduction in the primary composite endpoint (HR 0.71, 95% CI: 0.707–0.729). Secondary outcomes, including all-cause mortality, hospitalization, and the number of intubations, were also significantly lower in patients treated with SGLT-2 inhibitors. Beneficial effects of SGLT2-I were observed across all PH groups.
Conclusion:
This study demonstrates that SGLT2-I may be clinically beneficial in patients with PH by reducing all-cause mortality, RHF, and hospital admissions. Our findings support the role of SGLT2-I as a therapeutic option in PH and provide support for future randomized controlled trials using this treatment.
Keywords
Key findings
Treatment with SGLT2-I significantly reduced the composite outcome of all-cause mortality, right heart failure (RHF), and all-cause hospital admission in patients with pulmonary hypertension (PH) over a 365-day period. In addition, SGLT2-I improved secondary outcomes, including lower all-cause mortality, hospitalization, and intubation rates, as well as a lower rate of RHF and need for IV diuretics.
What is known?
Pulmonary hypertension (PH) is a complex disorder linked to various cardiac and respiratory diseases, classified into five groups. SGLT2-I are currently used in the treatment of chronic kidney disease, heart failure, and type-2 diabetes mellitus, where they slow disease progression, reduce hospitalizations, and lower mortality. Early studies suggest that SGLT2-I may have favorable effects on pulmonary hemodynamics in animal models of PH.
What is new?
Our research provides the first large-scale clinical evidence that SGLT2-I significantly reduce all-cause mortality, right heart failure (RHF), and all-cause admissions in patients with PH. The beneficial effects of SGLT2-I are consistent across all PH groups, prompting randomized controlled trials in various PH groups.
Implication
SGLT2-I could significantly improve clinical outcomes in patients with pulmonary hypertension (PH), offering benefits across all PH groups. This positions SGLT2-I as a potential new therapeutic option for PH that, for the first time, targets metabolic pathways.
What should change now?
Clinicians should consider risks/benefits of using SGLT2-I in PH patients until randomized controlled trials confirm our promising findings.
Introduction
Pulmonary hypertension (PH) is a disorder associated with various medical conditions, including cardiac and respiratory diseases. 1 A mean pulmonary arterial pressure (mPAP) of more than 20 mmHg is diagnostic of PH. 2 PH is classified into five groups depending on etiology, disease mechanism, hemodynamic data, and treatment options. 2 Group 1 PH is characterized by specific conditions that increase pulmonary vascular resistance (PVR) in the context of normal left ventricular diastolic pressure.1,2 Group 2 PH is the most common type of PH and is associated with elevated left ventricular diastolic pressure. 3 Group 3 PH is related to chronic lung disease or hypoxemia.4,5 Group 4 PH is predominantly attributed to chronic thromboembolic PH (CTEPH)6 –8 and group 5 PH refers to PH due to unclear or multifactorial mechanisms.9 –12
The sodium-glucose cotransport-2 inhibitors (SGLT2-I) are medications that lower glycemia by reducing glucose absorption from the kidneys. SGLT2-I are currently used in chronic kidney disease (CKD), left heart failure, and type-2 diabetes mellitus.13 –17 In these conditions, SGLT2-I slowed the progression of CKD, reduced hospitalization for heart failure, and decreased the risk of death, respectively. Therefore, SGLT2-I are part of the corresponding guidelines for the management of these diseases.18 –23 SGLT2-I may also have therapeutic implications in PH. PH has been associated with characteristic metabolic abnormalities, which include a shift toward aerobic glycolysis, mitochondrial dysfunction, disrupted fatty acid oxidation, and altered tricarboxylic acid (TCA) cycle; disorders that contribute to vascular remodeling and disease progression.24 –30 SGLT2-I exert metabolic effects that can counteract these metabolic disruptions, including a decrease in aerobic glycolysis, improvement in mitochondrial function, enhancement in fatty acid oxidation, and optimization of the TCA cycle.1,30 –34
In fact, SGLT2-I reduce vascular inflammation and oxidative stress and reverse vascular dysfunction.35 –37 SGLT2-I may decrease vascular remodeling and pulmonary artery smooth muscle (PASMC) proliferation by inducing apoptosis.38 –44 Up to this date, there are no clinical studies testing the effect of SGLT2-I in patients with PH. Based on the apparent beneficial effects described above, we hypothesize that SGLT2-I are effective metabolic modulators in PH, improving clinical outcomes. We, therefore, explore the clinical effects of SGLT2-I in patients with PH by analyzing a multicenter health research network (TriNetX Network) that includes a large number of medical records.
Methods
Data source
Data used in this study were obtained from the TriNetX research network, which contains information from the electronic health records of approximately 90 million patients from 72 health care organizations in North America, Europe, Asia, and other regions. This platform only has aggregate, deidentified records per the standard defined in section §164.514(a) of the Health Insurance Portability and Accountability Act Privacy Rule.
Study population and design
The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 45 We included patients 18 years or older with PH between January 1st 2012 and January 1st 2023. For the SGLT2-I group, we chose patients who were started on SGLT2-I after PH diagnosis, regardless of which type of SGLT2-I (i.e., canagliflozin, dapagliflozin, and empagliflozin) or dose was prescribed. We cluster patients based on the PH groups based on the 6th World Symposium in PH. 46 In the SGLT2-I group, time zero is defined as the initiation of SGLT2-I, provided that the patient had been diagnosed with pulmonary hypertension prior to starting the medication. For the non-SGLT2-I group, time zero is defined as the diagnosis of PH. Propensity score matching (PSM) was performed to reduce bias from uneven distribution of observed baseline characteristics by accounting for covariates that predict receiving the treatment.
Study endpoints
The primary end point was a composite outcome of all-cause mortality, diagnosis of RHF, and all-cause hospital admissions. Secondary outcomes were all-cause mortality, hospitalization, number of endotracheal intubations, diagnosis of acute RHF, diagnosis of RHF, need for IV diuretic, and plasma level of N-terminal pro-brain natriuretic peptide (NT pro-BNP). Primary and secondary end points were defined according to ICD 10 codes or whether IV diuretics were prescribed according to information reported in the medical records. Primary and secondary outcomes were analyzed over a 365-day period.
Statistical analysis
The entire cohort was divided into two groups based on the treatment with SGLT2-I. In addition, we created four cohorts based on the PH group patients belong (PH groups 1–4) based on the 6th World Symposium in PH, 46 and then we separated these patients based on whether they were prescribed SGLT2-I. Patients with group 5 PH were not included as a PH subgroup because of the lower number of subjects and the heterogeneity of their medical conditions. Continuous variables are presented as mean ± standard deviation (SD) and were compared between the cohorts using a t-test. Categorical variables are reported as n (%) and compared using the Chi-square (X2) test. To control for unbalanced baseline characteristics between patient cohorts, 1:1 PSM was performed using a built-in algorithm that applies the greedy nearest-neighbor method with a caliper of 0.1 pooled SDs. Any observed characteristic with a standardized mean difference of < 0.1 between groups was considered well matched. Odds ratios were adjusted for demographics, medications, and comorbidities, which are mentioned in Table 1. Adjusted odds ratios (ORs) with 95% confidence intervals (CIs) were calculated for primary and secondary outcomes. Survival analysis was conducted using Kaplan–Meier curves, and log-rank tests were used to compare the two groups. Statistical significance was defined as a two-sided p-value of < 0.05. All statistical analyses were performed using the TriNetX online platform, which uses the R survival package version 3.2-3.
Baseline patient characteristics before and after propensity score matching.
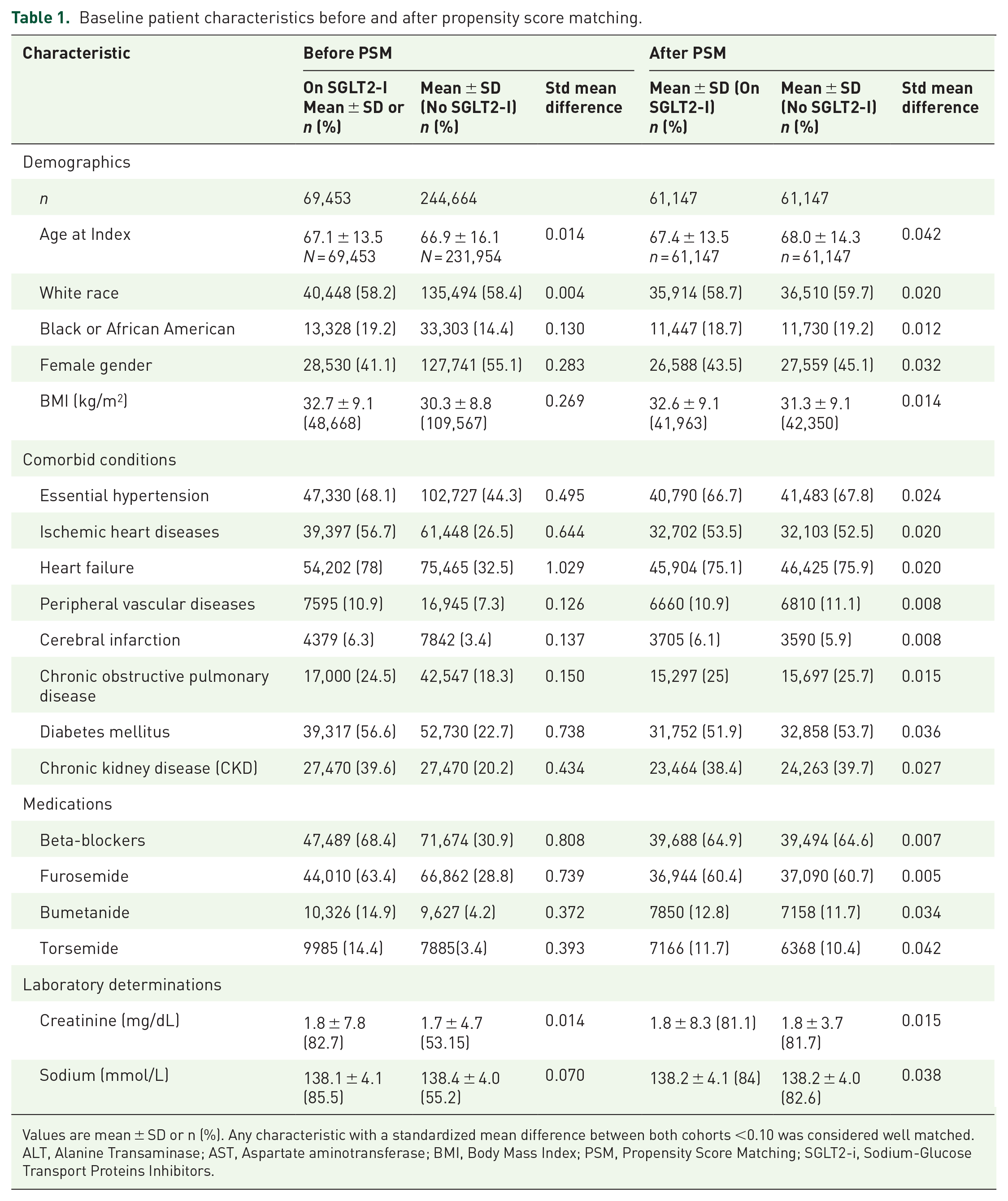
Values are mean ± SD or n (%). Any characteristic with a standardized mean difference between both cohorts <0.10 was considered well matched.
ALT, Alanine Transaminase; AST, Aspartate aminotransferase; BMI, Body Mass Index; PSM, Propensity Score Matching; SGLT2-i, Sodium-Glucose Transport Proteins Inhibitors.
Results
Study cohorts
A total of 771,490 patients diagnosed with PH were identified, among whom 58,303 received treatment with sodium-glucose co-transporter 2 inhibitors (SGLT2-I). After PSM, 58,302 patient pairs were matched between those treated and those untreated with SGLT2-I (Table 1). Among the identified PH subgroups, 72,211 patients were classified as Group 1 PH, with 2029 receiving SGLT2-I and 2021 matched for comparison (Table 2). In Group 2 PH, 56,642 patients were identified, with 8342 receiving SGLT2-I and an equal number matched (Supplemental File “PHT_SGLT_2_Grp_2”). A total of 34,905 patients were classified as Group 3 PH, with 2926 treated with SGLT2-I and matched (Supplemental File “PHT_SGLT_2_Grp_3”). Lastly, 11,949 patients were identified with Group 4 PH, of whom 767 were treated with SGLT2-I and 763 were matched (Supplemental File “PHT_SGLT_2_Grp_4”).
Baseline patient characteristics before and after PSM for PH group 1.

Values are mean ± SD or n (%). Any characteristic with a standardized mean difference between both cohorts <0.10 was considered well matched.
ALT, Alanine Transaminase; AST, Aspartate aminotransferase; BMI, Body Mass Index; PSM, Propensity Score Matching; SGLT2-i, Sodium-Glucose Transport Proteins Inhibitors.
Primary outcome
Over a 1-year follow-up period after PH diagnosis, treatment with SGLT2-I was associated with a significant reduction in the composite outcome of all-cause mortality, RHF, and all-cause hospitalization compared to those not receiving SGLT2-I (OR 0.68, 95% CI: 0.66–0.70, p < 0.001; Table 3). When analyzed by PH subgroups, patients with Group 1 PH had an OR of 0.71 (95% CI: 0.68–0.74), Group 2 PH had an OR of 0.65 (95% CI: 0.63–0.67), Group 3 PH had an OR of 0.70 (95% CI: 0.67–0.73), and Group 4 PH had an OR of 0.76 (95% CI: 0.72–0.80) (Table 4 and Supplemental Tables 1–3).
Primary and secondary outcomes for the entire cohort.

Values are % unless otherwise indicated. Outcomes compared within a year of starting SGLT2-I.
Acute RHF, Acute Right Heart Failure; IV diuretics, Intravenous Diuretics; OR, Odds Ratio; PH, Pulmonary Hypertension; RHF, Right Heart Failure; SGLT2-i, Sodium-glucose transport proteins inhibitors.
Primary and secondary outcomes for PH Group 1.

Values are % unless otherwise indicated. Outcomes compared within a year of starting SGLT2-I.
Acute RHF, Acute Right Heart Failure; IV diuretics, Intravenous Diuretics; OR, Odds Ratio; PH, Pulmonary Hypertension; RHF, Right Heart Failure; SGLT2-i, Sodium-Glucose Transport Proteins Inhibitors.
Secondary outcomes
Patients treated with SGLT2-I demonstrated significantly lower odds of mortality, hospitalization, and intubation. A total of 7432 deaths occurred in the SGLT2-I group compared to 20,086 in the non-SGLT2-I group, with an OR of 0.49 (95% CI: 0.47–0.51, p < 0.001). The odds of all-cause hospitalization were significantly lower among SGLT2-I users, with an OR of 0.72 (95% CI: 0.70–0.75, p < 0.001). The odds of requiring mechanical ventilation were reduced in the SGLT2-I group (OR 0.45, 95% CI: 0.43–0.48, p < 0.001). In addition, patients treated with SGLT2-I had lower odds of RHF and IV diuretic use (Table 3).
When analyzed by PH subgroups, SGLT2-I use significantly lowered the risk of mortality in each group separately (Figure 1 and Supplemental Figures 2–4). Patients with Group 1 PH treated with SGLT2-I had a significant reduction in intubation (OR 0.46, 95% CI: 0.43–0.49, p < 0.001). In Group 2 PH, all-cause hospitalization was reduced (OR 0.68, 95% CI: 0.65–0.71, p < 0.001), along with a lower RHF rate (OR 0.63, 95% CI: 0.61–0.66, p < 0.001; Table 1). Patients in Group 3 PH exhibited a significant reduction in acute RHF (OR 0.66, 95% CI: 0.63–0.69, p < 0.001) and intubation risk (OR 0.48, 95% CI: 0.45–0.51, p < 0.001; Table 2). In Group 4 PH, RHF-related hospitalizations were lower in the SGLT2-I group (OR 0.72, 95% CI: 0.69–0.75, p < 0.001), as was the need for intubation (OR 0.74, 95% CI: 0.71–0.78, p < 0.001; Table 4 and Supplemental Tables 1–3).

Kaplan–Meier curve comparing mortality for group 1 Pulmonary Hypertension between two cohorts: SGLT2-I users versus SGLT2-I Non-users.
NT-proBNP levels were significantly lower in patients with PH who were on SGLT2 inhibitors compared to those who were not (Supplemental Table 4). This effect was notable in Group 1, Group 2, and Group 3 showing statistical significance (p < 0.001, p < 0.001, p = 0.004). Group 4 had a trend toward lower levels but did not reach statistical significance (p = 0.052).
Sensitivity analyses
A sensitivity analysis was conducted in patients with Group 1 PH to control for confounding effects of PH-specific therapies, including nifedipine, diltiazem, amlodipine, sildenafil, tadalafil, bosentan, macitentan, and prostacyclins (Table 3). This analysis confirmed that SGLT2-I use remained significantly associated with a reduction in the composite primary outcome (OR 0.69, 95% CI: 0.67–0.72, p < 0.001). Mortality remained significantly lower (OR 0.52, 95% CI: 0.50–0.55, p < 0.001), as did hospitalization (OR 0.73, 95% CI: 0.71–0.76, p < 0.001) and intubation (OR 0.48, 95% CI: 0.45–0.51, p < 0.001; Table 4).
Discussion
Our study, which used the TriNetX research network, which contains clinical information from many PH patients, revealed a significant reduction in the combined outcome of all-cause mortality, RHF, and all-cause hospital admissions in patients with PH treated with SGLT2-I, irrespective of their PH group. Also, we observed a reduction in all-cause hospitalization as well as mortality and rates of endotracheal intubation in PH patients treated with SGLT2-I regardless of the PH group. Treatment with PH therapies does not negate the positive effect of SGLT2-I in patients with Group 1 PH.
The results of our study align with the favorable effect of SGLT2-I previously reported in animal models of PH. Studies performed in monocrotaline rat models of PH showed that SGLT2-I decreased pulmonary arterial pressure, right ventricular pressure, right ventricular myocardial fibrosis, and hypertrophy.44 –49 Empagliflozin decreased collagen deposition in the right ventricle and lessened the muscularization of the pulmonary artery. 49 These effects suggest that SGLT2-I may have favorable effects on critical cellular processes that impact pulmonary hemodynamics and the progression of the disease.
Favorable results of using SGLT2-I were also seen in randomized control studies that included patients with group 2 PH, that is, PH associated with left heart disease. The EMBRACE-HF trial showed that empagliflozin reduced pulmonary arterial pressure in patients with left heart disease. 50 In another placebo-controlled trial, patients with group 2 PH were randomized to receive dapagliflozin + sacubitril/valsartan or sacubitril/ valsartan alone. Dapagliflozin plus sacubitril/valsartan improved left heart function and exercise tolerance as well as reduced mean pulmonary artery pressures when compared to sacubitril/valsartan without dapagliflozin. 51 Similarly, our study showed better clinical outcomes in group 2 PH patients when treated with SGLT2-I.
Limitations
This study has several limitations inherent to observational research. There may be residual confounding factors, such as social determinants of health, that were not measured or controlled for. While propensity score matching helps reduce bias, it does not fully eliminate it. In addition, the data were obtained retrospectively from electronic medical records, which are prone to miscoding and documentation errors. The database used tracks outcomes only within participating healthcare organizations, meaning outcomes may be missing for patients transferred to non-participating institutions. Another key limitation is the inability to risk-stratify patients within each PH group due to the lack of granular data. Despite these limitations, it is likely that they equally impacted both groups in the study. In addition, the potential for incorrect CPT/ICD codes, missing data, and inadequate variable capture could introduce bias. Variability in data quality across institutions and the reliance on ICD codes alone may limit the ability to capture clinical nuances, affecting the accuracy and generalizability of the findings. Nonetheless, despite these limitations, our study is the first to demonstrate reductions in critical clinical outcomes, including all-cause mortality, hospitalization, intubation rates, incidence of acute RHF, RHF rates, and the need for intravenous diuretic use.
Conclusion
In conclusion, this retrospective, propensity score-matched analysis suggests that SGLT2-I PH showed beneficial effects in PH patients, by demonstrating a reduction in the combined primary endpoint of all-cause mortality, RHF, and all-cause admission. In addition, our analysis showed a reduction in all-cause mortality, hospitalizations, rate of intubations, the incidence of acute RHF, rate of RHF, need for IV diuretic use, and plasma level of NT-Pro-BNP. These findings support future randomized controlled studies using SGLT2-I in patients with various groups of PH.
Supplemental Material
sj-doc-1-tar-10.1177_17534666251351443 – Supplemental material for Effects of sodium-glucose cotransport-2 inhibitors treatment in patients with pulmonary hypertension
Supplemental material, sj-doc-1-tar-10.1177_17534666251351443 for Effects of sodium-glucose cotransport-2 inhibitors treatment in patients with pulmonary hypertension by Mohammed Obeidat, Aravinthan Vignarajah, Rashid Abdel-Razeq, Ala’eddien Nathir, Nishanthi A. Vigneswaramoorthy and Adriano R. Tonelli in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-docx-2-tar-10.1177_17534666251351443 – Supplemental material for Effects of sodium-glucose cotransport-2 inhibitors treatment in patients with pulmonary hypertension
Supplemental material, sj-docx-2-tar-10.1177_17534666251351443 for Effects of sodium-glucose cotransport-2 inhibitors treatment in patients with pulmonary hypertension by Mohammed Obeidat, Aravinthan Vignarajah, Rashid Abdel-Razeq, Ala’eddien Nathir, Nishanthi A. Vigneswaramoorthy and Adriano R. Tonelli in Therapeutic Advances in Respiratory Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
