Abstract
Background:
Acute exacerbations of bronchiectasis (AEB) are frequently caused by bacterial and/or viral infections. Rapid multiplex polymerase chain reaction (PCR) panels in respiratory specimens have significantly advanced microbial evaluation in patients with pneumonia. However, their clinical utility in patients with AEB remains unknown.
Objectives:
To investigate the microbial characteristics of AEB using FilmArray Pneumonia plus Panel (FAPP) and explore its clinical impact in a real-world setting.
Design:
A cross-sectional study.
Methods:
Spontaneous sputum samples of patients hospitalized for AEB were tested using FAPP in addition to standard-of-care testing. Microbial characteristics of AEB and the clinical impact of FAPP were evaluated.
Results:
Among 83 patients, FAPP detected ⩾1 bacterial pathogen(s) in 68 samples (81.9%), identifying 101 bacteria, with high abundance (106 to ⩾107 copies/ml) observed in 48 patients (57.8%). The most commonly detected bacteria were Pseudomonas aeruginosa (Pa) (37/83, 44.6%), Staphylococcus aureus (21/83, 25.3%), and Haemophilus influenzae (13/83, 15.7%). Respiratory viruses were identified in 21 patients (25.3), with Influenza A and Respiratory syncytial virus being the most common. Culture detected bacteria in significantly fewer samples (n = 25 [30.1%], p < 0.001). FAPP demonstrated 100% positive percent agreement and negative predictive value for all cultured bacteria, except for Corynebacterium striatum (n = 2), which was not included in the panel. FAPP shortened the time to bacterial results (mean: 10.8 h vs 70.8 h by culture, p < 0.001), and led to antimicrobial changes in 25 patients (30.1%) before the culture results were available. In multivariate analysis, chronic Pa infection (odds ratio (OR) 14.71), underweight status (OR 5.84), and cystic bronchiectasis (OR 5.26) were independent predictors of Pa detection by FAPP.
Conclusion:
The sputum multiplex PCR panel (FAPP) enables rapid identification of bacterial and viral pathogens in AEB, supporting early antimicrobial decision-making. Our findings highlight the potential utility of sputum multiplex PCR panels in improving the management of bronchiectasis exacerbations.
Plain language summary
Flare-ups of bronchiectasis, known as acute exacerbations of bronchiectasis (AEB), are often triggered by bacterial or viral infections. However, traditional methods struggle to quickly and accurately identify the specific pathogens responsible. This study assessed the use of the Filmarray Pneumonia plus Panel (FAPP) —a fast, advanced test that detects multiple bacteria and viruses at once—to diagnose infections in AEB patients in a real-world hospital setting. We tested sputum samples from 83 hospitalized AEB patients using both FAPP and conventional culture methods. FAPP identified bacteria in 82% of cases, compared to just 30% with conventional culture. It also detected respiratory viruses in 25% of cases. Common bacteria included Pseudomonas aeruginosa (45%), Staphylococcus aureus (25%), and Haemophilus influenzae (16%), while Influenza A and Respiratory syncytial virus were the most frequent viruses. Results from FAPP were faster (10.8 hours versus 70.8 hours for culture) and led to earlier adjustments in antibiotic treatment for 30% of patients before standard culture results were available. Factors associated with Pseudomonas aeruginosa detection included chronic infection by the same organism, being underweight, and cystic bronchiectasis. Our findings suggest that this advanced sputum test helps quickly identify bacterial and viral infections in AEB patients, allowing doctors to make faster and more informed treatment decisions. This highlights its potential to improve how bronchiectasis flare-ups are managed in hospitals.
Introduction
Non-cystic fibrosis bronchiectasis (hereafter referred to as bronchiectasis) is a chronic respiratory disease characterized by abnormal dilatation of bronchi, recurrent respiratory infections, and exacerbations. 1 Acute exacerbations of bronchiectasis (AEB) are significant events in the natural course of the disease. 2 Frequent exacerbations are associated with worse outcomes. 3 Exacerbations that require hospitalization are severe events and are recognized as major endpoints in clinical trials.4,5 Microbiological assessment is crucial in AEB. International guidelines suggest obtaining sputum for culture and sensitivity testing prior to commencing antibiotics. 6 However, traditional culture-based methods are time-consuming and have low positivity rates. Delays in providing appropriate pathogen-specific antimicrobial treatment are associated with poor outcomes in lower respiratory tract infections. 7 Since Pseudomonas aeruginosa (Pa) is frequently involved in AEB, some guidelines suggest the empirical use of anti-pseudomonal antibiotics (e.g., piperacillin-tazobactam) before culture results are available. 8 This practice raises concerns about the development of drug resistance. Recent data show that multidrug resistant (MDR) bacterial infections occur in 20% of AEB cases, 9 resulting in higher in-hospital and 3-year mortality rates. 10 Thus, there is an urgent need for rapid and accurate microbiological testing to guide antimicrobial use and minimize MDR infections in bronchiectasis patients. Recent advancements in molecular diagnostics enable rapid identification of causative pathogens of pneumonia in respiratory samples. The BioFire FilmArray Pneumonia plus Panel (FAPP; BioMérieux) is a multiplex polymerase chain reaction (PCR) assay that tests respiratory specimens for bacteria, viruses, and antimicrobial resistance markers. 11 It overcomes the limitations of single and narrow-target detection in earlier rapid diagnostics (e.g., urine pneumococcal antigen) and demonstrates higher sensitivity than conventional cultures in detecting bacterial pathogens.12,13 However, the performance and clinical impact of FAPP in AEB remains unknown.
Our hypothesis is that sputum FAPP enhances the detection of bacterial and viral pathogens and improves clinical management in patients with AEB. This study aims to describe the microbial characteristics of AEB using sputum FAPP and compare them with those of standard-of-care testing. In addition, the clinical impact of FAPP and risk factors for Pa detection in AEB were evaluated.
Methods
Study design and subjects
We performed a cross-sectional study of prospectively recruited, consecutive adult patients with bronchiectasis admitted to Pamela Youde Nethersole Eastern Hospital (PYNEH, an 1829-bed acute hospital and tertiary referral center), Hong Kong, due to an acute exacerbation between May 2022 and February 2024. Patients with no informed consent, age <18 years, pregnancy, known cystic fibrosis, bronchiectasis not being the main or co-dominant respiratory disease, traction bronchiectasis due to interstitial lung disease, or prior lung transplantation for bronchiectasis were excluded.
Bronchiectasis was defined according to international consensus recommendations, by the presence of cough, shortness of breath, chronic sputum production, recurrent respiratory infection, or a combination thereof, with computed tomography appearance of bronchiectasis, as indicated by bronchoarterial ratio >1, and/or lack of tapering, and/or airway visibility within 1 cm of the pleural surface. 14 AEB was defined by a change in bronchiectasis treatment usually involving the addition of antibiotic, with deterioration in three or more of the following key symptoms for at least 48 h: cough; increase in sputum volume and/or changes in consistency; sputum purulence; breathlessness and/or exercise intolerance; fatigue and/or malaise; and hemoptysis. 15 This study was reported according to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 16
Microbiological evaluation
Upon admission, each participant provided two consecutive expectorated sputum samples in sterile containers after deep coughing. The first sample was sent to the microbiology laboratory immediately for Gram stain microscopy and culture (standard practice), while the second sample was sent to the molecular laboratory immediately for FAPP. Sputum FAPP analysis was performed for each participant, 17 and the results were blinded to the staff performing sputum culture.
For sputum culture, specimens with a Gram stain showing ⩾25 squamous epithelial cells (SEC) per low-power field (LPF) were considered contaminated, and culture would not be performed. 18 All sputum samples with <25 SEC per LPF were cultured. The quality of cultured sputum was assessed using the Bartlett criteria: classifying into “good quality” if Bartlett score ⩾1, and “moderate quality” if Bartlett score <1.19,20 (Supplemental Material) Following Gram-stained smear examination, specimens were inoculated onto Blood, McConkey, and Chocolate agar and incubated at 37°C in 5% CO2 for 48 h. Colonies of suspected pathogens were identified using MALDI-TOF, with antibiotic susceptibility testing done by the Sensititre® MIC system (Thermo Scientific). Sputum culture was semiquantitative, with grading based on Clinical Microbiology Procedures Handbook (Fifth Edition) 21 : “Scanty”: at least one to five colonies, up to the first quadrant, ignoring a few colonies in the second quadrant; “Moderate”: up to second quadrant, ignoring a few colonies in the next quadrant; “Heavy”: up to third or fourth quadrant.
Regarding FAPP, the testing service operates daily from 9 am to 5 pm. Specimens are processed upon receipt, provided that staff are available. If immediate processing is not possible, specimens will be stored at 4°C, as per the package insert. The FAPP targets 27 pathogens (15 common bacteria, 3 atypical bacteria, and 9 viruses) and 7 antimicrobial resistance genes (Table 1). The relative abundance of standard bacterial organisms was reported as 104, 105, 106, or ⩾107 genomic copies per milliliter (ml). Bacteria detected at 106 to ⩾107 copies/ml were classified as “high abundance,” while those at 104 to 105 copies/ml were considered “low abundance.” 13
Microorganisms and antimicrobial resistance genes detected by FilmArray Pneumonia plus Panel.

Nasopharyngeal swabs (NPS) for viral study were performed at the discretion of the treating physician. The NPS multiplex PCR assay targeted Adenovirus DNA, parainfluenza virus (types 1–4) RNA, respiratory syncytial virus RNA, influenza A–C virus RNA, human metapneumovirus RNA, Human coronavirus OC43 and 229E RNA, and enterovirus/rhinovirus RNA. NPS and sputum samples were collected simultaneously. All microbial results were provided to the treating physician upon availability.
Clinical assessment and data collection
Baseline demographics and clinical characteristics were recorded at recruitment, including bronchiectasis etiology, chronic bronchial infection (⩾2 respiratory isolates of the same pathogen within a year, ⩾3 months apart), 22 exacerbation frequency, AEB-related hospital admissions, FEV1 (% predicted), and body mass index (underweight: <18.5 kg/m2). 23 Comorbidities were summarized using the Charlson comorbidity index. 24 Disease severity was estimated using the Bronchiectasis Severity Index 25 and FACED score (FEV1, Age, Chronic colonization, Extension, and Dyspnea). 26
Study outcomes
The primary outcome of the study was the prevalence of various microbial pathogens detected in sputum compared to standard-of-care methods in AEB. Secondary outcomes included the time to microbial results by FAPP versus standard-of-care, the rate of antibiotic adjustments following FAPP results, and predictors of Pa detection by FAPP.
Statistical analysis
Sample size was calculated to estimate the sensitivity and specificity for Pa, assuming an expected sensitivity of 99%, a specificity of 97%, 13 and a prevalence of 40%, 27 with an absolute precision of 5%, the necessary sample size was 75 patients. Allowing for a 10% dropout or unanalyzable data, a recruitment target of 83 patients was set. Descriptive statistics included frequency (percentages) for categorical variables, mean (standard deviation [SD]) or median (interquartile range) for continuous variables. Positive percent agreement (PPA), negative percent agreement (NPA), positive predictive value (PPV), and negative predictive value (NPV) of sputum FAPP for bacteria were reported, using culture as reference (Supplemental Material). PPA and NPA were used instead of sensitivity and specificity, respectively, as a non-perfect gold standard method (sputum culture in lower respiratory tract infection) was used as the reference test. Categorical variables were compared using the Chi-Square test or Fisher Exact test as appropriate, continuous variables were compared using the Student’s t test or Mann-Whitney U test as appropriate. Logistic regression was utilized with FAPP detection of Pa as the dependent variable. Independent variables identified through univariate analysis (p < 0.05) were included in the model, while highly correlated variables were excluded to avoid multicollinearity. p values were considered significant if <0.05. All statistical analyses were performed using computer programs Statistical Package for the Social Science (SPSS) version 23.0 (IBM Corp., Armonk, NY, USA, for Microsoft Windows) and Stata version 14.2 (College Station, TX, USA).
Results
Study population
Altogether, 83 bronchiectasis patients hospitalized for acute exacerbation were recruited. Baseline demographics and clinical characteristics are summarized in Table 2, while clinical presentations, severity, and treatment outcomes are summarized in Table S1. The study cohort had a female predominance (71.1%) with a mean age of 75 years. The most common etiologies of bronchiectasis were idiopathic (49.4%) and sequelae of tuberculosis (27.7%). Nine patients (10.8%) had chronic Pa infection. None were on long-term macrolide therapy. The most common symptoms were cough (96.4%) and an increase in sputum volume and/or change in consistency (92.8%). Forty-four patients had sputum samples collected before antibiotic administration, and of those collected afterward, the median time from antibiotic administration to sputum collection was 7 h. All sputum sent for culture had Gram stain showing <25 SEC per LPF and were cultured. Ninety percent of these sputum samples had a Bartlett score ⩾1 and were classified as “good quality,” whereas 10% had a Bartlett score <1 and were classified as “moderate quality.” Empirical antibiotics were prescribed for all patients, with anti-pseudomonal coverage in 29.
Baseline characteristics of the study population (n = 83).

Left lingula segment is counted as a separate lobe.
BMI, body mass index; BSI, bronchiectasis severity index; COPD, chronic obstructive pulmonary disease; FACED, FEV1, Age, Chronic colonization, Extension, and Dyspnea; FEV1, forced expiratory volume in 1 s; ICS, inhaled corticosteroid; IQR, interquartile range; LABA, long-acting beta agonist; LAMA, long-acting muscarinic antagonist; mMRC, modified medical research council; NTM, non-tuberculosis mycobacterium; Pa, Pseudomonas aeruginosa; PPI, proton pump inhibitors; SD, standard deviation.
Primary outcome: Microbial characteristics of AEB by sputum FAPP and standard-of-care tests
Among 83 patients, FAPP detected bacteria in 68 sputum samples (81.9%), identifying 101 bacteria, with high bacterial abundance (106 to ⩾107 copies/ml) observed in 48 samples (57.8%). Bacteria detected by FAPP were diverse, with Pa (37/83, 44.6%), Staphylococcus aureus (21/83, 25.3%), and Haemophilus influenzae (13/83, 15.7%) being the most common pathogens (Figure 1). Pa was also the most frequently detected pathogen at high abundance (Table 3). In contrast, culture identified only 28 bacteria in 25 samples (30.1%), significantly fewer than FAPP (p < 0.001). Pa was the most frequently cultured bacterial pathogen (Figure 2). Prior antibiotic use and sputum quality did not significantly affect the bacterial detection rate by FAPP. However, culture positivity was significantly reduced in those who received prior antibiotics (Table S2).

Bacteria and viruses detected by sputum FilmArray Pneumonia plus Panel.
Bacterial pathogens detected by sputum FilmArray Pneumonia plus Panel stratified into high (106 to ⩾107 copies/ml) and low abundance (104 to 105 copies/ml).


Bacteria detected by sputum culture and viruses detected by nasopharyngeal swab multiplex PCR in acute exacerbations of bronchiectasis.
The PPA, NPA, PPV and NPV of FAPP were calculated for bacterial pathogens, using culture as reference standard. Notably, FAPP had PPA and NPV of both 100% for all bacteria that grew on culture (Table 4) except for Corynebacterium striatum (n = 2), which was not included in FAPP. There was a positive correlation between the maximum copy number for the bacterial target detected by FAPP and the maximum semiquantitative growth for the same bacteria by culture (Spearman correlation 0.61, p < 0.001). Notably, when FAPP detected no targets or targets at low abundance (104–105 copies/ml), all sputum cultures had no growth; and conversely, when FAPP detected a target at 10⁶ copies/ml, 12% of cultures showed heavy growth of the same pathogen, increasing to 43.5% at 10⁷ copies/ml (Table S3).
Diagnostic performance of sputum FilmArray Pneumonia plus Panel in bacterial pathogen detection versus sputum culture.

Cannot be determined as the organism never grew on culture.
NPA, negative percent agreement; NPV, negative predictive value; PPA, positive percent agreement; PPV, positive predictive value.
Using FAPP, respiratory viruses were identified in 21 patients (25.3%), with Influenza A (n = 5) and Respiratory syncytial virus (n = 5) being the most common (Figure 1). All viruses were co-detected with at least one bacterial pathogen. Of 63 patients with NPS performed, a virus was detected in 12 (Figure 2). FAPP had PPA and NPV both of 100% for viral detection when compared with NPS (Table 5).
Diagnostic performance of sputum FilmArray Pneumonia plus Panel in viral pathogen detection versus nasopharyngeal swab multiplex.

NPA, negative percent agreement; NPV, negative predictive value; PCR, polymerase chain reaction; PPA, positive percent agreement; PPV, positive predictive value.
Secondary outcomes (I): Time to microbial result analysis
The mean (SD) time from sputum collection to microbial result was significantly shorter for FAPP compared to standard-of-care (10.8 [8.7] h vs 70.8 [24.3] h, p < 0.001).
Secondary outcomes (II): Antimicrobial changes following FAPP result
Antibiotics were prescribed to all patients. In addition, all patients with sputum FAPP positivity for Influenzae A (n = 5) were treated with oseltamivir. Antibiotic changes were observed in 25 patients (30.1%) following sputum FAPP results. Most changes were driven by the detection or non-detection of Pa: 5 patients had de-escalations from initial empirical anti-pseudomonal antibiotics, 16 had escalations or changes of antibiotics upon Pa detection, while 4 had resistance gene(s) detected (Table S4).
Secondary outcomes (III): Risk factors for Pa detection by FAPP
Clinical parameters of patients with and without Pa detection by FAPP were compared (Table S5). In multivariate analysis, chronic Pa infection (odds ratio (OR) 14.71, 95% CI 1.52–141.98), underweight status (OR 5.84, 95% CI 2.04–16.71), and cystic bronchiectasis (OR 5.26, 95% CI 1.08–25.63) were independent predictors of Pa detection by FAPP (Table 6). The model’s area under the receiver operating characteristic curve was 0.763 (95% CI 0.658–0.867) (Figure S1). The probability of Pa detection by FAPP increased with the number of risk factors (Figure 3).
Predictors of Pseudomonas aeruginosa detection by sputum FAPP in multivariate logistic regression analysis.
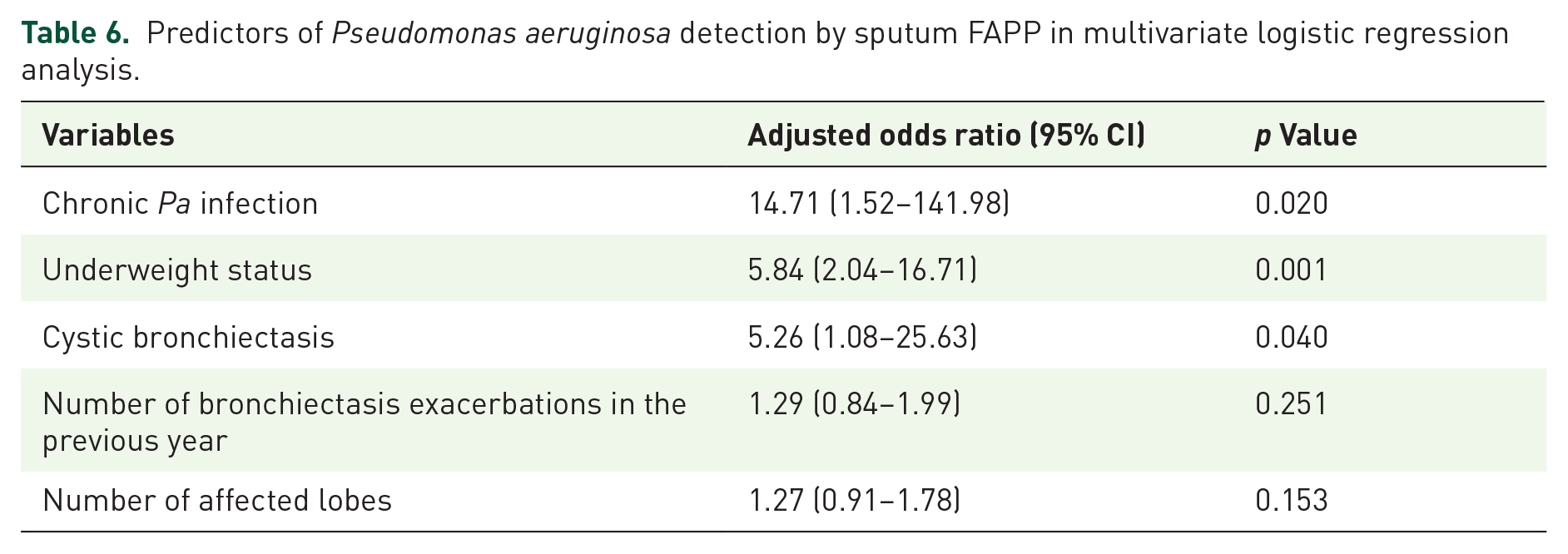
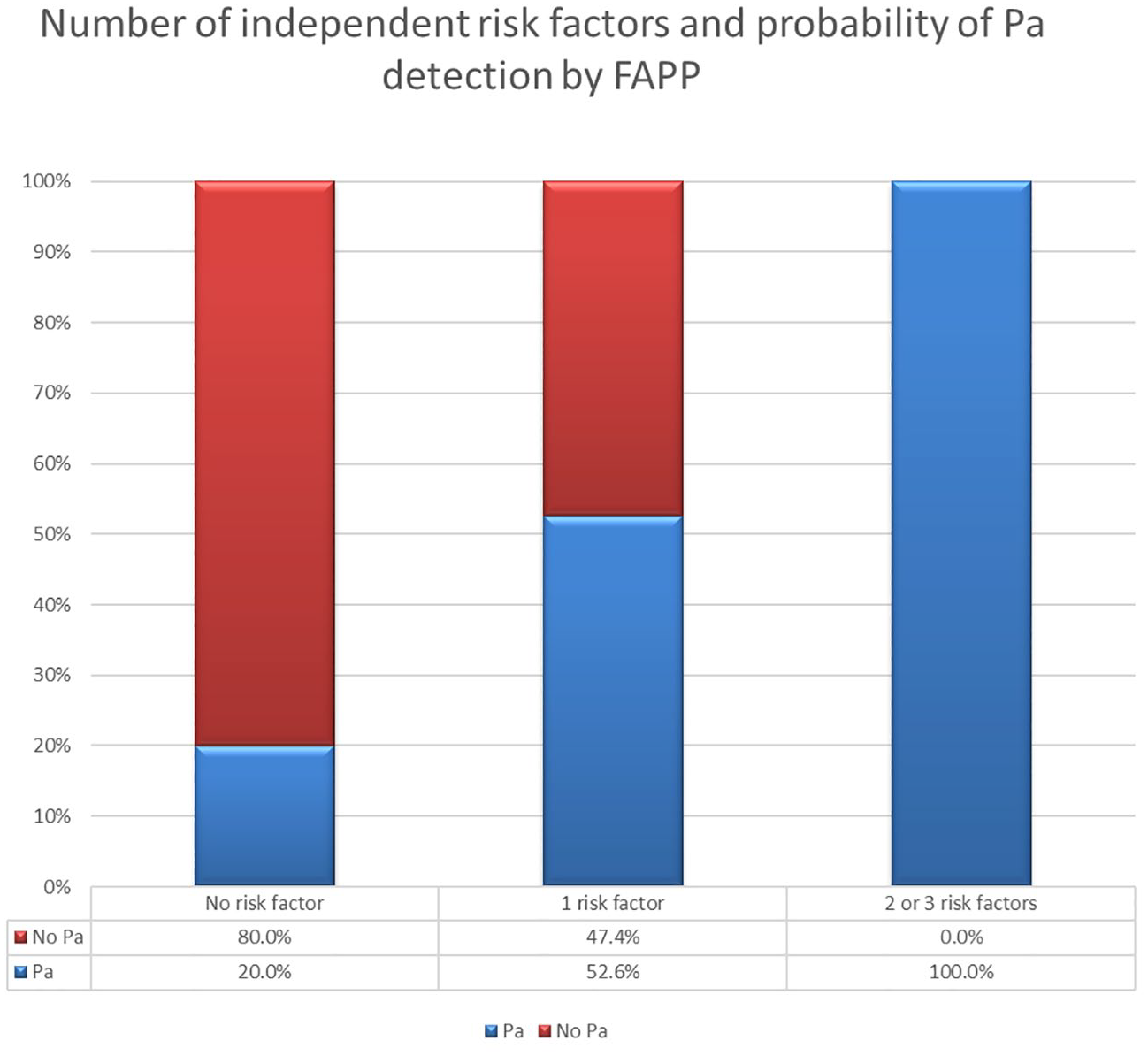
Probability of Pa detection by FAPP according to the number of risk factors.
Discussion
To the best of our knowledge, this study is the first to evaluate the clinical impact of FAPP and compare the microbial characteristics with culture-based method in AEB in a real-world setting. A recent study reported the endotypes of bronchiectasis exacerbations utilizing the same assay, further supporting its role in enhancing precision medicine in bronchiectasis exacerbations. 28 The predominance of Pa detection by FAPP in our study aligns with previous reports using culture methods. 27 The sputum FAPP had a significantly higher bacterial detection rate compared to culture in AEB, with excellent PPA and NPV, consistent with a recent study in patients hospitalized for respiratory illness. 13 However, it is important to consider potential pathogenic microorganisms not included in FAPP such as Corynebacterium striatum and Stenotrophomonas maltophilia. 28 Developing a bronchiectasis-specific panel with a broader spectrum in the future could potentially improve the diagnostic yield. Atypical bacteria are rarely detected in AEB, consistent with previous reports. 29 Regarding viruses, we observed a lower detection rate compared to previous studies,30 –32 which may be attributed to the study’s timeframe: conducted during and after the COVID-19 pandemic in Hong Kong, when masking and social distancing likely reduced respiratory virus transmission. Sputum FAPP demonstrated excellent PPA and NPV compared to NPS for viral detection, potentially saving the latter procedure.
The timely release of FAPP results to the treating physicians led to antibiotic adjustments in 30% of patients, mainly driven by the detection or non-detection of Pa. While Pa was the most common bacterium identified by both FAPP and culture, FAPP’s higher sensitivity resulted in a greater detection rate. Its rapid turnaround facilitated early appropriate anti-pseudomonal antibiotic adjustments in 9 patients (11%) (Table S4) before sputum culture grew Pa. However, for FAPP-positive cases involving non-culturable bacterial targets, further studies are needed to assess the impact of targeted treatment on patient outcomes. For patients whose FAPP results do not suggest Pa or resistant organisms, early and appropriate antimicrobial de-escalation may reduce the risk of developing MDR bacterial infections in the future.
In multivariate analysis, chronic Pa infection, underweight status, and cystic bronchiectasis were independent predictors of Pa detection by FAPP. These factors should be considered when determining antimicrobial treatment for AEB, especially when FAPP is not available, to guide the decision on the need for anti-pseudomonal antibiotics while awaiting culture results.
Our study has several limitations. First, the relatively small sample size from a single center limits generalizability. Validation in a larger multicenter cohort is desired. Second, sputum FAPP results during stable-state were not available. Thus, comparison of microbial patterns before and during AEB within the same patient is not possible. Third, as observed in previous real-world studies, prior antibiotic administration may reduce microbial yield. However, this reflects common practice, 13 where many AEB patients receive antibiotics before hospital admission or sputum collection. Despite this, FAPP still demonstrated a high bacterial detection rate, impacting clinical decisions. Finally, our study was limited to hospitalized patients (severe exacerbations) who could provide informed consent. Milder cases treated in outpatient settings or those unable to consent were not included. The utility of sputum FAPP in these patient populations remains to be explored.
Conclusion
The sputum multiplex PCR panel (FAPP) enables rapid identification of bacterial and viral pathogens in AEB, supporting early antimicrobial decision-making. Our findings highlight the potential utility of sputum multiplex PCR panels in improving the management of bronchiectasis exacerbations.
Supplemental Material
sj-docx-1-tar-10.1177_17534666251341751 – Supplemental material for Rapid microbial evaluation of acute exacerbations of bronchiectasis using FilmArray Pneumonia plus Panel in a real-world setting
Supplemental material, sj-docx-1-tar-10.1177_17534666251341751 for Rapid microbial evaluation of acute exacerbations of bronchiectasis using FilmArray Pneumonia plus Panel in a real-world setting by Charles Wong, Chun Wai Tong, Hei Shun Cheng, Pui Hing Chiu, Flora Pui Ling Miu, Yiu Wing Lam and Loretta Yin Chun Yam in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-docx-2-tar-10.1177_17534666251341751 – Supplemental material for Rapid microbial evaluation of acute exacerbations of bronchiectasis using FilmArray Pneumonia plus Panel in a real-world setting
Supplemental material, sj-docx-2-tar-10.1177_17534666251341751 for Rapid microbial evaluation of acute exacerbations of bronchiectasis using FilmArray Pneumonia plus Panel in a real-world setting by Charles Wong, Chun Wai Tong, Hei Shun Cheng, Pui Hing Chiu, Flora Pui Ling Miu, Yiu Wing Lam and Loretta Yin Chun Yam in Therapeutic Advances in Respiratory Disease
Footnotes
Acknowledgements
The authors would like to thank Ms. Miranda Yau and the other staff of the microbiology and molecular laboratories who have facilitated the laboratory tests for patients in this study. The authors would also like to thank Ms. Tsz-Ling Choi for facilitating patient recruitment.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
