Abstract
Background:
Bronchiectasis exacerbations are a significant contributor to morbidity and mortality. While environmental factors, such as viral infections, are well-established triggers for exacerbations, the role of intrinsic factors, particularly chronic bacterial infections, remains incompletely understood.
Objectives:
In this context, we sought to investigate the impact of chronic bacterial infections using the COVID-19 pandemic as a natural experiment, providing a unique opportunity to assess the effects of reduced external infections.
Design:
A retrospective observational cohort study was conducted involving patients with non-cystic fibrosis bronchiectasis.
Methods:
Data were collected via telephone interviews and medical record reviews, comparing exacerbation rates before (2019) and during (2020) the pandemic. The difference in exacerbation rates between 2020 and 2019 (delta exacerbations) served as the dependent variable in a multiple regression model.
Results:
Sixty-three patients were included in the analysis. Those without chronic bacterial infections showed a significant reduction in exacerbations during the pandemic: mean (SD) was 1.06 (1.3) versus 1.61 (1.3), respectively (p-value = 0.006). In contrast, no such reduction was observed in patients with chronic bacterial infections. Notably, chronic infection with Pseudomonas aeruginosa emerged as an independent predictor of sustained or increased exacerbations in 2020 (positive delta exacerbations), despite the implementation of social distancing measures.
Conclusion:
While social distancing effectively reduced bronchiectasis exacerbations in patients without chronic bacterial infections, those with Pseudomonas aeruginosa infections remained vulnerable to exacerbations, underscoring the importance of intrinsic disease/host factors. These findings highlight the need for targeted management strategies addressing chronic infections in patients with bronchiectasis.
Plain language summary
This study looked at how ongoing lung infections affect people with bronchiectasis, a lung condition that causes coughing and breathing problems. The researchers found that in people without long-term infections, the number of flare-ups (times when symptoms get worse) decreased when they wore protective devices like masks or avoided exposure to infections from others. But for those who had a long-lasting infection with a germ called Pseudomonas aeruginosa, flare-ups didn’t improve. This means that having a chronic infection makes the lung condition harder to manage. The findings show that people with these ongoing infections may need special treatments to keep their lungs healthier and reduce flare-ups.
Keywords
Introduction
Bronchiectasis exacerbations are a major cause of both morbidity and mortality. The primary challenge for clinicians managing bronchiectasis is to reduce the frequency of exacerbations and extend the time between episodes, as exacerbations significantly impact clinical outcomes and patient quality of life. Most therapies recommended by the European Respiratory Society (ERS) focus on preventive measures designed to delay the onset of subsequent exacerbations. 1 The onset of exacerbations in bronchiectasis is influenced by multiple factors. Extrinsic factors, such as environmental pathogens, can trigger exacerbations, while intrinsic factors, related to the host, remain incompletely understood and may play a pivotal role in exacerbation development.
Chronic bacterial infections, particularly Pseudomonas aeruginosa (PA), are common among patients with bronchiectasis and are known to exacerbate the disease, leading to worsened clinical outcomes and diminished quality of life.2–4 Respiratory viral infections, one of the key triggers of pulmonary exacerbations, are also frequently implicated, although the exact mechanisms remain elusive. 5 Viral infections not only precipitate exacerbations but can also contribute to increased mortality rates. During the COVID-19 pandemic, for instance, mortality among patients with bronchiectasis was found to be up to three times higher than in those without the condition. 6
The relative contribution of extrinsic versus intrinsic factors in triggering bronchiectasis exacerbations is still not fully understood. The COVID-19 pandemic provides a unique opportunity to investigate this issue, as it represents a period in which the influence of external infective agents, such as viruses and bacteria, was notably reduced.7–10 Despite the implementation of social distancing measures and widespread use of personal protective equipment, exacerbations in bronchiectasis patients continued to occur, albeit at a lower frequency. This observation suggests that host-related intrinsic factors may be critical in maintaining a residual rate of exacerbations, even in the absence of external infections.
Building on this premise, we hypothesized that chronic bacterial infections, as intrinsic factors, play a significant role in triggering acute exacerbations of bronchiectasis. Consequently, our study sought to investigate the relative contribution of chronic bronchial infections to exacerbation development in bronchiectasis, using the exceptional context of the SARS-CoV-2 pandemic as a natural experiment.
Materials and methods
Design
We conducted a retrospective observational cohort study involving telephone interviews with patients diagnosed with non-cystic fibrosis bronchiectasis, who were followed at the Respiratory Unit of the Policlinico Universitario in Palermo, Italy. This study follows the STROBE statement to ensure transparent and complete reporting of observational research. 11 This study qualifies as a retrospective cohort study because participants were grouped based on a preexisting exposure status (presence or absence of chronic bacterial infection) at a defined baseline (the first year, 2019). Their outcomes (number of disease exacerbations) were assessed over a subsequent period (two consecutive years: 2019 and 2020). Although data collection occurred after the outcomes had already taken place, the temporal relationship between exposure and outcome was preserved, and no intervention was applied. Moreover, the cohorts were followed over time retrospectively, through existing clinical records and structured patient interviews. This distinguishes the design from other observational studies and aligns it with the characteristics of a cohort study, as defined by STROBE guidelines. 11 A completed STROBE checklist is provided in the Supplemental Material.
Subjects
The study included adult patients with non-cystic fibrosis bronchiectasis and clinically significant disease of varying severity. Diagnosis of bronchiectasis was confirmed based on both clinical features (history, symptoms, and physical examination findings) and high-resolution computed tomography of the chest, consistent with the disease, in accordance with ERS guidelines. 12
Patients attending the outpatient clinic of the Division of Respiratory Diseases at the University of Palermo, Italy, for at least 1 year prior to the onset of the pandemic were eligible for inclusion. Exclusion criteria included patients with bronchiectasis secondary to cystic fibrosis and those with traction bronchiectasis resulting from interstitial lung diseases.
Measurements
Two data sources were used: (i) Medical records for the first year (2019), (ii) Structured telephone interviews (provided in the Supplemental Materials) with patients for the second year (2020). Anthropometric and functional data were retrieved from the outpatient clinic database. In-depth telephone interviews were conducted during 2020 and 2021 to gather data on exacerbations and clinical parameters, including symptoms, access to healthcare services, respiratory treatments, and patients’ general health status.
The first interview, conducted between June and July 2020, focused on the period of COVID-19-related restrictions, from March to June 2020. A subsequent phone interview or in-person consultation was conducted between January and February 2021 to collect data regarding the exact number of exacerbations in 2020.
Mild-to-moderate exacerbations managed at home were self-reported during a structured telephone interview conducted by a physician. Whereas, in cases of severe exacerbations, hospital admissions were considered valid documentation.
Potential sources of bias were identified and addressed as follows. Recall bias was minimized through the use of a structured interview protocol, which included a predefined set of standardized questions aimed at consistently assessing the occurrence of exacerbations. To further aid memory recall, participants were prompted with temporal anchors, such as references to the lockdown and pre-lockdown periods. Selection bias was reduced by including all eligible patients consecutively, without applying any sampling procedures.
Additional data, including exacerbation rates from the pre-pandemic year (2019), microbiological data, functional parameters, and radiological findings, were obtained from patients’ clinical records archived in the hospital’s system. Chronic bacterial infection was defined as the isolation of potentially pathogenic bacteria in sputum cultures on at least two separate occasions, at least 3 months apart within a year.
Statistical analysis
The study size was a convenience sample based on all eligible patients within the study period. No formal sample size calculation was performed.
Data were expressed as mean ± standard deviation (SD), median (interquartile range, IQR), or frequencies (proportions), as appropriate. The chi-square test of independence was employed to assess the relationship between categorical variables. Comparisons of patient characteristics were made using non-parametric tests. Group differences were evaluated using the Kruskal–Wallis test and the Mann–Whitney U test for independent groups, while paired comparisons were assessed using the Wilcoxon signed-rank test. No specific methods were applied to address missing data. Cases with missing values were included in the analysis as available, without imputation or exclusion.
The exposure was the presence of chronic bacterial infection at baseline (year 2019). The change in the number of exacerbations (Delta Exacerbations) was calculated as the difference between the number of exacerbations in 2020 and those in 2019. A negative value for “Delta Exacerbations” indicates a reduction in exacerbations in 2020 relative to 2019. To identify independent factors associated with Delta Exacerbations (the dependent variable), a robust multiple linear regression was performed. For this purpose, we first conducted a hierarchical regression using classical linear models. Predictors were added sequentially in the following order: age, sex, PA chronic infection, and bronchiectasis severity index (BSI). This approach allowed us to assess the incremental contribution of each variable to the explained variance in Delta Exacerbations. Afterward, we performed a robust multiple linear regression that accounts for potential violations of the assumptions of ordinary least squares regression, specifically non-normality of residuals and the presence of outliers.
Statistical significance was defined as a p-value < 0.05. All analyses were conducted using R (R Core Team 2022; www.R-project.org).
Results
A total of 79 patients were identified, of whom 63 provided their oral consent to participate in the study, including answering the phone questionnaire and allowing access to their medical records. The mean age of the patients was 64.1 ± 15.0 years, with 36 (57.1%) females and 27 (42.9%) males (Table 1).
Patients characteristics.
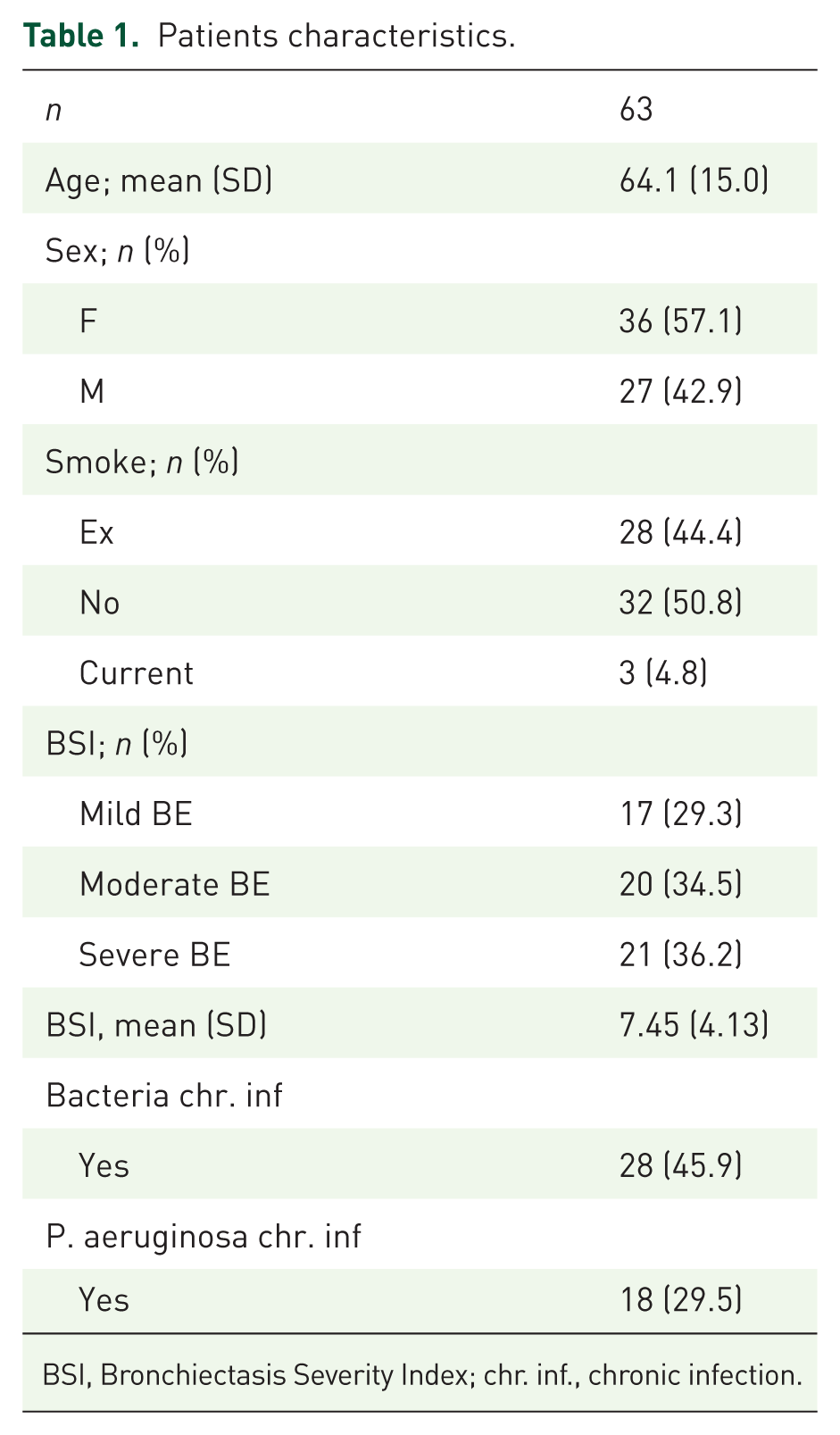
BSI, Bronchiectasis Severity Index; chr. inf., chronic infection.
In 2019, 25 patients (39.7%) experienced three or more exacerbations, and 28 patients (45.9%) had chronic infections, predominantly (n = 18) PA. During the period from March to June 2020, most patients (66.7%) reported never leaving their homes.
Patients with severe bronchiectasis demonstrated a positive difference in the number of exacerbations (Delta Exacerbations) in 2020 compared to 2019. Specifically, the delta was significantly higher (i.e., more exacerbations) in patients with severe bronchiectasis compared to those with moderate or mild forms of the disease: the median (IQR) of the delta in severe, moderate, and mild bronchiectasis was 0.5 (0.0–1.0), 0.0 (−0.25 to 0.0), and −0.5 (−1.0 to 0.0), respectively (p-value = 0.024).
Interestingly, patients without chronic infection showed a reduction in the number of exacerbations in 2020 compared to 2019: the mean (SD) was 1.06 (1.3) versus 1.61 (1.3), respectively (p-value = 0.006), Figure 1. Conversely, no such difference was observed in patients with chronic infection: 3.96(3.38) in 2020 and 3.86(3.48) in 2019 (p = 0.336), Figure 1.

Trend of the absolute number of exacerbations between 2019 (Exac.2019) and 2020 (Exac.2020), depending on whether patients had chronic bacterial infection or not.
The change in exacerbation rate was even more pronounced in patients with chronic PA infection. In patients without chronic Pseudomonas, the mean delta was −0.52 (1.13) compared with 1.00 (1.66) in those with chronic PA infection (p < 0.001).
In the multiple regression model, age, sex, and PA chronic infection were identified as statistically significant predictors of Delta Exacerbations (Table 2). Age and sex had moderate effects, with advancing age and female sex being associated with an increase in Delta Exacerbations. More importantly, patients with chronic PA infection exhibited a strong positive association with Delta Exacerbations, indicating that PA chronic infection is strongly linked to exacerbation frequency. On the contrary, the BSI score did not show a significant relationship with Delta Exacerbations in this model. The model accounted for approximately 50% of the variance in Delta Exacerbations, reflecting a moderate predictive power.
Multiple regression model with Delta Exacerbations as dependent variable.
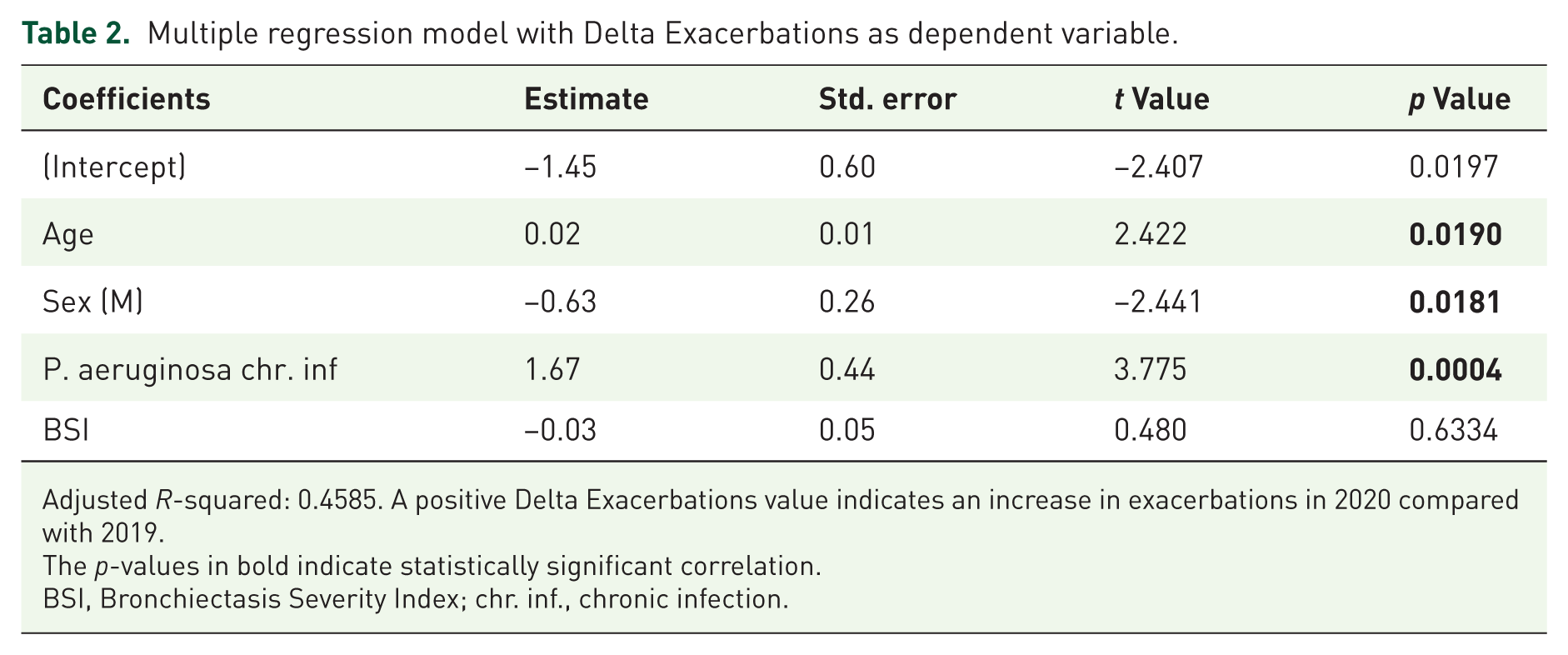
Adjusted R-squared: 0.4585. A positive Delta Exacerbations value indicates an increase in exacerbations in 2020 compared with 2019.
The p-values in bold indicate statistically significant correlation.
BSI, Bronchiectasis Severity Index; chr. inf., chronic infection.
Discussion
This study demonstrates that patients with bronchiectasis and chronic bacterial infection can experience exacerbations even in the absence of external infectious exposures, such as during a lockdown. In contrast, in patients without chronic infection, exacerbation frequency decreases. Clinically, this suggests that while preventing contact with external agents is essential for reducing exacerbations in bronchiectasis patients, it may not be sufficient in more severe cases with chronic bacterial infection.
Our findings are novel, as the role of chronic bacterial infections in triggering exacerbations has not been fully explored. While previous studies have shown a reduction in exacerbations during lockdowns, our study adds the key insight that patients with chronic PA infection were relatively unaffected by social isolation. These findings align with prior research, 7 which reported a decline in exacerbations without significant changes in chronic respiratory symptoms. Notably, not only did the frequency of exacerbations decrease, but their severity also declined compared to the pre-pandemic period. 13
We conducted a detailed regression analysis to identify clinical predictors of change in exacerbation frequency. Robust modeling confirmed that PA chronic infection, age, and sex are independently associated with increased exacerbations. Although BSI was not statistically significant in multivariable models, we retained it due to its established clinical role in assessing disease severity and its significant association with outcomes in exploratory analysis.
As for underlying mechanisms, we hypothesize that patients without chronic infection, particularly those with mild disease, are more susceptible to exacerbations triggered by new infections or external factors. A thorough and rigorous analysis of our statistical data leads to the conclusion that the observed mechanism is, in fact, a reduction in exacerbations in the group without chronic bacterial infection, and a relative stability in the group with chronic infection. In contrast, the chronic presence of PA may itself act as a trigger, even in patients who limit external exposure, when the host-bacterium balance shifts in favor of increased bacterial replication. This is consistent with previous observations showing that PA exhibits an increased number of virulence factors during exacerbations compared to stable phases. 14 Moreover, data from the ORBIT-3 and ORBIT-4 trials showed that increased exacerbations in chronic PA infection were linked to higher Pseudomonas abundance. This was also associated with neutrophilic inflammation, reduced diversity of commensal bacteria such as Rothia, and changes in B-cell responses, highlighting the role of microbial imbalance in disease severity. 15 Also, in alpha-1 antitrypsin deficiency (a risk factor for bronchiectasis development), bacterial colonization, including Pseudomonas species, significantly increases the risk of exacerbations. 16 In this context, the presence of Pseudomonas during stable disease was independently associated with nearly doubled odds of a subsequent exacerbation. This highlights how Pseudomonas contributes to inflammation and worsens disease outcomes. 16 Recent evidence suggests that the management of patients experiencing acute exacerbations of bronchiectasis may benefit from the innovative application of multiplex polymerase chain reaction assays, which analyze respiratory specimens for bacteria, viruses, and antimicrobial resistance markers. In these cases, the detection of PA was found to be associated with chronic infection by the same organism, underweight status, and the presence of cystic bronchiectasis. 17
It is possible to postulate that a similar mechanism might be generalizable to other populations, such as patients with bronchiectasis and chronic infection due to nontuberculous mycobacteria (NTM). However, as this study did not include any patients with NTM pulmonary disease, this consideration remains speculative.
In addition to exposure to external viral or bacterial triggers, a potential role may also be attributed to other external factors, like environmental pollutants, to which patients were less exposed during the COVID-19 pandemic. However, during the pandemic, reduced exposure to environmental factors affected both groups, with and without chronic PA infection. Consequently, if exacerbations were solely triggered by external factors, a similar outcome would be expected in both cohorts. For this reason, we hypothesize that the absence of chronic infection itself may have contributed to the observed reduction. Poor or reduced adherence to therapeutic programs during the pandemic may also have played a role. However, it is important to note that none of the patients included in the study were receiving treatment with inhaled antibiotics. Additionally, participation in pulmonary rehabilitation programs was generally low in our cohort, primarily due to logistical barriers and limited healthcare resources in the city of Palermo. Therefore, the impact of these factors was likely limited or negligible.
In our study, the BSI was not an independent predictor of exacerbation changes. It is possible that, within the context of social isolation and the short observation period, the impact of chronic PA infection outweighed the predictive value of the BSI.
This study has some limitations, including its single-center design and relatively small sample size. However, it provides valuable insights. It is a real-world study with a novel analytical perspective—rather than being just another study on the reduction of exacerbations during the COVID-19 pandemic, it leverages that period to uncover the role of factors that remain poorly understood in the pathogenesis of bronchiectasis exacerbations.
Another limitation of the study is the inability to account for other potential extrinsic factors that may have influenced exacerbations. The complexity of the disease extends well beyond the scope of this analysis and includes both pharmacological interventions (e.g., inhaled antibiotics) and non-pharmacological approaches (e.g., respiratory physiotherapy programs) that could affect the occurrence of exacerbations. However, as previously discussed, the impact of these variables is likely limited in our local setting. Nonetheless, it remains important that such factors be evaluated in future studies conducted in more appropriate contexts.
The single-center design represents a limitation, potentially affecting the generalizability of our findings, particularly given the known geographical variation in the distribution of PA. Indeed, differences in the microbiological profiles of bronchiectasis patients have been documented across European countries. 18 PA appears to be significantly more prevalent in Southern Europe, suggesting a strong geographical influence potentially related to environmental and climatic factors, as this pathogen is typically acquired from environmental sources rather than through person-to-person transmission. These considerations further emphasize the need for multicenter studies across diverse regions to validate and extend our observations.
Conclusion
Our findings suggest that patients with chronic bacterial infections, particularly PA, may experience exacerbations independently of external triggers such as new infections or environmental factors. In contrast, patients without chronic infection may require an external infection to trigger an exacerbation. While these insights emerged during the COVID-19 pandemic, they provide a broader understanding of bronchiectasis exacerbations beyond that specific period.
The evidence highlights the interplay between external (infectious or environmental) and internal factors in exacerbation pathogenesis. However, it also underscores the critical role of intrinsic, host-related mechanisms—still poorly understood—such as chronic bacterial infection and persistent inflammation. Further research is needed to clarify their contribution and develop targeted therapeutic strategies.
Our observations may offer useful insights into the behavioral recommendations clinicians should provide to this patient population, supporting the idea that healthcare providers should encourage the use of personal protective equipment, such as face masks, particularly in crowded settings, as a preventive measure. However, although preventive measures like hand hygiene, mask use, and seasonal vaccinations remain essential, not all patients benefit equally. Those with chronic bacterial infections may require additional interventions tailored to their specific needs.
Supplemental Material
sj-doc-2-tar-10.1177_17534666251376501 – Supplemental material for Unveiling the causes of bronchiectasis exacerbations: insights from a single-center study
Supplemental material, sj-doc-2-tar-10.1177_17534666251376501 for Unveiling the causes of bronchiectasis exacerbations: insights from a single-center study by Martina Lo Casto, Carlo Chessari, Stefania Marino, Maria Fulvia Di Grado, Anna Isabella Memmo, Stefania Principe, Nicola Scichilone and Salvatore Battaglia in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-pdf-1-tar-10.1177_17534666251376501 – Supplemental material for Unveiling the causes of bronchiectasis exacerbations: insights from a single-center study
Supplemental material, sj-pdf-1-tar-10.1177_17534666251376501 for Unveiling the causes of bronchiectasis exacerbations: insights from a single-center study by Martina Lo Casto, Carlo Chessari, Stefania Marino, Maria Fulvia Di Grado, Anna Isabella Memmo, Stefania Principe, Nicola Scichilone and Salvatore Battaglia in Therapeutic Advances in Respiratory Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
