Abstract
In this review, we discuss the risks and adverse effects reported for the current Food and Drug Association (FDA)-approved biologics used in the management of asthma, including omalizumab, benralizumab, dupilumab, mepolizumab, reslizumab, and tezepelumab. Our review focuses on the risk of hypersensitivity reactions, infection, and malignancy. Where relevant, we have included information regarding the risk of cardiovascular disease and eosinophilia, and we have included specific information regarding vaccine use among patients receiving the above biologics. We also review currently available data regarding the use of biologics in the context of pregnancy. Our goal is to provide a comprehensive resource for providers utilizing these agents, so that they may adequately counsel patients about the risks of therapy and identify adverse events if they occur.
Introduction
Biologic agents have revolutionized the treatment of asthma, offering a targeted and personalized approach for patients with uncontrolled severe asthma. They have been shown to reduce asthma exacerbations, improve lung function, reduce oral corticosteroid use, and improve quality of life for appropriately selected patients. 1 Several factors must be considered when initiating a biologic and monitoring response to therapy, including risks of biologic therapy. A comprehensive understanding of the risks and adverse effects is important to counsel patients adequately and to monitor adverse events appropriately.
In this review, we provide a comprehensive summary of risks and adverse effects reported for the current Food and Drug Association (FDA)-approved biologics used in the management of asthma, including omalizumab, mepolizumab, reslizumab, benralizumab, dupilumab, and tezepelumab. Please see Table 1 for a complete list of FDA-approved indications and mechanisms of these agents.
FDA-approved indications, dosing of biologics used in asthma.
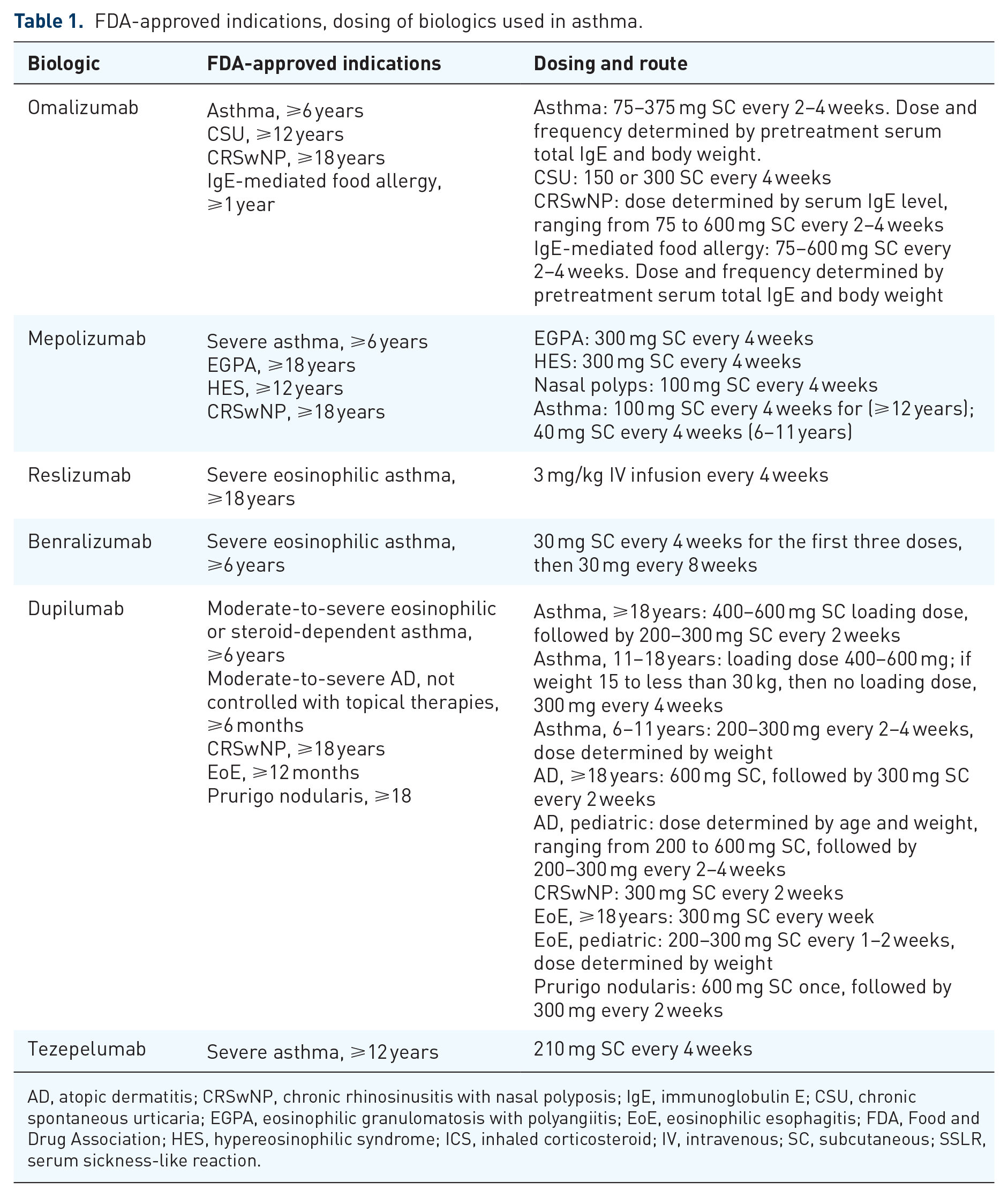
AD, atopic dermatitis; CRSwNP, chronic rhinosinusitis with nasal polyposis; IgE, immunoglobulin E; CSU, chronic spontaneous urticaria; EGPA, eosinophilic granulomatosis with polyangiitis; EoE, eosinophilic esophagitis; FDA, Food and Drug Association; HES, hypereosinophilic syndrome; ICS, inhaled corticosteroid; IV, intravenous; SC, subcutaneous; SSLR, serum sickness-like reaction.
The above biologics are generally well-tolerated. According to a large meta-analysis of randomized controlled trials that assessed the efficacy of biologics approved to treat asthma, biologics have been associated with fewer adverse effects, serious adverse events, deaths, and pneumonia, compared to standard of care. 2 The most common adverse effects are minor, benign, and common to all agents, and include nasopharyngitis, upper respiratory infection, and reactions at the injection site.3 –8
Our review will focus on major adverse events and risks, including immediate and delayed hypersensitivity reactions, risk of infection, and risk of malignancy. We review drug-specific adverse events, like cardiovascular events and peripheral eosinophilia. We also provide a summary of available data regarding biologic use during pregnancy. Please see Table 2 for a summary of adverse events attributed to each biologic agent.
Overview of adverse effects of biologics used in asthma.

Cardiovascular death, myocardial infarction, ischemic stroke, transient ischemic attack, and unstable angina.
Cardiovascular-related death, nonfatal myocardial infarction, arrhythmia, thromboembolism, and stroke.
CI, confidence interval; CV, cardiovascular/cerebrovascular; EM, erythema multiforme; HR, hazard ratio; HSR, hypersensitivity reaction; LTE, long-term extension; OLE, open-label extension; PI, package insert; PY, person years; RDB, randomized double-blind; RDBPCT, randomized double-blind placebo-controlled trial; RZV, recombinant zoster vaccine; SSLR, serum sickness-like reactions.
Omalizumab
Hypersensitivity reactions
Immediate and delayed allergic reactions have been attributed to omalizumab. The risk of anaphylaxis with omalizumab was identified in the early clinical trials that led to its FDA approval for the treatment of asthma in 2003. As a result, omalizumab’s product label includes a boxed warning for anaphylaxis.9,10
Subsequent studies identified that many cases of anaphylaxis occurred with the first dose and were delayed beyond 2 h; both are atypical of immediate-type allergic reactions and raise questions about underlying pathophysiology. 11 In a retrospective case-control study designed to further characterize and identify risk factors for anaphylaxis to omalizumab, 51% of 123 reported cases occurred after the first dose; 72% occurred during the first three doses. 12 Those with a history of anaphylaxis to other agents, including foods and other medications, had an increased risk of anaphylaxis (odds ratio (OR) of 8.1 (95% confidence interval (CI) 2.7–24.3)).12,13 The majority of reactions (63%) occurred within an hour of omalizumab administration, but about 10% of cases were significantly delayed, occurring between 90 min and 24 h. 12 The overall rate of omalizumab-related anaphylaxis is low, occurring in approximately 0.1%–0.2% of patients.9,10
The American Academy of Allergy, Asthma and Immunology/American College of Allergy, Asthma, and Immunology Joint Task Force has outlined recommendations for the administration of omalizumab, which includes a recommendation that patients receiving omalizumab have an epinephrine auto-injector.9,10 The Joint Task Force initially recommended that all patients be observed after each injection, for a duration of 2 h after the first three injections and 30 min after all subsequent injections.9,10 The FDA now advises that omalizumab may be self-administered at home after receiving and tolerating the first three doses under observation if the patient has no prior history of anaphylaxis. 5
In early clinical trials, serum sickness-like reactions were reported among three patients receiving omalizumab. 14 Signs and symptoms included arthritis, rash, fever, and lymphadenopathy, which developed 1–5 days after the first or subsequent injections of omalizumab. Each case was resolved despite the continuation of treatment. A few cases have since been described wherein discontinuation of omalizumab was required for symptom resolution. 15
Malignancy
In early clinical trials of patients >12 years old, malignancy occurred in 20 of 4127 (0.5%) of patients receiving omalizumab compared to 5 of 2236 (0.2%) of controls. 16 An analysis of pooled data from 67 randomized double-blind placebo-controlled trials found no association between omalizumab use and malignancy. 16 In a subsequent phase IV 5-year safety study (EXCELS), the malignancy rate per 1000 patients years was found to be 16 in the omalizumab group (95% CI 14.2–17.9) and 19.1 in the non-omalizumab group (95% CI 16.5–22.0), with a hazard ratio of 1.09 (95% CI 0.87–1.38). 17 Omalizumab’s package insert includes the risk of malignancy and highlights limitations of the EXCELS study, including the exclusion of patients with a history of cancer or premalignancy and a high discontinuation rate of 44% in omalizumab-treated cohort versus 35.2% of non-omalizumab–treated cohort.5,18
Infection and vaccination
There is insufficient data to make conclusions about the risk of parasitic infections in patients receiving omalizumab. 19 One double-blind, placebo-controlled study assessed the risk of helminth infection among individuals treated with omalizumab who were determined to have a high risk of helminth infection. All individuals received antihelmintic treatment, followed by 52 weeks of omalizumab or placebo. Those receiving omalizumab did not have a significantly higher risk of helminth infection (OR 1.47, 95% CI 0.74–2.95), and response to appropriate treatment was similar. 20 There are no published contraindications or recommendations regarding vaccination.
Cardiovascular events
In a 5-year observational cohort study (EXCELS), a higher incidence rate of cardiovascular and cerebrovascular events was identified among patients receiving omalizumab for allergic asthma compared to the non-omalizumab cohort. 21 A total of 5007 omalizumab-treated and 2829 non-omalizumab–treated patients were enrolled. The cohorts were demographically similar; however, severe asthma was more common in the omalizumab versus the non-omalizumab group (50% vs 23%). 21 Serious adverse cardiovascular events of interest included cardiovascular death, myocardial infarction, ischemic stroke, transient ischemic attack, and unstable angina. 21 Patients treated with omalizumab had a higher rate of cardiovascular and cerebrovascular events (13.4 per 1000 person years (PYs)) compared to non-omalizumab (8.1 per 1000 PYs). 21 It was concluded by the authors that differences in baseline risk factors for cardiovascular disease (asthma severity) contributed to the risk differences, but a contribution from omalizumab cannot be ruled out. 21 A pooled analysis of 25 randomized double-blind, placebo-controlled trials was conducted to further assess this risk. 22 Most patients received omalizumab for allergic asthma (72%). 22 Rates of adverse cardiovascular and cerebrovascular events were similar between the omalizumab (N = 3342) and placebo (N = 3895) groups. 22 However, as the median duration of treatment was 154 days for omalizumab and 161 days for placebo, and patients were overall younger compared to the previous cohort study (mean of 38 years vs 44 years), it was concluded that results were insufficient to reject findings of the cohort study.21,22
Eosinophilia
Multiple case reports describe the diagnosis of eosinophilic granulomatosis with polyangiitis (EGPA) after treatment with omalizumab for severe asthma, and this risk is included on omalizumab’s product label.23 –28 Similar cases have been reported among asthmatics receiving dupilumab and anti-IL-5 biologic agents. 29 In most cases, EGPA presents during systemic corticosteroid taper, and it has been suggested that this may reflect an “unmasking” of EGPA. 29 Routine monitoring of eosinophil counts is not required, but there should be a low threshold to investigate eosinophil counts if a patient develops worsening respiratory symptoms, rash, myalgias, neuropathy, or other potential manifestations of hypereosinophilia while on omalizumab.
Mepolizumab
Hypersensitivity reactions
No episodes of anaphylaxis were reported among those randomized to mepolizumab for asthma in double-blind, placebo-controlled studies.30,31 In three open-label extension trials, the rate of hypersensitivity reactions was reported at 1%–2%, and reactions included rash, pruritus, headache, and myalgia.32 –34 There were no reports of serious hypersensitivity reactions, including anaphylaxis.32 –34 Postmarketing studies have reported anaphylaxis. 35 The frequency of anaphylaxis among patients receiving mepolizumab is not known.
Malignancy
There is no evidence that patients receiving mepolizumab are at increased risk of developing malignancy. In one open-label extension trial, with a maximum exposure duration of 4.8 years, 8 (2%) patients receiving mepolizumab for severe asthma developed malignancy. 33 Reported malignancies did not indicate a clear pattern, and included basal cell carcinoma, prostate cancer, breast cancer, colon adenocarcinoma, unspecified colon neoplasm, and melanoma. 33
Infection and vaccination
Opportunistic infection occurred in 4%–7% of patients receiving mepolizumab in two 52-week, open-label extension trials for severe eosinophilic asthma.32,33 Most of these cases (n = 11) were herpes zoster infections. 33 Candida (n = 3) and pulmonary tuberculosis (n = 1) were also reported. 33 One episode of herpes zoster was considered serious; all other episodes resolved despite the continuation of mepolizumab treatment.32,33 Mepolizumab’s package insert recommends herpes zoster vaccination for eligible patients prior to starting therapy, and we typically recommend this approach to our patients. 36
Reslizumab
Hypersensitivity reactions
Reslizumab features a boxed warning for anaphylaxis. 8 In placebo-controlled studies, three cases of anaphylaxis occurred with reslizumab administration (0.3%). 37 All events occurred shortly after initiating or completing the infusion, and all affected patients were discontinued from the study. 37 In an open-label extension trial, no cases of anaphylaxis were reported among 1051 patients who received reslizumab. 38 Unlike omalizumab, there are no specific guidelines to attempt to mitigate the risk of anaphylaxis with reslizumab. The package insert recommends observation after reslizumab infusion for “an appropriate period of time.” 8
Malignancy
In a pooled analysis of randomized controlled trials, there was not a significant difference in malignancy occurrence between patients receiving reslizumab compared to placebo. 37 Malignancy was identified in 2 of 730 (0.27%) patients on placebo, compared to 6 of 1028 (0.58%) on reslizumab (OR 2.14 (95% CI 0.38–21.70)). 37 There was no reported difference in the incidence of malignancy among patients on treatment for longer than 12 months compared to shorter durations of treatment. 37
Infections and vaccinations
There were no reported helminth infections or opportunistic infections in a 24-month open-label extension trial. 37 There are no specific recommendations or restrictions regarding vaccination. 8
Benralizumab
Hypersensitivity reactions
Hypersensitivity reactions, primarily urticarial rash, were reported in 1%–3% of patients in placebo-controlled trials.39 –41 Anaphylaxis did not occur in randomized, controlled, double-blind studies among patients receiving benralizumab, though it was reported in one patient in the open-label extension trial.42,43
Malignant neoplasms
As eosinophils are thought to play a role in immunomodulation, concern regarding the potential impact of eosinopenia induced by benralizumab on the risk of malignancy has been raised. 44 There is insufficient evidence to suggest an increased risk of malignancy among patients receiving benralizumab. In a 1-year open-label extension study, new malignancy occurred in 12 of 1576 (1%) patients, and one case was felt by the investigator to be related to benralizumab. 42 This patient had a history of prostatic hypertrophy and elevated prostate-specific antigen and developed prostate cancer 3 days after the second dose of benralizumab. 42 A subsequent 5-year extension did not demonstrate an increased risk of malignancy compared to placebo; malignancy was reported in 3 (0.7%) of patients receiving benralizumab compared to 1 (0.5%) of patients receiving placebo. 45
Infections and vaccination
As benralizumab is associated with more significant eosinophil depletion than mepolizumab and reslizumab, there was concern that this could result in increased adverse events. Across all trials, patients receiving benralizumab had no increased risk of infection, and there were no reported helminth infections.39 –42 There was one reported case of herpes zoster. 39 Subsequently, a case report published in 2019 describes a 61-year-old patient who developed disseminated herpes zoster after initiation of benralizumab. 46 There are no published contraindications or recommendations regarding vaccination. Subsequent 1- and 5-year extension trials similarly did not demonstrate an increased risk of infection.45,47
Dupilumab
Hypersensitivity reactions
Hypersensitivity reactions, inclusive of urticaria, serum sickness, rash, erythema nodosum, and anaphylaxis, were reported in approximately 1% of participants in early clinical trials, with no significant difference in rates of hypersensitivity reaction between placebo and dupilumab.48 –52 Serum sickness reactions and erythema multiforme have been described in case reports. 15
Malignancy
There is insufficient evidence to suggest a causal relationship between dupilumab and malignancy. In an open-label extension trial, five patients developed malignancy. Two were felt to be related to the study drug, including one patient with Hodgkin’s disease and one patient with prostate cancer. 53 In another open-label extension trial assessing the safety of dupilumab use among asthmatics, the incidence of serious emergent adverse events, including malignancy, was low and did not differ between treatment groups. 54
Several case reports have associated the use of dupilumab with the development or progression of cutaneous T-cell lymphoma. 55 This has largely been described among patients receiving dupilumab for atopic dermatitis. 55
Infections and vaccinations
There was no evidence of increased infection incidence in any trial, and there were no reported helminth infections.48 –52,56 Among patients receiving dupilumab for atopic dermatitis, 0.4%–1% of patients developed herpes zoster infection.57,58 Notably, oral herpes and other herpes simplex viral infections were not reported among patients receiving dupilumab for asthma. 7
Patients who received a live vaccine within 12 weeks of the initial visit were excluded from randomized placebo-controlled trials. 58 Thus, the product’s package insert suggests that patients complete all age-appropriate vaccines prior to initiating dupilumab, including vaccination for herpes zoster, if indicated, and recommends avoidance of live vaccines. 7 There have subsequently been efforts to clarify the risks surrounding vaccination in patients receiving dupilumab, particularly as this agent is approved for use among patients 6 months and older.
A randomized, placebo-controlled study sought to assess the impact of dupilumab on vaccination by checking pre- and post-vaccination antibody titers to tetanus and meningococcal vaccines in adult patients receiving dupilumab versus placebo for 16 weeks. 59 Dupilumab did not impact response to the vaccine in this study, though the participants’ prior vaccination status was not known. 59 There are few studies assessing the use of live and inactivated vaccines in pediatric patients receiving dupilumab. 60
An expert Delphi panel systemically reviewed available literature on patients who received vaccinations while using dupilumab. 61 Available data suggests that the use of live vaccines in patients receiving dupilumab is safe, and dupilumab does not seem to impact the efficacy of the vaccines. 61 They concluded that patients receiving dupilumab may receive live vaccines without discontinuing therapy and recommended a shared decision-making approach to this issue. 61
Eosinophilia
Blood eosinophilia was initially reported as an adverse event of dupilumab in the Liberty Asthma Quest trial. 62 Of the 1902 patients with uncontrolled asthma who were enrolled in the study, 52 (4.1%) developed transient blood eosinophilia on dupilumab compared to 4 (0.6%) on placebo. 62 The highest mean percent change in eosinophils from baseline occurred between 16 and 20 weeks, and the greatest change occurred in patients receiving a higher dose of dupilumab, 300 mg every 2 weeks versus 200 mg every 2 weeks. 62
The majority of these patients did not develop end-organ involvement of eosinophilia; four (0.2%) of patients developed symptomatic eosinophilia, including chronic eosinophilic pneumonia. 62 All patients experienced resolution of eosinophilia and related symptoms, and one patient was able to continue dupilumab.
In the subsequent Liberty Asthma Venture trial, asymptomatic eosinophilia was reported as an adverse event in 14% of patients receiving dupilumab versus 1% placebo. 48 An open-label extension trial (TRAVERSE) found that 1.4%–6.4% of patients receiving dupilumab 300 mg every 2 weeks developed eosinophilia. 54 Most patients remained asymptomatic with a resolution of eosinophilia by week 96 without intervention; five were diagnosed with EGPA.
Eosinophilia may occur as a result of reduced eosinophil migration due to the blockage of IL-4/IL-13 and subsequent downstream inhibition of eotaxin-3 and other chemokines that facilitate eosinophil migration. 29 Routine monitoring of eosinophil counts for patients on dupilumab is not required, but there should be a low threshold to investigate eosinophil counts if a patient develops worsening respiratory symptoms, rash, myalgias, neuropathy, or other potential manifestations of hypereosinophilia while on dupilumab.
Cardiovascular events
In a 1-year placebo-controlled trial assessing the safety and efficacy of dupilumab for asthma (QUEST), a difference in the number of serious cardiovascular adverse events was noted between dupilumab and placebo groups. 62 Serious cardiovascular events, including cardiovascular-related death, nonfatal myocardial infarction, arrhythmia, thromboembolism, and stroke, were reported in 1 (0.2%) of patients receiving dupilumab 200 mg every 2 weeks, 4 (0.6%) of patients receiving dupilumab 300 mg every 2 weeks and 2 (0.3%) of the placebo group. 62 An expert panel assessed this data and concluded that there was no evidence of a significant risk difference between groups. 62 No cardiac events were associated with eosinophilia. 62 A potential risk of cardiovascular disease is included on the product label in response to this data. 7
Conjunctivitis
Conjunctivitis is a frequent adverse effect among patients receiving dupilumab for atopic dermatitis. In an open-label extension trial, 10.7% (n = 160) of patients developed conjunctivitis (i.e., allergic conjunctivitis, bacterial conjunctivitis, viral conjunctivitis), and 80% of these cases resolved while continuing dupilumab. 53 Most cases developed early in the study period, during weeks 0–12. 53 An increased risk of other ocular surface disorders has been reported among patients receiving dupilumab for atopic dermatitis, including blepharitis and keratitis, though less frequently. 63
Notably, an increased risk of conjunctivitis was not found in clinical trials using dupilumab for asthma or chronic rhinosinusitis with nasal polyps, indicating that the underlying barrier plays an important role in the development of this adverse effect. 47
Arthralgia
Dupilumab-associated arthritis and arthralgia were not noted in clinical trials; however, this has been described in several case reports and case series, and it has since been added to the product label.7,64 In most reported cases, patients who developed arthralgia were receiving dupilumab for atopic dermatitis.64,65 In one case series, onset varied from after the first dose to after 19 months of therapy, and presentations included generalized joint pain, oligoarthritis, enthesitis, tenosynovitis, joint swelling, and joint stiffness. 65 A cohort study compared the risk of joint pain/arthralgia among patients receiving dupilumab versus cyclosporine or mycophenolate for atopic dermatitis using US insurance claims data. 64 The primary outcome was a physician visit for joint pain while on dupilumab versus cyclosporine or mycophenolate, and they found no increased risk over a 6-month period of dupilumab use compared to the other agents. 64
Tezepelumab
Hypersensitivity reactions
In randomized, double-blind, placebo-controlled studies (PATHWAY, NAVIGATOR, and SOURCE) and subsequent long-term extension studies, there were no reported cases of anaphylaxis among patients receiving tezepelumab.66 –69 Per tezepelumab’s package insert, postmarketing cases of anaphylaxis have been reported. 4
Malignancy
There is no evidence to suggest an increased risk of malignancy among patients receiving tezepelumab. In a long-term extension study (DESTINATION), the incidence of malignancy was not significantly different between tezepelumab and placebo groups over the 104-week study period. 67
Infection and vaccination
There was no increased risk of helminthic or opportunistic infection noted among patients receiving tezepelumab in randomized clinical trials or the long-term extension study.66 –69 There was one case of pneumonia in the randomized clinic trial PATHWAY, which was thought to be related to the study drug. 69 There were no cases of pneumonia attributed to tezepelumab in the long-term extension study. 67 Patients with recent receipt of live vaccines were excluded from clinical trials, so avoidance of live vaccines is recommended. 4
Cardiovascular events
A potential risk of cardiovascular events is listed on tezepelumab’s product label. 4 In a randomized, double-blind long-term extension trial, patients with severe asthma received tezepelumab 210 mg q4w versus placebo for up to 104 weeks. 67 The incidence of cardiac adverse events and cardiovascular deaths was similar in those receiving tezepelumab compared to placebo (incidence rate difference of 0.88 (95% CI 0.24, 1.530)). 67 A variety of cardiovascular disorders were reported, and there was no discernable pattern regarding the timing of the cardiac events in relation to the receipt of tezepelumab. 67
Use of biologics for asthma in the context of pregnancy
Pregnant patients with poorly controlled asthma are at risk of adverse maternal and fetal outcomes, including low birth weight, small size for gestational age, preterm delivery, and preeclampsia. 70 Biologics can play an essential role in controlling severe asthma, and understanding the risks of these agents in the context of pregnancy is crucial to inform treatment decisions. In general, because monoclonal antibodies transport across the placenta in a linear fashion as pregnancy progresses, it is thought that potential effects on a fetus are likely to be greater during the second and third trimesters of pregnancy. In animal reproduction studies, no evidence of fetal harm has been observed. 71
Of the biologics included in this review, omalizumab is the most studied agent in the context of pregnancy. The Observational Study of the Use and Safety of Xolair during Pregnancy (EXPECT) reported outcomes in 250 pregnant women with asthma who were exposed to at least one dose of omalizumab within 8 weeks of conception or anytime during pregnancy. 72 Nearly all (98.4%) patients were exposed to omalizumab in the first trimester, and 81.8% continued omalizumab throughout pregnancy (median exposure, 8.8 months). 72 Results demonstrated a rate of major congenital anomalies of 8.1%, live birth rate of 99.1%, premature birth rate of 15%, and small for gestation age of 9.7%. 72 In a subsequent study, these results were compared to a disease-matched external comparator cohort, which consisted of 1153 pregnant women with moderate-to-severe asthma and without exposure to omalizumab, called the Quebec External Comparator Cohort (QECC). 73 There was no evidence of increased risk of major congenital anomalies, prematurity, low birth weight, or small size for gestational age among pregnant patients receiving omalizumab compared to QECC. 72 The strength of these results is limited by the observation nature of the study and the small size of the cohorts.
A review of 25 case reports, case series, and cohort studies, which describe outcomes among patients exposed to biologics during pregnancy, found that biologics do not seem to impact maternal or fetal outcomes. 70 The majority of cases identified (298 of 313) describe outcomes after exposure to omalizumab; other cases describe outcomes after exposure to dupilumab, mepolizumab, and benralizumab. 70 There was no published pregnancy data on reslizumab or tezepelumab. 70
Overall, there is insufficient data to inform drug-associated risk with the use of all biologics approved to treat asthma. There are ongoing pregnancy exposure registries for dupilumab, mepolizumab, and benralizumab; there are currently no registries for reslizumab or tezepelumab (Table 3). Further research in this area is needed. Providers who have prescribed biologics to pregnant patients should report adverse outcomes, if they occur, and alert pregnant patients of the option to enroll in pregnancy exposure registries.
Ongoing pregnancy exposure registries for biologics used to treat asthma, as of 10/2024. 74

Summary
Biologics used to treat asthma are generally well-tolerated, with a low risk of serious adverse reactions. The risk of anaphylaxis with the use of these agents is low. Omalizumab and reslizumab have a boxed warning for anaphylaxis, which occurs in 0.2% and 0.3% of patients, respectively. Delayed allergic reactions are infrequently reported. Serum sickness and serum sickness-like reactions have been reported to omalizumab and dupilumab, and erythema multiforme has been reported in a patient receiving dupilumab.
There is no definitive evidence for increased risk of malignancy with any of the biologics reviewed. Studies are limited by the duration of extension trials peaking at 5 years.
Herpes zoster was reported in a small number of patients receiving dupilumab, mepolizumab, and benralizumab in the context of clinical trials. There is otherwise no increased risk of opportunistic or serious infection with the use of biologics reviewed. There is no data confirming an increased risk of helminth infection. A warning to treat helminth infection, if present, prior to starting therapy is included on the product label for each agent in this review.
Package inserts for dupilumab and tezepelumab recommend avoidance of live vaccines.4,7 This recommendation is based on a lack of data confirming the safety of concomitant use of these agents and life vaccines. An expert Delphi panel performed a systemic review of the literature on the safety and efficacy of vaccines in patients who are receiving dupilumab and found that dupilumab does not appear to impact the safety or efficacy of vaccines. 61 The panel advises a shared decision-making approach to the use of vaccines in patients receiving dupilumab. 61
There is insufficient evidence to determine the risks associated with biologic use in pregnant patients. The most studied agent in the context of pregnancy is omalizumab, and results indicate no increased risk of major congenital anomalies, prematurity, low birth weight, or small size for gestational age among pregnant patients receiving omalizumab.
Conclusion
Biologic agents are an effective therapeutic option for many patients who have moderate-to-severe asthma, and they are generally well-tolerated with a low risk of serious adverse effects. It is important to understand the risks associated with the use of these agents, to adequately counsel patients, and to monitor the development of adverse events. Additional research is needed regarding risks associated with live vaccine use among patients receiving certain biologics, as well as risks associated with the use of biologics in the context of pregnancy, to help guide shared decision-making and avoid unnecessary interruptions in therapy.
