Abstract
Asthma is a disease characterised by heterogeneous and multifaceted airway inflammation. Despite the availability of effective treatments, a substantial percentage of patients with the type 2 (T2)-high, but mainly the T2-low, phenotype complain of persistent symptoms, airflow limitation, and poor response to treatments. Currently available biologicals target T2 cytokines, but no monoclonal antibodies or other specific therapeutic options are available for non-T2 asthma. However, targeted therapy against alarmins is radically changing this perspective. The development of alarmin-targeted therapies, of which tezepelumab (TZP) is the first example, may offer broad action on inflammatory pathways as well as an enhanced therapeutic effect on epithelial dysfunction. In this regard, TZP demonstrated positive results not only in patients with severe T2 asthma but also those with non-allergic, non-eosinophilic disease. Therefore, it is necessary to identify clinical features of patients who can benefit from an upstream targeted therapy such as anti-thymic stromal lymphopoietin. The aims of this narrative review are to understand the role of alarmins in asthma pathogenesis and epithelial dysfunction, examine the rationale underlying the indication of TZP treatment in severe asthma, summarise the results of clinical studies, and recognise the specific characteristics of patients potentially eligible for TZP treatment.
Introduction
Asthma is a disease characterised by heterogeneous and complex airway inflammation. Approximately 70% to 80% of inhaled corticosteroid (ICS)-naïve asthmatic patients and 50% of ICS-treated patients have an elevated sputum eosinophil count, 1 which is associated with increased expression of type 2 (T2) cytokines. The process involves interleukin (IL)-4, IL-5, and IL-13, 2 an augmented exhaled nitric oxide (FeNO) fraction 3 and blood eosinophil count (BEC), and a T2 inflammatory gene signature. 3 The main feature of this “T2-high” phenotype is the response to ICSs and, in the most severe stage, to monoclonal antibodies (mAbs) against these T2 cytokines. 4 Despite the availability of these effective treatments, a significant percentage (varying between 15% and 25%) of patients with T2-high disease, as well as many patients with T2-low disease (the latter characterised by normal eosinophil counts in both the sputum and peripheral blood as well as low FeNO and T2 cytokine levels), have persistent symptoms associated with airflow limitation and a poor response to inhaled and/or systemic corticosteroids.5,6 Because of the currently limited therapeutic options, these patients have often represented a clinical challenge. Furthermore, numerically normal sputum eosinophil counts have been detected in approximately 25% of patients with untreated symptomatic asthma 7 and in 40% to 50% of patients with asthma requiring therapy with high ICS doses. 8 T2-low asthma is usually more common in mild to moderate disease (64% to 73% of cases).9,10
Currently available biological agents target single or multiple T2 cytokines (as in the case of dupilumab), while no mAbs or other specific therapeutic options are available for non-T2 asthma. However, targeted therapy against epithelial cytokines (the so-called alarmins), including IL-25, IL-33, and especially thymic stromal lymphopoietin (TSLP), is radically changing this perspective.11 –13 Given its upstream position in the inflammatory cascade, TSLP may exert a broad influence on inflammation through its impact on numerous cells and different pathways. Therefore, treatments targeting TSLP provide an innovative and very promising approach to modulate airway inflammation. 12
The aims of this narrative review were to highlight the role of alarmins in asthma pathogenesis and epithelial dysfunction, examine the rationale underlying the treatment of severe asthma (SA) with the first anti-TSLP mAb, tezepelumab (TZP), a human mAb that binds and inhibits TSLP, summarise the results of clinical studies, and, above all, identify particular characteristics of patients potentially eligible for TZP treatment.
Methods
Data source and study selection
We conducted a review of English language literature regarding epithelial dysfunction in SA, clinical trials, and reviews on TZP published from the beginning of the databases examined until January 2024. The biomedical bibliographic databases consulted included MEDLINE (PubMed), Scopus, Web of Science, Google Scholar, and Embase. The search terms used included “severe asthma”, “epithelial dysfunction”, “alarmins”, “TSLP”, “biologics”, “monoclonal antibodies”, “biomarkers”, “clinical studies”, “exacerbations”, and “lung function”. This article is guided by the Scale for the Assessment of Narrative Review Articles (SANRA). 14
Alarmins and their role in the immune system
The alarmin family is composed of a series of multifunctional, structurally and evolutionarily unrelated endogenous molecules. These molecules can be released by necrotic cells in the case of infection or tissue injury or secreted by leukocytes and following epithelial stimulation or damage. 15 When released extracellularly, alarmins result in the activation of innate immune cells and the recruitment and activation of antigen-presenting cells. 16 These cells are involved in host defence and increased susceptibility to infections as well as tissue repair through pattern recognition receptors, such as toll-like receptors, which play an important role in pathogen detection. 17 As potent mediators of inflammation, alarmins play a key role in the pathogenesis of a wide range of immune and inflammatory disorders also induced by infections. In particular, their ability to enhance the adaptive immune response through their effects on antigen-presenting cells, including dendritic cells, makes them a critical link between the innate and adaptive arms of the immune response. 18 Numerous studies have shown that lower respiratory tract infections such as those caused by respiratory syncytial virus represent a significant risk factor for the onset of respiratory diseases in childhood as well as in adulthood. 19 Other authors revealed that respiratory syncytial virus infection leads to airway epithelial barrier dysfunction in the absence of cell death. 20
Role of alarmins in the pathogenesis of asthma
T2-high asthma
TSLP and IL-33 play key roles as drivers of T2 inflammation. These “alarmins” are released mainly by ciliated epithelial cells, mast cells, macrophages, and endothelial cells at the initial site of the inflammatory trigger, 21 where local and systemic adaptive and innate immune responses are activated. 22 Epithelial cells produce and are themselves a target of TSLP. This alarmin belongs to the IL-2 family, and its structure is related to that of IL-7. 23 There are two isoforms of TSLP; the first is characterised by a longer sequence (159 amino acids), and the second has a shorter sequence (60 amino acids). 24 The short variant is the constitutive form and exerts homeostatic functions, while the longer variant exhibits inducible TSLP synthesis, stimulated by inflammatory factors, and it is up-regulated in asthma. 25 The positive charges of the long TSLP surface interact with the negative charges of the TSLP receptor (TSLPR), and the TSLP/TSLPR complex incorporates the α subunit of the IL-7 receptor (IL-7Rα). 26 The result of this sequential process leads to the assembly of the ternary TSLP/TSLPR/IL-7Rα molecular complex. The latter activates an intricate network of signalling pathways, including signal transducers and activators of transcription 3 and 5 (STAT3/5), Janus kinases 1 and 2 (JAK1/2), mitogen-activated kinase (MAPK), phosphoinositide 3 kinase (PI3K), and nuclear factor κB (NF-κB) (Figure 1).
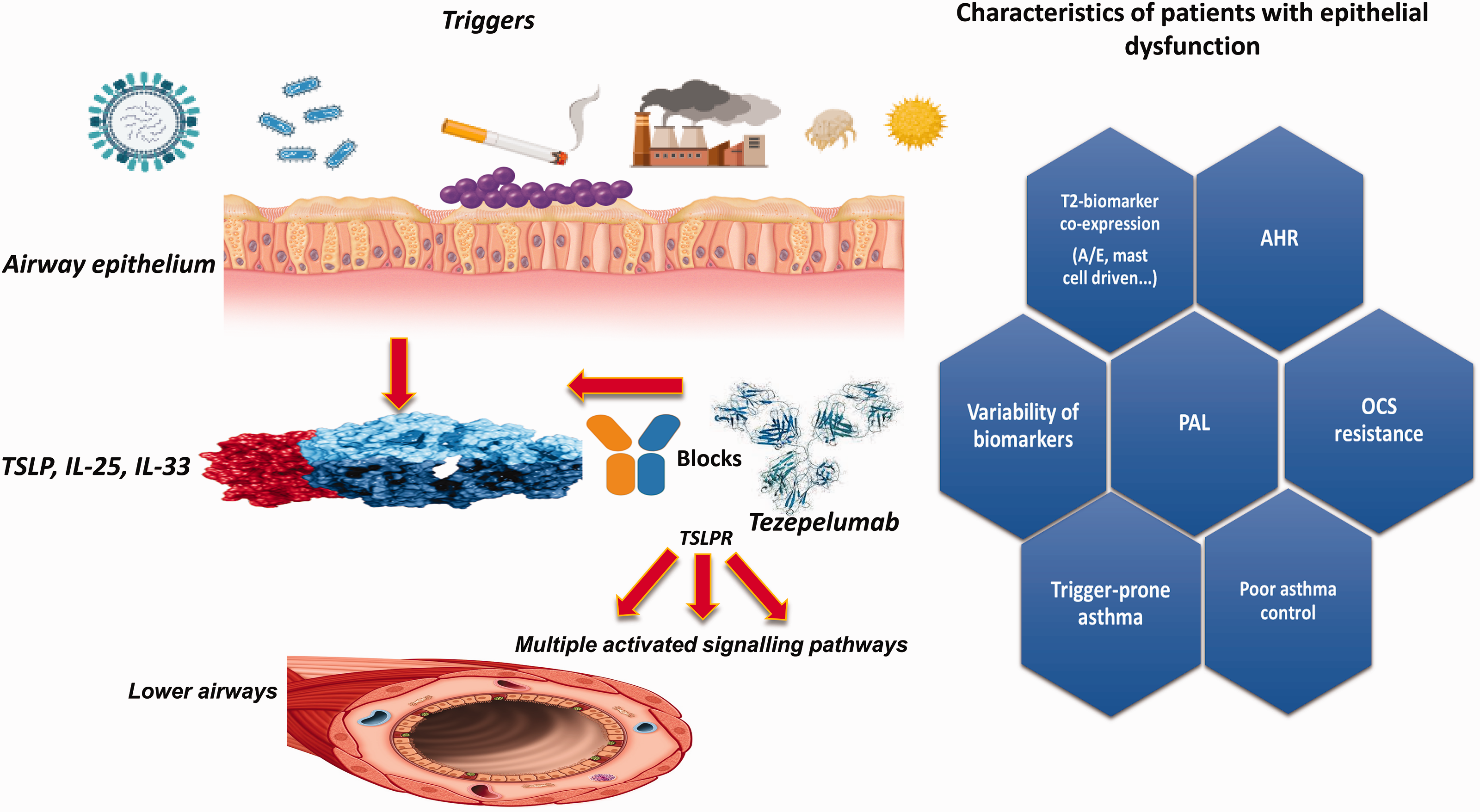
Mechanism of action of tezepelumab and clinical phenotype characteristics of patients with epithelial dysfunction. TSLP, thymic stromal lymphopoietin; TSLPR, thymic stromal lymphopoietin receptor; IL, interleukin; AHR, airway hyperresponsiveness; PAL, persistent airflow limitation; T2, type 2; OCS, oral corticosteroid; A/E, allergic/eosinophilic.
Patients with allergic and eosinophilic asthma phenotypes have higher IL-33 serum levels (belonging to the IL-1 family) than those with different phenotypes. 27 In patients with allergic asthma, the expression of ST2, the soluble receptor for IL-33, on eosinophils in blood and sputum is significantly upregulated after allergen inhalation. 28 Higher levels of ST2 in the serum correlate with an increased risk of exacerbation. 29 TSLP has been shown to play a crucial role in various steps of asthma pathophysiology, including the aforementioned remodelling, airway hyperresponsiveness (AHR), and mucus production. During T2 inflammatory responses, TSLP potently activates dendritic cells and induces the production of chemokines that attract T helper cell type 2 (Th2) lymphocytes, thymus and activation-regulated chemokines (TARC, also known as CCL17), and macrophage-derived chemokines. TSLP-activated dendritic cells drive naïve T helper cells to produce several inflammatory mediators, most notably IL-4, IL-5, and IL-13. 30 TSLP activates not only Th2 cells but also many other cell types that release IL-13, including group 2 innate lymphoid cells (ILC2s), mast cells, and basophils.31,32 IL-33 stimulates AHR through IL-13-mediated crosstalk between mast cells and smooth muscle cells. Furthermore, human pulmonary mast cells are stimulated by IL-33 to release histamine and IL-13, with subsequent airway constriction. 33 IL-33 also determines the survival of mast cells, 34 eosinophils, and basophils, as well as the release of IL-13 by ILC2s. 35
Of the three alarmins, the role of IL-25 is the least known. IL-25 appears to play an important role in modulating allergic inflammation, especially as a driver of the T2 inflammatory response during virus-induced asthma exacerbations. 36 Elevated serum levels of IL-25 are not only associated with the allergic asthma phenotype, the concentration of IL-25 in sputum also correlates with disease severity. TSLP remains the most important and most widely produced alarmin, while the others are quantitatively produced to a lesser extent and only by a limited number of cells, with the main ones being epithelial cells.37,38
T2-low asthma
The role of alarmins is still debated in T2-low asthma. Most of the available data were generated by animal studies, whereas relatively few studies have been carried out on human cells or humans. Among the alarmins, TSLP is the most relevant to T2-low asthma. This evidence is derived mainly from the demonstration that anti-TSLP treatment reduces exacerbations in patients with severe refractory asthma, even those with low blood eosinophil and FeNO levels. 39 TSLP may play a role in T2-low neutrophilic airway inflammation by activating dendritic cells, resulting in polarisation of naïve T-cells towards a Th17 phenotype. 40 Th17 polarisation and the release of IL-17 promote neutrophilic inflammation and the T2-low response. 40 Expression of TSLP, but not IL-33 or IL-25, in human bronchoalveolar lavage (BAL) has been shown to correlate closely with neutrophil infiltration. 41 However, further studies are needed to better understand the role of alarmins in the pathogenesis of T2-low asthma.
Epithelial dysfunction as a risk factor for SA
The epithelial lining of the respiratory tract forms a solid physical-chemical barrier that protects against inhaled toxic and irritant particles and pathogens. Inhalation of these environmental substances or physical triggers can impair the physiological function of the airway epithelial barrier and lead to exaggerated inflammatory responses and long-term airway remodelling.42,43 Consequently, this epithelial barrier dysfunction increases susceptibility to respiratory infections, leading to an increased risk of exacerbations and triggering further inflammation in a vicious circle. Furthermore, airway epithelial cells are essential components and structurally part of the lung’s innate immune system. The resulting inflammation leads to the release of a wide variety of cytokines and chemokines that cause profound damage to the respiratory epithelia, also leading to thickening of the airway wall and the restriction of airflow. The damage to the epithelial layer of the airways in asthma is characterised by the detachment of hair cells, the presence of epithelial cell aggregates (“Creola bodies”) in sputum, increased permeability to allergens, and reduced expression of hair cells and the cell–cell adhesion molecule E-cadherin.44,45 These pathological features are present in all asthma phenotypes 46 and are observed very early, even in paediatric age, before the clinical diagnosis of asthma. 47
Th1, Th2, and Th17 cytokines play a pivotal role in the pathogenesis of asthma, contributing to epithelial damage.20,48 Exposure to air pollutants activates these adaptive immune responses and increases the release of their mediators, including pro-inflammatory cytokines such as interferon-γ, tumour necrosis factor-α, IL-13, IL-17A, and IL-22. 48 Increased IL-17A levels suppress E-cadherin expression during asthma exacerbations and promote mitochondrial dysfunction, leading to increased mitochondrial reactive oxygen species levels and a further reduction in cellular integrity. 49 Impaired barrier function is then a consequence of altered expression of E-cadherin, β-catenin, and the transmembrane proteins zona occludens-1 (ZO-1), occludin, and junction adhesion molecules, which are the main controllers of the permeability of the airway epithelium of asthmatic patients.50,51 However, despite these data, the mechanisms leading to increased sensitivity of the airways to environmental factors have not been fully explained. Damaged epithelium in response to allergens, pollutants, and viral respiratory infections releases three alarmins, IL-25, IL-33, and TSLP. Systemic, especially inhaled, glucocorticoids influence inflammation by modulating cytokine production by immune cells and promoting the recovery of barrier function by the airway epithelium.52 –54 The development of specific molecular-targeted therapies, such as TZP, will offer a more targeted action on inflammation and a more specific therapeutic effect against epithelial dysfunction.
Mechanism of action and role of TZP in asthma
TZP is a first-in-class fully human IgG2λ mAb that specifically binds to TSLP at its binding site for cognate heterodimeric TSLPR, antagonising the human TSLP–TSLPR interaction (Figure 1). 55 Both TSLPR and IL-7R are co-expressed on the membrane of eosinophils, which thus become sensitive to the activating and anti-apoptotic effects of TSLP, which stimulates the release of cytotoxic proteins and chemokines from several cells.56,57 The biological actions of TSLP are mediated by TSPLRs via MAPK and NF-kB activation-based signalling pathways. 57 As a consequence of this antagonising effect, TZP inhibits the production of numerous cytokines and the activation of different types of inflammatory cells. In particular, this mAb has been shown to reduce the tissue eosinophil count and BEC, IgE, IL-5, IL-13, and FeNO, in addition to restoring the physiological epithelial barrier function. 58
In 2018, the US Food and Drug Administration granted TZP a breakthrough therapy designation for patients with SA, without a high T2 phenotype, treated with ICSs/long-acting beta2-agonists with or without oral corticosteroids (OCSs) and additional asthma controllers.
59
This award was based on data from phase IIb of the PATHWAY study.
39
In this randomised clinical trial (RCT), TZP significantly reduced annual asthma exacerbation rates by 62%, 71%, and 66% in the TZP active arms receiving three dosing regimens—70 mg or 210 mg every 4 weeks or 280 mg every 2 weeks compared with placebo (
Innovative evidence from clinical trials of TZP
After the PATHWAY study, two other phase II RCTs were carried out that proved interesting and had conceptually innovative results. The first was the phase II UPSTREAM trial. 60 In this study, adult patients with asthma and AHR to mannitol received 700 mg TZP or a placebo intravenously at 4-week intervals for 12 weeks. The primary outcome was the change in provoking dose that caused a 15% decrease (PD15) in forced expiratory volume in the first second (FEV1)—expressed as doubling doses—to inhaled mannitol from baseline to week 12. Moreover, bronchoscopy was performed at baseline and after 12 weeks to assess secondary outcomes (changes in airway inflammation). AHR to mannitol improved from baseline to treatment week 12 in patients treated with TZP compared with those treated with placebo, with a mean change in PD15 of 1 to 9 dose doublings (95% confidence interval (CI): 1–2 to 2–5). The improvement in PD15 was more noticeable in patients with eosinophilic asthma. From baseline to week 12, eosinophil levels in airway tissue were reduced by 74% (95% CI: 46%–87%) in the TZP-active group. Treatment with TZP reduced total tissue mast cells by 25% (95% CI: −47%–6%), compared with that of the placebo group, and eosinophils in BAL, sputum, and blood samples were significantly reduced with TZP compared with those in the placebo arm. Exhaled FeNO decreased by 48% (95% CI: −33% to −60%) in patients treated with TZP. In contrast, FEV1 and forced expiratory flow at 25% to 75% of the vital capacity did not improve significantly compared with those at baseline in either group.
As a whole, these results contrast with other studies on anti-IL-5 biologics, which did not show an improvement in allergen-induced bronchoconstriction, and strongly support the hypothesis that TZP may inhibit TSLP-related mast cell activation and thus attenuate AHR. 61
The second trial was the CASCADE study.
62
In this RCT, adult patients aged 18 to 75 years with uncontrolled moderate SA were randomly assigned (1:1) to receive 210 mg TZP or placebo administered subcutaneously every 4 weeks up to 52 weeks. The primary endpoint was the change from baseline until the end of treatment in the number of airway submucosal inflammatory cells in bronchoscopic biopsy specimens, individually assessing eosinophils, neutrophils, CD3+ T cells, CD4+ T cells, and mast cells with tryptase and chymase. This endpoint was also examined by stratification into subgroups according to the baseline levels of T2 inflammatory biomarkers, including the BEC. Airway remodelling was assessed through secondary endpoints, such as changes from baseline in reticular basement membrane thickness and epithelium integrity. TZP treatment resulted in a significant reduction in submucosal airway eosinophils compared with that of the placebo at baseline (95% CI: 0.05–0.41; nominal
The NAVIGATOR study was a phase III, multicentre, double-blind, placebo-controlled RCT with a primary objective of determining the annual rate of exacerbation of 1061 subjects with SA after subcutaneous administration of 210 mg TZP every 4 weeks.
63
At baseline, enrolled patients had at least two exacerbations in the previous year (40% reported more than three exacerbations) and a homogeneously distributed BEC greater than or less than 300 cells/µL (25% < 150/µL and 25% > 450/µL). The annual exacerbation rate was 0.93 for patients treated with TZP compared with 2.10 for the placebo group (
This finding might better predict the results in ongoing RCTs with the same primary endpoint, the evaluation of efficacy in reducing OCS use in adult patients with OCS-dependent asthma, including the WAYFINDER (ClinicalTrials.gov number NCT05274815) and SUNRISE (ClinicalTrials.gov number NCT05398263) trials. Of the two studies, only the WAYFINDER trial has preliminary results: the maintenance dose of OCS was reduced from 10.9 mg/day to ≤5 mg/day in 90.6% of patients who reached week 20 of the study compared with the total 52 weeks planned, without loss of asthma control. 65
Regarding the safety profile, phase II and III studies reported no noteworthy adverse reactions. As with other biologics, it is crucial to assess safety in the long term, which was the aim of the Destination extension study. 66 In this RCT, after 104 weeks of treatment with TZP, the same results were reported as in the NAVIGATOR and SOURCE studies with regard to safety (low incidence of side effects) and efficacy (sustained reduction in the rate of exacerbations). These data were independent of the baseline eosinophil count, FeNO level, and atopy status, although patients with high levels of T2 biomarkers or chronic rhinosinusitis with nasal polyposis (CRSwNP) exhibited better results in terms of reduced exacerbations. Notably, a numerical imbalance in serious cardiac events was found in the TZP group compared with the placebo group. However, a cause–effect relationship has not been established between TZP and these events, nor has a subpopulation of patients at increased risk been identified. In any case, the incidence rates of serious cardiac adverse events with TZP were comparable to those estimated from published data with other biologics for SA. 67
Position of TZP and main characteristics for appropriate patient selection
Because the target and therapeutic effect of TZP is caused by the upstream location of the inflammatory cascade, and according to the results in both T2-high and T2-low asthma, the candidate profile for treatment with TZP varies greatly and has not been fully defined. In fact, the clinical and biologic features may differ from those of target patients for currently registered biologics but may also share elements in common with them. Ongoing studies and future real-life evidence will allow us to better define the role of this biologic in the treatment regimen of SA. However, possible preliminary indications have been obtained from RCTs (Table 1). Because TZP acts by antagonising the main epithelial cytokine, it is crucial to define the possible clinical features of patients with epithelial dysfunction who can be considered among those evaluated for biologic treatment. In particular, data on its effects on AHR are also useful in providing information to guide treatment choices.
At-a-glance overview on published RCTs on tezepelumab.
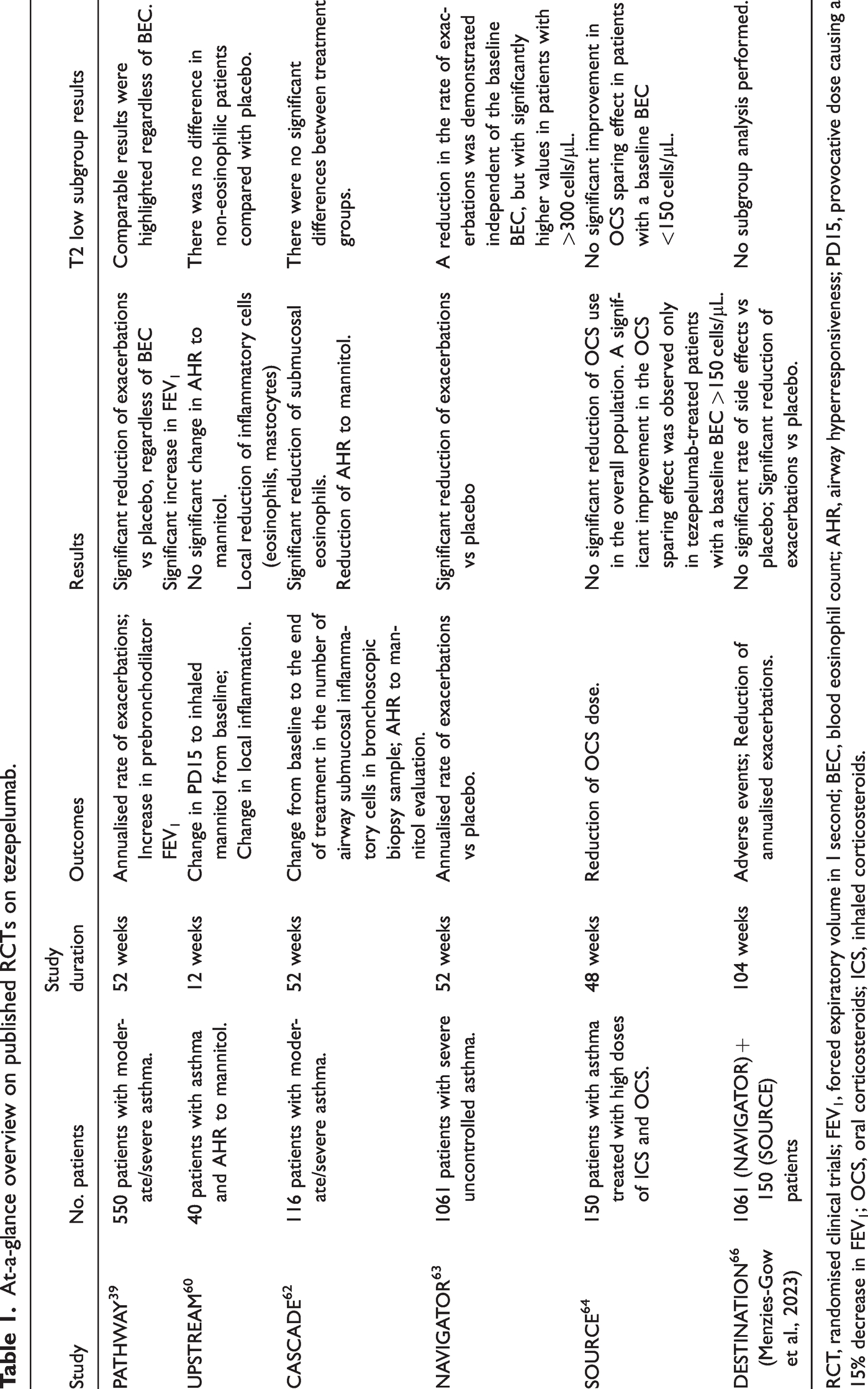
RCT, randomised clinical trials; FEV1, forced expiratory volume in 1 second; BEC, blood eosinophil count; AHR, airway hyperresponsiveness; PD15, provocative dose causing a 15% decrease in FEV1; OCS, oral corticosteroids; ICS, inhaled corticosteroids.
T2 non-allergic eosinophilic asthma
The PATHWAY and NAVIGATOR studies confirmed a significant reduction in exacerbations (61.7%–66%) and a similar favourable result for TZP for allergic and non-allergic phenotypes. A post hoc analysis of the PATHWAY study showed no difference in the reduction of exacerbations according to eosinophilic phenotype.39,63 In both studies, TZP reduced eosinophils, FeNO, and total IgE, demonstrating its action on multiple T2 pathways, including IL-5, IL-4, and IL-13. Furthermore, in a pooled analysis of data from the PATHWAY and NAVIGATOR studies, TZP reduced the rate of severe exacerbations requiring hospitalisation or an emergency room visit by 90% in the subgroup of patients with a BEC of at least 300 cells/μL. 68 A confirmation of the high efficacy in the eosinophilic phenotype also emerged from the results of the SOURCE study, in which the subgroup of subjects with a baseline BEC ≥ 150 cells/µL alone exhibited higher odds of achieving a reduction in daily OCS dose with TZP treatment than with placebo. 64
T2 allergic asthma
The results of the PATHWAY and NAVIGATOR RCTs showed that TZP significantly reduced exacerbations in patients with a BEC <250 cells/µL (up to <150 cells/µL in the NAVIGATOR RCT).39,63 However, this favourable improvement was lower than that observed in patients with eosinophilic asthma. In fact, among the subgroup of patients with perennial aeroallergen sensitisation, a pooled analysis of PATHWAY and NAVIGATOR data reported a reduction in the annualised asthma exacerbation rate of 62% and a reduction in the rate of exacerbations requiring hospitalisation or emergency room admission of 80% at the end of 52 weeks of treatment.68,69
T2-low asthma
TZP showed positive results in patients with severe non-allergic, non-eosinophilic asthma. In the pooled analysis of PATHWAY and NAVIGATOR data, 210 mg TZP every 4 weeks reduced the annualised asthma exacerbation rate over 52 weeks by 54% in patients without sensitivity to aeroallergens and by 48% in patients with a BEC of less than 300 cells/μL or less than 150 cells/μL, compared with the placebo.
68
Similar results were observed in patients without sensitivities and with a low BEC at baseline. Exacerbations requiring hospitalisation or emergency room admission decreased by 74% in patients without sensitivity to perennial aeroallergens and by 67% (95% CI: 41–88) in patients with a BEC of less than 300 cells/μL or less than 150 cells/μL, compared with the placebo group. Evaluation of the results in the group of patients with eosinophils >300 cells/μL showed that the annual exacerbation rate was 0.93, compared with 2.10 for placebo (
Clinical and phenotypic characteristics of patients with epithelial dysfunction
Despite the existence of numerous clinical endotypes, most patients with asthma present with a dysregulated epithelial barrier. It is important to define the main characteristics of patients in whom epithelial dysfunction has the greatest impact on the pathogenesis of asthma, which can help identify those who can most benefit from the action of a drug that acts directly at that level. Many clinical features of asthma are associated with TSLP, but it should be noted that the damage begins very early in life. Histopathological data on endobronchial biopsies taken from paediatric patients have revealed changes indicative of airway remodelling, characterised by basement membrane thickening and smooth muscle hypertrophy, with varying degrees of goblet cell and submucosal gland hyperplasia. 70
Various types of environmental insults on airway epithelial cells can initiate apoptosis or programmed cell death of the epithelium. 71 This is accompanied by the release of soluble paracrine factors, such as transforming growth factor β, which can initiate the tissue regenerative process to restore homeostasis. 72 As a consequence, persistent damage and prolonged stimulation by growth factors result in aberrant tissue repair, leading to airway remodelling. In this process, in addition to epithelial cells playing a role as “initiators”, immune cells intervene as “amplifiers” and mesenchymal cells act as “effectors” of remodelling; the latter cells differentiate into myofibroblasts. 73 An increase in the expression of TSLP and IL-33 in smooth muscle was detected in these patients, confirming the close correlation with the epithelial trigger. 74 TSLP promotes asthmatic airway remodelling also through activation of the p38 MAPK-STAT3 axis and crosstalk between epithelial cells and fibroblasts. 75 As a further insight into this notion, using BAL, one study demonstrated that median concentrations of IL-33, TSLP, IL-4, IL-5, and IL-13 were increased in patients with persistent airway inflammation and impaired lung function despite intensive corticosteroid therapy. 76
Another study showed that ILC2s detected in BAL from asthma patients with increased TSLP levels were resistant to corticosteroids. 77 The existence of patients with barrier damage and increased TSLP levels induced by stimulation with toll-like receptor 3 ligand and T2 cytokines implies increased susceptibility to viral infections and amplification of Th2 inflammation. 78 Following a viral infection, TSLP release from bronchial epithelial cells of patients with asthma increases, resulting in an exaggerated T2 response to viral infections. These findings may explain how viruses can cause exacerbations in asthma patients with or without atopy but in the presence of epithelial barrier damage. 79
In an interesting study, BAL and bronchial epithelial cells were obtained from patients with uncontrolled asthma before and after 12 weeks of TZP treatment. 80 The cells were cultured in vitro and exposed to the viral infection mimic poly(I:C) or rhinovirus mimic viral infection. Blocking TSLP with TZP in vivo in patients with asthma significantly reduced the inflammatory response of the airway epithelium, including T2 cytokines and IL-33, without affecting the host’s antiviral response. This result confirms that TSLP blockade improves the immune response of the bronchial epithelium to respiratory viruses. Confirming this claim, the interesting phase III Vector study showed that after influenza vaccination in adolescent and young adult subjects with moderate to SA treated with TZP, the lack of suppression of the humoral immune response was highlighted. 81
Based on the data presented to date and on other important evidence, it is possible to explain the precise characteristics of patients who can benefit from an upstream targeted treatment, such as the anti-TLSP biologic (Figure 1):
Poor asthma control despite inhalation therapy with ICS-long-acting beta(2)-agonists (GINA step 4/5, 2023). Steroid resistance. Presence of persistent airflow limitation. Susceptibility to viral and/or bacterial infections (innate immunity deficiency). AHR. Co-expression of T2 biomarkers: This type of patient typically has multiple activated T2 pathways; for example, (a) patients with allergic eosinophilic disease, for whom it can be difficult to discern whether exacerbations are primarily driven by allergen exposure or are related to eosinophilic inflammation; (b) subjects with predominantly mast cell-driven asthma or those without a clear T2 inflammation pattern; (c) patients with eosinophilic inflammation without allergy but who concurrently have a very high FeNO level, indicating concomitant activation of both IL-5 and IL-13, perhaps through activation of ILC2s; and (d) patients with severe eosinophilic asthma and comorbidities such as CRSwNP, who may also have co-activation of cells releasing both IL-5 and IL-4/IL-13.
82
In these cases, it is plausible that broader suppression of T2 pathways is needed. Furthermore, in patients with SA and a predominant T2-high phenotype, concomitant neutrophilic inflammation is the hallmark of more severe disease. In this regard, recent data have demonstrated the coexistence of neutrophils and eosinophils in SA
41
and that a more rapid decline in respiratory function occurs in patients with mixed neutrophilic and eosinophilic inflammation. Other studies have reported that rhinovirus-induced extracellular neutrophil trapping is involved in exacerbations of T2 asthma.
83
Patients with variability or iatrogenic suppression of biomarkers. The efficacy demonstrated in pivotal studies and post hoc analyses independent of biomarker expression also makes patients with variability or suppression of different biomarkers, such as in the case of blood eosinophils during OCS therapy, eligible for TZP therapy.
Conclusion
Biological drugs represent an extraordinary innovation and an opportunity that has truly improved the quality of life of many patients. Unfortunately, the biological drugs currently available for SA cannot achieve significant clinical improvement in all patients with T2-high phenotypes. In addition, there are no head-to-head studies between mAbs, and only indirect comparisons and real-world studies are available. The burden of disease is also exacerbated in patients with T2-low asthma, for whom TZP, as mentioned above, represents the first biologic option. This important limitation of current biological agents is probably because of the selective inhibition of a single (anti-IL-5, anti-IgE) or double pathway (anti-IL-4/IL-13), leaving others still active. The upstream block of alarmins including TSLP may have a wide-ranging inflammatory effect (not only targeting T2-high phenotypes). Therefore, this new and promising approach seems to offer effective treatment to a larger and more heterogeneous patient population with different phenotypes. Another possible advantage of TZP is its use in cases of SA, despite the detection of T2 biomarkers within the normal range. Nonetheless, the future availability of specific biomarkers for TSLP-mediated inflammation could better support the choice of this mAb. Furthermore, the identification of trigger-prone patients based on clinical history is an additional indication of TZP, despite the difficulty of assessing bronchial hyperreactivity with specific tests in patients with SA. However, further studies on the role of alarmins in the pathogenesis of asthma are needed and may improve our understanding of the immunological pathways in the different endotypes and clinical phenotypes of asthma.
Another aspect that is currently an unmet need is the insufficiency of data on TZP in asthma and comorbidities, such as CRSwNP and bronchiectasis. Such studies are crucial to further refine the role of this biologic and increasing its indications (as has occurred previously with other mAbs). These studies may improve our ability to correctly select the most appropriate treatment given the growing number of different therapeutic options, for which it is necessary for clinicians to have in-depth knowledge of the mechanisms of action and immunomodulatory effects. It is hoped that biomarkers will soon be available; however, currently available information can allow us to adequately manage SA. In this regard, studies using real-world evidence settings and head-to-head prospective trials could also be useful for improving the application of new anti-alarmin agents. Among these, TZP is the first effective and safe mAb to be available and represents another promising option for a more personalised approach in the treatment of severe refractory asthma. Prospectively, additional studies are needed to better assess the safety and clinical efficacy in real life, supported by better knowledge of the role of single alarmins in the pathogenesis of asthma at an experimental level. Such studies should be conducted considering the outcomes emerging from pivotal studies of other biologics. 84
Footnotes
Author contributions
FM conceptualised and planned the article and wrote the draft of the manuscript. SM, LC, ST, WC, AB, AF, and CD contributed to, reviewed, and edited the manuscript. GES contributed to, reviewed, and revised the final manuscript. All the authors read and approved the final version.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
