Abstract
Background:
Impaired quality of life (QoL) among pulmonary arterial hypertension (PAH) patients has been often attributed to increased symptomatology, functional disability, and poor mental health; however, the unique impact that PAH treatments may exert on the daily lives of patients remains underexplored.
Objectives:
To gain insight into the day-to-day challenges associated with receiving non-oral PAH treatments, and the specific impact these may exert on patients’ QoL, above and beyond that exerted by PAH itself.
Design:
Explanatory sequential mixed methods design.
Methods:
Eighty-three PAH patients provided information on demographic and clinical characteristics, and completed measures of symptomatology, functional disability, QoL, psychological well-being, and perceived stress. Nine of these patients also participated in a focused group discussion and interviews.
Results:
No group differences in symptomatology and functional disability were observed between patients receiving oral-only and non-oral PAH therapy; however, patients on non-oral therapy reported poorer QoL, after taking into consideration the effect of relevant covariates (i.e., age, level of symptomatology and functional disability, psychological well-being, and perceived stress) that could have confounded the observed group differences in QoL. Participants who started on non-oral medications acknowledged they had experienced significant improvements in health status. However, they also stated that transitioning from oral to non-oral therapy elicited great apprehension and that non-oral therapy regimens interfered with daily activities, resulted in added difficulties for self-management, and negatively impacted their subjective well-being.
Conclusion:
Non-oral therapy regimens may pose challenges beyond those posed by PAH itself, potentially resulting in an added burden to the QoL of PAH patients.
Keywords
Introduction
Pulmonary arterial hypertension (PAH) is a progressive and incurable condition characterized by increased pulmonary arterial pressure and pulmonary vascular resistance, leading to right ventricular failure and premature death if untreated. Patients with PAH frequently report shortness of breath, fatigue, weakness, and chest pain, which can result in considerably impaired physical function. 1
The introduction of PAH-specific medications during the early 1990s and their subsequent development resulted in considerable improvements in health status and survival.1,2 However, these advances have also increased the salience of issues related to quality of life (QoL), long-term self-management, and the impact PAH may exert on different life areas beyond health.3–7 Previous research has revealed that PAH patients are afflicted by elevated rates of depressive and anxiety symptomatology and impaired QoL, most often attributed to increased PAH symptomatology, functional disability, and psychosocial issues.8,9 To date, however, the day-to-day challenges associated with receiving PAH treatment, and the specific impact they may exert on QoL, remain underexplored.
A variety of therapeutic agents are used as PAH-specific therapy. These comprise calcium channel blockers, endothelin receptor antagonists, phosphodiesterase type 5 inhibitors, and guanylate cyclase stimulators—all orally administered— and prostacyclin analogs and prostacyclin receptor agonists—most often administered by means of subcutaneous or intravenous infusion or using a nebulizer.1,10 Upon confirmation of a PAH diagnosis, the implementation of PAH therapy is based on risk assessment, whereas patients at relatively low risk may be started on oral therapy, initial combination therapy with intravenous or subcutaneous prostacyclin is recommended for those at high risk for adverse outcomes.1,11 Despite its critical role in successfully managing PAH, recent studies have revealed that adherence to PAH-specific therapy is suboptimal. Higher administration frequency, longer time on therapy, and concomitant therapy are some of the factors that have been associated with lower adherence, specifically to oral medications.12,13 Further, poorly tolerated side effects have been observed to lead to treatment discontinuation.14,15
The administration of medication through non-oral routes can bear added challenges for the self-management of PAH and result in severe health consequences. For example, the intravenous or subcutaneous administration of medication is achieved by means of a catheter and an infusion pump that must be worn permanently. These routes of administration are associated with an increased risk of infections; also, the medication delivery system itself may lead to further complications, as infusion pumps may malfunction and catheters may become obstructed. 16 The subcutaneous infusion of medication can further result in significantly high levels of site pain. 14 Patients taking their medication by means of inhaled administration must take several doses per day, normally between six and nine 17 ; this may cause interruptions in daily activities and/or require that patients plan their day around their medication schedule.
The present study
The objective of this study was to gain insight into the day-to-day challenges associated with receiving non-oral PAH treatments and the specific impact these may exert on patients’ QoL, above and beyond that exerted by PAH itself. Based on patients’ accounts, the burden associated with receiving non-oral medications constitutes a highly relevant issue.3,14 However, previous research focused on highlighting the perspective of PAH patients has not addressed this issue in detail.
First, this study assessed the magnitude of differences in QoL between participants receiving oral-only and non-oral PAH-specific therapy. Second, a focus group discussion and interviews were performed to shed light on differences in QoL that may be attributed to the type of treatment (i.e., oral vs non-oral). With these objectives in mind, it was hypothesized that participants receiving non-oral therapy would report lower QoL than those receiving oral therapy alone.
Methods
Study design and procedure
This study followed an explanatory sequential mixed method design. 18 Inclusion criteria comprised patients of at least 18 years of age, with a confirmed diagnosis of PAH, and receiving PAH-specific treatment. Participants were recruited from the Spanish National Association of Pulmonary Hypertension. The study was introduced to participants at annual meetings held by the association and was further promoted to other potential participants using the snowball sampling method. Data were collected from 2016 to 2021. Participants self-reported demographic and clinical information (Table 1) and completed the instruments described below. In addition, a focus group discussion and interviews (moderated by the author) were conducted with a subgroup of nine participants; the selection of these participants followed purposive sampling, 19 and was based on good communication skills and experience with oral and non-oral PAH treatments. The use of a qualitative approach was deemed necessary to complement quantitative analyses with elaborate patient accounts of the challenges associated with receiving non-oral treatments and the impact they may have on daily life. 18 This study adhered to the Strengthening the Reporting of Observational Studies in Epidemiology guidelines (STROBE) and the Consolidated Criteria for Reporting Qualitative Research (See Supplemental File).19,20
Demographic and clinical characteristics of participants (N = 83).
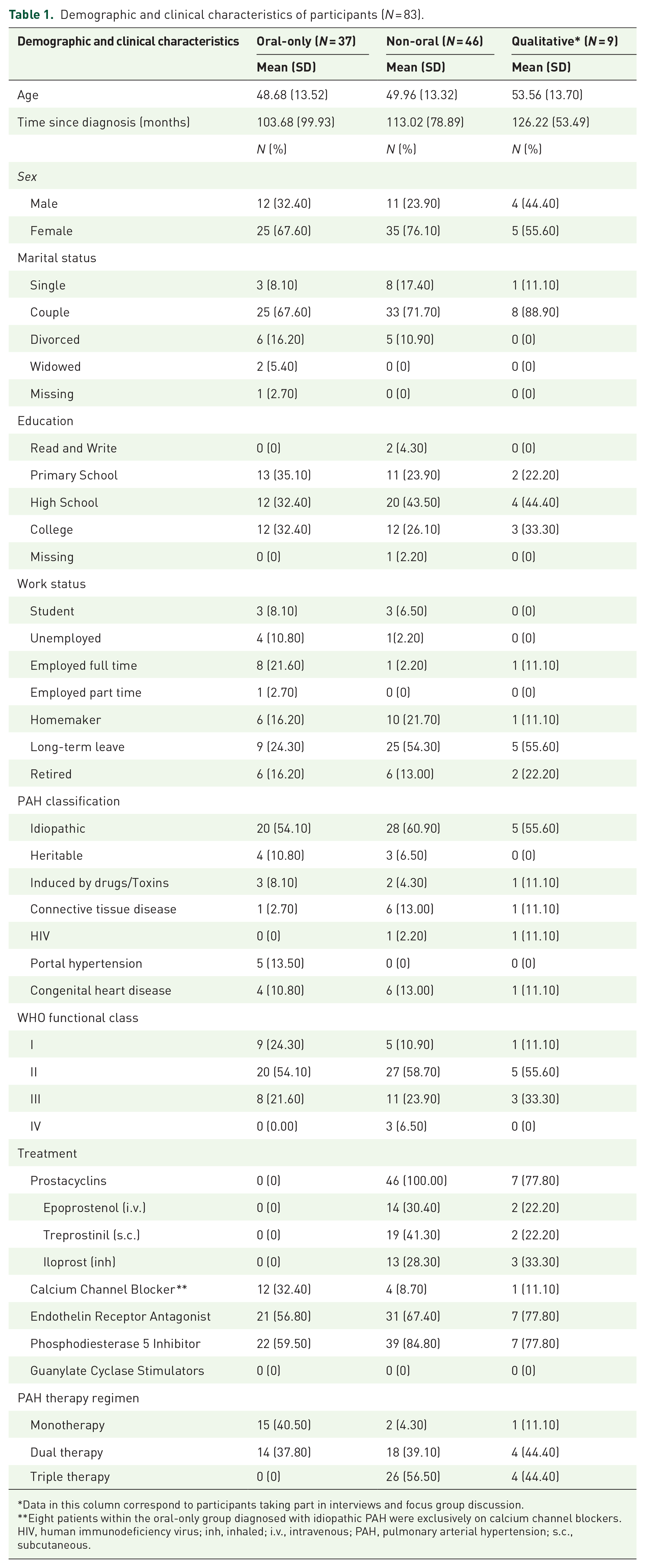
Data in this column correspond to participants taking part in interviews and focus group discussion.
Eight patients within the oral-only group diagnosed with idiopathic PAH were exclusively on calcium channel blockers.
HIV, human immunodeficiency virus; inh, inhaled; i.v., intravenous; PAH, pulmonary arterial hypertension; s.c., subcutaneous.
Instruments
Cambridge Pulmonary Hypertension Outcome Review (CAMPHOR).21,22 The CAMPHOR is divided into three subscales. The symptoms subscale contains 25 items answered using a “yes”/”no” format, which results in a score ranging from 0 to 25, and evaluates the presence of symptoms commonly present in patients with PAH. The activities subscale evaluates functional disability by inquiring whether patients can perform a series of activities using three response options: “able to do on own without difficulty,” “able to do on own with difficulty,” and “unable to do on own”; this subscale comprises 15 items and results in a score ranging from 0 to 30. The third subscale, QoL, comprises 25 questions answered using a “true”/”false” format and leads to a score ranging from 0 to 25; these questions inquire about needs that are commonly frustrated by PAH. Higher scores on each of the three subscales indicate higher levels of symptomatology and functional disability, and lower levels of QoL. The CAMPHOR minimal clinically important difference (i.e., clinical significance) has been assessed using different methods; when averaged, clinically significant changes correspond to a 4-point difference on the symptoms and activities subscales and a three-point difference on the QoL subscale. 23 The internal consistency (Cronbach’s α) obtained from this sample on the symptoms, activities, and QoL subscales were 0.90, 0.91, and 0.87, respectively.
Acceptance and Action Questionnaire II (AAQ-II).24,25 The AAQ-II was originally designed as a measure of psychological flexibility; however, recent studies have found that the AAQ-II may constitute a broader measure of general distress, neuroticism, and negative affect26–28; therefore, the AAQ-II has been used in this study as a proxy measure of psychological well-being. The AAQ-II comprises seven items, answered on a 7-point Likert scale ranging from 1 (“never true”) to 7 (“always true”), which results in a total score ranging from 7 to 49. Higher scores on the AAQ-II denote lower levels of psychological well-being. The internal consistency (Cronbach’s α) obtained from this sample was 0.92.
Perceived Stress Scale (PSS).29,30 The PSS evaluates how unpredictable, uncontrollable, and overloaded respondents find their lives to be. Responses to the PSS are provided using a 5-point Likert scale ranging from 0 (“Never”) to 4 (“Very often”), which results in a score ranging from 0 to 40. Higher scores indicate higher levels of perceived stress and lower levels of perceived control. The internal consistency (Cronbach’s α) obtained from this sample on the PSS was 0.89.
A license was obtained from Galen Research to use the CAMPHOR; no license or permissions were required to use the rest of the questionnaires (see Supplemental File for AAQ-II and PSS items).
Statistical analyses
Data analyses were performed using IBM SPSS Statistics version 23. Frequencies and descriptive statistics were used to obtain information about the sample’s demographic and clinical characteristics and the study variables. An analysis of covariance (ANCOVA) was carried out to assess the magnitude of differences in QoL between patients receiving oral-only and non-oral treatment for PAH, while statistically adjusting for relevant clinical, individual, and psychosocial factors included in the model as covariates (i.e., age, level of symptomatology, functional disability, psychological well-being, and perceived stress).31,32 Based on the ANCOVA, sample size calculations using G*Power 33 indicated that a sample size of 121 would be required to provide 80% power to detect a medium effect size (f = 0.25), with an alpha level of .05. Before conducting the statistical analyses, the data were screened for missing values and outliers, as well as examined for compliance with the statistical assumptions of ANCOVA.
Qualitative data analyses were carried out following the phases of thematic analysis proposed by Braun and Clarke. 34 First, the recordings from the focus group discussion and interviews were transcribed and read several times. Second, codes were generated by two coders (i.e., the author and a collaborator listed under Acknowledgments) following a process of open coding. 35 Third, the codes were organized into themes, which were discussed and refined to get the final naming and description to be included in this manuscript. Last, participants reviewed a draft that included some of their responses organized into themes for comments and correction.
Results
Participants
Eighty-three patients were enrolled in this study; 37 were receiving oral-only treatments and 46 were receiving non-oral treatments. Detailed information about the demographic and clinical characteristics of the sample, and the subsample of participants taking part in the focus group discussion and interviews, can be found in Table 1. Table 2 shows detailed information about the study variables, also divided by group. A few things are worth noting. First, the majority of participants (i.e. oral-only 78.40% and non-oral 69.60%) were in functional class I and II. Second, participants receiving non-oral therapy reported higher perceived levels of symptomatology and functional disability than those receiving oral-only therapy, however, these differences did not reach statistical significance. Third, eight patients within the oral-only group were exclusively on calcium channel blockers. Fourth, no patients were on selexipag, the oral selective prostacyclin receptor agonist. 36
Means and standard deviations of study variables divided by group.

AAQ-II, Acceptance and Action Questionnaire II; CAMPHOR, Cambridge Pulmonary Hypertension Outcome Review; PSS, Perceived Stress Scale.
Preliminary data analyses
Missing data analyses revealed a negligible amount of missing data among the study variables (i.e., 1.35% overall, in 19.28% of participants), which were imputed using expectation maximization after confirming that they were missing completely at random (χ2 = 989.23, df = 995, p = 0.546). No univariate or multivariate outliers were detected using the outlier labeling rule and Mahalanobis distance, respectively. The assumption of normality was satisfied, as values of skewness and kurtosis were in all cases within the recommended boundaries of |2| and |9|, respectively. The result of Levene’s test of equality of variances was nonsignificant, F (1, 81) = 2.03, p = 0.158, which confirms that the assumption of equality of variances was satisfied. The assumption of homogeneity of regression slopes was evaluated by running ANCOVAs with group, covariates, and interaction terms (i.e., group × covariates) as predictors of QoL. The results showed that no interaction terms reached statistical significance, which suggests this assumption was also met.
Differences in QoL between patients receiving oral-only and non-oral treatment
The results of the ANCOVA revealed a significant main effect of treatment type on QoL, after statistically adjusting for the effects of age, level of symptomatology, functional disability, psychological well-being, and perceived stress; this effect represented a medium effect size. Post hoc analyses (Fisher’s LSD) indicated that participants receiving non-oral treatments reported a statistically significant higher level of impairment in their QoL than those only receiving oral treatment. The difference in adjusted means (i.e., 1.87) fell below the three-point threshold indicating clinical significance using the CAMPHOR. 23 The results also revealed that the level of symptomatology, physical disability, and perceived stress were statistically significant predictors of QoL (Table 3).
Mean adjusted differences in QoL between patients receiving oral-only (N = 37) and non-oral PAH treatment (N = 46) while statistically adjusting for age, level of symptomatology, functional disability, psychological well-being, and perceived stress.

CV, covariate; IV, independent variable; PAH, pulmonary arterial hypertension; QoL, quality of life.
Qualitative data analysis
The results of the qualitative data analyses based on the focus group discussion and interviews were organized into four themes: apprehension about starting on non-oral treatments, difficulties associated with self-management, treatment-related interference in daily life, and low subjective well-being.
Apprehension about starting on non-oral medications
Participants expressed absolute consensus that they felt great anguish when they were first informed about the need to receive non-oral treatments. For patients just receiving the diagnosis this news resulted in added distress and was often taken as an early glimpse of the potential seriousness of their condition. One patient who was prescribed inhaled iloprost narrated his experience: When I first saw the nebulizer, and they told me, you have to take this 6 times per day, I was like, this won’t be easy . . . it’s not too bad now, but at first this seemed like a world to me. I thought it was a life changer. (Participant 1)
Having to start on non-oral treatments was seen as something requiring great psychological adjustment, as expressed by another patient who had just received the diagnosis: What they told me, and maybe it’s a bit harsh, was that I had to make friends with it [i.e. with the epoprostenol infusion pump], because that was what would keep me alive. You have no other choice, for me it was the only option. You either accept it or accept it. (Participant 2)
Another patient said: I felt pretty depressed, because previously I would just get sick from hearing that I might need to wear a pump, just like everyone else . . . because taking a pill is not the same as always carrying something with you, the quality of life is not the same. . . (Participant 3)
Further, and from a psychological point of view, patients thought that starting on non-oral treatments was even more difficult for patients who had been only taking oral medications for some time after their diagnosis. Some of these patients acknowledged they had delayed as much as possible giving their consent to start on non-oral treatments, even though they realized that their health was deteriorating: I dreaded the [treprostinil infusion] pump! I was putting it off . . . and had quite a few syncope . . . because one day the doctor introduced me to another patient, and they showed me how to wear the pump, how you carried it with you, and I felt such a strong sense of rejection that I kept taking the pills until the very last moment, which again that’s why I had so many syncope. That was one thing that I didn’t want to accept . . . I didn’t want to wear a pump. (Participant 4)
Difficulties associated with self-management
In comparison to taking oral medications alone, participants agreed that being on non-oral treatments (taken concurrently or not with oral medications) resulted in extra challenges for self-management. Some patients stated that it had taken them some time to properly operate the infusion pumps. One patient remembered his experience: It took me about 6 months to get used to it, not only because of the transition [from oral-only medication], but above all because I didn’t handle the pump well, the treprostinil pump . . . I don’t know, you think it’s working and then you realize that actually all the medication is coming out . . . because I didn’t handle it correctly. . . (Participant 5)
Patients who were or had been on treprostinil also agreed that the site pain resulting from the subcutaneous infusion could be quite disabling and lead them to spend up to a week in bed every month.
Another patient complained about his day-to-day experience with iloprost: I wish they would get me off the nebulizer, because when the nebulizer doesn’t work the way it should then I’m not well. When I have been going at it for 10 minutes, what happens? Well, that I get breathless, that it takes so long that I have to stop taking it. It’s not the same as taking a pill . . . after 10 minutes with the nebulizer my body says “I can’t take this anymore,” and sometimes I try to go for a little longer . . . for 11 or 12 minutes, and maybe the nebulizer hasn’t stopped and I keep going, but I feel I get worse than I was, from breathing through the nebulizer while covering my nose. Sometimes I get angry and feel like throwing the nebulizer against the wall! (Participant 1)
Further, patients found it especially difficult to deal with emergency situations, to some extent because sometimes it was difficult to find specialized medical assistance due to the lack of knowledge about PAH among most health practitioners. As conveyed by one patient: It was a Sunday and I was feeling terrible, but I waited until the following day because I can’t just go through the emergency room, because 90% of the doctors don’t know about this, they haven’t seen this in their lives. This one time that I went through the emergency room I was like “have you ever seen this [i.e. the illoprost nebulizer]?” And I took it out and put it on the table and showed it to [the doctor]. Because it’s a completely unknown disease, only PAH experts know about it. Regular doctors haven’t heard about it. My GP doesn’t know about this. She is learning with me, with the information that I give her. (Participant 1)
In addition to a potentially critical event creating an emergency, knowing that obtaining help could be difficult greatly aggravated the experience of distress. Another patient narrated her experience one time when her epoprostenol infusion pump stopped working: Four days ago my pump stopped working, and the thing is that I depend on the pump! So I switched to the spare one and fortunately everything went well. But until I put it on I was freaking out . . . what if the catheter gets blocked or anything else happens to you!? I mean, the first thing they tell you is that you cannot be more than half an hour without this medication! You either get your spare pump working or you run to the hospital. But at midnight you can’t just go to any hospital. We are supposed to go to [name of PAH reference center] but that late at night you call and there is no one there who knows about PAH. The PAH nurses are there only during the morning. Can you imagine the feeling!? So the other night I just called [name of other patient] to ask her what to do. (Participant 2)
Interference with daily life activities
Participants agreed that non-oral treatments often significantly interfered with daily life activities. For patients wearing infusion pumps, for example, even simple activities like showering or sleeping require that they make adjustments as a result of their treatment: At first you don’t even know how to sleep, how to position yourself in bed, what to do with it [i.e. with the infusion pump], because you feel you are going to break it, that it’s going to fall off the bed, that it’s going to get obstructed. At first you just don’t sleep at all. . . (Participant 2)
Another patient said: The [infusion] pump is with you 24/7. So, any verb you do, the pump is with you. Any verb: walking, jumping, and some other more private verbs . . . the pump is with you. (Participant 6)
Patients who were on iloprost agreed that they often had to plan their day around their nebulizer schedule: The nebulizer is a huge burden, because it prevents me from going places, from sleeping, because now . . . look, I have to take my medication at 9.00, 11.15, 13.30, 15.45, 18.00, 20.15, 22.30 and 00.45. I can never go to bed before 1.00. Never! And then I have to wake up at 8.45. I have two hours and 15 minutes between each dose, and out of this time it takes me about 10 minutes to take the medication and then 5 minutes between setting up the nebulizer and cleaning it after each use. (Participant 1)
Low subjective well-being
Most patients taking part in the focus group discussion acknowledged that they experienced improvements in their physical function when they started on non-oral treatments. Nonetheless, starting on non-oral treatments was often seen as a sign that their health was slowly worsening, which negatively impacted their subjective well-being: You see that at first they give you oral treatment, and from there one day instead of one pill they give you two, or something else, three, and when they start you on the pump, then you say “this is not going well.” From time to time they give you one more thing, they add more, and you are left to assume that it is because things are not going well. (Participant 7)
This perception was aggravated in patients who had not experienced noticeable improvements in their health status after starting non-oral treatments: I don’t feel I have really improved. Even though the test results are better, I feel the same, and on top of that now I have to wear this [i.e. the infusion pump], and before I didn’t have to . . . and I can’t get used to carrying this with me, I can’t fit this with how I see myself. (Participant 3)
Further, being on medications requiring the use of an infusion pump often evoked feelings of “being too dependent on a machine.” Again, this had a negative impact on patients’ subjective well-being and added to the uncertainty of living with PAH: You feel completely lost, out of control. You have the threat of the disease itself and on top of that you feel you are 100% dependent on a machine. (Participant 2)
Further, patients stated that wearing an infusion pump constituted a continuous reminder of their condition: As long as I wear this [i.e. the infusion pump] I can’t forget, and that’s very hard. The pump doesn’t let you forget about the disease. It’s not like taking a pill and walking out the door and sometimes you can forget about it, at least for a little while, but I, wherever I am, I am with this thing and can’t forget that I have PAH. (Participant 3)
Patients who wore infusion pumps also stated that the pumps made PAH difficult to conceal, and that made them feel stigmatized. In the words of one patient: And in terms of feeling rare, well, when you say you have PAH you are rare, but at least you can hide it if you want to. But of course, being rare and on top of that wearing that [i.e. an infusion pump] then you can no longer hide it, which is yet another handicap. (Participant 6)
Discussion
The results of the ANCOVA revealed that participants receiving non-oral treatments reported significantly greater impairment in their QoL in comparison to participants receiving oral-only treatments, after taking into consideration the effect of relevant covariates that could have confounded the observed group differences in QoL. A couple of aspects are worth highlighting regarding these results. Most participants in both groups reported relatively low levels of symptomatology and functional disability; also, group differences in symptomatology and functional disability were nonsignificant (Table 2). Therefore, this suggests that the observed differences in QoL may have been associated with differences in the medication regimen. It is worth noting, however, that such observed differences did not reach the threshold for clinical significance. Therefore, these results question whether the challenges associated with receiving non-oral therapy—relative to oral therapy—may have been sufficiently salient to patients within this sample, as to make a difference in their QoL.
Insights into the challenges associated with receiving non-oral therapy were obtained by means of a focus group discussion and interviews with nine PAH patients.
They highlighted that non-oral treatments often caused significant interference in their day-to-day lives, relative to oral treatments. The experience of site pain resulting from the subcutaneous infusion of treprostinil was highly disabling and often confined them to bed rest for a few days, which interfered with the fulfillment of important responsibilities (e.g., house chores and work). This is congruent with previous studies showing that infusion site pain constitutes the most common adverse side effect attributed to treprostinil, often leading to treatment discontinuation. 14 Having to wear an infusion pump was also identified as an aspect interfering with performing ordinary daily activities (e.g., sleeping and taking a shower). This aligns with the results from a recent study showing that PAH patients often delayed parenteral prostacyclin treatment over concerns about side effects and interference with daily life activities. 14 Similarly, treatment with iloprost often required that patients plan their daily routines around their medication schedule; the number of required daily doses (i.e., up to nine) and the time devoted to preparing and taking the medication was also reported to interfere with daily activities (e.g., going out).
Patients expressed concern about effectively self-managing their non-oral treatment. Some patients acknowledged that it took them some time to learn to operate the different devices (i.e., iloprost nebulizers, infusion pumps) to properly self-administer their medication. Most importantly, they stated that it had been often quite difficult to obtain specialized help when they needed it most, that is, when suddenly feeling sick or dealing with a pump malfunction; the potentially significant consequences of going through one of these experiences elicited great apprehension. This is congruent with research showing that PAH patients often find it difficult to obtain specialized medical care, and this causes great frustration and contributes to a sense of social isolation. 3 In general, patients agreed that being on non-oral treatments was not the same as “just taking a pill,” given that non-oral treatments brought added challenges for self-management.
The transition from oral-only to non-oral medications marked an inflection point for patients in their journey with PAH. This event often elicited great apprehension, and some patients admitted they tried to delay such a transition as much as possible. There was consensus among patients that they often felt “dependent on a machine,” and this made it difficult to accept the change in treatment. Even though most patients acknowledged they had noticed an improvement in their health status after starting on non-oral treatments, some of them also stated that they had also experienced a decline in their subjective well-being: the transition to non-oral medications was often perceived as a sign that their health was deteriorating. Similarly to previous patient reports, 14 non-oral treatments were often seen as a tradeoff between extended survival and higher QoL. Some patients also viewed non-oral treatments as a continuous reminder of PAH because they made it difficult to conceal the disease, which also created a social stigma.
Overall, these findings reveal that patients receiving non-oral PAH therapy face unique challenges in their day-to-day lives. To some extent, some of these challenges are psychosocial in nature, for example, those related to feeling apprehension, managing pain, and experiencing low subjective well-being; others are simply directly related to the nature of non-oral medication regimens (e.g., wearing a pump and frequent dosing of iloprost), which makes adjustment to PAH therapy more difficult. On one hand, this highlights the need to look beyond health-related issues to attain a more comprehensive understanding of the QoL of PAH patients. Doing so seems ever more pertinent as advances in therapy and clinical management continue to extend life expectancy, and potentially, the time PAH patients may live afflicted by challenges such as these. As revealed by the quantitative data analyses, participants appraising their lives as relatively unpredictable, uncontrollable, and overloaded (i.e., as more stressful) were more likely to report lower QoL; this is highly congruent with previous research attesting to the many hurdles related to living with PAH.4,37 On the other hand, these findings highlight the need for multidisciplinary work devoted to assist PAH patients self-manage their disease and treatment, wherein non-pharmaceutical interventions play a more prominent role. This, for example, may include psychological support to assist patients to manage their pain, 38 reduce apprehension, modify behavior to more effectively deal with everyday problems, 39 and build valued lives in the face of adversity. 40
Limitations
The results of this study should be interpreted with caution. First, the majority of participants from both groups self-reported relatively low levels of symptomatology and functional disability; this may have been the result of self-selection bias: participants with higher symptomatology and functional disability may have felt less motivated to participate. Therefore, the observed differences in QoL and their potential association with the type of therapy (i.e., oral-only vs non-oral) should be interpreted in the context of similar samples of PAH patients. Second, the qualitative analyses were based on the responses of nine participants, and even though thematic saturation was reached 19 the obtained results may be biased; for example, it is unclear whether other groups of PAH patients (e.g., those in different societies) may identify the same challenges associated with receiving non-oral therapy. Third, all participants were members of a patient association, and it is unclear whether the higher potential for social support and shared information may have had an impact on variables relevant to this study (e.g., self-management and psychological well-being). Fourth, even though relevant covariates were included in the ANCOVA, it is unclear whether the uneven between-group distribution of patients with different diagnoses may have had an impact on group differences in QoL, given that different types of PAH may affect QoL differently. Fifth, adherence to PAH-specific treatments was not assessed, even though it may be associated with symptomatology levels, functional disability, and QoL. Sixth, the statistical analyses were not sufficiently powered to detect meaningful effects due to a relatively small sample size. Relatedly, it is worth noting that the CAMPHOR was designed to assess “needs” that are frustrated as the result of PAH, 21 not as the result of its treatment; therefore, the impact that non-oral treatment may have had on QoL may have been underestimated. Last, the data on symptomatology and functional disability were based on patients’ self-reports, rather than clinical measures. Even though the interest in this study was on the patient perspective, including the way patients may perceive their own health status, the data provided by clinical measures may have been useful in complementing the self-reports with indicators of disease severity (e.g., mean pulmonary arterial pressure and 6-minute walking distance).
Conclusion
The results of this study suggest that the QoL and daily lives of PAH patients with relatively mild levels of symptomatology and functional disability can be affected by the challenges associated with following non-oral treatment regimens, above and beyond the effects of PAH itself. Most participants taking part in the focused group discussion acknowledged that their health status had significantly improved after starting non-oral treatments. Nonetheless, they also stated that non-oral treatment regimens exerted a negative impact on their daily lives and subjective well-being, due to their interference across multiple life areas.
Supplemental Material
sj-docx-1-tar-10.1177_17534666241289807 – Supplemental material for Patients’ perspectives on the challenges associated with receiving non-oral pulmonary arterial hypertension treatment: a mixed methods study
Supplemental material, sj-docx-1-tar-10.1177_17534666241289807 for Patients’ perspectives on the challenges associated with receiving non-oral pulmonary arterial hypertension treatment: a mixed methods study by Aldo Aguirre-Camacho in Therapeutic Advances in Respiratory Disease
Footnotes
Acknowledgements
The author thanks Dr. Daniel García Pérez for his assistance in data analyses. The author also thanks the Spanish National Association of Pulmonary Hypertension (i.e., Asociación Nacional de Hipertensión Pulmonar), and its members, for their support and involvement in this study.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
