Abstract
Background:
It remains unclear whether erythrocyte sedimentation rate (ESR) accurately predicts prognosis during treatment and how ESR changes.
Objectives:
We aimed to assess the predictive values of ESR as a prognostic factor of Mycobacterium avium complex pulmonary disease (MAC-PD) while on anti-mycobacterial treatment and its changes according to the treatment responses.
Design:
This study is a retrospective cohort study.
Methods:
This study included patients aged 18 years or older who initiated anti-mycobacterial treatment for MAC-PD at Seoul National University Hospital between January 1, 2009 and March 31, 2022. ESR should be measured at least twice, with a minimum interval of 3 months, during the initial 12 months from the commencement of antibiotic treatment. A mixed linear regression and Cox proportional-hazards models were used to analyze repeated ESR data and the association with patient survival.
Results:
Of a total of 825 patients who initiated antibiotic treatment for MAC-PD, 369 patients were included in the analysis. Increased levels of ESR during the treatment process were associated with a higher risk of mortality (adjusted hazard ratio 1.03; 95% confidence interval, 1.02–1.03) after adjusting age, sex, comorbidities, presence of cavity, acid-fast bacilli smear positivity, and culture conversion at 12 months. During the treatment, ESR at 12 months of treatment significantly decreased compared to baseline ESR in both the culture-converted and not-converted groups, which was categorized based on whether the culture conversion was achieved within the 12 months after treatment initiation.
Conclusion:
ESR predicted mortality during treatment and decreased over time, regardless of treatment outcomes. Our results underscore the importance of administering anti-mycobacterial treatment even in patients who did not achieve a microbiological cure.
Introduction
Nontuberculous mycobacteria (NTM) are ubiquitous organisms found in soil, dust, and municipal water. NTM can lead to chronic infection in humans, with pulmonary disease (PD) being the most common manifestation. 1 Over the past decades, the burden of NTM-PD has increased globally. In South Korea, between 2010 and 2022, the prevalence of NTM infection increased approximately fivefold. 2 This rise in burden has led to NTM-PD being recognized as an emerging threat associated with significant mortality and morbidity. 3
The treatment of NTM-PD typically requires the administration of at least 2–3 antibiotics for 12 months or longer, after achieving culture conversion.1,4 The assessment of treatment response during antibiotic therapy relies on sputum acid-fast bacilli (AFB) culture. 5 However, this method takes up to 2 months to confirm the results and many patients fail to expectorate sputum while on treatment. 6 Therefore, it is imperative to identify biomarkers that can change during treatment, offering a more efficient means of monitoring treatment response.
Low body mass index (BMI), male sex, old age, presence of cavity, and respiratory comorbidities at the time of diagnosis have been identified as factors that increase the risk of mortality in patients with NTM-PD.7–9 Furthermore, elevated erythrocyte sedimentation rate (ESR) levels have been linked to an increased risk of mortality.7,10 However, the prognostic impacts of these factors after initiating anti-mycobacterial treatment have not been elucidated. For instance, while ESR levels can fluctuate over time, it remains unclear how ESR changes with treatment and whether it can accurately predict prognosis during treatment.
In this study, we aimed to assess whether ESR, an easily measured biomarker, can be used to predict the risk of mortality, as well as how ESR changes by treatment response during treatment of Mycobacterium avium complex (MAC)-PD, which represents the majority of NTM-PD. 11
Methods
Study design
This retrospective cohort study includes patients aged 18 years and above who initiated anti-mycobacterial treatment for MAC-PD at Seoul National University Hospital between January 1, 2009 and March 31, 2022. The causative pathogen was identified as M. avium complex by 16S rRNA and rpoB gene sequencing.12–14 Patients who had ESR measurements at least twice, with a minimum interval of 3 months, during the initial 12 months from the commencement of antibiotic treatment were included. Patients with antibiotic treatment duration of less than 3 months or with ESR measurements conducted fewer than two times were excluded from the study. Patients with a follow-up period of less than 1 year were also excluded to enhance the reliability of the survival analysis. The reporting of this study conforms to the Standards for Reporting Diagnostic Accuracy statement. 15
ESR measurement
ESR values were measured using the closed automated analyzer TEST-1 (Alifax; Padova, Italy). TEST-1 aspirates the patient’s blood, distributes it into capillaries, applies a centrifugal force of 20 g, and then detects electrical resistance by passing infrared light at 950 nm through the sample. ESR values are measured by converting the detected electrical resistance into Westergren units using a linear regression model.16,17
Data collection
Baseline characteristics were collected, including demographic factors, comorbidities, cavity in chest computed tomography scans, AFB smear results, and laboratory findings. ESR values were collected at 3-month intervals from the initiation of antibiotic treatment up to 12 months. A window period of 1 month before and after each time point was allowed at each time point. Culture conversion, defined as having at least three consecutive negative mycobacterial cultures from respiratory specimens collected at a minimum interval of 4 weeks, 5 was assessed 12 months after treatment initiation. All patients were followed up until May 31, 2022, and the date of death was confirmed using the Ministry of the Interior and Safety database in South Korea.
Statistical analysis
Patients were divided into two groups based on their culture conversion status at the 12 months after treatment initiation. Continuous variables were presented as mean and standard deviation, while categorical variables were presented as numbers and percentages. Continuous variables were compared using Student’s t-test and Mann–Whitney U test, while categorical variables were compared using Pearson’s chi-squared test and Fisher’s exact test. A mixed linear regression model was used to analyze repeated ESR data and to account for missing values. The model included fixed effects for gender, age, BMI, presence of cavity, and AFB smear positivity as covariates. ESR values were estimated every 3 months according to the groups and compared for differences within each group and between the groups.
Furthermore, the Cox proportional-hazards model was used to analyze the association between repeatedly measured ESR values during the treatment course and patient survival after clustering by each patient. Kaplan–Meier curves were plotted, and a log-rank test was conducted to analyze statistical differences between the two groups after dividing patients into groups whose ESR increased or did not increase by the difference between the baseline and last measured ESR. A p-value below 0.05 was considered statistically significant. All statistical analyses were conducted using Stata 17.0 (StataCorp., College Station, TX, USA).
Results
During the study period, 825 patients initiated antibiotic treatment for MAC-PD. After excluding 456 patients (154 patients who received treatment for less than 3 months, 269 patients with less than two ESR measurements, and 33 patients with a follow-up period of less than 1 year), 369 patients were included in the analysis. The mean age of the patients was 63.5 ± 10.7 years, and 113 (30.6%) were male. The AFB smear was positive in 76 patients (20.6%). The detailed baseline characteristics of patients are presented in Table 1.
Baseline characteristics of the patients with MAC-PD at treatment initiation
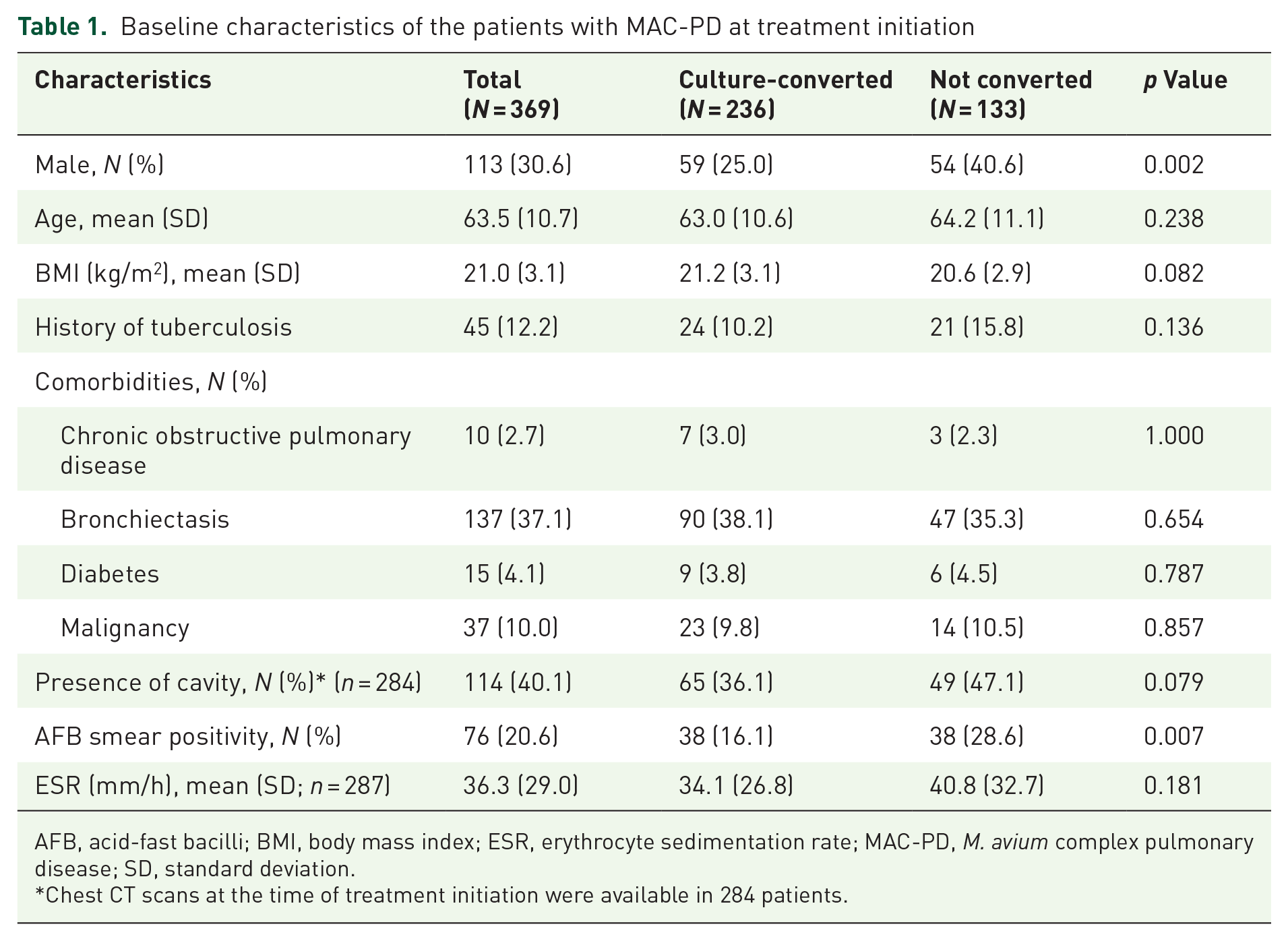
AFB, acid-fast bacilli; BMI, body mass index; ESR, erythrocyte sedimentation rate; MAC-PD, M. avium complex pulmonary disease; SD, standard deviation.
Chest CT scans at the time of treatment initiation were available in 284 patients.
During the 12 months after the initiation of treatment, ESR was measured 1523 times (mean 4.1 ± 1.0) in those patients. The mean value of ESR at the time of treatment initiation was 36.3 ± 29.0 mm/h. The median follow-up period was 45.5 months (interquartile range 28.0–92.8 months). During the follow-up period, 51 patients (13.8%) died, and the mortality rate was 7.29 deaths per 100,000 person-years. Clustered survival data showed that increased levels of ESR during the treatment process as well as at the time of treatment initiation were associated with a higher risk of mortality (adjusted hazard ratio 1.03; 95% confidence interval [CI], 1.02–1.03) after adjusting age, sex, BMI, comorbidities, presence of cavity, AFB smear positivity, and culture conversion at 12 months (Table 2). When dividing patients into groups based on whether their ESR increased or not after treatment initiation, the probability of survival significantly increased in the group where ESR did not increase (log-rank p = 0.031; Figure 1). In addition, ESR levels were significantly higher in non-survivors compared to survivors throughout the entire 12-month treatment period (p < 0.001; Figure 2).
Factors associated with mortality in patients with anti-mycobacterial treatment for MAC-PD.
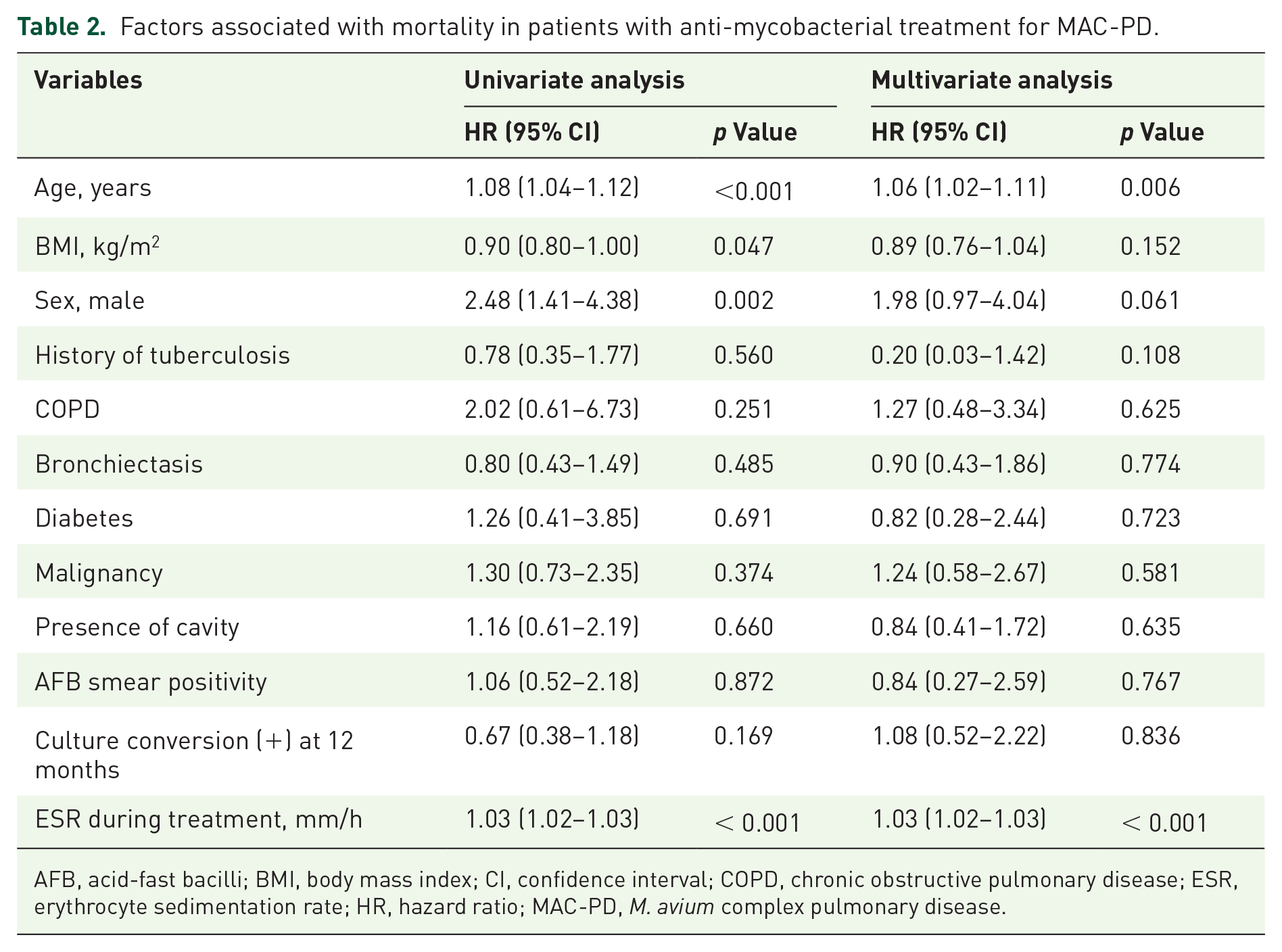
AFB, acid-fast bacilli; BMI, body mass index; CI, confidence interval; COPD, chronic obstructive pulmonary disease; ESR, erythrocyte sedimentation rate; HR, hazard ratio; MAC-PD, M. avium complex pulmonary disease.
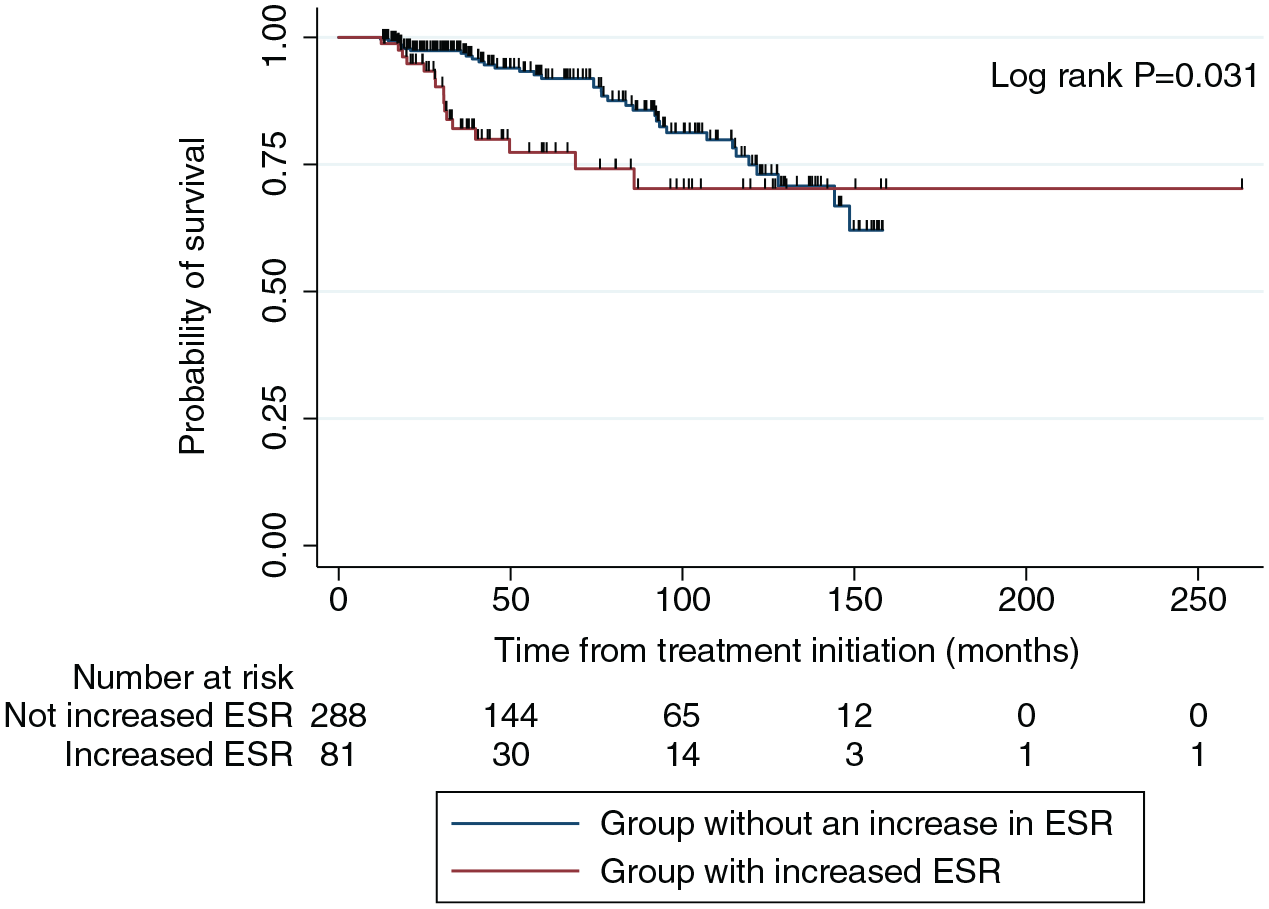
Kaplan–Meier curve for survival according to whether there is ESR elevation after anti-mycobacterial treatment initiation.

Longitudinal changes in serum ESR during anti-mycobacterial treatment stratified by survival. There were statistically significant differences in estimated ESR between the two groups at each time point based on survival (p < 0.001).
Culture conversion was achieved in 236 patients (64.0%; Culture-converted group), while 133 patients (36.0%; Not-converted group) failed to achieve culture conversion. Male patients were more common in the not-converted group (40.6%) than in the culture-converted group (25.0%; p = 0.002). AFB smear positivity was also more common in the not-converted group (28.6%) than in the culture-converted group (16.1%; p = 0.007). The mean value of ESR at the time of treatment initiation was 34.1 ± 26.8 in the culture-converted group and 40.8 ± 32.7 in the not-converted group, respectively (p = 0.181). In addition, the mean baseline ESR of patients with cavities was significantly higher than those without (46.0 ± 32.2 vs 29.3 ± 24.9, p < 0.001). The mean baseline ESR of patients with positive AFB smear was 44.0 ± 35.7, while that of patients with negative AFB smear was 34.5 ± 26.9, with no statistically significant difference between the two groups (p = 0.210).
Throughout the treatment period, both the culture-converted and not-converted groups exhibited a decreasing trend in ESR. In the culture-converted group, the mean values of ESR were 26.8 (95% CI 22.7–30.9) at 3 months, 24.7 (95% CI 20.5–28.8) at 6 months, and 22.5 (95% CI 18.2–26.8) at 9 months after treatment, respectively. At 12 months of antibiotic treatment, ESR significantly decreased to 22.2 (95% CI 17.9–26.6), compared to baseline (p < 0.001). In the not-converted group, the mean values of ESR were 30.7 (95% CI 25.1–36.2) at 3 months, 26.1 (95% CI 20.4–31.7) at 6 months, and 26.1 (95% CI 20.2–32.0) at 9 months after treatment, respectively. Likewise, the level of ESR at 12 months of treatment (24.8, 95% CI 18.8–30.8) also decreased significantly compared to baseline in the not-converted group (p < 0.001; Table 3). However, there were no significant differences in ESR between the groups at each point in time, and the slopes of ESR reduction were also not significantly different between the groups (Figure 3).
Mean value of ESR according to culture conversion over time.
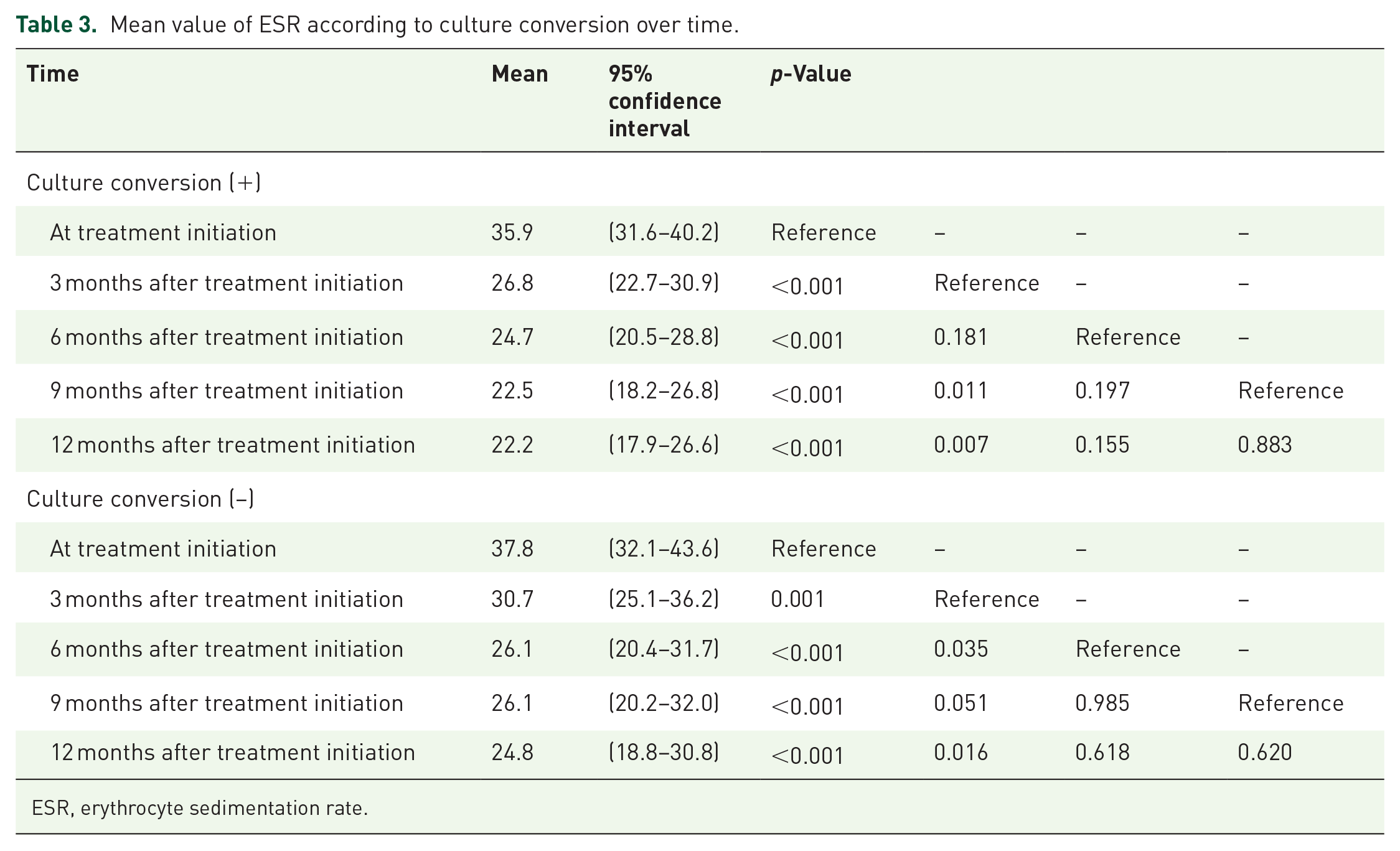
ESR, erythrocyte sedimentation rate.
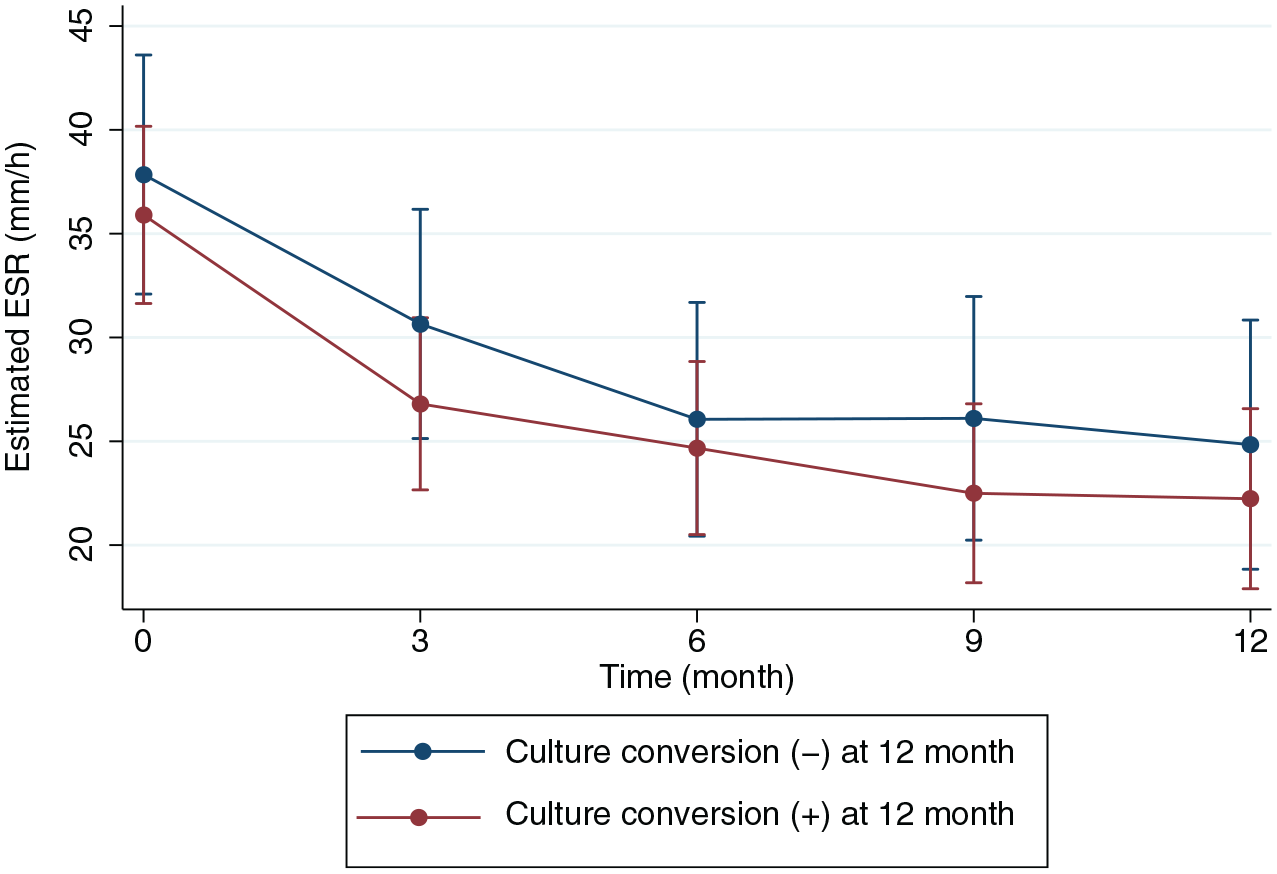
Longitudinal changes in serum ESR during anti-mycobacterial treatment according to the treatment responses. There was no statistically significant difference in estimated ESR between the two groups at each time point based on treatment response.
Discussion
Several factors, including old age, male sex, lower BMI, or the presence of a cavity at the time of diagnosis, predict the prognosis of NTM-PD. 7 However, the clinical significance of these factors during treatment has not been investigated. In this study, we demonstrated that serum ESR could predict mortality in patients with MAC-PD receiving anti-mycobacterial treatment. Elevated ESR values during treatment were associated with a higher risk of mortality. Nonetheless, ESR levels decreased over time during treatment, regardless of the microbiological response.
ESR, one of the oldest measures of inflammation, reflects the disease severity and treatment responses in mycobacterial infection.18,19 In cases of pulmonary tuberculosis, the level of ESR increased in patients with a more advanced extent. 18 Moreover, higher ESR was associated with a longer time to sputum culture conversion. 19 Our previous study showed that ESR measured at the time of diagnosis could function as a serum biomarker predicting the prognosis of NTM-PD. 7 However, its role during anti-mycobacterial treatment for NTM-PD remains unclear. Furthermore, the association between ESR and treatment responses has yet to be studied.
In this study, ESR during the treatment course was associated with mortality risk. The changes in ESR during the treatment period accurately predicted mortality risk at each point in time. In other words, ESR could serve as a valuable biomarker to predict the prognosis of patients while on treatment. If ESR decreases during the treatment period, it can be predicted that the patient will benefit in terms of survival, regardless of microbiological responses.
In our study, the administration of anti-mycobacterial treatment led to a decrease in ESR in the not-converted group as well as the culture-converted group. During anti-mycobacterial treatment, the levels of ESR decreased over time in the culture-converted group. Notably, a similar decrease in ESR was also observed in the not-converted group. Our results suggest that even in treatment-refractory patients, the administration of anti-mycobacterial treatment reduced the degree of inflammation as reflected by ESR. This implies a beneficial effect of anti-mycobacterial treatment on survival, even in patients experiencing treatment failure. 20
The beneficial effect of anti-mycobacterial treatment, even in treatment-refractory patients, has been proven in several studies. Anti-mycobacterial treatment can improve health-related quality of life, even in patients with treatment failure. 21 In addition, a substantial proportion of patients with treatment failure have improved in disease extent, as measured by chest X-ray. 22 Finally, prolonged treatment could improve the survival of patients with cavitation or AFB smear positivity. 20 These findings indicate that anti-mycobacterial treatment can suppress inflammation to some extent and benefit patients while not completely eradicating mycobacteria.
Our study has several limitations. First, we were unable to analyze the long-term outcomes of treatment. Second, the analysis to calculate sample size was not performed due to the retrospective cohort study’s nature. Third, ESR was not routinely measured from the time of treatment initiation, and many patients who did not have ESR measured more than twice during treatment were excluded. Fourth, while various conditions such as bacterial exacerbation or chronic fungal infection can influence the change of ESR, we could not adjust for all of them except for age, BMI, sex, comorbidities, presence of cavity, and AFB smear positivity at treatment initiation. Our study was unable to obtain repeatedly measured data beyond ESR. Variables such as cavities and AFB smear results during the treatment process were not included in the adjustment, considering it would be inaccurate to include them. Lastly, although the association between increased ESR levels during the treatment process and increased mortality risk was significant, the HR of 1.03 was not high, necessitating cautious interpretation in clinical practice. Nevertheless, our study identified a biomarker that can be used during treatment. Furthermore, we have once again demonstrated the need for treatment even in patients who experience treatment failure.
Conclusion
In conclusion, ESR, which predicted the mortality of patients with MAC-PD during treatment, decreased over time regardless of treatment outcomes. Our results underscore the importance of administering anti-mycobacterial treatment even in patients who did not achieve a microbiological cure.
Supplemental Material
sj-docx-1-tar-10.1177_17534666241282272 – Supplemental material for Change of erythrocyte sedimentation rate as prognostic biomarker for Mycobacterium avium complex pulmonary disease through anti-mycobacterial treatment
Supplemental material, sj-docx-1-tar-10.1177_17534666241282272 for Change of erythrocyte sedimentation rate as prognostic biomarker for Mycobacterium avium complex pulmonary disease through anti-mycobacterial treatment by Hyeontaek Hwang, Joong-Yub Kim, Jae-Joon Yim and Nakwon Kwak in Therapeutic Advances in Respiratory Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
