Abstract
Blastomycosis can result in lung injury with high mortality rates. The literature on veno-venous extracorporeal membrane oxygenation (VV-ECMO) used as a rescue therapy is limited to case reports and small case series collected over extended time periods. This report describes the clinical course and post-hospitalization outcomes among patients with blastomycosis-induced respiratory failure requiring VV-ECMO in the most recent time frame. The data were collected retrospectively from the health records of eight patients with blastomycosis-induced respiratory failure admitted to a tertiary care center between 2019 and 2023. The mean time from the start of mechanical ventilation to ECMO initiation was 57 h. All patients survived to ECMO decannulation, and seven of them survived to hospital discharge. All six patients whose post-discharge follow-up information was available were weaned from mechanical ventilation and lived at home while two required supplemental oxygen. This includes a case where the provision of adequate ECMO support was challenging due to the patient’s morbid obesity. The most common residual imaging abnormalities included pulmonary infiltrates and pneumatoceles. The study demonstrates the feasibility of VV-ECMO as a rescue therapy in patients with blastomycosis-related refractory respiratory failure. Rapid initiation of ECMO support in eligible patients may have contributed to the good outcomes.
Introduction
Blastomycosis is a potentially life-threatening infection caused by the fungus Blastomyces which is considered endemic to the south-central, southeastern, and midwestern US states, particularly those bordering the Ohio and Mississippi Rivers and in parts of the United States and Canada surrounding the Great Lakes and the Saint Lawrence River. 1 The clinical presentations are variable and range from subclinical infection to fatal disseminated disease. 2 The first report of the use of veno-venous extracorporeal membrane oxygenation (VV-ECMO) in a patient with severe acute respiratory distress syndrome (ARDS) due to blastomycosis pneumonia who did not survive was published in 1999. 3 In a retrospective study of 43 patients, blastomycosis-related ARDS was described as mild, moderate, and severe in 2 (5%), 12 (28%), and 29 (67%) patients, respectively; the overall mortality was 40%, but all four patients treated with VV-ECMO survived. 4 One of the largest reports on 84 patients hospitalized over a 14-year period found that approximately one-third required intensive care unit (ICU) admission, of whom 69% required mechanical ventilation, 10.7% tracheostomy, 9.5% renal replacement therapy, and 75.9% vasopressors. Overall mortality was 14.3% and ICU mortality was 41.4%. Only one of the four patients treated with VV-ECMO survived. 5 Subsequent case series reported prolonged VV-ECMO courses and survival in four out of five patients with respiratory failure due to blastomycosis, two of whom had significant long-term functional and neuropsychological limitations. 6 A multi-center study of patients with fungal infections treated with VV-ECMO included 16 cases of blastomycosis over a 10-year period and reported 67% survival to hospital discharge, but it is unclear whether it differed between the centers, and no long-term outcomes were reported. 7 The literature on VV-ECMO utilization in blastomycosis-related respiratory failure remains limited to case reports and case series. This report describes a single-center experience with the clinical course, imaging characteristics, and post-hospitalization outcomes among patients with blastomycosis requiring VV-ECMO.
Materials and methods
This retrospective case series included patients with blastomycosis-induced refractory respiratory failure treated with VV-ECMO between June 2019 and September 2023 at an urban tertiary care center in Minneapolis, MN. Patients who were <18 years old or had not consented to the use of their medical records for research were excluded. The eligible cohort was identified from the hospital’s ECMO program database. The CARE Case Report Guidelines were followed. 8 Primary outcomes included survival to VV-ECMO decannulation, in-hospital mortality, and oxygen dependence at post-discharge follow-up.
Study data were manually extracted from the electronic medical record. Variables included baseline sociodemographic characteristics and health history (age, gender, weight, body mass index (BMI), comorbidities), state of potential exposure, laboratory methods of infection confirmation, imaging abnormalities, time from symptoms to hospital admission, date/time stamps for mechanical ventilation initiation/discontinuation, ECMO cannulation/decannulation, ICU admission/discharge, hospital admission/discharge, and post-discharge follow-up. We collected respiratory parameters at the time of cannulation for ECMO (peak, plateau and driving pressure, positive end-expiratory pressure, partial pressure of arterial oxygen to fraction to inspired oxygen ratio (P/F), partial pressure of arterial carbon dioxide (PaCO2)), Sequential Organ Failure Assessment (SOFA) score, and ECMO parameters (location and size of ECMO cannulas, initial pump and sweep flows). We documented survival to decannulation and in-hospital mortality as well as post-hospital discharge parameters (oxygen dependence, readmissions for respiratory cause, and computed tomography (CT) imaging abnormalities). Complications (hemorrhage, bacterial pneumonia, and acute kidney injury) were defined according to the Extracorporeal Life Support Organization Registry data definitions; ultrasound confirmation was required for the diagnosis of deep venous thrombosis. 9
Data were analyzed using Microsoft Excel. Using date-time stamps we calculated the time from ventilator start to ECMO cannulation, duration of mechanical ventilation, duration of ECMO, ICU length of stay (LOS), hospital LOS, time from discharge to an outpatient follow-up visit, and time from discharge to repeat CT imaging. We computed summary statistics – frequencies, proportions, means, and/or ranges – to characterize the sample.
Results
Eight patients with confirmed diagnosis of blastomycosis required treatment with VV-ECMO during the study period. The mean age was 46.1 years, and five (62.5%) were male (Table 1). The mean BMI was 32.6 and one patient had a BMI of 60.8. Three patients had a history of hypertension and two patients had diabetes; none of the patients was immunosuppressed. Known or highly likely environmental exposure (work in the wooded areas or soil digging) was reported in three cases. Five patients were from Minnesota, two from North Dakota, and one from Wisconsin. Radiologic abnormalities included infiltrates and/or nodular changes in all cases while cavitation was present in six, and mediastinal lymphadenopathy was present in three patients. Extrapulmonary (skin) lesions were present in one patient. All six patients with the available respiratory parameters prior to ECMO initiation had severe gas exchange abnormalities, with a mean P/F ratio of 71.8, pCO2 67.3 mmHg, and pH 7.18. The average SOFA score was 7.75. The average interval between the initiation of mechanical ventilation and ECMO cannulation was 57 h. The diagnosis of blastomycosis was established by bronchoalveolar lavage culture and cytology in all patients—prior to ECMO initiation in six of them and while on ECMO support in the remaining two cases. The most common ECMO circuit configuration included drainage from the femoral vein with a return into the right internal jugular vein. One patient (case 8) with extremely high BMI required the addition of a second circuit (drainage from the right and left femoral veins with the return into the right jugular and left subclavian veins) to provide sufficient flow and oxygen delivery. One patient (case 2) was decannulated after a week of ECMO support but had to be re-cannulated for an additional VV-ECMO run due to severe hypoxemia.
Characteristics of blastomycosis patients treated with ECMO.

BAL, bronchoalveolar lavage; DM, diabetes mellitus; DVT, deep venous thrombosis; ECMO, extracorporeal membrane oxygenation; HTN, hypertension; LSC, left subclavian vein; N/A, not available; N, no; RFV, right femoral vein; RIJ, right internal jugular vein; TBI, traumatic brain injury; Y, yes.
All patients were treated with amphotericin, comprehensive ICU support including lung-protective mechanical ventilation, and all received steroids (Table 2). Tracheostomy was required in seven and percutaneous feeding gastrostomy in four patients. Four patients required renal replacement therapy. The most common complications during VV-ECMO included acute kidney injury, bacterial pneumonia, deep vein thrombosis, bleeding, and paroxysmal atrial fibrillation/flutter (Table 2). One patient (case 1) developed ischemic bowel requiring multiple surgical interventions.
Treatments and complications of patients with blastomycosis treated with ECMO (n = 8).
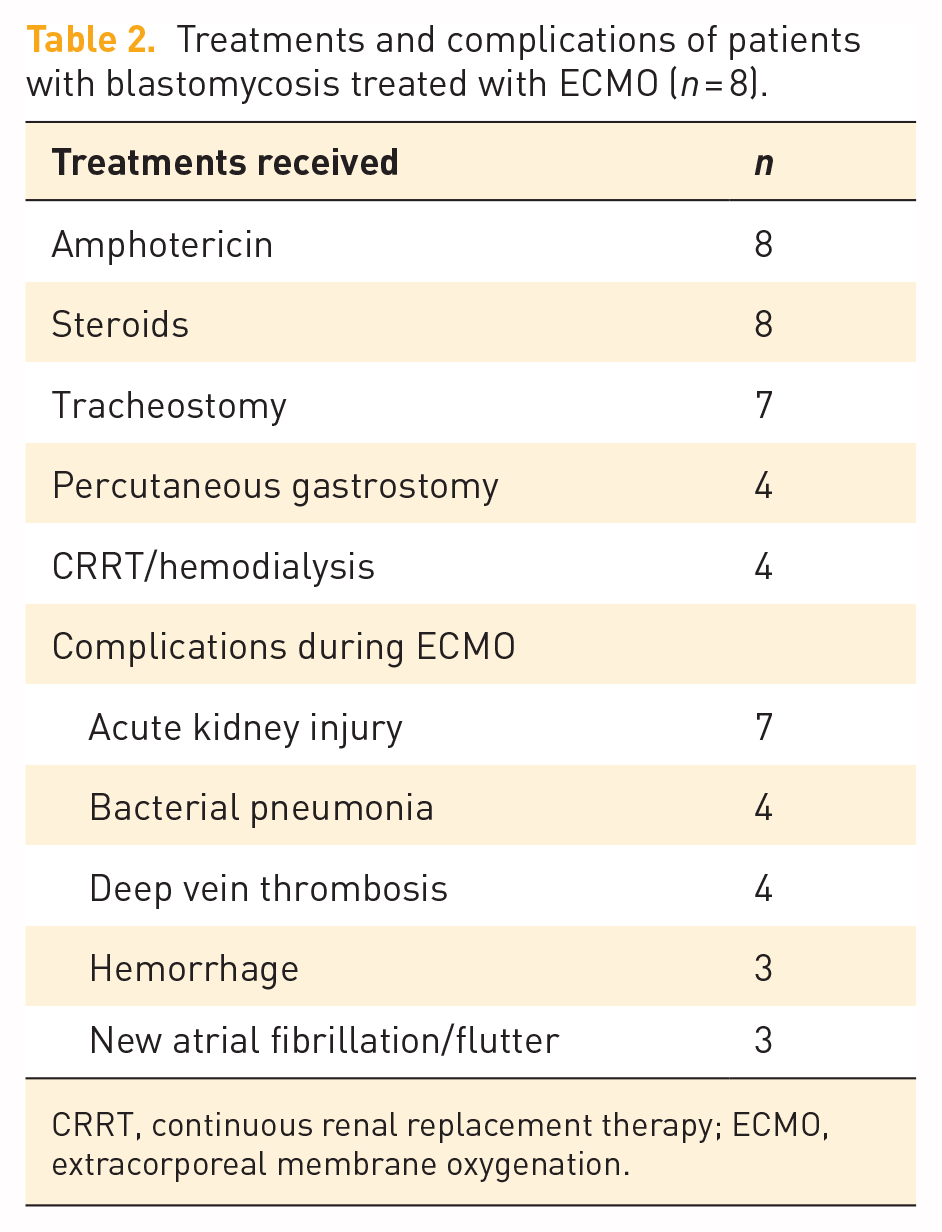
CRRT, continuous renal replacement therapy; ECMO, extracorporeal membrane oxygenation.
All patients survived to ECMO decannulation, and seven (87.5%) survived to hospital discharge (Table 3). The overall survival rate in our VV-ECMO population for the same time period was 61% (100 cases total with 49 of them related to COVID-19). One patient (case 6) died of septic shock and bleeding complications that developed after ECMO removal. The mean mechanical ventilation time was 29.4 days, ECMO duration 18.6 days, ICU LOS 38.9 days, and hospital LOS 43.4 days. All seven survivors were discharged to a rehabilitation facility. The six patients whose follow-up information was available (follow-up time range 1–12 months) were independent of mechanical ventilation and reported improved respiratory symptoms. Two patients continued to require home oxygen, and one patient with multiple pneumatoceles (case 4) was readmitted for pneumothorax requiring chest tube placement 2 months after the hospital discharge.
Outcomes of patients with blastomycosis treated with ECMO (n = 8).

CT, computed tomography; ECMO, extracorporeal membrane oxygenation; ICU, intensive care unit.
Five patients had a follow-up chest CT within the 13 months post-discharge (Figure 1). Overall improvement in radiologic abnormalities was reported in all patients but none had a completely normal CT. Residual infiltrates (n = 5) and pneumatoceles (n = 4) were the most common persistent abnormalities (Table 3).
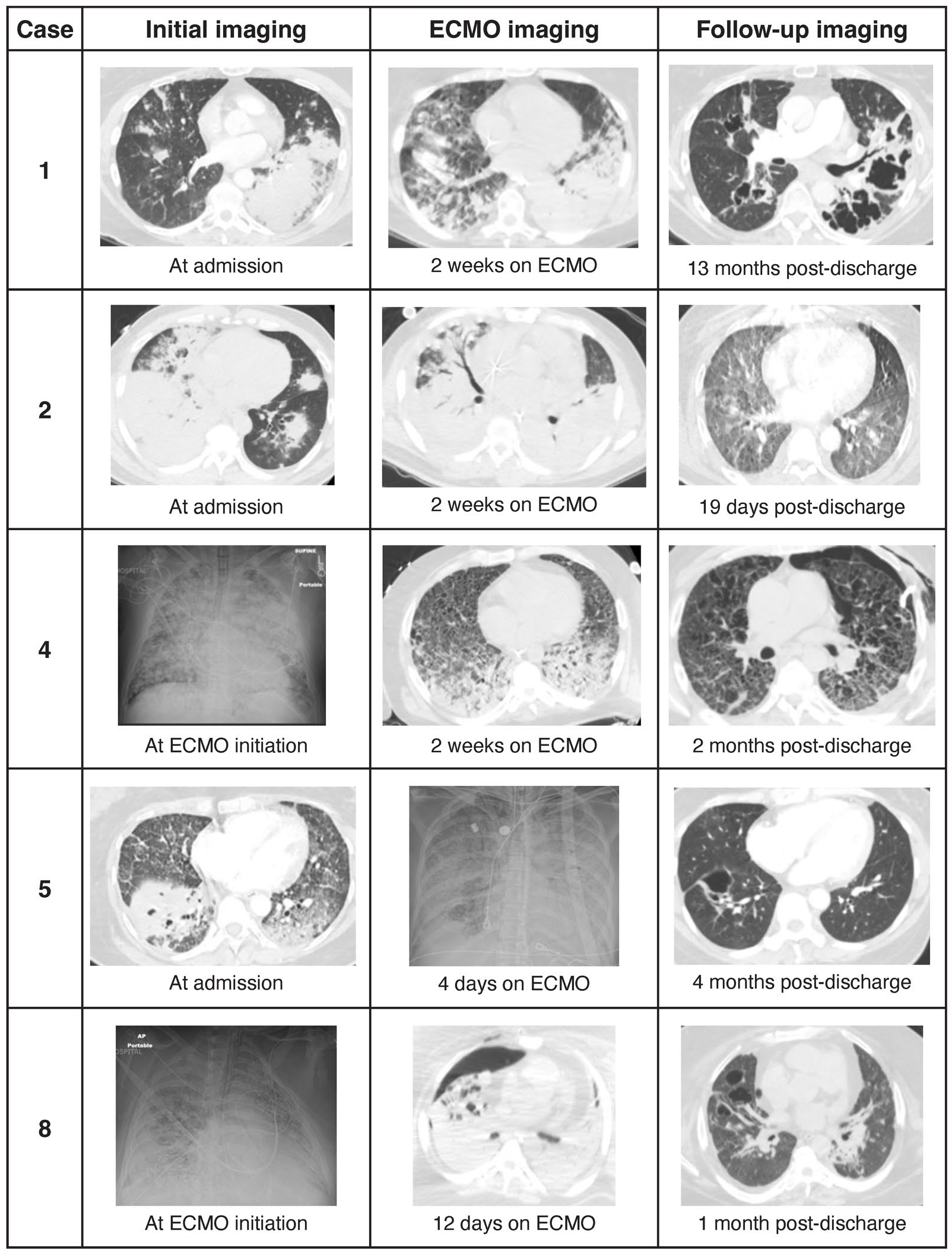
Examples of the initial, during ECMO course, and post-hospital discharge lung images
Discussion
This report demonstrates the feasibility of VV-ECMO use as a rescue therapy in patients with blastomycosis-related respiratory failure. While cases from non-endemic areas have been described, most originate from endemic areas which is consistent with our observations and reflects our center’s referral base.5,6,10,11 We identified the likely exposure in only three of eight cases, confirming the difficulty in determining the specific source and the possibility of the disease without known risk factors.11,12 While most patients with proven diagnoses of blastomycosis are not immunocompromised, those with immunosuppression have been reported to have higher rates of severe infection, respiratory failure, and mortality.5,7,10 The absence of immunosuppression in any of our patients could have been a factor positively affecting their outcomes. The duration of mechanical ventilation, VV-ECMO support, and hospital LOS in our cohort were consistent with another report on VV-ECMO in patients with refractory blastomycosis-associated ARDS. 13 However, the decision to initiate ECMO support in our study was relatively expeditious, with an average of 57 h from intubation to ECMO cannulation, with the longest case of 96 h. While it is difficult to compare mortality rates from various publications due to the likely differences in patient selection and other confounding factors, the relatively rapid decision to implement ECMO support may have contributed to the high survival rate in our study.
The literature on long-term radiologic abnormalities in patients who survived life-threatening lung injury due to blastomycosis is very limited, especially in those treated with VV-ECMO.14,15 Consistent with another report showing overall improvement but the persistence of some abnormalities (interlobar septal thickening, bronchiectasis, and nodularity) on chest CT obtained within 1- to 2-year time frame, we also demonstrate significant improvements on the follow-up CT imaging. 6 However, the most common post-discharge CT imaging findings in our patients likely reflect a shorter time to follow-up and include residual infiltrates and pneumatoceles, and pneumothorax in one of the cases. Despite the presence of radiologic abnormalities, all survivors were weaned from mechanical ventilation, functioning independently, and only the minority required supplemental oxygen at the time of follow-up. This is consistent with another report showing recovery of pulmonary function in survivors of severe disease at long-term follow-up. 16
Our study demonstrates that despite the complex and resource-intensive ECMO course, survival to hospital discharge is possible, and patients’ respiratory status can improve over time. We demonstrate the possibility of VV-ECMO implementation and a favorable outcome in the setting of severe obesity which, to our knowledge, has not been previously reported in the literature for this indication.
The strengths of this report include the relatively large number of cases treated within the most recent time frame as well as the ability to provide information on post-hospital discharge follow-up, including imaging. The study was limited by the retrospective design, limited number of cases reflecting the rarity of the extreme presentation of the disease, and a single-center population. The post-hospitalization follow-up was not protocolized, and the inclusion of imaging studies was based on their availability.
Conclusion
In patients with blastomycosis-related respiratory failure, support with VV-ECMO is feasible and may result in favorable clinical outcomes. VV-ECMO can be used as a rescue therapy in patients with refractory blastomycosis-related ARDS, including those with morbid obesity. Rapid initiation of ECMO support in eligible patients may have contributed to the good outcomes. Significant clinical improvement is possible despite the persistence of the residual radiologic abnormalities. Further research is necessary to determine the optimal time of ECMO initiation and strategies to improve outcomes in this patient population.
