Abstract
Re-expansion pulmonary edema is defined as pulmonary edema that occurs when a chronically collapsed lung rapidly re-expands, most commonly following chest tube placement for pneumothorax, re-expansion of severe atelectasis, and evacuation of pleural effusion. Though it is very rare, the sudden onset and clinical features of re-expansion pulmonary edema make it a lethal complication that requires urgent treatment. We present a 60-year-old patient who underwent an aortic valve replacement with pre-existing large bilateral pleural effusions. Intraoperatively, upon evacuation of the pleural effusions, the patient developed worsening lung compliance, refractory hypoxemia, and hypercapnia that required emergent veno-venous extracorporeal membrane oxygenation support.
Keywords
Introduction
Re-expansion pulmonary edema (RPE) is a rare but lethal complication that occurs when a chronically collapsed lung re-expands rapidly.1–3 RPE is associated with a relatively high mortality rate of 15%–20% due to sudden onset and severe clinical features.4,5 RPE was first described in 1853 when Pinault described an occurrence of pulmonary edema after evacuation of a pleural effusion. 6 Although the exact pathophysiology of RPE remains undefined, it is thought to be caused by pulmonary microvasculature abnormalities in the setting of chronic lung collapse and mechanical stress upon rapid re-expansion. 1 The onset of RPE is often sudden and immediate, with most patients experiencing symptoms of significant pulmonary edema within 1 h following re-expansion. 1 This severe edema would create a shunt that poses a challenge to oxygenation that is refractory to the conventional first-line therapy. It also worsens lungs compliance, causing an increase in airway pressure, making ventilation insufficient. Furthermore, this increase in airway pressure would decrease in venous return to the heart, adding hemodynamic instability to the overall clinical picture. 7 Urgent temporary measures for circulatory and respiratory support are often required for these patients, including endotracheal intubation with ventilator support, with veno-venous extracorporeal membrane oxygenation (V-V ECMO) being the next option. 4 The most common radiographic finding are ground-glass opacities on chest x-ray and smooth muscle thickening, and consolidation on computed tomography. 8 In most cases, symptoms of RPE often resolve within 24–72 h but can last up to 4 days or more from the onset. 1 Here we report a case of severe RPE during a cardiac surgery that required an emergent V-V ECMO intraoperatively.
Case report
A 60-year-old male with a past medical history of aortic valve endocarditis in the setting of an infected pacemaker lead, hypertension, hyperlipidemia, coronary artery disease, peripheral vascular disease, sick sinus syndrome, abdominal aortic aneurysm, type 2 diabetes, and tobacco use presented to the operating room for a surgical aortic valve replacement. Preoperative chest X-ray, taken approximately 1 month prior to the surgery, was unremarkable, except for a dull costophrenic angle on the left lung, suggesting a pre-existing left pleural effusion (Figure 1). Standard anesthetic care for cardiac surgery was provided. Induction of anesthesia was uneventful. Post-induction intraoperative transthoracic echocardiography showed severely decreased left ventricular ejection fraction of 30% with severe aortic insufficiency, a mobile vegetation on the aortic valve, a moderate pericardial effusion, and large bilateral pleural effusions (Figure 2). The patient required a transfusion of two units of packed red blood cells at the beginning of surgery due to the chronic and expected dilutional anemia with the initiation of cardiopulmonary bypass (CPB). The aortic valve was successfully replaced under CPB, and the patient was smoothly separated from CPB with minimum inotropic support. The patient remained hemodynamically stable over the subsequent hour. Prior to sternal closure at the conclusion of the surgery, the surgeons removed a total of 1500 mL of serous pleural fluid from the bilateral chest cavities. The evacuation of the pleural effusions was completed within several minutes; thereafter, Valsalva maneuvers were applied to help expand the atelectatic lungs. A few minutes after this event, the patient’s oxygen saturation began to drop gradually. Targeted tidal volumes became unachievable with increased peak airway pressures of 50 cmH2O, which was significantly increased from 16 cmH2O. Oxygen saturation continued to drop precipitously despite the maximal effort of ventilation support. The lowest oxygen saturation recorded during this event was in the low 70s %. Upon checking the endotracheal tube, visible serous frothy fluid was pouring out. Aggressive pulmonary toilet, including tracheal suctioning, the Valsalva maneuver, and positive end-expiratory pressure, was attempted without improvement. Endotracheal tube suction continued to yield copious amounts of secretions. The surgical team was immediately notified, and after discussion, it was decided to place the patient on V-V ECMO emergently. While waiting for the ECMO team to arrive and prepare, inhaled nitric oxide was empirically administered to attempt improving ventilation with minimal improvement. Furosemide 40 mg was also administered intravenously to promote diuresis. The patient’s arterial blood gas values changed dramatically from near-normal to a pH of 7.08, PaO2 of 69 mmHg, PaCO2 of 70 mmHg, and HCO3 of −9 mEq/L, while ventilation effort was maximized with 100% oxygen. The ECMO team arrived in the operating room approximately 10 min after the consultation. The patient’s respiratory condition further deteriorated within the next several minutes. Within the first minute of completing V-V ECMO cannulations via the common femoral veins, ventilation became impossible to generate any tidal volumes due to the extremely low lung compliance. The V-V ECMO was promptly initiated, and both severe hypoxia and hypercarbia were markedly improved.

Preoperative chest X-ray taken approximately 1 month prior to surgery. Unremarkable findings except for a dull costophrenic angle on the left lung, indicative of a pre-existing left pleural effusion.

Intraoperative transesophageal echocardiography shows bilateral pleural effusions and atelectasis in the lungs. (a) Large right pleural effusion. (b) Large left pleural effusion. (c) Reduced right pleural effusion after evacuation. (d) Reduced left pleural effusion after evacuation.
The patient was transferred to the cardiovascular intensive care unit (CVICU) in a stable condition and remained on V-V ECMO. A bronchoscopy was performed in the CVICU, resulting in the removal of 1000 mL of serous fluid. A chest X-ray revealed diffuse patchiness in both lungs, with the right lung being worse than the left (Figure 3). The patient was then aggressively diuresed for the following 2 days. De-cannulation of V-V ECMO cannulas occurred on postoperative day (POD) two and the patient was successfully extubated on POD three. The remaining of his recovery was uneventful.
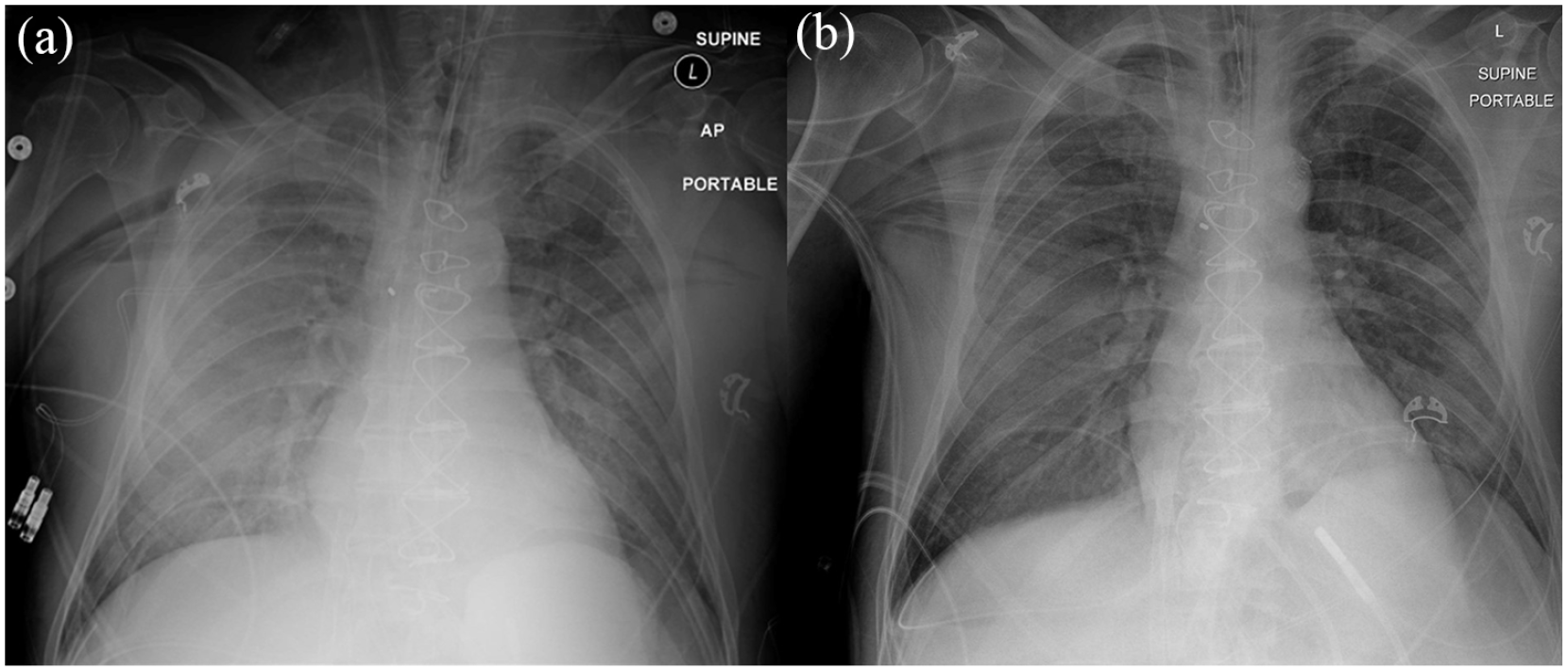
Postoperative chest X-rays. (a) Diffuse ground-glass opacity representing pulmonary edema, with the right lung worse than the left lung on postoperative day (POD)1. (b) Resolving pulmonary edema on POD3.
Discussion
The development of RPE following the re-expansion of a chronically collapsed lung is a rare but potentially life-threatening complication. This condition can result in significant morbidity and mortality if not promptly recognized and managed. The sudden onset and severe clinical features of RPE, such as refractory hypoxemia, hypercapnia, and hemodynamic instability, pose a considerable challenge, necessitating rapid decision-making to provide the necessary support to the patient. 1
There have been few reports that mention the requirement to initiate V-V ECMO for RPE during the intraoperative period. In such situations, effective communication between the anesthesia and surgical teams is crucial to ensure that appropriate interventions are implemented quickly. In this presented case, the decision to consult the ECMO team was made expeditiously, ultimately saving the patient’s life. We believe that the patient would not have survived had the ECMO team not arrived promptly due to rapid deterioration of the patient’s respiratory status, including worsening oxygenation and ventilation despite maximum ventilation efforts. By the time of ECMO initiation, ventilation became impossible to generate any tidal volumes.
While there are no randomized studies investigating methods to prevent the onset of RPE, the American College of Chest Physicians recommends against draining more than 1000 mL of fluid at once to minimize the risk of RPE development.2,5 This recommendation highlights the significance of adopting appropriate strategies for managing patients with chronically collapsed lungs who are undergoing re-expansion procedures. Examples of these strategies may include gradual fluid removal, avoidance of acute lung expansion with forced Valsalva maneuver, and close monitoring of patient’s vital signs and symptoms during the re-expansion process. In addition to these preventive measures, early recognition of RPE signs and symptoms is crucial for initiating timely interventions, as this can significantly impact patient outcomes.
Conclusion
Although uncommon, the sudden onset and clinical features of RPE make it a fatal complication that requires urgent treatment. Prompt recognition and management of RPE are critical in preventing severe complications and improving patient outcomes. Effective communication, collaboration, and adherence to established guidelines can help ensure optimal management of patients with chronically collapsed lungs undergoing re-expansion procedures. Healthcare providers should remain vigilant for the potential development of RPE in vulnerable patients and be prepared to act swiftly in the event of its occurrence, as timely intervention can make a life-saving difference.
Footnotes
Acknowledgements
None.
Author contributions statement
N.B. Cavanaugh wrote the initial draft and contributed to manuscript writing. L.H.N. contributed to manuscript writing. L.A. was the primary intensivist for the case and contributed to manuscript writing. A.S. was the primary surgeon for the case and contributed to manuscript writing. S.H. was the primary cardiac anesthesiologist for the case and contributed to manuscript writing. All authors approved the final version of the manuscript and have ensured the integrity and accuracy of the work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Department of Anesthesia at the University of Iowa Carver College of Medicine.
Ethics approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the patient for their anonymized information to be published in this article.
