Abstract
Background:
Acute exacerbation (AE) refers to rapidly progressive respiratory deterioration in the clinical course of interstitial lung disease (ILD). Progressive pulmonary fibrosis (PPF) is the chronic progressive phenotype of ILD. No study has investigated the relationship between AE and PPF in ILD.
Objectives:
We aimed to determine the association between AE and PPF in ILD patients.
Design:
A retrospective cohort study.
Methods:
A total of 414 patients hospitalised for ILD were included in our study. The clinical presentations, radiographic features and laboratory findings of the patients were reviewed.
Results:
AE was present in 120 (29.0%) ILD patients and was associated with a higher risk of death than non-AE patients in the whole cohort (HR 2.893; 95% CI, 1.847–4.529; p < 0.001). However, the significant difference disappeared when stratified by PPF (HR 1.192; 95% CI, 0.633–2.247; p = 0.586) and non-PPF (HR 1.113; 95% CI, 0.384–3.223; p = 0.844). In addition, the adverse effect of PPF on prognosis remained consistent in both AE and non-AE patients. Multivariable logistic regression analysis showed that compared with non-PPF patients, only age was a risk factor for PPF in AE-ILD, while the risk factors for PPF in the non-AE group were age, definite usual interstitial pneumonia and mediastinal lymph node enlargement.
Conclusion:
In the context of ILD, both AE and PPF were found to be associated with poor prognosis. However, the adverse effect of AE on prognosis disappeared when PPF was considered as a stratification feature, whereas the adverse effect of PPF on prognosis persisted in both AE and non-AE individuals. Therefore, it is important to investigate effective strategies to prevent disease progression after AE. Increased recognition and attention to PPF and early antifibrotic therapy at the appropriate time is also warranted.
Plain language summary
Why was the study done? Acute exacerbation (AE) is an acute respiratory worsening of interstitial lung disease (ILD). Progressive pulmonary fibrosis (PPF) is a chronic progressive-fibrosing form of ILD. The relationship between AE and PPF in ILD remained unclear. We aimed to determine the association between AE and PPF in ILD patients.
What did the researchers do? The researchers studied 414 patients with ILD to see how AE and PPF affect the outcome of ILD and explored the risk factors for PPF in ILD.
What did the researchers find? AE was present in 120 (29.0%) ILD patients and was associated with higher risk of death than non-AE patients in the whole cohort. However, the significant difference disappeared when stratified by PPF and non-PPF. In addition, the adverse effect of PPF on prognosis remained consistent in both AE and non-AE patients. In AE-ILD patients, age was the only risk factor for PPF. In the non-AE group, age, definite usual interstitial pneumonia and mediastinal lymph node enlargement were risk factors for PPF.
What do the findings mean? The findings suggest that it is important to investigate effective strategies to prevent disease progression after AE. Increased recognition and attention to PPF and early antifibrotic therapy at the appropriate time is also necessary.
Introduction
Interstitial lung diseases (ILD) encompass a group of wide and heterogeneous pulmonary parenchymal disorders including more than 200 acute and chronic diseases with known or unknown aetiology.1,2 AE refers to an acute, clinically significant respiratory deterioration characterised by evidence of new widespread alveolar abnormality in the clinical course of ILD. 3 Progressive-fibrosing ILD (PF-ILD) is the name given to patients with fibrosing ILD who develop a progressive phenotype. PF-ILD comprises a diverse group of ILD subtypes. Among these, all idiopathic pulmonary fibrosis (IPF) patients develop a progressive fibrosing phenotype, which can be regarded as the prototype of PF-ILD. However, progressive fibrosis only occurs in a proportion of patients with other ILD subtypes. 4 Progressive pulmonary fibrosis (PPF) is a new terminology recently used to describe the chronic progressive phenotype of ILD, the difference is that PPF excludes IPF patients compared to PF-ILD. 5 Both AE and PPF have been identified as poor prognostic factors for ILD. 6 One study reported that the proportion of PF-ILD just before AE was 20% in systemic autoimmune disease-related ILD. 7 There is no relevant study on the relationship between AE and PPF in ILD, so we believe that further research is necessary to reveal the significance of PPF and AE in ILD.
Methods
Study design and patient selection
This study retrospectively reviewed 414 patients hospitalised for ILD at the affiliated hospital of Qingdao University, Shandong, Qingdao, China, from December 2013 to December 2020. All subjects had a confirmed multidisciplinary diagnosis of ILD according to guidelines.8–11 We used the revised definition for AE proposed by an international working group in 2016 in patients with IPF for all ILD patients. 3 The diagnostic criteria for AE-ILD were as follows: (1) previous or concurrent diagnosis of ILDs; (2) acute worsening or development of dyspnoea typically of less than 1-month duration; (3) computed tomography (CT) with new bilateral ground-glass opacity and/or consolidation superimposed on a background pattern consistent with ILDs; and (4) deterioration not fully explained by cardiac failure or fluid overload. AE was further categorised as triggered or idiopathic AE depending on whether an underlying trigger (e.g. infection, drug toxicity, aspiration and mechanical stretch) for AE is found. Although the 2016 AE-IPF criteria 3 no longer require the exclusion of triggers such as infection or other potential triggers, the patients with simple infection and no AE (identified causative organism and rapid improvement and response to antibiotic treatment alone) were categorised as an acute worsening of dyspnoea due to infection rather than AE.12,13 In addition, pulmonary embolism was excluded by negative CT pulmonary arteriography. Heart failure was excluded by normal echocardiography, normal levels of B-natriuretic peptide and a negative clinical evaluation. PPF was defined as the presence of at least two of the following three criteria other than IPF: (1) worsening respiratory symptoms; (2) physiological evidence of disease progression (one of the following): absolute decline in forced vital capacity (FVC) >5% predicted within 1 year of follow-up, or absolute decline in diffusion capacity of the lung for carbon monoxide (DLCO) (corrected for haemoglobin) >10% predicted within 1 year of follow-up; (3) radiological evidence of disease progression (one or more of the following): (i) increased extent or severity of traction bronchiectasis and bronchiolectasis, (ii) new ground-glass opacity with traction bronchiectasis, (iii) new fine reticulation, (iv) increased extent or increased coarseness of reticular abnormality, (v) new or increased honeycombing or (vi) increased lobar volume loss. 5
The inclusion criteria for this study were patients who had a multidisciplinary diagnosis of ILD and baseline chest CT scan obtained within 1 year of ILD diagnosis. The exclusion criteria were patients who had confirmed or suspected malignancy, concurrent pulmonary infection, uncontrolled heart disease, sarcoidosis, occupational lung disease or radiation pneumonitis. Patients without chest CT scans for review, or lost to follow-up were also excluded. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 14
Data collection
The medical records of each patient’s initial clinic visit, including demographic characteristics, clinical symptoms and signs, comorbid conditions, smoking history, laboratory and chest CT imaging findings, ultrasonic cardiogram and pulmonary function test, were reviewed. Blood samples were collected on an empty stomach in the early morning after hospitalisation. Biochemical parameters were measured using the Beckman AU5811 automatic biochemical analyzer (Beckman Coulter Inc, Krefeld, Germany) with commercially available kits (Leadman Biochemistry Co., Beijing, China). Spirometry was performed using a Jaeger Medical spirometer (Jaeger, Omaha, NE, USA) as per standard guidelines.
Chest CT evaluation
Two radiologists blinded to clinical and outcomes data independently measured all the chest CT scans. In case of discrepant readings, readings were re-evaluated by a third radiologist with the greatest experience in pulmonary imaging. The degree of fibrosis was evaluated using semiquantitative image analysis.15,16 Briefly, CT images were scored at five levels (the origin of great vessels, the main carina, the pulmonary venous confluence, halfway between the third and fifth sections and immediately above the right hemidiaphragm) and disease extent was visually scored in each of the five CT sections (0, absent; 1, 1%–25%; 2, 26%–50%; 3, 51%–75%; and 4, 76%–100%). The total extent of fibrosis was calculated as the mean extent score in the five scored CT sections. The modified coarseness of reticular disease (MCRD) was also calculated in each of the five sections as follows: 0, normal; 1, ground-glass opacity alone; 2, fine intralobular fibrosis; 3, microcystic honeycombing (⩽4 mm); 4, macrocystic honeycombing (>4 mm). MCRD was estimated based on the summed score for all five levels. The total fibrosis score was the product of the total extent of fibrosis and MCRD. All CT images were reviewed at lung window settings (with a window centre of −500 to −600 HU and a window width of 1600 HU). MLNs were assessed using soft tissue windows only (level 35 Hounsfield unit (HU), width 450 HU) based on the International Association for the Study of Lung Cancer (IASLC; Denver, CO, USA) nomenclature. 17 MLN measurements were provided by radiologists from the reformatted imaging data using virtual calipers and enlargement was defined as short-axis diameters ⩾10 mm.18,19
Follow-up and endpoint of the study
The primary endpoint of our study was transplant-free survival, defined as the time from the diagnosis of ILD to death or lung transplantation. Each patient was followed up until the occurrence of death, lung transplantation, end of the study period or loss of follow-up. Follow-up time was censored on 14 September 2023. The secondary endpoints included physician-diagnosed AE and PPF of ILD.
Statistical analysis
The ILD patient group was divided and compared according to whether the patients had AE. Categorical variables were processed using the χ2 test or Fisher’s exact tests, as appropriate. The Shapiro–Wilk W-test was used to assess the normality of the data. Student’s t-test and the Mann–Whitney test were used for parametric and non-parametric distribution variables, respectively. After adjusting for study covariates, multivariate logistic regression analysis was performed to explore the relevant risk factors in ILD patients. Kaplan–Meier survival analysis and Cox proportional hazards regression analysis were used to compare all-cause mortality. Analysis was performed using SPSS v26.0 (IBM Corporation, Armonk, NY, USA). The statistical significance of the difference was set at 0.05.
Results
Comparison of characteristics according to the presence of AE
Baseline characteristics of 414 patients with ILD included in this study are shown in Table 1. AE was present in 120 (29.0%) patients. Study participants with AE were significantly older (66.0 years vs 64.0 years), had a longer time to be diagnosed with ILD (181.25 days vs 90.00 days) and had lower albumin/globulin ratio than patients without AE. In addition, the AE group had significantly worse lung function, higher total fibrosis score (19.70 vs 16.00; p = 0.014), mortality (37.3% vs 13.5%; p < 0.001) and prevalence of mediastinal lymph node enlargement (MLNE; 45.8% vs 29.3%; p = 0.002) and PPF (70.0% vs 11.9%; p < 0.001) compared with the non-AE group.
Characteristics of patients with ILD, according to the presence of AE.

AE, acute exacerbation; CA125, carbohydrate antigen 125; CEA, carcinoembryonic antigen; CHD, coronary heart disease; CT, computed tomography; DLCO, lung diffusion capacity for carbon monoxide; FEV1, forced expiratory volume in 1 s; FVC, forced vital capacity; ILD, interstitial lung disease; IQR, interquartile range; MCRD, modified coarseness of reticular disease; MLNE, mediastinal lymph node enlargement; PPF, progressive pulmonary fibrosis; RV, residual volume; SCC-Ag, squamous cell carcinoma antigen; VA, alveolar ventilation; VC, vital capacity; TLC, total lung capacity.
Based on the above results, multivariate logistic regression analysis was performed to show that age (OR 1.030, 95% CI 1.006–1.055; p = 0.014) and MLNE (OR 1.785, 95% CI 1.128–2.825; p = 0.013) were risk factors for AE in ILD patients. However, the total fibrosis score (OR 1.012, 95% CI 0.996–1.028; p = 0.132) was not a risk factor (Table 2).
Risk factors for AE in ILD patients.
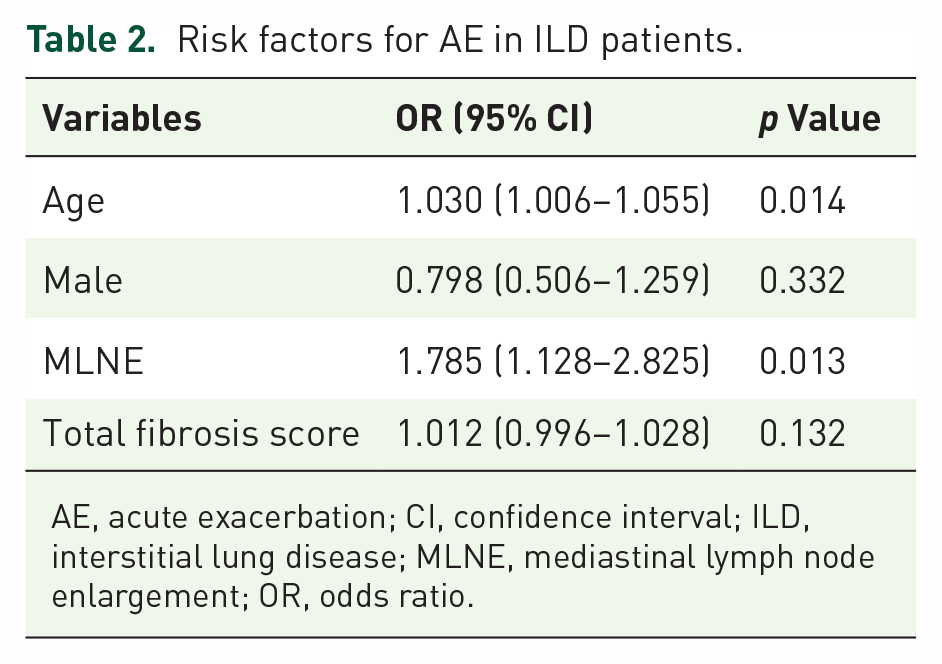
AE, acute exacerbation; CI, confidence interval; ILD, interstitial lung disease; MLNE, mediastinal lymph node enlargement; OR, odds ratio.
Survival analysis of AE in ILD patients stratified by progressive phenotype
First, we performed Kaplan–Meier survival analysis and Cox proportional hazards regression analysis to find that ILD patients with AE had a higher risk of death compared to patients without AE in the whole cohort (HR 2.893; 95% CI, 1.847–4.529; p < 0.001). However, the significant difference disappeared when we further evaluated the survival of AE-ILD stratified by PPF (HR 1.192; 95% CI, 0.633–2.247; p = 0.586) and non-PPF (HR 1.113; 95% CI, 0.384–3.223; p = 0.844). (Figure 1)

Survival analysis of AE-ILD stratified by progressive phenotype.
Survival analysis of PPF in ILD patients with or without AE
Kaplan–Meier survival analysis and Cox proportional hazards regression analysis were performed and found that PPF had a higher risk of death compared with non-PPF in ILD patients with (HR 5.254; 95% CI, 1.868–14.775; p = 0.002) or without AE (HR 4.514; 95% CI, 2.245–9.075; p < 0.001; Figure 2).

Survival analysis of PPF in ILD patients with or without AE.
Risk factors for PPF in ILD patients with or without AE
In the AE group, we found that the PPF patients were older, more frequently combined with hypertension and had higher pulmonary arterial systolic pressure (PASP), prevalence of MLNE and mortality than non-PPF patients. Multivariable logistic regression analysis showed that only age (OR 1.046; 95% CI, 1.002–1.091; p = 0.038) was a risk factor for PPF compared to non-PPF. See Supplemental Table S1 and Table 3 for details.
Risk factors for PPF in ILD patients with or without AE.

AE, acute exacerbation; CI, confidence interval; ILD, interstitial lung disease; MLNE, mediastinal lymph node enlargement; OR, odds ratio; PPF, progressive pulmonary fibrosis; UIP, usual interstitial pneumonia.
In the non-AE group, we found that the PPF patients were older, had a longer time to diagnose and higher prevalence of MLNE, definite usual interstitial pneumonia (UIP) and mortality than non-PPF patients. Multivariable logistic regression analysis showed that age (OR 1.045; 95% CI, 1.001–1.091; p = 0.044), definite UIP (OR 3.473; 95% CI, 1.384–8.711; p = 0.008) and MLNE (OR 3.353; 95% CI, 1.466–7.670; p = 0.004) were risk factors for PPF compared to non-PPF. See Supplemental Table S2 and Table 3 for details.
Discussion
The frequency of AE in patients with ILD was reported to range from 3.19% to 55.6%.20–26 In our cohort, AE was present in 29.0% of patients and had significantly older age, longer time to diagnose, lower albumin/globulin ratio, worse lung function, higher total fibrosis score, mortality and prevalence of MLNE and PPF than patients without AE. As studies reported, older age, UIP pattern, low %FVC or %DLCO, high serum C-reactive protein or D-dimer level, monocyte count and hypoalbuminemia were associated with the development of AE in patients with ILD.27–30 Our analysis showed that older age and MLNE were independent predictive factors for AE in ILD patients.
Almost all literature had reported that AE adversely affected the prognosis of ILD.21–23,25,27 We also confirmed this point that ILD patients with AE had a higher risk of death compared to patients without AE in the whole cohort. However, the significant difference disappeared when stratified by PPF and non-PPF. As our previous study reported, PPF was associated with poor prognosis in ILD patients. 31 We further compared the survival of PPF with non-PPF stratified by the presence of AE in ILD patients and found that the significant difference remained. These seemed to indicate that PPF was more important than AE for the prognosis of ILD patients. Therefore, we must concentrate on the risk factors for PPF in ILD patients. Multivariable logistic regression analysis showed that the only risk factor for PPF in the AE group was age. In the non-AE group, age, definite UIP and MLNE were all found to be risk factors for PPF compared to non-PPF. Different factors may be of concern for PPF in ILD patients with or without AE. The relationship between MLNE and PPF was discussed in our previous article. 31
To our knowledge, the present study is the first to investigate the association between acute exacerbation and PPF of ILD. Our findings suggest that the adverse effect of AE on prognosis disappears after stratification by whether ILD is chronic progressive. So AE is not important in ILD patients? We cannot generalise that. The proportion of PPF in AE-ILD patients is still large at 70.0%, which was much higher than 11.9% in ILD patients without AE. Therefore, it is imperative to acknowledge that AE holds significance in ILD patients and to remain vigilant towards risk factors for AE-ILD. Meanwhile, more attention should be paid to the chronic progression of ILD patients and close follow-up is essential.
Currently, very few therapeutic options (including pulse corticosteroids) are available for the treatment of AE-ILD, which are mainly based on the correction of hypoxemia, palliation of symptoms and looking for reversible aetiologies of respiratory decline.32–34 Investigating effective treatment strategies for AE-ILD to prevent disease progression represents a future research direction. On the other hand, regarding the potential persistent adverse effects of PPF on prognosis in ILD patients independent of the occurrence of AE, it would be advantageous to allocate greater attention towards the identification of PPF and implementing antifibrotic therapy appropriately at an early stage.
The main limitation of our study is due to its retrospective and single-centre design. Furthermore, while the majority of CT scans were high-resolution ILD protocol images, a few patients had standard chest CT scans only. As only hospitalised patients were included, this may introduce a bias (e.g. overestimation of AE). Finally, our analysis results had limited generalisability to other countries.
Conclusion
In the context of ILD, the detrimental effect of AE on prognosis disappeared when considering PPF as a stratification factor; however, the adverse effect of PPF on prognosis remained consistent in both AE and non-AE patients. Therefore, future studies should focus on investigating effective strategies for preventing disease progression after AE. Increased recognition and attention to PPF and early antifibrotic therapy at the appropriate time is also warranted.
Supplemental Material
sj-docx-1-tar-10.1177_17534666241276800 – Supplemental material for Association between acute exacerbation and progressive pulmonary fibrosis in interstitial lung disease: a retrospective cohort study
Supplemental material, sj-docx-1-tar-10.1177_17534666241276800 for Association between acute exacerbation and progressive pulmonary fibrosis in interstitial lung disease: a retrospective cohort study by Liying Zhai, Zhiqiang Wang and Wencheng Yu in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-docx-2-tar-10.1177_17534666241276800 – Supplemental material for Association between acute exacerbation and progressive pulmonary fibrosis in interstitial lung disease: a retrospective cohort study
Supplemental material, sj-docx-2-tar-10.1177_17534666241276800 for Association between acute exacerbation and progressive pulmonary fibrosis in interstitial lung disease: a retrospective cohort study by Liying Zhai, Zhiqiang Wang and Wencheng Yu in Therapeutic Advances in Respiratory Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
