Abstract
Background:
The association between interstitial lung disease (ILD) and antineutrophil cytoplasmic antibody (ANCA)–associated vasculitis (AAV) has been increasingly recognized in recent years. The clinical features and prognostic differences between AAV-associated ILD and isolated ANCA-positive idiopathic interstitial pneumonias (IIPs) remain unclear. The purpose of this study was to determine the clinical significance and prognosis of ANCA-positive ILD to further guide clinical management.
Methods:
This study retrospectively reviewed the data of 379 ILD patients with available ANCA results and ultimately analysed 49 ANCA-positive patients. AAV diagnosis was based on the 2012 revised Chapel Hill Consensus Conference (CHCC) criteria, and 33 of 49 patients were diagnosed with microscopic polyangiitis (MPA). The baseline clinical information and laboratory parameters were collected and analysed at each patient’s initial diagnosis.
Results:
Among 49 ANCA-positive ILD patients, the high-resolution computed tomography (HRCT) pattern was mainly usual interstitial pneumonia (UIP) (59.18%), followed by nonspecific interstitial pneumonia (NSIP) (26.53%). The C-reactive protein (CRP) level (43.89± 40.61 versus 18.74± 20.05, p = 0.028) and erythrocyte sedimentation rate (ESR) (71.97± 42.73 versus 40.69± 28.46, p = 0.011) were significantly higher in the MPA-ILD group than in the ANCA-IIP group. Haemoglobin (113.09 ± 24.47 versus 132.19± 13.34, p = 0.006) and albumin (32.95± 5.84 versus 36.52± 3.94, p = 0.032) levels were significantly lower. Survival was shorter among MPA-ILD patients than among ANCA-IIP patients [hazard ratio (HR) 3.38, 95% confidence interval (CI) 1.32–8.67, p = 0.040]. In the multivariable Cox analysis, a diagnosis of MPA (HR 3.91, 95% CI 1.07–14.08, p = 0.038) and acute exacerbation (AE) of ILD (HR 9.43, 95% CI 2.89–30.30, p < 0.001) were significantly independently associated with shorter survival in ANCA-positive ILD patients, and the NSIP pattern (HR 0.07, 95% CI 0.01–0.41, p = 0.003) was independently associated with prolonged survival.
Conclusion:
ANCA-ILD patients mostly have myeloperoxidase (MPO)-ANCA positivity and an MPA diagnosis. Survival was shorter among MPA-ILD patients than among ANCA-IIP patients. Respiratory failure and AE were associated with poorer prognosis. Early antifibrotic treatment may be a reasonable treatment option in fibrotic ILD patients with ANCA positivity.
Keywords
Introduction
Antineutrophil cytoplasmic antibodies (ANCAs) are a family of autoantibodies that are specific for antigens that are predominantly expressed in cytoplasmic granules of neutrophils and the lysosomes of monocytes. Myeloperoxidase (MPO) and proteinase 3 (PR3) are the two major ANCA antigens. Testing for ANCA directed towards MPO and PR3 is commonly performed to support the diagnosis of ANCA-associated vasculitis (AAV). 1 AAV is a group of systemic vasculitides affecting small blood vessels and is characterized by the presence of circulating ANCAs. Clinical disease phenotypes include microscopic polyangiitis (MPA), granulomatosis with polyangiitis (GPA) and eosinophilic granulomatosis with polyangiitis (EGPA).2,3
Interstitial lung diseases (ILDs) are a group of diffuse parenchymal lung disorders of variable aetiologies, including idiopathic interstitial pneumonias (IIPs), connective tissue disease–associated interstitial lung diseases (CTD-ILD), interstitial pneumonia with autoimmune features (IPAFs) and so on. 4 The association between ILD and ANCA or AAV has been increasingly recognized in recent years. Previous studies have shown that MPO-ANCA and MPA have the strongest association with ILD.5–7 In clinical practice, ILD patients with ANCA positivity and extrapulmonary involvement meet the AAV diagnostic criteria and receive a diagnosis of AAV-ILD, but some ILD patients present with isolated ANCA positivity without manifestations of extrapulmonary systemic vasculitis. This group of patients can be initially diagnosed with idiopathic pulmonary fibrosis (IPF), IIP or IPAF before systemic vasculitis manifestations occur in some patients.
The clinical features and prognostic differences between AAV-ILD and isolated ANCA-positive IIP and whether ILD patients with isolated ANCA-positive IIP should be treated as AAV-ILD or IIP remain unclear, however. Therefore, we retrospectively analysed the baseline clinical data of patients diagnosed with ANCA-positive ILD in the past 8 years. This study aims to assess the clinical significance and prognosis of ANCA-positive ILD to further guide clinical management.
Methods
Study design
We retrospectively reviewed the medical records of patients diagnosed with ILD who had available ANCA testing results between March 2014 and June 2022 at the Affiliated Hospital of Medical School of Ningbo University. AAV diagnosis was based on the 2012 revised International Chapel Hill Consensus Conference (CHCC) Nomenclature of Vasculitides. 2 Patients who met MPA criteria could be included in the MPA-ILD group. On the contrary, patients with ANCA positivity who did not fulfil the AAV criteria were classified into the ANCA-IIP group. Acute exacerbation of interstitial lung disease (AE-ILD) was defined as an acute worsening or development of dyspnoea with a duration of <1 month, chest high-resolution computed tomography (HRCT) with new bilateral ground-glass opacity and consolidation superimposed on fibrosis, and deterioration not fully explained by cardiac failure or fluid overload.8,9 Patients with ILD secondary to CTD, hypersensitivity pneumonitis, sarcoidosis, drug and occupational exposure were excluded. The flow chart of patient screening and classification for this study is presented in Figure 1. The study was approved by the Ethics Committee of the Affiliated Hospital of Medical School of Ningbo University [institutional review board (IRB) no. KS20228007].

Flow chart of the study.
Data collection
The clinical data – including detailed patient histories, clinical manifestations, laboratory results and chest HRCT – were obtained from the medical records from the patients’ initial diagnoses, and the data included basic patient information (age, sex, medical history, diagnosis, smoking status, occupational history) and laboratory results [arterial blood gas analysis, lactate dehydrogenase, erythrocyte sedimentation rate (ESR), alanine aminotransferase, aspartate aminotransferase, creatine kinase, C-reactive protein (CRP), rheumatoid factor, serologic autoantibodies, ANCA, etc.]. ANCAs were assessed by enzyme-linked immunosorbent assay (ELISA) or indirect immunofluorescence assay determination of their specificity antigen (anti-MPO or PR3), according to the practice of each immunology laboratory.
Pulmonary function test (PFT) data – including forced vital capacity (FVC), FVC% predicted, forced expiratory volume in 1 s (FEV1), FEV1/FVC, diffusing capacity of lung for carbon monoxide (DLCO%) predicted – were also noted. Furthermore, the echocardiographic estimate of the systolic pulmonary artery pressure (SPAP) was also recorded. Radiographic information, including all previous chest CT (computed tomography) and HRCT scans, was collected. Independent, separate, retrospective reviews of the chest HRCT images of each patient were performed by two professional and experienced thoracic radiologists. The criteria of the 2013 American Thoracic Society/European Respiratory Society (ATS/ESR) classification of IIP were used to classify ILD patterns, 10 and the criteria of the 2018 ATS/ERS/Japanese Respiratory Society (JRS)/Latin American Thoracic Association (ALAT) collaboration were used to classify the usual interstitial pneumonia (UIP) pattern to develop clinical practice guidelines for the diagnosis and management of IPF. 11 The treatment regimens were recorded. The last follow-up was in June 2022, and the outcomes were defined as death from all causes.
Statistical analysis
Continuous variables are presented as the mean value ± standard deviation (SD). Continuous, nonnormally distributed data are presented as the median with interquartile range (IQR). For the two-group comparisons of binary data, the Chi-square test or Fisher’s exact test was used, and a t test or the Mann–Whitney U test was used to compare continuous variables between two groups. The Kaplan–Meier survival analysis was performed to estimate the survival rates and plot survival curves, and the log-rank test was used to compare the intergroup differences. A multivariate Cox regression model was used to identify variables independently associated with death and variables associated with a given outcome at p < 0.1 in the Kaplan–Meier analysis. Statistical analysis was performed using IBM SPSS 26 (SPSS Inc., Chicago, IL, USA) and GraphPad Prism 9 software (GraphPad Software, San Diego, CA, USA). A p value < 0.05 was considered to indicate statistical significance.
Results
Study design
Three hundred seventy-nine ILD patients were screened, and 175 patients with CTD-ILD, 12 with occupational ILD, 6 with chronic hypersensitivity pneumonitis (CHP), and 2 with sarcoidosis were excluded from the study. Forty-nine patients had findings of ANCA positivity at the final diagnosis, with 33 patients in the MPA-ILD group and 16 patients in the ANCA-IIP group (Figure 1). A total of 12.93% (49/379) of patients with ILDs and 26.63% (49/184) of patients with IIPs were ANCA-positive.
Demographic and clinical characteristics
The demographic and main characteristics of ANCA-positive ILD are summarized in Table 1. There were 49 patients with ANCA-positive ILDs, 30 males and 19 females, with an average age of 72 years. Among them, 22 patients had a history of smoking. There were no significant differences in age, sex or smoking status between the MPA-ILD group and the ANCA-IIP group. The follow-up time was significantly longer for the ANCA-IIP group than for the MPA-ILD group (40.5 months versus 25.0 months).
Comparison between the MPA-ILD group and ANCA-IIP group in terms of demographic and clinical characteristics.

ANCA, antineutrophil cytoplasmic antibody; F, female; ILD, interstitial lung disease; IIP, idiopathic interstitial pneumonia; M, male; MPA, microscopic polyangiitis.
Data are presented as the median [IQR] or mean value ± SD or number (%).
p < 0.05 was considered significant.
The significance of bold values in Table 1 are meaning statistically differences.
The main clinical manifestations in our cohort included cough (61.22%), dyspnoea (55.10%), fever (36.73%) and arthralgia (28.57%). However, there were no significant differences in symptoms. Systemic AAV manifestations, such as involvement of the kidneys and nervous system, were significantly more common in the MPA-ILD group. ILD antedated MPA in 39.4% (13/33) of patients. A total of 11.11% (2/18) of ANCA-IIP patients who subsequently developed the clinical features of MPA were included in the MPA-ILD group. Cyclophosphamide was used significantly more often in the MPA-ILD group (48.48% versus 12.50%, p = 0.014).
Laboratory findings, PFTs and HRCT patterns
The laboratory tests, PFTs and HRCT patterns of ANCA-positive ILD are summarized in Table 2. Compared with those in the ANCA-IIP group, the white blood cell count (8.25 ± 2.36 versus 6.84 ± 1.75, p = 0.039), CRP level (43.89 ± 40.61 versus 18.74 ± 20.05, p = 0.028), ESR (71.97 ± 42.73 versus 40.69 ± 28.46, p = 0.011) and beta 2-microglobulin (β2-MG) level (5.15 ± 4.24 versus 4.29 ± 1.00, p = 0.018) were significantly higher in the MPA-ILD group. However, haemoglobin (113.09 ± 24.47 versus 132.19 ± 13.34, p = 0.006) and albumin (32.95 ± 5.84 versus 36.52 ± 3.94, p = 0.032) levels were significantly lower in the MPA-ILD group. Of the ANCA antibodies, perinuclear antineutrophil cytoplasmic antibody (P-ANCA) and MPO-ANCA had positivity rates of 81.63% and 65.31%, respectively. Only two patients were classic antineutrophil cytoplasmic antibody (C-ANCA) positive, and four patients were PR3-ANCA positive.
Comparison between the MPA-ILD group and ANCA-IIP group in terms of laboratory tests, PFTs and HRCT patterns.
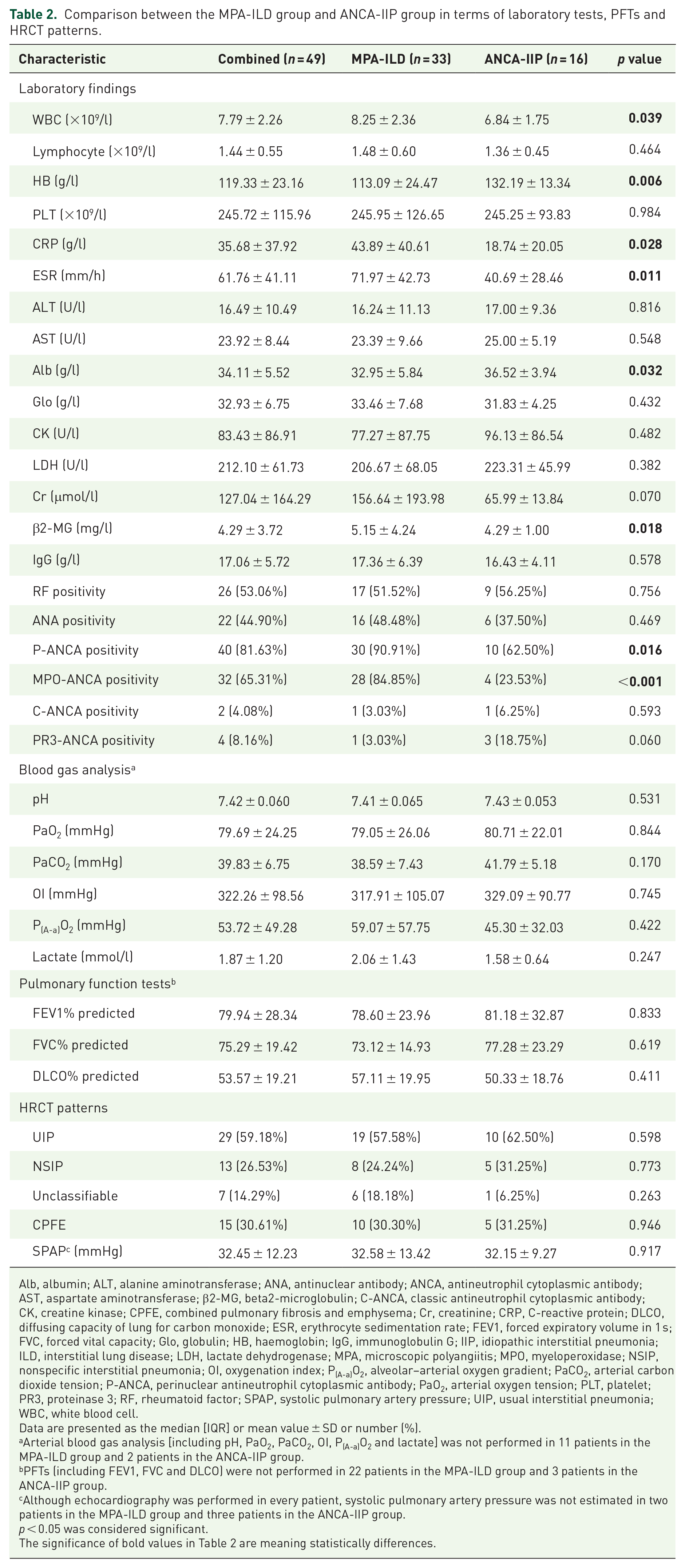
Alb, albumin; ALT, alanine aminotransferase; ANA, antinuclear antibody; ANCA, antineutrophil cytoplasmic antibody; AST, aspartate aminotransferase; β2-MG, beta2-microglobulin; C-ANCA, classic antineutrophil cytoplasmic antibody; CK, creatine kinase; CPFE, combined pulmonary fibrosis and emphysema; Cr, creatinine; CRP, C-reactive protein; DLCO, diffusing capacity of lung for carbon monoxide; ESR, erythrocyte sedimentation rate; FEV1, forced expiratory volume in 1 s; FVC, forced vital capacity; Glo, globulin; HB, haemoglobin; IgG, immunoglobulin G; IIP, idiopathic interstitial pneumonia; ILD, interstitial lung disease; LDH, lactate dehydrogenase; MPA, microscopic polyangiitis; MPO, myeloperoxidase; NSIP, nonspecific interstitial pneumonia; OI, oxygenation index; P(A-a)O2, alveolar–arterial oxygen gradient; PaCO2, arterial carbon dioxide tension; P-ANCA, perinuclear antineutrophil cytoplasmic antibody; PaO2, arterial oxygen tension; PLT, platelet; PR3, proteinase 3; RF, rheumatoid factor; SPAP, systolic pulmonary artery pressure; UIP, usual interstitial pneumonia; WBC, white blood cell.
Data are presented as the median [IQR] or mean value ± SD or number (%).
Arterial blood gas analysis [including pH, PaO2, PaCO2, OI, P(A-a)O2 and lactate] was not performed in 11 patients in the MPA-ILD group and 2 patients in the ANCA-IIP group.
PFTs (including FEV1, FVC and DLCO) were not performed in 22 patients in the MPA-ILD group and 3 patients in the ANCA-IIP group.
Although echocardiography was performed in every patient, systolic pulmonary artery pressure was not estimated in two patients in the MPA-ILD group and three patients in the ANCA-IIP group.
p < 0.05 was considered significant.
The significance of bold values in Table 2 are meaning statistically differences.
Twenty-four of the 49 patients underwent PFTs at initial presentation, and the most common manifestation was restrictive ventilation dysfunction with diffusion dysfunction. The HRCT pattern was mainly UIP or UIP-like (59.18%), followed by nonspecific interstitial pneumonia (NSIP) (26.53%). There was no significant difference in the baseline PFTs and HRCT patterns between the two groups, however.
Survival
Univariate and multivariate analyses of the risk of all-cause mortality in ANCA-positive ILD patients are summarized in Table 3. The median follow-up time of 49 patients with ANCA-positive ILDs was 32 (IQR 16.5–49.5) months, and 18 (36.73%) patients died from all causes during the follow-up. The 1-, 3- and 5-year overall survival rates were 87.55%, 69.92% and 58.74%, respectively, for ANCA-positive ILD cases. The 1-, 3- and 5-year overall survival rates were 84.85%, 54.53% and 48.47% for MPA-ILD patients compared with 100%, 100% and 80% for ANCA-IIP patients, respectively. In the univariate analysis, survival was significantly shorter for patients who smoked and had a diagnosis of MPA (Figure 2(a)), renal involvement, AE-ILD (Figure 2(b)), elevated serum LDH levels, PaCO2 > 45 mmHg, oxygenation index < 300 mmHg or SPAP ⩾ 35 mmHg at their ANCA-positive ILD diagnosis. The NSIP pattern (Figure 2(c)), however, was found to prolong survival among all ANCA-positive patients.
Univariate and multivariate analyses of the risk of all-cause mortality in ANCA-positive ILD patients.

ANCA, antineutrophil cytoplasmic antibody; CI, confidence interval; CRP, C-reactive protein; ESR, erythrocyte sedimentation rate; HR, hazard ratio; HRCT, high-resolution computed tomography; ILD, interstitial lung disease; LDH, lactate dehydrogenase; MPA, microscopic polyangiitis; MPO, myeloperoxidase; NS, nonsignificant; NSIP, nonspecific interstitial pneumonia; OI, oxygenation index; P(A-a)O2, alveolar–arterial oxygen gradient; PaCO2, arterial carbon dioxide tension; PR3, proteinase 3 antibody; RF, rheumatoid factor; SPAP, systolic pulmonary artery pressure; UIP, usual interstitial pneumonia.
Owing to the lack of some data, PaCO2, OI, P(A-a)O2 and SPAP were not included in the multivariate Cox regression model.
p < 0.05 was considered significant.
The significance of bold values in Table 3 are meaning statistically differences.

Kaplan–Meier survival curves of ANCA-positive ILD patients with different variables.
In the multivariable Cox analysis, a diagnosis of MPA [hazard ratio (HR) = 3.91, 95% confidence interval (CI) = 1.07–14.08, p = 0.038] and AE-ILD (HR = 9.43, 95% CI = 2.89–30.30, p < 0.001) remained significant factors that were independently associated with shorter survival, and the NSIP pattern (HR = 0.07, 95% CI = 0.01–0.41, p = 0.003) was independently associated with prolonged survival.
Discussion
As reported previously,6,7,12–15 ANCA-ILD patients were mostly MPO-ANCA positive (65.3%), and 67.3% had a diagnosis of MPA, with a UIP radiologic pattern (59.2%). A total of 12.9% of patients with ILDs and 26.6% of patients with IIPs were ANCA-positive. Compared with the ANCA-IIP group, patients in the MPA-ILD group had a higher CRP level and ESR. Haemoglobin and albumin levels were lower, however. The survival of the MPA-ILD patients was shorter than that of the ANCA-IIP group. In addition, our results highlight that a diagnosis of MPA and AE-ILD were independent risk factors associated with shorter survival and that those with the NSIP pattern had prolonged survival compared with those with the non-NSIP pattern on chest HRCT.
Whether there is a pathogenic relationship between ANCA and ILD or whether ILD is a disease manifestation of AAV versus an independent comorbidity remains unclear. MPO-ANCA probably contributes to the association between AAV and ILD, as the latter is exceedingly rare in PR3-ANCA-positive patients, and observational studies also did not find that these PR3-ANCA-positive ILD patients developed AAV.6,14,16,17 In recent years, some scholars considered that ILD could induce MPO-ANCA production and then promote the development of MPA, mainly based on the following findings. First, ILD usually occurs before AAV14,18,19 and 25–40% of ANCA-IIP patients develop clinical manifestations of vasculitis during follow-up.20–22 This study also showed that ILD antedated MPA in 39.4% of patients and 11.1% of ANCA-IIP patients subsequently developed the clinical features of MPA. Therefore, ANCA needs to be part of initial serologic testing in the work-up of a patient presenting with ILD at the baseline evaluation, and it is necessary for ANCA-IIP patients to closely monitor routine urine, renal function and other systemic involvement manifestations. Second, previous studies evaluating surgical lung biopsy findings in ANCA-ILD patients mainly showed a UIP histologic pattern, with accompanied areas of NSIP, organizing pneumonia, bronchiolitis, lymphoid hyperplasia and lymphoid follicles. Interestingly, none of the patients had findings of vasculitis.22–24 Furthermore, ILD could enhance the release of neutrophil extracellular traps (NETs) due to hypoxia,25,26 and NETs cause neutrophil cell death, named NETosis, resulting in the release of locally released enzymes (including MPO) and contributing to pulmonary tissue injury and fibrosis.27–29 Therefore, ILD itself could induce MPO-ANCA production as a result of neutrophil destruction during the chronic inflammation process, perhaps explaining the appearance of ANCA after the onset of ILD. 30 Thus, we do not consider ILD as a priori disease manifestation of MPA but rather a pre-existing comorbidity, and antifibrotic drugs can be considered a reasonable treatment option for fibrotic ILD patients with ANCA positivity.
Studies concerning ANCA-ILD, ANCA-IIP and AAV-ILD are limited. A previous study showed that patients in the MPA-ILD group were older and were more likely to have fever and a higher ESR and CRP level than patients in the ANCA-IIP group. 16 Furthermore, the multivariable analysis showed that the ESR was an independent risk factor for mortality in ANCA-ILD patients. 16 This study also revealed that patients in the MPA-ILD group had a more severe systemic inflammatory response, including an increased likelihood of fever and a higher WBC count, ESR and CRP level. Through the univariate survival analysis, however, this study did not find that an elevated ESR or CRP level was a risk factor for shortened survival, which may be related to the lack of systemic inflammation in some UIP-pattern patients. In addition, this study also showed that the most frequent radiologic pattern in ANCA-ILD is UIP, followed by nonspecific interstitial pneumonia (NSIP). This finding is consistent with prior studies.7,14,21,22,31 According to our univariate and multivariate survival analyses, the NSIP pattern was independently associated with prolonged survival, and the UIP pattern conferred a relatively high risk with shorter survival, but the difference did not reach statistical significance. This result may be related to the light degree of pulmonary fibrosis in some UIP-pattern patients.
Acute exacerbations (AEs) are the most extreme form of IPF and have a high mortality rate; the median survival of patients with IPF who experience an AE is approximately 3–4 months.8,32 Accumulating available evidence indicates that patients with ILD other than IPF also develop AE during their clinical course and have an extremely poor prognosis,33,34 and the highest AE rates are reported in patients with a histological or radiological pattern of UIP.33–37 This study showed that the 90-day mortality of ANCA-ILD patients who experienced an AE was 50%. Multivariate Cox regression analyses also indicated that AE was a significant factor independently associated with shorter survival. In addition, it has been proposed that lower baseline lung function parameters, impair oxygenation and extensive fibrosis on chest HRCT increase the risk of AE in fibrotic ILD patients.34,38,39 In the univariate analysis, our results showed that PaCO2 > 45 mmHg, OI < 300 mmHg or SPAP ⩾ 35 mmHg were significantly associated with increased mortality. These indicators often indicate extensive pulmonary fibrosis and poor pulmonary function parameters in ILD patients. Taken together, these findings indicate that more attention should be given to patients who have poor pulmonary function, respiratory failure or SPAP ⩾ 35 mmHg during their clinical course, especially if the patient develops AE.
As a retrospective cohort study, there were still some limitations. First, this was a single-centre study in China with a small sample size, which was representative of only a fraction of the Chinese population. Second, in the process of data collection and follow-up, data loss and follow-up bias were inevitable. Third, due to an inadequate understanding of ANCA-ILD in the past, 38.8% of patients received no treatment, which may lead to bias in the univariate and multivariate survival analysis results. Finally, PFTs and arterial blood gas analysis were performed for some patients, and related variables were not analysed by univariate and multivariate survival analysis. The results of the multivariable analysis of risk factors for prognosis should be carefully interpreted. Thus, larger-scale prospective studies or clinical randomized controlled trials are needed for further evaluation.
Conclusion
In conclusion, ANCA-ILD patients mostly have MPO-ANCA positivity, and with a diagnosis of MPA, the most frequent radiologic pattern is UIP, followed by NSIP. Compared with patients in the ANCA-IIP group, those in the MPA-ILD group had higher levels of inflammation markers. The survival of MPA-ILD patients was shorter than that of ANCA-IIP patients. Respiratory failure, SPAP ⩾ 35 mmHg and AE were found to be risk factors that were associated with shorter survival, and more attention should be given to patients with these characteristics. Early antifibrotic treatment may be considered a reasonable treatment option in fibrotic ILD patients with ANCA positivity.
