Abstract
Severe asthma (SA) poses a significant challenge to management and treatment, leading to a reduced quality of life and a heavy burden on society and healthcare resources. Bronchial thermoplasty (BT) has emerged as a non-pharmacological intervention for SA, demonstrating its efficacy and safety in improving patients’ quality of life and reducing exacerbation rates for over a decade. In particular, BT encounters various obstacles in its clinical application. Since asthma is characterized by high heterogeneity, not all patients derive effective outcomes from BT. Furthermore, current knowledge of markers that indicate response to BT remains limited. Recent research has shed light on the intricate mechanism of action of BT, which extends beyond simple smooth muscle ablation. Therefore, to enhance the clinical practice and implementation of BT, this paper aims to elucidate the mechanism of action and identify potential markers associated with BT response.
Plain language summary
A non-drug treatment for asthma: Severe asthma leads to a low quality of life and places a heavy burden on society and healthcare resources. A non-drug treatment for asthma called bronchial thermoplasty is a technique that uses high temperatures to remove excess airway muscle. Its efficacy and safety in improving asthma patients’ quality of life have been demonstrated for more than a decade. Barriers to the treatment development: When it comes to clinical application, bronchial thermoplasty faces several barriers. Asthma is characterized by high variability, and not all patients have good outcomes from bronchial thermoplasty. Understanding of the effectiveness of bronchial thermoplasty remains limited. New insights into how the treatment works: Recent research has found that the effect of bronchial thermoplasty is complicated and goes beyond simple muscle removal. What is the aim of this review: To improve the use of bronchial thermoplasty by discussing its effects on the body and identifying ways to measure how effective the treatment is.
Introduction
Asthma, a heterogeneous disease characterized by airway inflammation, bronchial hyperresponsiveness, and variable airflow obstruction, 1 afflicts over 300 million individuals globally and accounts for approximately 250,000 deaths annually. 2 In China, an estimated 30 million individuals suffer from asthma, with a prevalence rate of 1.24% among those aged 14 and above. 3 Among asthma patients, severe asthma (SA) represents a prominent cause of disability and death associated with asthma, which significantly burdens both society and healthcare systems. 4
First approved in the United States in 2010, bronchial thermoplasty (BT) offered a practical solution for SA patients above the age of 18 who were unable to adequately manage their symptoms with inhaled corticosteroids and long-acting β-agonists. BT was subsequently authorized for clinical use in China in 2014. The long-term efficacy and safety of BT have been demonstrated in real-world studies and clinical trials. Patients with SA exhibited sustained beneficial effects of BT for over a decade, resulting in reduced exacerbation rates and improved quality of life.5–16 Although head-to-head comparisons between BT and biological therapy have not been performed in randomized controlled trials, observational studies and meta-analyses have shown that BT provides clinical improvements comparable to biological therapy.17–19 In addition, when compared with biological therapy and conventional treatments, BT therapy has shown sustained clinical and economic benefits over time, despite its higher up-front costs.20–22
However, full acceptance of BT has been hampered by the lack of a comprehensive understanding of its mechanism and the challenges associated with identifying predictors of BT treatment response. Consequently, this paper aims to review the mechanism of action and explore potential response markers of BT as a means of establishing a solid theoretical foundation for its future clinical applications.
Mechanism of BT therapy
Increasing evidence has shown that the mechanism of action of BT does not lie solely in the ablation of airway smooth muscle (ASM). It is believed that BT targets various components, including ASM, reticular basement membrane (RBM), extracellular matrix (ECM), airway epithelial cells (AEC), neural innervation, and inflammation (Figure 1).
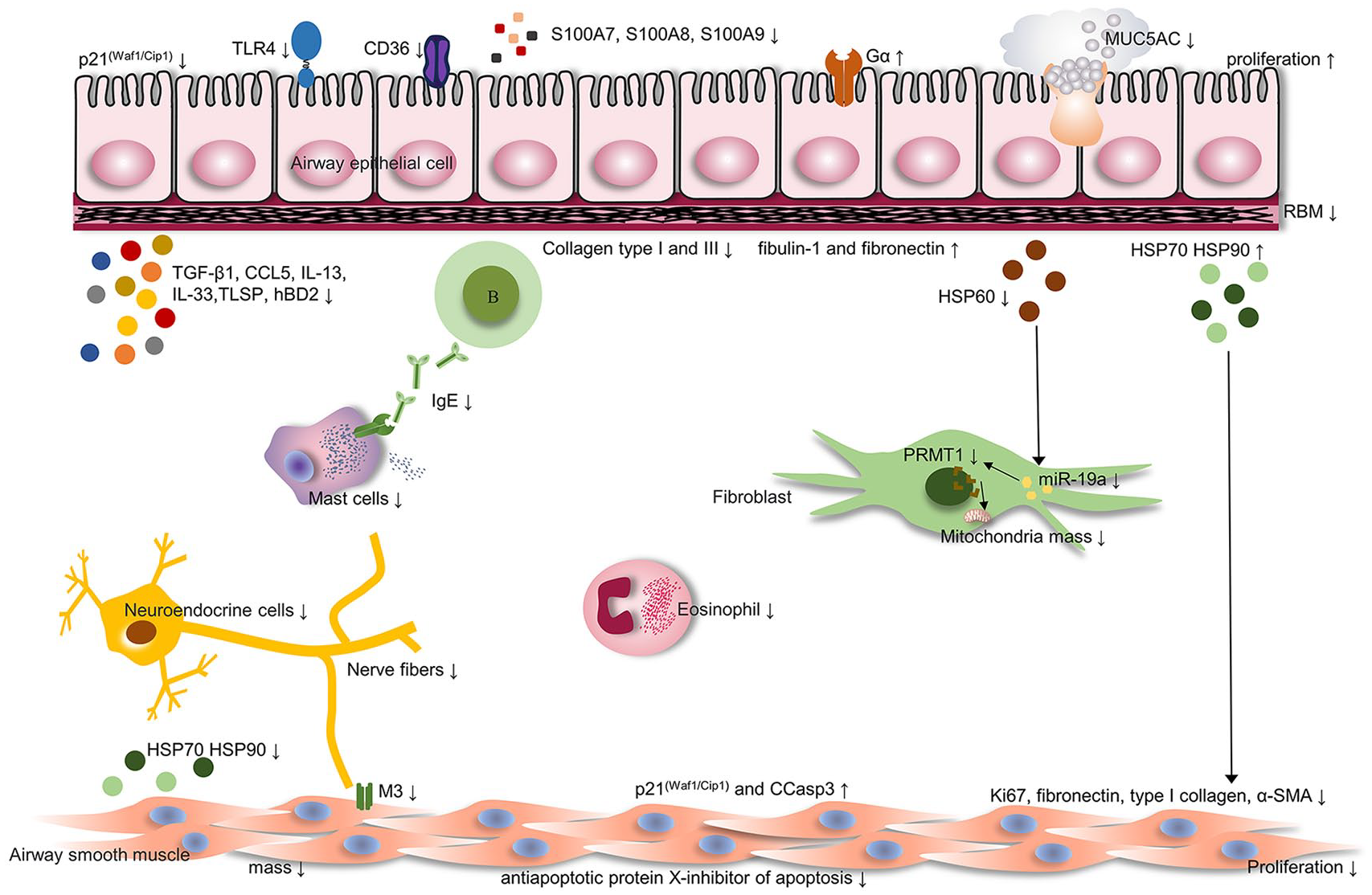
Main mechanisms of BT action in severe asthma. Potential targets for BT are thought to involve changes in ASM, AEC, neural innervation, and inflammation.
Impact of BT on changes in ASM
ASM governs bronchial motor tension and has pro-inflammatory and immunomodulatory functions.23,24 Extensive research has shown that BT has a profound influence on various aspects of ASM.
BT has demonstrated the ability to reduce ASM mass, regardless of whether the ASM is located within the treated region. There is increasing evidence of a reduction in ASM mass in patients with SA after BT.8,25–33 Gagnon et al. 26 found a sustained reduction in ASM mass in an observational study of 10 subjects ⩾12 years after BT. The limitation of this study is that ASM mass was not measured longitudinally in the same patients. Wijsman et al. 8 demonstrated that the reduction in ASM mass was still greater than 50% after 2.5 years, which is the longest follow-up for the effect of ASM mass after BT by paired longitudinal analysis. Surprisingly, the expected effect of BT in ASM goes beyond the treated area.26,27,31 Pretolani et al. 31 initially found that 70% of patients had a noteworthy reduction in ASM mass in the untreated middle lobe 3 months following BT. The therapeutic impact of BT extends beyond the treated area, potentially due to the diffusion of heat generated in the treated lobes during the BT procedure to untreated lobe. 31 Goorsenberg et al. 34 further demonstrated that the decrease in ASM mass in the untreated area was more prominent in segmental than subsegmental airways. Recently, Gagnon et al. 26 showed that the effects of BT on the untreated areas are as durable as those on the treated areas, confirming that ASM mass reduction could last up to 30 months. Interestingly, Donovan et al. 35 found a reduction in ASM mass can potentially influence changes in the flow patterns throughout the lung. This phenomenon can be attributed to structural modifications in the central airway ASM, which lead to the reopening of small airways and a reduction in the spatial heterogeneity of flow patterns.34 Konietzke et al. 36 demonstrated in an imaging study that dilates the most after BT are the small airways. Henry et al. 37 recently confirmed physiological evidence of small airway reopening after BT, supporting the computational findings of Donovan et al. Taken together, although BT is a treatment directed against large airways, the clinical improvements may be due to a reduction in small airway closure.
The association between ASM mass reduction and clinical improvement remains controversial. d’Hooghe et al. demonstrated that reduction in ASM mass was inversely correlated with baseline FEV1 6 months post-BT. 38 Pretolani et al. 28 revealed that ASM mass was correlated significantly with ACT scores and numbers of severe exacerbations 12 months after BT. Wijsman et al. 8 further found an association between the remaining ASM mass and exacerbation rate, as well as the post-bronchodilator FEV1(%predicted) 2.5 years after BT. 8 However, several studies failed to correlate reducing ASM mass and clinical improvement.32,39,40 Further studies with larger sample sizes and longer follow-ups are needed to clarify the relationship between clinical benefit and ASM mass reduction. Unfortunately, several studies reported that some patients continue to experience uncontrolled symptoms, with no decrease in ASM mass post-BT.31,41 Such variations might be attributed to individual differences and different ASM phenotypes. Patients with thicker ASM may require higher temperatures for successful ablation. Additionally, ASM displays phenotypic plasticity, exhibiting two distinctive phenotypes: synthetic and contractile.42–44 Future work should investigate whether different ASM phenotypes exhibit different responses to thermal stimulation.
BT can affect the proliferation and differentiation of ASM. Jendzjowsky et al. 30 found that simulating BT thermal stimulation in vitro had significant deleterious effects on ASM morphology and viability. Furthermore, Fang et al. 45 demonstrated that BT can inhibit ASM proliferation and remodeling through direct thermal stimulation or indirectly by upregulating the expression of HSP70 and HSP90 in AEC. In in vitro experiments, the proliferation of ASM exhibited clear signs of degeneration after 10 s of heat exposure at 65°C. This heat exposure resulted in increased expression of the cell cycle inhibitor p21(Waf1/Cip1) and pro-apoptotic cleavage caspase-3 (CCasp3), alongside decreased expression of the anti-apoptotic protein X-inhibitor of apoptosis protein in ASM. 45 In addition, in in vitro studies, HSP70 and HSP90 derived from AEC significantly reduced the expression of fibronectin, type I collagen, and α-SMA, three proteins associated with remodeling and cell differentiation in ASM. HSP70 and HSP90 also decreased the expression of the proliferation marker Ki67 and significantly upregulated the expression of the cell cycle inhibitor p21(Waf1/Cip1) in ASM. 45 The long-term effects of BT on ASM proliferation and differentiation should be investigated in the future.
BT can have a significant impact on ASM contractility. Dyrda et al. 46 concluded that the extreme temperatures used in BT directly disrupt actin-myosin interactions in bovine ASM, leading to an immediate loss of ASM function. However, it is unclear whether this effect can be observed in human ASM. In addition, it is not known whether the reduction in airway contractility after BT is permanent.
In conclusion, BT has the potential to decrease ASM mass and influence various aspects of ASM function. The relationship between ASM mass reduction and clinical improvement remains controversial. In addition, further investigations are needed to explore the impact of BT on the pro-inflammatory and immunomodulatory functions of ASM.
Effects of BT on changes in RBM thickness and ECM composition
The thickened RBM is a histopathological feature of asthma that correlates with FEV1 and disease severity.47,48 Most studies reported a reduction in RBM thickness after BT,25,28,49,50 while three studies reported no change in RBM thickness following BT.27,30,32 Differences in results may be related to differences in the methodology used and the number of patients included.
Accumulation of ECM in the RBM, lamina propria, and submucosa is a hallmark of asthmatic airways. Several studies reported that BT can alter ECM composition. Chakir et al.38,39 showed that the type I collagen deposition underneath the RBM decreased 3 weeks post-BT. Jendzjowsky et al. 30 found a significant reduction in collagen types I and III in the RBM and submucosa at 6 weeks post-BT, which then returned to baseline levels by 12 months post-BT. However, Facciolongo et al. 51 reported an increase in collagen type I gene expression (COL1A1, COL1A2) in bronchial biopsies after a third BT session by analyzing the total content of collagen. Similarly, Wijsman et al. 50 found an increase in total collagen, with a change in collagen fiber structure by examining the full airway biopsy area. Differences between gene and protein expression of collagen may be related to the location of the selected airways and the length of follow-up. In addition, the changes in other ECM components after BT have also been described. Wijsman et al. 50 found an increase in fibulin-1 and fibronectin with no change in elastin after BT. Interestingly, changes in total collagen and fibulin-1 area are associated with a reduction in FEV1 reversibility. It is believed that BT increases airway wall stability by altering ECM composition. 50
In conclusion, in addition to the reduction of ASM, BT can alter ECM composition and RBM thickness to influence airway remodeling. Future research should investigate the impact of BT on other components of the ECM. In addition, it would be of great interest to explore whether the changes in the ECM after BT have an impact on the inflammatory state of the airways.
Impact of BT on changes in AEC
AEC is the initial structure exposed to thermal stimulation during the BT process. There are indications that AEC may be a target of BT.
BT has been found to induce AEC proliferation and inhibit apoptosis. Endzjowsky et al. 30 found that simulating BT thermal stimulation in vitro had no effects on AEC morphology and viability. Chernyavsky et al. 33 showed significantly improved epithelial integrity after BT in vivo biopsies. Fang et al. 45 revealed that the expression of Ki67 was increased in AEC 1 month following BT. In addition, they demonstrated that heat exposure at 65°C for 10 s significantly stimulated AEC proliferation without affecting morphology in vitro. Furthermore, the expression of the cell cycle inhibitor p21(Waf1/Cip1) decreased, while the expression of the anti-apoptotic protein X-apoptotic protein and CCasp3 remained unchanged in vitro. However, despite the proliferative effects of BT on AEC, no significant impact on the ratio of regenerative epithelium or different types of epithelial cells was observed.28,52,53 Further investigation is warranted as epithelial repair may have an impact on other changes in airway wall structure following BT.
Protein expression in AEC can be altered by BT. Several studies have shown that the expression of heat shock proteins (HSPs) in AEC was significantly altered following BT. Sun et al. 54 found that BT can decrease airway remodeling by reducing the secretion of HSP60 in AEC and suppressing the expression of miR-19a in fibroblasts, which in turn reduces fibroblast proliferation and mitochondrial stimulation. Fang et al. 45 revealed that increased secretion of HSP70 and HSP90 promotes AEC regeneration. Moreover, Papakonstantinou et al. 27 proved the elevated expression of HSP70 and HSP90 in AEC can enhance hormone sensitivity through continuous increases in the expression of glucocorticoid receptor (Gα) and the promotion of Gα nuclear transfer. However, the specific targets and pathways through which BT improves hormonal sensitivity are still unknown. Further investigations are required to elucidate the connection between HSPs and ASM ablation as well as matrix deposition pathways.
AEC metabolism can be normalized by BT (Table 1). Ravi et al. 55 observed that BT significantly increased the expression of OXPHOS genes while reducing the expression of genes involved in fatty acid metabolism in AEC after 6 months. Gagnon et al. 56 revealed that a large portion of differentially expressed genes (DEG) was involved in antiviral response, anti-microbial response, and pathogen-induced cytokine storm signaling pathway. The study showed that BT downregulated S100A7, S100A8, and S100A9 gene expressions, which was further validated at gene and protein levels in AEC and bronchial biopsies. 56 Further transcriptomic analysis of AEC by Liao et al. 57 revealed alterations in neurophysiological processes, Cystic Fibrosis Transmembrane Conductance Regulator activity, and Rho GTPase during BT in the untreated area. Notably, the selected AEC samples for this study did not undergo heat treatment with BT, indicating that the impact of BT on AEC may extend throughout the entire lung. Future investigations should focus on exploring the long-term effects of BT on AEC metabolism. The specific metabolomic and lipidomic differences between BT responders and non-responders should be investigated in further studies to identify potential markers of BT.
Summary of BT-related omics analysis.

ACTA2, actin alpha 2; AEC, airway epithelial cell; BALF, bronchoalveolar lavage fluid; BT, bronchial thermoplasty; CFTR, cystic fibrosis transmembrane conductance regulator; COL1A1, collagen type I alpha 1.; COL1A2, collagen type I alpha 2; DEGs, differentially expressed genes; DEPs, differentially expressed proteins; FAP, fibroblast activation protein; HSP, heat shock protein; OXPHOS, oxidative phosphorylation.
In summary, BT can promote AEC proliferation and influence protein expression and metabolism in AEC. However, further research is needed to explore additional mechanisms by which BT affects AEC and to determine the sustainability of these effects.
Impact of BT on changes in neuroregulation
The density of sensory nerves is notably elevated in asthmatic patients, correlating with impaired lung function and heightened sensitivity to environmental triggers.59,60 Several studies have shown that BT can influence neuromodulation and impact the abundance of airway neuroendocrine cells (NEC) and nerve fibers.
A substantial decrease in the number of NEC and nerve fibers was observed following BT. Pretolani et al. 28 revealed a significant reduction in autonomic nerve fibers both in the bronchial submucosa and within the ASM bundles 3 months post-BT, corresponding to a reduction in the number of severe exacerbations. Furthermore, the study also found that BT reduced the number of NEC by approximately 95%, which was highly correlated with the improvement in SA control, number of exacerbations, hospitalizations for asthma and ICU stays, and emergency department visits. 28 Jendzjowsky et al. 30 found that neural bundles were significantly reduced at 6 weeks and 12 months post-BT. Notably, this study showed that the reduction in the number of NEC was not limited to the treated middle lobe but exhibited a lung-wide effect, highlighting the widespread impact of BT on neuromodulation. Facciolongo et al. 61 revealed that the reduction of nerve fibers in AEC and ASM can persist for up to 1 year following BT treatment. They also observed that BT reduced PGP9.5 mRNA expression at the third BT session compared to baseline. 51 Luo et al. 62 further showed that BT-induced bronchial denervation in both treated large airways and untreated small airways by reducing the airway nerve distribution (marked by PGP9.5) and muscarinic receptor 3 (M3 receptor). However, as the results of this study were based on a healthy beagle model, further studies should be carried out to establish an asthmatic canine or porcine model receiving BT treatment to explore the effects of BT on neuromodulation. Moreover, transcriptomic analyses of AEC have demonstrated alterations in neurophysiological processes, involving genes such as syntaxin-binding protein 1 (STXBP1), lending further support to the idea that BT has the capacity to modify neurophysiological functions. 57
In further studies, the effect of BT on neuromodulation should be assessed in larger cohorts with longer follow-ups. In addition, it is worth investigating whether differences in neuromodulation account for differences in response to BT.
Impact of BT on changes in inflammation
Asthma is characterized by the infiltration and increased presence of inflammatory cells. 63 Studies indicated that inflammation regulation may also be a valuable hallmark of the beneficial clinical effects of BT.
BT is capable of modulating T2-type allergic responses in the airways and affecting immune cells. T2 markers such as mucosal and blood eosinophils, Immunoglobulin E (IgE) levels, IL-13 and IL-33 positive cell counts, and epithelial IL-13 receptor α2 expression were reduced in responders 12 months after BT. 49 Particularly, there was also a reduction in mast cells (MCs) in the mucosa and ASM. 49 BT may inhibit the production of MC chemokines by various airway cell types, thus reducing the overall inflammatory response. The impact of BT on other immune cells is not well understood. Several studies found no significant change in inflammatory cell numbers (including eosinophils, neutrophils, MCs, and lymphocytes) in bronchial biopsies after BT.27,28,32 However, Facciolongo et al.51,61 found an increase in CD68 expression post-BT, indicating a potential increase in monocytes, macrophages, and dendritic cells. Further investigation is required to understand the effect of BT on airway immune cell function, proliferation, and differentiation.
Cytokine levels associated with airway inflammation were observed to change after BT. Denner et al. 53 revealed that the levels of TGF-β1 and (RANTES)/CCL5 in the BALF fluid decreased significantly, while the levels of tumor necrosis factor-associated apoptosis-inducing ligand (TRAIL) increased substantially 6 weeks after BT. Recently, Gagnon et al. 56 investigated the effect of BT on the expression of alarmins in AEC and found that TSLP and human β-defensin 2 (hBD2) were significantly decreased while no significant change was observed in IL-25 and IL-33 post-BT. However, Wijsman et al. 64 found that no differences were observed in cytokines, neither from the untreated compared to treated airways, nor before and after treatment. The impact of BT on MUC5AC and MUC5B secretion remains controversial. Ladjemi et al. 49 found a noteworthy increase in the epithelial expression of MUC5AC post-BT, which was negatively correlated with disease exacerbation, whereas MUC5B expression did not increase. 49 Consistent with the research of Ladjemi et al., Sun et al. detected increased levels of alveolar MUC5AC after BT using proteomic analysis. 54 Nonetheless, Salem et al. 65 discovered reduced MUC5AC expression in SA patients following BT, which did not correlate with clinical outcomes. The variation in cytokine results between different experiments may be attributed to differences in study design, timing of sample collection, and sample location.
The effects of BT on systemic inflammation have rarely been studied. Chupp et al. 66 found significant downregulation of pathways associated with eosinophilic inflammation, T cell activation, and neuronal function by examining gene expression in the blood following BT. Furthermore, a reduction in peripheral blood eosinophil count and total IgE levels was observed 1 year after BT, although no notable change in blood neutrophil count was found.49,67 Subsequent investigations should include transcriptomic analysis of peripheral blood to explore the effects of BT on systemic inflammation. In addition, future studies should establish a correlation between systemic inflammatory changes and clinical symptoms and search for peripheral blood BT response markers.
Markers of BT response
BT is beneficial for patients with different asthma phenotypes. 16 However, identifying specific biological, genetic, or clinical characteristics that may indicate a better clinical response is crucial for patient selection and reducing the medical burden (Table 2).
Summary of BT-related markers.

ACT, asthma control test; ASM, airway smooth muscle; BT, bronchial thermoplasty; COL1A2, collagen type I alpha 2; CTGF, connective tissue growth factor; FEV1, forced expiratory volume in 1 s; GC, glucocorticoid; OCLN, occluding; SLPI, secretory leukocyte peptidase inhibitor.
Some baseline clinical characteristics of BT patients may be predictors of BT response. No association between the baseline ASM mass and the response to BT has been found in previous studies.34,72 Therefore, selecting patients solely based on baseline ASM mass assessment may not be the answer. Sarikonda et al. 68 suggested that a shorter duration of asthma, a history of severe exacerbations, impaired asthma-specific quality of life, and a higher baseline dose of corticosteroids could potentially serve as predictors of BT response. Kashizaki et al. 73 showed that the pre-BT ACT score could predict the response to BT in younger adult patients with SA and preserved lung function. Wang et al. 74 found that a higher Asthma Control Questionnaire (ACQ) score and better lung function before BT were significantly associated with a better response to BT, indicating that lung function should be improved as much as possible before BT. Additionally, a study by Postigo et al. 71 reported that the most significant improvement was observed in patients with the most severe symptom at baseline, having the highest ACQ score and the highest frequency of exacerbations. It is recommended that an ACQ cut-off score of 2.7 has the best sensitivity and specificity for the prediction of responder/non-responder status. d’Hooghe et al. 38 found that patients with FEV1 < 80% tended to have a larger ASM mass at baseline and experienced the greatest decrease in ASM mass following BT. Nevertheless, confirming whether SA patients with low FEV1 are the most responsive to BT requires verification in a larger sample size. Recently, research by Wang et al. 74 revealed that the number of activations in BT, especially in the third BT session, was highly relative to and predictive of the clinical response to BT, while main bronchial ablation did not markedly affect clinical response to BT. It is advised to boost activations in the third BT session without main bronchial ablation.
Baseline differences in airway inflammation and gene levels were observed between BT responders and non-responders. Ladjemi et al. 49 revealed that BT responders were younger, had a higher incidence of atopy, elevated blood eosinophil counts and IgE levels, increased epithelial IFN-α expression, and higher numbers of mucosal eosinophils and IL-33-positive cells. Ano et al. 58 revealed 67 DEGs among responders and non-responders. These genes are associated with inflammation, matrix regulation, surfactant metabolism, and airway mucus secretion and are expected to be potential baseline markers for predicting BT response, particularly Secretory leukoprotease inhibitor (SLPI) gene expression. 58 Facciolongo et al. 51 identified Occludin (OCLN), CD68, Connective tissue growth factor (CTGF), and SLPI transcript levels as potential indicators for a higher risk of disease exacerbation after BT. However, further research and clinical validation are necessary to determine whether the identified transcriptome changes can effectively serve as markers for BT response.
Several studies investigated the predictive value of markers of airway inflammation, such as periostin, sputum differential cell count, and exhaled nitric oxide, for a favorable response to BT. Svenningsen et al. 69 demonstrated that following normalization of sputum cell count, BT can significantly improve patient prognosis and reduce airway hyperresponsiveness. Iikura et al. 75 found that BT responders had lower serum periostin and exhaled nitric oxide levels at baseline. Additionally, Yamamura et al. 70 found that increased sensitivity of cough receptors may also serve as a potential predictor of BT response. Nishiyama et al. 76 revealed that BT may be effective for cough in severe uncontrolled asthma by improving capsaicin cough sensitivity. However, a recently published study by Yamamura et al. 70 overturned the results of their previous study, which was limited by a short follow-up period. They believed that the capsaicin cough reflex sensitivity (CRS) at baseline could not predict the BT efficacy after 2 years and the patients who responded to BT exhibited desensitization of CRS to capsaicin. 77 Further validation through larger, long-term, and multi-center studies is necessary to settle these disputes.
With the increasing utilization of imaging techniques in BT, studies are underway to explore optical coherence tomography (OCT), computed tomography (CT), and Endobronchial Ultrasound (EBUS) as potential non-invasive methods for identifying predictors of response to BT.78–81 Donovan et al. 82 demonstrated that the measurement of total lung volume by CT as a means of assessing pretreatment airway dimensions can feasibly predict response to BT and inform subject-specific computational models. Soja et al. 32 found that EBUS can assess bronchial structural changes related to BT; however, it did not predict a favorable clinical response to therapy. These methods are still validated in relatively small study groups. More studies should use these imaging techniques to assess the effects of BT treatment in larger study groups and to explore whether the response to BT can be predicted.
A current ongoing biometric study (clinicaltrials.gov NCT01185275) aims to determine whether baseline clinical, physiological, biological, and imaging markers can effectively identify patients most likely to benefit from BT. The examination of BT markers has long-term clinical value and will enhance the targeted clinical application of BT and improve the efficiency of diagnosis and treatment.
Conclusion
Since its initial clinical approval in 2010, BT therapy has advanced into its second decade. Over the past decade, research has demonstrated the long-term efficacy and safety of BT treatment for SA, showcasing its ability to reduce the rate of SA exacerbations and improve symptoms. Moreover, the last decade has witnessed a renewed understanding of the mechanisms underlying BT. It extends beyond the ablation of ASM and involves neuromodulation, affecting the function of AEC, and impacting airway and systemic inflammation. Despite the progress made, the clinical implementation and widespread adoption of BT still encounter various challenges. To facilitate the promotion and maturation of the clinical application of BT, it is necessary to further explore its mechanisms and to search for reliable markers of BT. With these efforts, it is anticipated that BT will continue to deliver more significant clinical benefits to patients with SA in its second decade.
