Abstract
Published information on the effectiveness of bronchial thermoplasty (BT) for severe asthma in ‘real life’ patients is limited. We compared safety and efficacy outcomes 12 months post procedure in 10 clinic patients and 15 patients recruited to clinical trials of BT at the same centre. Baseline asthma severity was greater in the clinic group. Adverse events were similar. Clinical improvements occurred in 50% of the clinic patients compared with 73% of the research patients.
Introduction
Bronchial thermoplasty (BT) involves delivery of radiofrequency energy to the airways during flexible bronchoscopy, and possibly exerts its effect by reduction of airway smooth muscle mass [Thomson et al. 2012]. Clinical trials of BT have shown benefits in the treatment of patients with moderate to severe asthma [Cox et al. 2007; Pavord et al. 2007, 2013; Castro et al. 2010; Torrego et al. 2014; Thomson et al. 2011; Wechsler et al. 2013]. Little is known about the safety and efficacy of BT when performed in clinical practice [Doeing et al. 2013]. We describe our experience of introducing BT into a clinical service for severe asthma and compare demographics and clinical outcomes with patients who participated in three randomized controlled trials of BT at the same centre [Cox et al. 2007; Pavord et al. 2007; Castro et al. 2010].
Methods
Patients from the Difficult Asthma service in Glasgow were selected for the procedure using criteria broadly similar, but not identical to, those employed in clinical trials of BT [Cox et al. 2007; Pavord et al. 2007; Castro et al. 2010]. The main difference was that patients on all asthma medications, including omalizumab and high-dose oral prednisolone, and those with high frequency of exacerbations were not excluded. All patients had severe asthma and were on Step 4 or 5 of British Thoracic Society (BTS) guidelines, with inhaled corticosteroids dose at least 1000 μg beclometasone equivalent daily and additional preventer medications, and had no contraindication for bronchoscopy. Approval was obtained from the Institutional Review Board for the audit of clinical patients and local data from research patients were extracted only after the clinical trials had been published. The procedures were performed as previously described [Bicknell et al. 2014]. Tests performed at baseline and at 12 months after BT were spirometry, asthma control questionnaire (ACQ) [Wechsler et al. 2013] and Asthma Quality of Life Questionnaire (AQLQ) [Pavord et al. 2013]. The number of oral steroid courses, hospital admissions and adverse events were recorded during the periprocedure period (defined as the time from the first BT to 6 weeks after the last procedure) [Cox et al. 2007; Pavord et al. 2007; Castro et al. 2010] and at 6- and 12-month clinic visits during the post-treatment period (defined as the time from 6 weeks after the last procedure until 12 months).
Efficacy and safety outcome data were abstracted from 15 patients recruited to clinical trials of BT at the same centre [Asthma Intervention Research Trial (AIR), n = 5; Research in Severe Asthma Trial (RISA), n = 3; AIR2 Trial, n = 7] [Cox et al. 2007; Pavord et al. 2007; Castro et al. 2010] for the above endpoints where data were available. Clinical improvement was defined as achieving one or more of the following outcomes during the post-treatment period:
Reduction by at least one severe exacerbation (requirement for high-dose oral corticosteroids) or hospital admissions for asthma.
Improvement in ACQ or AQLQ score by the minimum clinical important difference (MCID), without a worsening of the other (ACQ score decrease by ⩾0.5; AQLQ score increase by ⩾0.5 is the MCID).
Stepdown in treatment: half the maintenance oral prednisolone dose or stop omalizumab without loss of asthma control (no increase in hospitalization/asthma exacerbations by one or worsening of ACQ/AQLQ scores by the MCID).
Statistical analysis
Comparison of demographic and outcome variables between and within clinical and research patients was by Student’s t test, Mann–Whitney U test, Wilcoxon’s signed rank test or χ2 test, depending on data distribution.
Results
Ten clinic patients underwent bronchial thermoplasty (Table 1). Four of 10 patients were taking oral prednisolone daily and two of 10 were receiving omalizumab treatment (for over 3 years). Baseline forced expiratory volume in 1 s (FEV1) % predicted pre salbutamol ranged from 45% to 96%. Asthma control was poor (mean ACQ score 3.3). Exacerbations in the previous year ranged from zero to eight (mean three) and hospital admissions ranged from zero to five (mean one). The demography of clinic patients bore the closest resemblance to those in the RISA study where Step 5 asthmatics were included [Pavord et al. 2007], but we extended this to include patients currently on omalizumab treatment.
Baseline demography and efficacy outcomes following bronchial thermoplasty in clinic patients and research patients from the same centre in Glasgow.
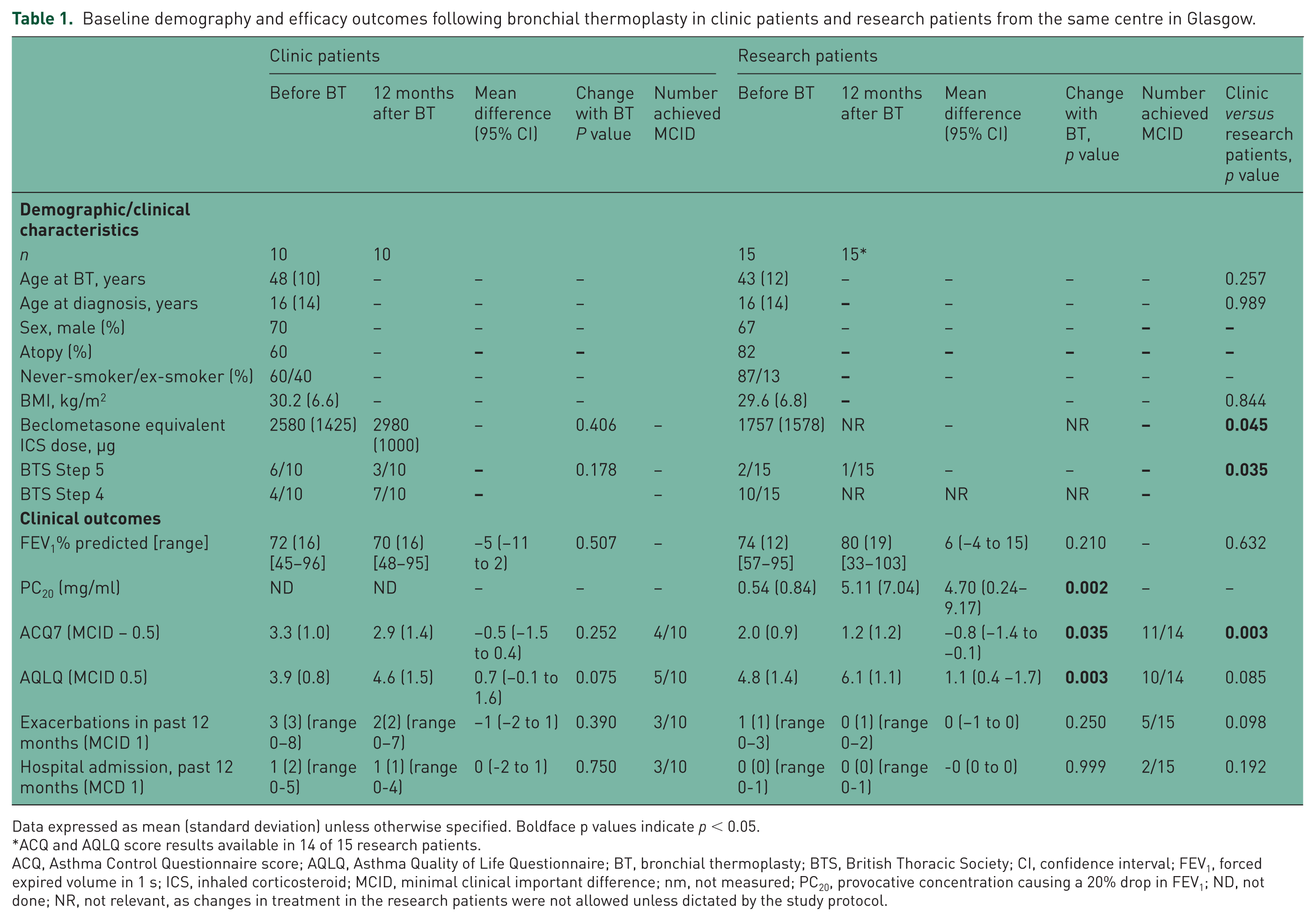
Data expressed as mean (standard deviation) unless otherwise specified. Boldface p values indicate p < 0.05.
ACQ and AQLQ score results available in 14 of 15 research patients.
ACQ, Asthma Control Questionnaire score; AQLQ, Asthma Quality of Life Questionnaire; BT, bronchial thermoplasty; BTS, British Thoracic Society; CI, confidence interval; FEV1, forced expired volume in 1 s; ICS, inhaled corticosteroid; MCID, minimal clinical important difference; nm, not measured; PC20, provocative concentration causing a 20% drop in FEV1; ND, not done; NR, not relevant, as changes in treatment in the research patients were not allowed unless dictated by the study protocol.
All procedures were performed as day cases. In the research trials and in the clinical patients, each subject had three treatment sessions, minimum 3 weeks apart. One of the clinical patients had only two treatment sessions as the clinical decision was made to avoid treating the left lower lobe due to stenosis of the lower lobe bronchus. This patient had a clinical improvement despite missing one treatment session. Treatment sessions were uneventful and adverse events similar to those in clinical trials [Cox et al. 2007; Pavord et al. 2007; Castro et al. 2010]. Most patients reported mild worsening of asthma symptoms for a few days after each procedure. During the periprocedure period, hospital admissions occurred in three patients (two for asthma; one for a partial lung atelectasis that responded to routine medical treatment).
Five of the 10 clinic patients met the criteria for clinical improvement at 12 months as defined above (Tables 1 and 2). Asthma medications were reduced in three patients without deterioration in asthma control: two discontinued omalizumab and one discontinued prednisolone. Halving the dose of oral corticosteroids was used as one of the indicators of improvement with BT as this is the same level achieved with a newer biological agent for asthma [Bel et al. 2014]. ACQ score improved by at least 0.5 in 4 of 10 patients and AQLQ score by at least 0.5 in 5 of 10 patients (Table 1). In the postprocedure period, there was a mean of two severe exacerbations (standard deviation two) and one hospital admission (standard deviation one). The number of severe exacerbations and hospitalizations was reduced in three of 10 patients.
Change in asthma control score, asthma quality of life score, severe exacerbations and hospital admissions plus stepdown in treatment in clinic patients 1 year after bronchial thermoplasty treatment.

↑ increase by MCID; ↓ decrease by MCID; ↔ no change.
Patients who benefited from bronchial thermoplasty.
ACQ, Asthma Control Questionnaire (reduction by 0.5 is a clinical improvement); AQLQ, Asthma Quality of Life Questionnaire (increase by 0.5 is a clinical improvement); BT, bronchial thermoplasty; MCID, minimal clinically important difference.
Asthma severity was greater in clinic patients compared with research patients with six of 10 (60%) clinic patients at BTS Step 5 treatment, compared with two of 15 (13%) research patients (Table 1). Baseline asthma control and number of exacerbations were worse in the clinic group (Table 1). In the research patients, there were five periprocedure hospital admissions (in three patients), four for asthma and one for lobar atelectasis. Post procedure, a clinical improvement in ACQ occurred in 11 of 14 patients and in AQLQ score in 10 of 14 patients. Post procedure, there were four severe exacerbations (in three patients); one of these led to a hospital admission.
Discussion
Data on safety and efficacy obtained from carefully controlled trials [Cox et al. 2007; Pavord et al. 2007; Castro et al. 2010] may not be replicated in ‘real life’ patients in clinical practice. We audited the introduction of bronchial thermoplasty in 10 patients with moderate to severe asthma attending a severe asthma service. The main findings were that half of these patients obtained clinical benefit, either by a stepdown in treatment, or an improvement in current asthma control, or a reduction in exacerbations at 12 months post procedure. Adverse events were similar to those reported in clinical trials. Current standard of care treatment options had been tried in the clinic patients with severe asthma and an improvement in 50% of the patients with BT is a clinically worthwhile benefit.
Clinical improvement with BT occurred in 73% of the research patients, higher than that in the clinic group. A possible explanation for this is likely to be due to greater severity of asthma in the clinic patients. At least seven of the 10 clinic patients would have failed prescreening for the AIR2 trial [Castro et al. 2010], mainly due to the high levels of asthma therapy, frequency of exacerbations and high use of inhaled salbutamol.
A Cochrane review pooled analysis of three randomized controlled trials on BT demonstrated a modest benefit in quality of life (mean difference in AQLQ 0.28, 95% confidence interval 0.07–0.50), and a significant reduction in exacerbation rates 12 months after treatment. There was no significant change in asthma control scores. The risk of adverse events increased during the treatment period with no further increase in the post-treatment period. In our clinic patients, there was a trend for AQLQ to improve by a mean of 0.7 after treatment, with half of the patients reaching the clinically relevant threshold of 0.5. There was no significant improvement in ACQ [Torrego et al. 2014].
The patients most suitable for the procedure are those with moderate to severe asthma, symptomatic despite maximal asthma therapy, with an FEV1 above 60% predicted and no contraindication to bronchoscopy. Details are described elsewhere [Bicknell et al. 2014]. There are currently no data on factors that predict a good response to BT. This treatment is licensed in the UK and training is required before individuals can perform the procedure. Costs include the one-off purchase of a controller (approximately £25,000), catheters (approximately £6000 per patient) and additional staff costs.
There are several limitations to the findings. The sample size is small which limits data analysis. The data are observational and treatment was unblinded. As it was performed as part of a routine clinical service and not part of a strict research protocol, data collected are ‘real life’ with their own imperfections. Therefore, these data can only serve to provide information about the feasibility of setting up a BT service in a clinical setting in the UK.
In conclusion, BT can be safely delivered in a clinical setting to patients with severe asthma with limited treatment options, although clinical benefits are variable.
Footnotes
Acknowledgements
We are grateful to the patients who participated both in the research studies and in the clinical audit and to Dr David Raeside, Dr David Stewart, Catherine McFarlane and Mary Mackenzie (NHS Greater Glasgow and Clyde) for their support.
SB and RC contributed equally (SB performed all the bronchial thermoplasty procedures and RC managed the asthma, and both contributed equally to concept, design and writing of the paper). Conception and design: SB, RC, NL, NCT; analysis and interpretation: SB, RC, NL, NCT, MS, MS,NP,EC,DC, JN,JT,CM; drafting the manuscript for important intellectual content: SB, RC, NL, NCT, MS, MS, NP, EC, DC, CM.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: NCT received funds for his institute to participate in research studies on bronchial thermoplasty and lecture fees from Boston Scientific. The other authors report no potential conflict of interest related to this manuscript.
