Abstract
Background:
Bronchial thermoplasty (BT) is a recently developed non-pharmacological therapy for refractory bronchial asthma. Although increasing evidence has suggested that BT is effective for various phenotypes of severe asthma, its safety and efficacy in patients with severe irreversible impaired lung function are unclear.
Objectives:
To assess the efficacy and safety of BT in patients with refractory asthma, including patients with a severely impaired forced expiratory volume in 1 second (FEV1).
Design:
This was a single-center, retrospective, observational cohort study.
Methods:
We retrospectively reviewed the medical records of 15 patients with refractory asthma (Global Initiative for Asthma step 4 or 5), including patients with severely impaired airflow limitation (% predicted pre-bronchodilator FEV1 <60%), who had undergone BT between June 2016 and January 2022. We analyzed the efficacy (change in asthma symptoms, exacerbation rate, pulmonary function, asthma medication, and serum inflammatory chemokine/cytokines before and after BT) and complications in all patients. We compared these data between patients with severe obstructive lung dysfunction [group 1(G1)] and patients with FEV1 ⩾ 60% [group 2 (G2)].
Results:
Six patients were in G1 and nine were in G2. Clinical characteristics, T2 inflammation, and concurrent treatment were equivalent in both groups. BT significantly improved asthma-related symptoms (measured using the Asthma Control Test and Asthma Quality of Life Questionnaire scores) in both groups. FEV1 was significantly improved in G1 but not in G2. Four patients in G2, but none in G1, experienced asthma exacerbation requiring additional systemic corticosteroids (including two requiring prolonged hospitalization) after BT. Long-term responders (patients who reduced systemic or inhaled corticosteroid without newly adding biologics in a follow-up > 2 years) of BT were identified in G1 and G2 (n = 2, 33.3% and n = 4, 44.4%, respectively).
Conclusion:
BT in patients with refractory asthma and severe airflow limitation is equally safe and efficacious as that in patients with moderate airflow limitation.
Introduction
More than 200 million individuals worldwide have bronchial asthma. Approximately 5%–10% of those patients have severe and refractory asthma, which is defined as still uncontrollable even after receiving appropriate asthma controller therapies. 1 Severe asthma is an emerging serious medical concern requiring newly developed therapeutic strategies. Bronchial thermoplasty (BT) is a relatively newly developed non-pharmacological therapy for severe asthma. International guidelines, such as the Global Strategy for Asthma Management and Prevention (GINA) guideline, do not recommend BT as a first-line treatment option for severe asthma. 2 However, there is growing evidence regarding the efficacy and safety of BT for severe asthma.3,4
Several prospective studies, such as the Asthma Intervention Research (AIR), 5 Research in Severe Asthma (RISA), 6 and Asthma Intervention Research 2 (AIR2) studies, 7 have reported the efficacy and safety of BT. These studies showed that BT resulted in a reduction of asthma symptoms, improvement in quality of life, and reduction in asthma exacerbation rate, as well as the safety of BT, with no cases of death reported. However, these studies did not recruit subjects with severely impaired obstructive lung dysfunction. The AIR and AIR2 trials included only patients with a pre-bronchodilator forced expiratory volume in 1 second (FEV1) ⩾60%, and the RISA study included patients with a pre-bronchodilator FEV1 ⩾50%. 8 In real-world clinical situations, we often experience refractory asthma cases with severely declined FEV1 in whom BT would be efficacious for controlling asthma symptoms. We have performed BT in our hospital in 20 cases of refractory asthma since the Japanese insurance reimbursement in 2015. We retrospectively reviewed the clinical records of 15 patients who underwent BT and whose clinical data were available 6 months after BT. Our cases included six with a pre-bronchodilator % predicted FEV1 <60%.
This study aimed to assess the efficacy and safety of BT in patients with refractory asthma and to compare patients with a severely impaired FEV1 (pre-bronchodilator predicted FEV1 <60%) with those with a predicted FEV1 ⩾60%. We also examined changes in serum asthma-related cytokines/chemokines induced by BT.
Methods
Study design and patients
We retrospectively reviewed the medical records of 19 consecutive patients who underwent BT at Fujita Health University Hospital from June 2015 to December 2021. Among these patients, four who had been transferred to other hospitals were excluded from this study. All patients enrolled in this study had uncontrollable asthma symptoms despite combined extensive asthma therapy according to the GINA guideline (step 4 or 5). Before the BT, four patients had used biologics. Biologics that had been started in these patients at least 6 months before the BT were judged ineffective after at least 4 months of observation. To analyze the efficacy of BT, we collected clinical data, such as asthma medication, the exacerbation rate, blood test results including serum total immunoglobulin E concentrations and the peripheral blood eosinophil count, chest X-ray images, chest computed tomography images, pulmonary function test results, the Asthma Quality of Life Questionnaire (AQLQ) score, 9 and the Asthma Control Test (ACT) score, 10 before and 6 months after BT. Exacerbations were defined as episodes in which patients reported a short-term burst of oral prednisolone or had unscheduled hospital visits (including ER visits) and received systemic corticosteroids. In this study, the pulmonary function test, including spirometry, was performed according to the American Thoracic Society/European Respiratory Society technical standard method 11 by trained technicians using the CHESTAC-8900 (CHEST, Tokyo, Japan). The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 12
Bronchial thermoplasty
BT was performed under moderate sedation using midazolam and was adjusted by age and body weight according to a protocol described elsewhere. 13 A subcutaneous injection of pethidine hydrochloride (50 mg) was provided just before the procedure. We also performed continuous intravenous dexmedetomidine administration (10 min of loading with 4–6 µg/kg/h) followed by maintenance infusion with 0.4–0.5 µg/kg/h). After educational training in the procedure, bronchoscopists (H.I., S.I., Y.N., H.T., Y.K., and T.H.) performed BT under the review of a qualified instructor (K.I.) using the Alair™ Bronchial Thermoplasty System (Boston Scientific Corporation, Marlborough, MA, USA), which includes the Alair RF controller® and Alair RF catheter®. We used the BF p290F (outer diameter: 4.2 mm, working channel diameter: 2.0 mm; Olympus, Japan) for all BT procedures. BT was performed according to the standard clinical protocol. 14 In brief, 50 mg of prednisolone was administered orally for 5 days from 3 days before BT. Under a bronchoscopic view, accessible bronchi beyond the lobar bronchus were treated with the Alair RF catheter®. The electrode array at the tip of the catheter was expanded and attached to the bronchial wall. The catheter was energized for 5 s (activation), moving from distally to proximally. The activation time in each BT session was recorded. BT was scheduled to be repeated three times with an interval of at least 3 weeks. BT was performed on the right lower lobe bronchus in the first session. The left lower lobe bronchus was treated at the second session, and both upper lobes were treated at the final session. The right middle lobe was excluded from BT treatment. All patients were intubated and underwent continuous positive airway pressure (5–8 cm H2O) ventilation assistance during the BT treatment.
Cytokine/chemokine measurement
We measured serum transforming growth factor beta (TGF-β), regulated on activation, normal T cell expressed and secreted (RANTES), thymic stromal lymphopoietin (TSLP), tumor necrosis factor-related apoptosis-inducing ligand (TRAIL), and periostin concentrations just before each session of BT and 6 months after BT. Patients’ serum samples were obtained and stored at −80°C until we assessed cytokines/chemokines using the corresponding ELISA kit (R & D Systems Inc., Minneapolis, MN, USA) according to the manufacturer’s instructions.
Statistical analysis
All statistical analyses were performed using IBM SPSS version 24 (International Business Machines Corp., Armonk, NY, USA). The paired t-test or Wilcoxon signed-rank test was used to compare data obtained before and 6 months after BT. A two-sided p value <0.05 was considered statistically significant.
Results
Patients’ characteristics
Fifteen patients (nine men and six women) completed BT therapy (three sessions of BT). We obtained clinical data and serum samples just before each of the three BT sessions and 6 months after the accomplishment of BT from all 15 patients. The median age of the patients was 57 years (range: 35–74 years; Table 1). One patient was a current smoker, and three patients were ex-smokers. The median exacerbation rate/year was 10. The median ACT and AQLQ scores were 10 and 3.8, respectively, which indicated that the patients showed poorly controlled asthma symptoms despite GINA step 4/5 treatment (step 4, n = 5; step 5, n = 10). A total of 40% (6/15) of the patients regularly used oral corticosteroids (median dose: 10 mg/day), and 33% (n = 5) of the patients used biologics. Six patients showed <60% of the predicted pre-bronchodilator FEV1 before the BT treatment [group 1 (G1)]. In particular, four of the six patients in G1 showed <50% of the predicted FEV1 at baseline (32.2%–40.0%). The other nine patients showed ⩾ 60% of the predicted FEV1 before BT [group 2 (G2)]. The % predicted FEV1 was significantly lower in G1 than in G2 (p = 0.028). The background characteristics, symptom scores, T2 inflammatory markers (eosinophil count in peripheral blood, serum immunoglobulin E concentrations, and fractional exhaled nitric oxide), and treatment details were not significantly different between the groups (Table 1). We defined clinically significant T2 inflammation as blood eosinophilia (⩾300/μL), increased serum immunoglobulin E concentrations (⩾150 U/L), or increased fractional exhaled nitric oxide (⩾25/ppb) at the initial assessment before BT. Twelve patients had T2 inflammation (T2 phenotype), and three did not. All three patients with the non-T2 phenotype were included in G2 (Table 2).
Characteristics of the patients.

Values are expressed as the median with range unless otherwise indicated.
ACT, Asthma Control Test; AQLQ, Asthma Quality of Life Questionnaire; BMI, body mass index; BT, bronchial thermoplasty; FENO, fractional exhaled nitric oxide; FEV1, forced expiratory volume in 1 second; GINA, Global Initiative for Asthma; ICS, inhaled corticosteroid; LABA, long-acting beta-agonist; LAMA, long-acting muscarinic antagonist; N.S., not significant; OCS, oral corticosteroid; pts, patients.
Effect of bronchial thermoplasty on asthma-related medication and the long-term response in individual patients in groups 1 and 2.
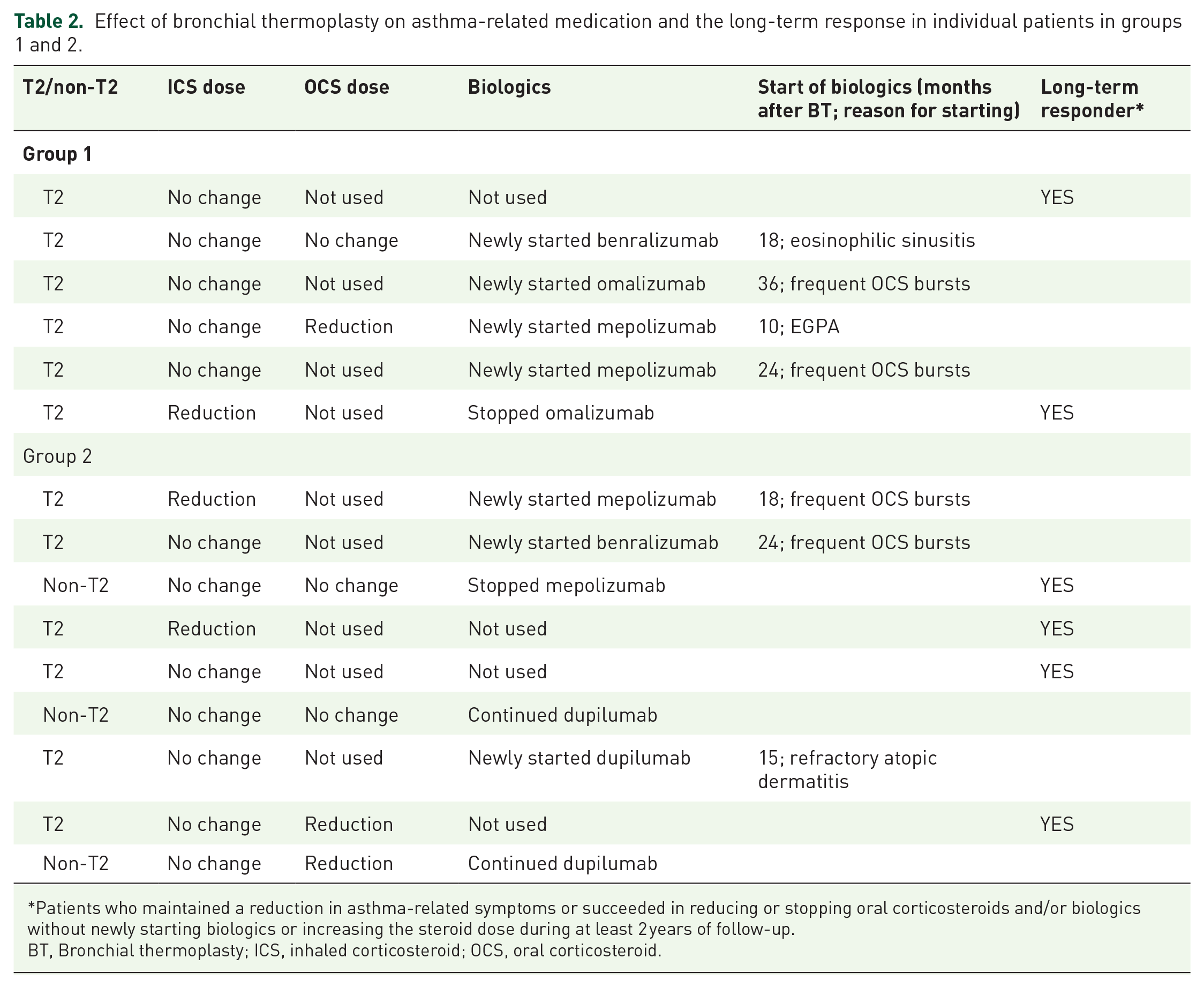
Patients who maintained a reduction in asthma-related symptoms or succeeded in reducing or stopping oral corticosteroids and/or biologics without newly starting biologics or increasing the steroid dose during at least 2 years of follow-up.
BT, Bronchial thermoplasty; ICS, inhaled corticosteroid; OCS, oral corticosteroid.
Efficacy of BT
Changes in asthma-related symptoms and frequency of exacerbation
BT significantly improved asthma-related symptoms and the quality of life (Figure 1). In all patients, ACT and AQLQ scores (higher scores indicated better asthma symptom control or asthma-related quality of life) were significantly increased 6 months after the completion of BT compared with those before BT. The mean (±standard error) ACT score increased from 9.33 ± 1.18 before BT to 21.1 ± 0.8 at 6 months after BT (p < 0.0001). The mean AQLQ score also increased from 3.43 ± 1.21 before BT to 5.28 ± 1.3 after BT (p < 0.0001). These effects of BT on improving the ACT and AQLQ scores were observed regardless of airflow obstruction (similarly observed in G1 and G2). BT also significantly reduced the frequency of asthma exacerbation in both groups (Figure 2).

BT significantly improves asthma-related symptoms and the quality of life in patients with severe asthma, regardless of the level of airflow limitation. ACT scores (a, b, c) and AQLQ scores (d, e, f) before BT and 6 months later were compared using the Wilcoxon signed-rank test. (a, d) The total patients, (b, e) patients with a predicted FEV1 <60%, (c, f), and patients with a predicted FEV1 ⩾ 60% are shown.

BT significantly reduces the frequency of acute asthma exacerbation, regardless of the level of airflow limitation. (a) Total patients, (b) patients with a predicted FEV1 <60%, and (c) patients with a predicted FEV1 ⩾ 60%.
Change in pulmonary function
We compared pulmonary function before and 6 months after BT [Figure 3(a)–(c)]. Forced vital capacity did not significantly change between before and 6 months after BT in the total patients or in G1 or G2. However, in some cases, FEV1 was increased 6 months after BT [Figure 3(d)]. Especially in G1, FEV1 was significantly increased 6 months after BT compared with that before BT [Figure 3(e), p = 0.0248]. In contrast, FEV1 did not change after BT in G1.

BT changes pulmonary function in patients with severe airflow limitation. FVC did not change between before and 6 months after BT in the total patients (a), patients with a predicted FEV1 <60% (b), or in patients with a predicted FEV1 ⩾60% (c). However, FEV1 was increased 6 months after BT in some patients (d). Especially in patients with a predicted FEV1 <60%, FEV1 was significantly increased after BT compared with before BT (p = 0.0248) (e). However, FEV1 did not change in patients with a predicted FEV1 ⩾60%.
Changes in asthma medication
BT also affected asthma medication (Table 2). The inhaled corticosteroid dose was reduced in three patients (one in G1 and two in G2). Among six patients (two in G1 and four in G2) who used regular oral corticosteroids (OCS) before BT, three (one in G1 and two in G2) reduced the dose of corticosteroids. No patients increased the dose of inhaled corticosteroids or newly started regular OCS. However, one patient (G2) who reduced the inhaled corticosteroid dose and another (G1) who declined OCS started biologics 18 and 10 months after BT, respectively. No change in the dose of long-acting beta-agonists, long-acting muscarinic antagonists, or leukotriene receptor antagonists was observed after BT.
Complications
BT transiently worsened asthma-related symptoms in some patients (Table 3). Two patients in G2 experienced asthma exacerbation with bacterial broncho-pneumonia, requiring additional systemic corticosteroids, antibiotics, and prolonged hospitalization (within 1 week). Another two patients in G2 experienced asthma exacerbation, requiring additional systemic corticosteroids without prolonged hospitalization. Surprisingly, no apparent asthma exacerbation occurred after BT in G1, except for one patient with an unscheduled emergency room visit between the first and second BT sessions. Thermal lung injury (transient lung infiltration observed in surrounding areas along the treated bronchi) was frequently observed in G1 and G2 (100% and 77.8%, respectively, no significant difference between the groups). There were no other serious complications related to BT, such as pneumothorax, arrhythmia, or heart failure. No deaths were reported. The activation times (frequency) of BT were equivalent between both groups.
Complications in individual patients in groups 1 –and 2.

AE, acute exacerbation of asthma symptoms; BT, Bronchial thermoplasty; ER; emergency room; STH, corticosteroids; TI, thermal injury.
Mean activation time in three BT sessions.
Changes in serum cytokines/chemokines and T2 mediators
To investigate whether BT affects systemic inflammation, we measured serum cytokines and mediators, which comprised TGF-β, RANTES, TRAIL, TSLP, and periostin, just before the first, second, and third procedures and at 6 months after the third session. Two patients were excluded from this analysis because they continued biologics before BT and immediately after the third procedure of BT. TGF-β, RANTES, TSLP, and periostin concentrations did not significantly change during three times of BT procedures and 6 months later (Figure 4). TRAIL concentrations did not increase during BT but were significantly increased 6 months after the final BT compared with those before the first BT (p = 0.0067). These changes in cytokines/chemokines or mediators were equivalent between both groups.

Changes in serum cytokines/chemokines and T2 mediators during and after BT. TGF-β (a), RANTES (b), TRAIL (c), TSLP (d), and periostin (e) concentrations just before the first, second, and third procedures and 6 months after the third session of BT. These cytokines/mediators did not show a significant change during three times of BT procedures and 6 months later, except for TRAIL concentrations, which were significantly increased 6 months after the final BT compared with those before the first BT (p = 0.0067).
Long-term effectiveness of BT
Some patients newly started biologics or continued systemic corticosteroids after BT because their asthma symptoms deteriorated again in the long-term follow-up after BT. Among them, seven patients (four patients in G1 and three patients in G2) started biologics. Four patients (two patients in G1 and two in G2) experienced apparent exacerbation of asthma symptoms, which required frequent bursts of systemic corticosteroid. In addition, three patients (two in G1 and one in G1) developed additional refractory allergic disorders, which comprised severe eosinophilic sinusitis, eosinophilic granulomatosis with eosinophilia, and uncontrollable atopic dermatitis in one patient each (Table 2). However, we identified six (40.0%) patients who maintained a reduction in asthma-related symptoms or succeeded in reducing or stopping OCS and/or biologics without newly started biologics or an increase in the steroid doses during at least 2 years of follow-up as long-term responders of BT (Table 2). There were two (33.3%) long-term responders among the six patients in G1 and four (44.4%) responders among the patients in G2 (not significantly different, p = 0.32). One (33.3%) patient among three non-T2 patients was a long-term responder, whereas 41.7% (5/12) of T2 patients showed long-term effectiveness of BT.
Discussion
Recently, therapy for uncontrollable asthma has been improving along with the introduction of biologics. Highly effective biologics are available, and systemic corticosteroids (oral and parental) are recommended to be avoided wherever possible. There has been increasing interest in precision medicine and a multidimensional therapeutic approach for severe asthma.15,16 Although biologics are effective in some patients, they do not show an improvement in asthma symptoms in all patients. Alternative therapeutic options need to be established for patients who do not respond to biologics. BT is another therapeutic option for patients with severe asthma.
Some clinical studies have already reported the efficacy and safety of BT, and more recent reports have shown a long-term effect of BT (8). However, most large-scale studies did not include patients with severely impaired respiratory function. The AIR and AIR2 trials included patients whose predicted FEV1 was >60%.5,7 The efficacy and safety of BT in patients with severe obstructive airflow limitation (predicted FEV1 <60%) have yet to be fully determined. Our study showed that BT was equally effective and safe in patients with a predicted FEV1 <60% and in patients with less damaged air flow limitation (predicted FEV1 <60%). A few studies have reported successful BT in patients with severe airflow limitation.17–19 A study by Langton et al., which included 68 patients (36 patients with a % predicted FEV1 <50%), showed no significant improvement in FEV1 after BT and no difference in % predicted FEV1 before BT between responders and non-responders. 18 Iikura et al. reported their BT experience in 12 patients, including five patients with a predicted FEV1 <60%. 19 They showed a significant improvement in FEV1 after BT, which differs from the result of other trials. They suggested that their ‘patient selection criteria’, which included patients with severe airflow limitation (predicted FEV1 <60%), might be one of the reasons for the difference between studies. Our study focused on the difference in the effect and safety of BT by comparing two groups (patients with and without severe airflow limitation). We found a significant improvement in FEV1 only in patients with a predicted FEV1 <60%, while asthma-related symptoms improved, regardless of the % predicted FEV1 before BT. Therefore, our study clearly showed the benefit of BT for patients with refractory asthma and severe airflow limitation, as well as the safety of BT. Our study suggests that we should not exclude patients from the indication of BT only by the degree of airflow limitation.
In this study, BT transiently improved asthma-related symptoms and the frequency of asthma exacerbation. However, almost 40% of the studied patients experienced re-deterioration of asthma symptoms, and they newly started biologics. We detected six (40.0% of the total patients and 33.3% of G1 patients) long-term responders of BT. There was no predictable biomarker to select patients who would show an extended response to BT. The six long-time BT responders comprised patients with or without elevated T2 inflammatory markers. Serial serum cytokines or mediators did not significantly change in our study, except for an elevation in TRAIL concentrations 6 months after BT. However, the mechanism of this finding is unknown. Although predicting the efficacy of BT is challenging, some patients with severely limited airflow could be long-time BT responders.
Our study has several limitations. First, this was a single-center study with a limited number of patients. Therefore, the evidence of the effectiveness and safety of BT in our study might be weak. However, an increasing number of studies have shown that BT is an important alternative for refractory asthma. The results of our study are consistent with the results of other studies. Second, because our study was retrospective, additional therapies other than BT were administered depending on attending physicians’ decisions. These additional therapies might have made the net effect of BT in our study unclear. However, our study showed the efficacy and safety of BT in patients with severe airflow limitations in a real-world setting. Finally, we cannot exclude the possible influence of the expertise at our institute. However, the years of experience of bronchoscopists who performed BT in our study ranged from 3 to 7 years. Therefore, general bronchoscopists might not have to be experts to safely perform BT.
At the end of 2022, Boston Scientific Co. informed us that they had decided to stop providing equipment (radiofrequency probes) of BT soon because of material supply chain defects. This potential loss of an effective therapeutic alternative for severe bronchial asthma other than biologics is disappointing. We hope that this situation will change in the future.
Conclusion
BT appears to be an effective and safe therapeutic method, even for patients with severe asthma and advanced airflow limitation. BT should be an essential alternative for patients who are refractory to biologics or inadaptable patients.
Supplemental Material
sj-doc-1-tar-10.1177_17534666241254980 – Supplemental material for Safety and efficacy of bronchial thermoplasty in refractory asthma with severe obstructive respiratory dysfunction
Supplemental material, sj-doc-1-tar-10.1177_17534666241254980 for Safety and efficacy of bronchial thermoplasty in refractory asthma with severe obstructive respiratory dysfunction by Takahiro Inoue, Sumito Isogai, Naoki Yamamoto, Noriko Hiramatsu, Yoshikazu Niwa, Hideaki Takahashi, Yutaro Kimura, Tomoya Horiguchi, Yasuhiro Goto, Naozumi Hashimoto and Kazuyoshi Imaizumi in Therapeutic Advances in Respiratory Disease
Footnotes
Acknowledgements
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
